Abstract
Background:
A critical factor in healing diabetic foot ulcers is patient adherence to offloading devices. We tested a smart offloading boot (SmartBoot) combined with a smartwatch app and cloud dashboard to remotely monitor patient adherence and activity. In addition, the impact of SmartBoot on balance, gait, and user experience was investigated.
Methods:
Fourteen volunteers (31.6±8.7 years; 64% female) performed natural activities (eg, sitting, standing, walking) with and without the SmartBoot for approximately 30 minutes. All participants completed balance tests, 10-meter walking tests at slow, normal, and fast pace while wearing the SmartBoot, and a user experience questionnaire. The accuracy of real-time adherence reporting was assessed by comparing the SmartBoot and staff observation. Center of mass (COM) sway and step counts were measured using a validated wearable system.
Results:
Average sensitivity, specificity, and accuracy for adherence and non-adherence were 90.6%, 88.0%, and 89.3%, respectively. The COM sway area was significantly smaller with the SmartBoot than without the SmartBoot regardless of test condition. Step count error was 4.4% for slow waking, 36.2% for normal walking, 16.0% for fast walking. Most participants agreed that the SmartBoot is easy to use, relatively comfortable, nonintrusive, and innovative.
Conclusions:
To our knowledge, this is the first smart offloading system that enables remote patient monitoring and real-time adherence and activity reporting. The SmartBoot enhanced balance performance, likely due to somatosensory feedback. Questionnaire results highlight SmartBoot’s technical and clinical potential. Future studies warrant clinical validation of real-time non-adherence alerting to improve wound healing outcomes in people with diabetic foot ulcers.
Introduction
Diabetic foot ulcers (DFUs) are the most severe end-stage complication of diabetic feet, 1 primarily resulting from peripheral neuropathy and ischemia.2,3 Diabetic foot ulcers occur in up to 26.1 million people with diabetes worldwide annually and have a recurrence rate of about 40% within 1 year after ulcer healing. 4 More than 50% of DFUs become infected, 5 and approximately 15% of DFUs lead to lower extremity amputations.6-8 Indeed, every 1.2 seconds someone in the world develops an ulcer, and every 20 seconds someone loses a lower limb due to diabetes. 4 A population-based cohort study has reported that the development of DFUs is associated with a 5% mortality within the first year and a 42% mortality within 5 years. 9 Indeed, patients with DFUs have 2.5 times risk of death than patients without DFUs. 9 Patients with DFUs generally experience significant morbidity, reduced quality of life, and poor psychosocial adjustment. 10 Health care costs and interactions are often burdensome.11,12 For instance, it is estimated that one third of all diabetes-related costs are spent on diabetic foot care in the United States, with two thirds of these costs incurred in the inpatient settings, constituting a substantial cost to society.13,14
Protective offloading of the diabetic foot remains the key principle of basic DFU management.15-18 There are four offloading intervention categories: (1) offloading devices (removable or non-removable and ankle-high or knee-high devices); (2) footwear (shoes, insoles, socks, etc); (3) surgery (silicone injections, tissue augmentation, tenotomy, etc); and (4) other offloading techniques (bed rest, wheelchairs, braces, etc). In 2020, a thorough systematic review reported that offloading devices are more effective in the prevention and healing of DFUs than the other categories. 17
Offloading devices relieve and redistribute the mechanical stresses of plantar pressure (acting normal to the skin surface) and shear stresses (acting tangential to the skin surface) during weight-bearing activities. 19 Adherence to wearing offloading devices improves their effectiveness in preventing and healing DFUs. Unfortunately, most activity taken in patients with open neuropathic diabetic foot wounds is unprotected. 20 This has led many clinicians to promote the use of non-removable offloading devices. While a non-removable offloading device is considered as a gold standard to treat DFU, there are several limitations including poor acceptability due to its irremovability during daily activity (eg, shower and sleep), poor scalability (eg, less than 2% of US clinics regularly prescribe irremovable offloading), 21 poor patient-centered outcomes (eg, poor sleep quality), 22 and high likelihood of frailty/leg muscle atrophy due to prolonged immobilization of the ankle joint leading to high recurrence rate of DFU.11,15,17
To address the limitations of non-removable offloading devices, we proposed the use of a smart offloading which enables reinforcement of adherence via smartwatch and gamification as well as remote patient monitoring including remote monitoring of adherence and weight-bearing physical activity dosage. More specifically, this study has three objectives: (1) design and test a smart removable offloading boot system (SmartBoot) that will remotely measure and monitor the level of adherence and provide real-time alerts and behavioral feedback to reinforce adherence, through a 4G LTE-enabled smartwatch application, clinician dashboard, cloud storage, and web services; (2) quantitatively define an accuracy measure for monitoring adherence; and (3) evaluate SmartBoot’s influence on patient activity, balance control, gait performance (ie, step count), and user experience.
Methods
Smart Offloading Boot System
Figure 1 shows the SmartBoot, developed by Sensoria Health Inc., Redmond, WA, USA, that consists of: (1) an offloading boot equipped with Sensoria Core microelectronics; (2) a smartwatch with dedicated patient monitoring app and 4G LTE Internet of Things (IoT) sim card enabled; and (3) a cloud-based clinical dashboard. The offloading boot is a commercially available removable knee-high footwear equipped with two Velcro straps for the foot and three Velcro straps for the lower leg (Figure 1a). The Sensoria Core microelectronics include a microcontroller unit, a miniaturized 6 degrees-of-freedom inertial measurement unit (IMU) including a tri-axial accelerometer and gyroscope, a Bluetooth Low Energy (BTLE) communication module, and a rechargeable battery (Figure 1a). The patient’s smartwatch has a dedicated mobile application installed, monitoring the level of adherence to wearing the boot and providing real-time alerts and behavioral feedback depending on daily patient adherence and activity (Figure 1b). The clinician dashboard provides web services with cloud storage, which allows the care team to monitor activity and adherence remotely and in near real time (Figure 1c).
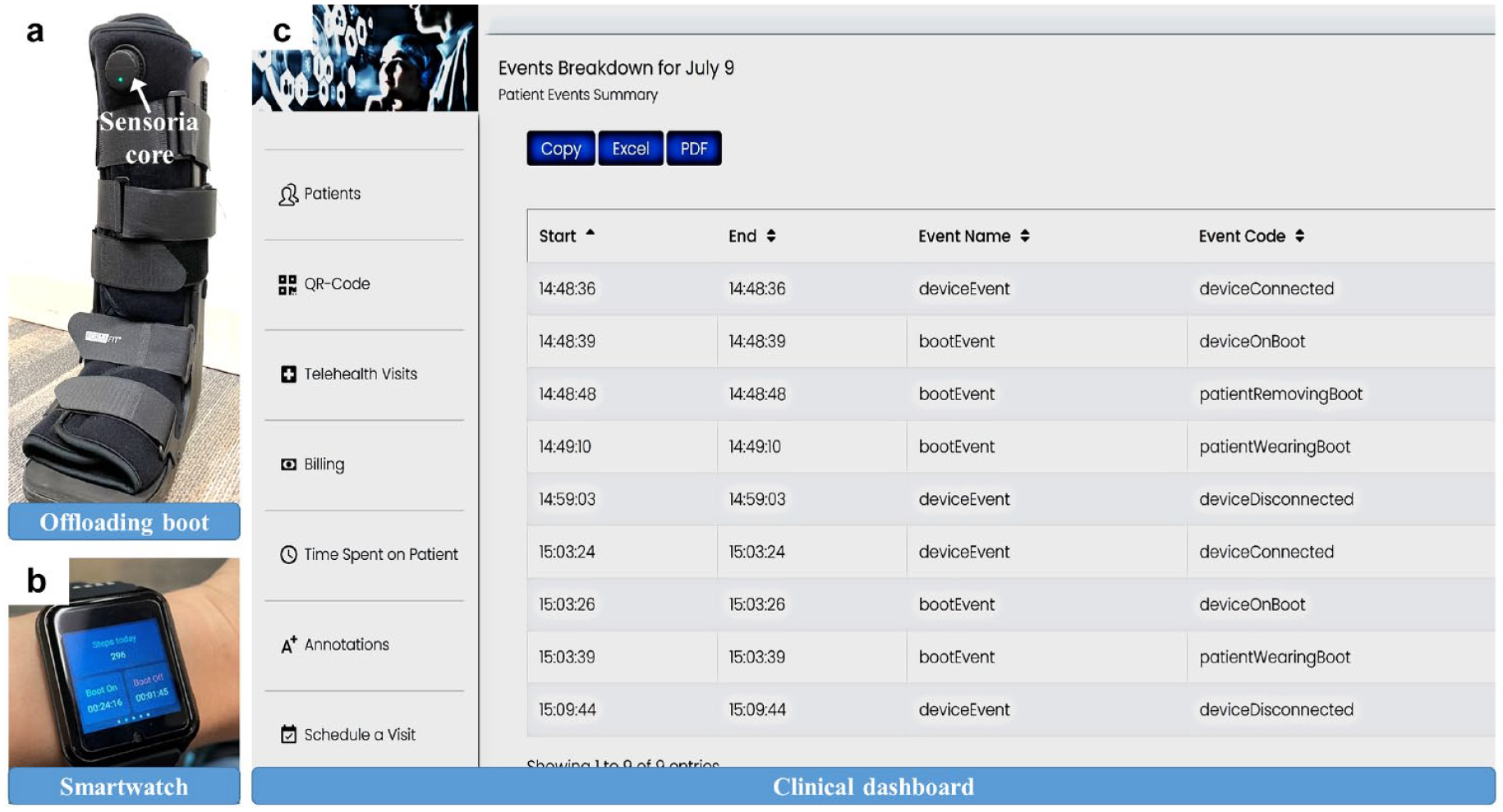
SmartBoot. (a) Offloading boot equipped with Sensoria Core. (b) Smartwatch. (c) Clinical dashboard. In the clinical dashboard, "deviceEvent” indicates whether the Sensoria Core is connected to the boot (“deviceConnected”) or not (“deviceDisconnected”). “bootEvent” indicates a status of the boot (the Sensoria core is on [“deviceOnBoot”], the boot is worn by a user (“patientWearingBoot”), the boot is not worn by a user (“patientRemovingBoot”)).
IMU data were processed using a set of Butterworth filters to remove movement artifacts by reducing errors for estimation of steps and adherence to offloading. To detect whether a user was wearing the boot or not, the accelerometer was leveraged: a detection algorithm detected movements on a scale typical of a human being wearing a boot in predefined positions or activity-related conditions, that is, sitting, standing, walking, lying down. In addition, a set of peak detection algorithms using accelerometer and gyroscope data measured step counts taken by a user while wearing the boot and/or the watch. If no movement at all was detected for a minimum of 10 minutes, the boot was classified as removed, and a real-time alert signal was sent to a user through the mobile application on the smartwatch. The real-time alert signal was also recorded into cloud infrastructure to inform the care team that the boot was removed via the clinician dashboard. Once the algorithm automatically detected the typical movements of a user wearing the boot, a notification appeared on the mobile application on the smartwatch to inform the user that the boot was worn. The application featured a second mechanism to detect boot removal exploiting the watch step detector. If at least eight steps were detected from the watch, no steps were detected from the boot, and no movement was registered for the boot; the boot was considered to be not worn.
The clinician dashboard allows a care team, including researchers and clinicians, to monitor the amount of time that a user has been wearing the boot. The clinician dashboard also shows additional events (connection/disconnection of the boot to the smartwatch and charging/discharging of the battery embedded in Sensoria Core microelectronics). Using the clinical dashboard, the care team can evaluate if the boot has been worn as prescribed. In addition, an adherence score is automatically calculated based on the amount of time that a user has been wearing the boot. Through the clinical dashboard, a patient can consult with their clinician about adherence to wearing the boot, and the care team can reinforce user adherence. If necessary, then the care team has the option to interact with a patient via telehealth or in-person to make the boot non-removable for patients that are unable to comply with the minimum level of expected adherence required for the expected DFU healing outcome (reinforcement).
Participants
Eligible participants were recruited from Baylor College of Medicine. The validation of the SmartBoot required significant weight-bearing activities (eg, walking and standing) that could have been risky for people with DFUs or at risk of DFUs. Therefore, for this phase of the study, we limited inclusion to healthy adults. Fourteen healthy adults (31.6 ± 8.7 years; 9 females, 5 males) were eligible to participate in this study. Inclusion criteria included healthy adults aged 18 and over. Exclusion criteria included: (1) musculoskeletal dysfunction; (2) neurological disorder (eg, stroke, Parkinson’s disease); (3) use of any walking aid; and (4) unwillingness to participate.
The Baylor College of Medicine Institutional Review Boards approved the experimental protocol, which conformed to the Helsinki Declaration (protocol: H-40765). Informed consent was obtained from each participant prior to the start of the experiment.
Experimental Protocol
The experimental design included an accuracy measure for adherence to wearing the boot, and balance and gait assessments. For the accuracy measure, all participants were asked to perform daily activities (eg, sitting, standing, walking) while wearing and not wearing the boot. During daily activities, all participants wore the smartwatch that measures the time to wear the boot and a time not to wear the boot, as shown in Figure 2a.

(a) A representative measured time to wear the SmartBoot (ie, “Boot On”) and not to wear the SmartBoot (ie, “Boot Off”) displayed on the smartwatch. (b) A representative image showing that a participant performed a balance assessment while wearing the smartwatch, boot, and five wearable sensors attached on the lower back, upper thighs, and lower shins.
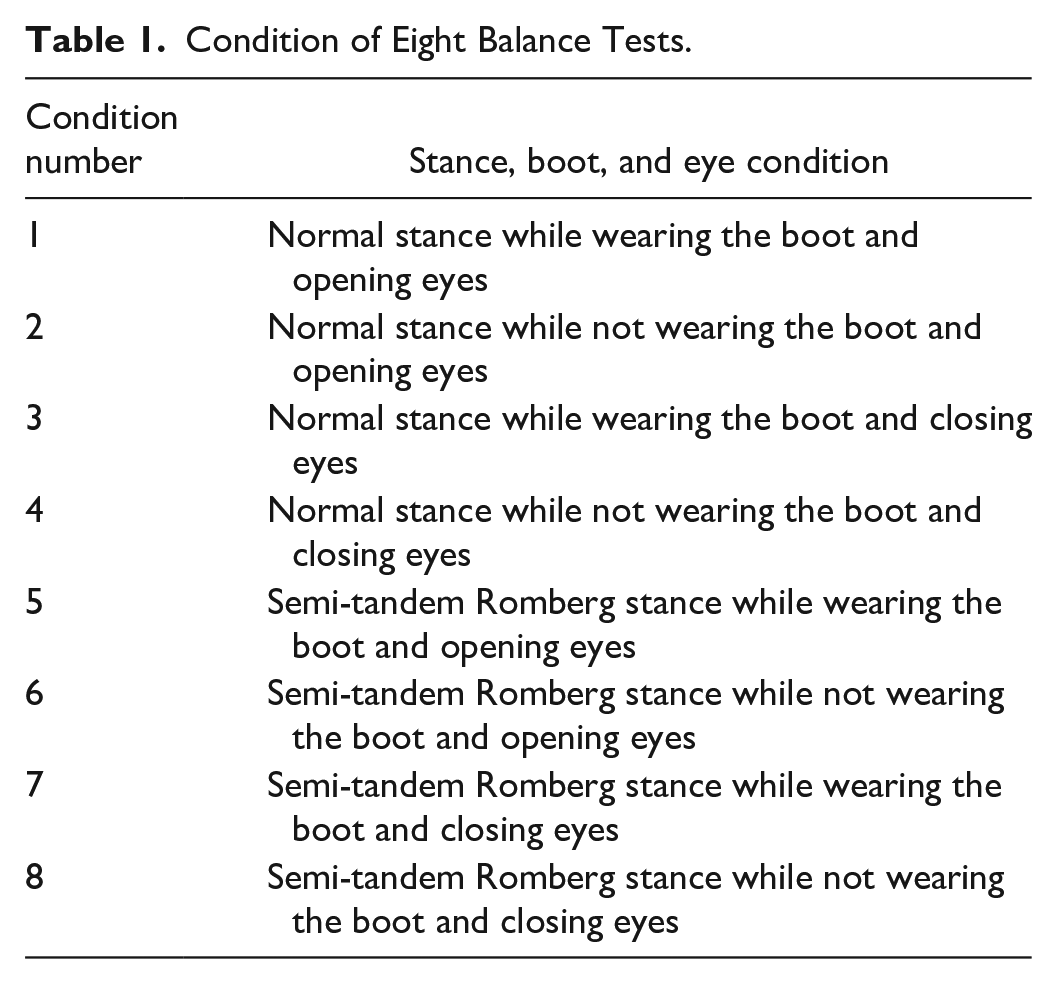
For balance and gait assessments, a validated wearable system (ie, BalanSens and LegSys, BioSensics, Watertown, MA, USA) with five wearable sensors attached to the lower back, upper thighs, and lower shins of all participants, as shown in Figure 2b, was used. After the instrumentation, all participants completed a total of eight balance tests (two stance conditions [normal and semi-tandem Romberg] × two boot conditions (wearing and not wearing the boot) × two eyes conditions [open and closed]), as listed in Table 1. The normal stance was defined as having the feet hip-width apart and parallel. Semi-tandem Romberg stance was defined as placing one foot in front of the other foot, with the big toe of the back foot in the groove of the front foot. While participants were performing the balance tests, they were instructed to stand upright, keep their knees extended, cross their arms at their chest, and breathe normally for 30 seconds. The order of the stance, eyes, and boot condition was randomized. Consecutive balance tests were separated by an approximately 20-second rest period. Participants were instructed to relax during the rest period by bending the torso and shaking their upper and lower legs. After the completion of all eight balance tests, participants were asked to walk 10 meters walking at a slow, normal, and fast pace while wearing the SmartBoot. All participants self-selected walking pace for slow, normal, and fast walking. The validated wearable system measured the area of body’s center of mass (COM) sway during balance assessments and step counts during gait assessments.23-25
Condition of Eight Balance Tests.
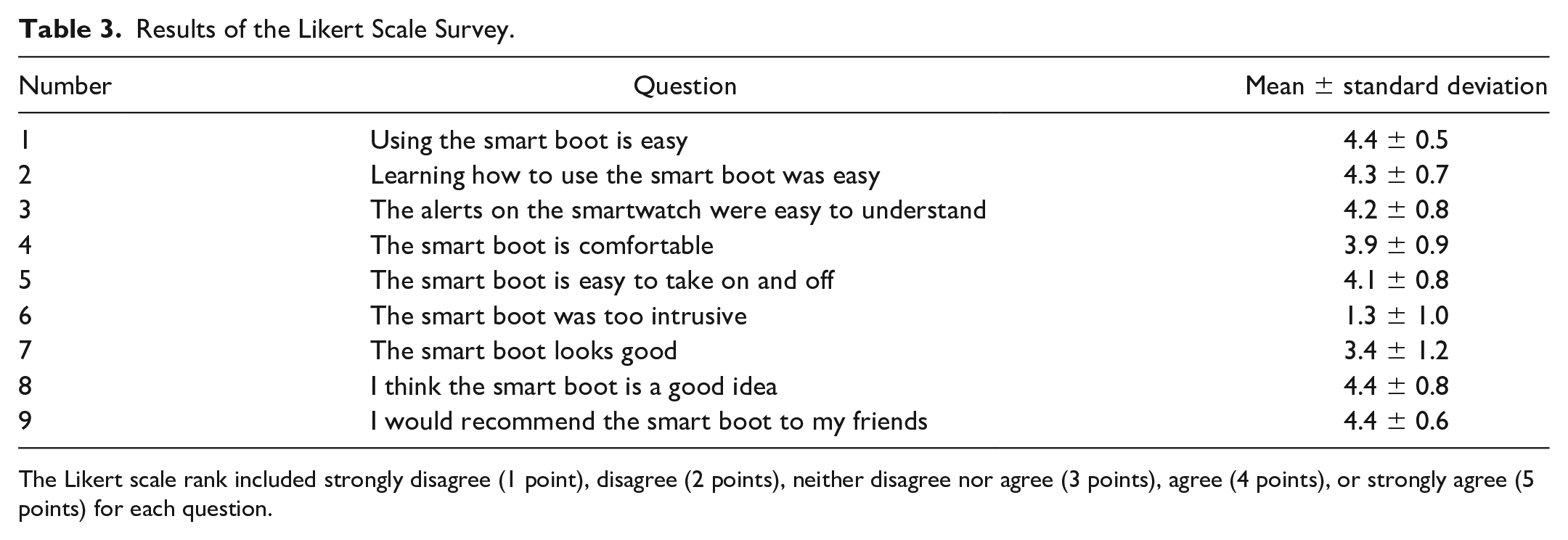
Following the completion of the experimental protocol, each subject was asked to fill out a Likert scale survey that was designed based on the technology acceptance model (TAM). 26 The TAM includes two major components: perceived usefulness and perceived ease of use, which both tailored for the use of novel footwear as suggested by Wang et al. 25 The survey questions were (Q1) Using the smart boot is easy; (Q2) Learning how to use the smart boot was easy; (Q3) The alerts on the smartwatch were easy to understand; (Q4) The smart boot is comfortable; (Q5) The smart boot is easy to take on and off; (Q6) The smart boot was too intrusive; (Q7) The smart boot looks good; (Q8) I think the smart boot is a good idea; and (Q9) I would recommend the smart boot to my friends. The Likert scale (5-point scale from 1-5) was used to quantify how much they disagreed with each statement, including “strongly disagree,” “disagree,” “neither disagree nor agree,” “agree,” and “strongly agree,” respectively. In this article, responses were only categorized as negative (1-2 points) or positive (4-5 points) attitudes toward the use of the SmartBoot.
Data and Statistical Analysis
The main outcome measure was time for wearing and not wearing the SmartBoot, measured by the smartwatch at the second unit. Figure 2a shows measured time to wear the boot as “Boot On” and not to wear the boot as “Boot Off.” Using measured time, the accuracy measure for adherence was evaluated by the sensitivity, specificity, and accuracy. The sensitivity, specificity, and accuracy were computed as:
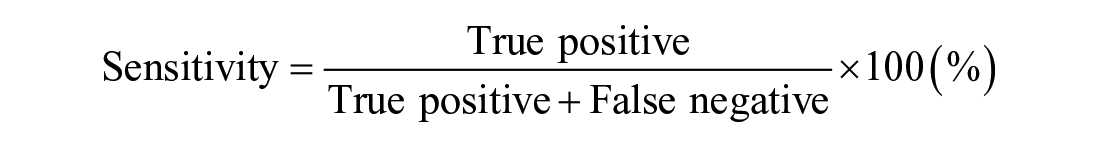
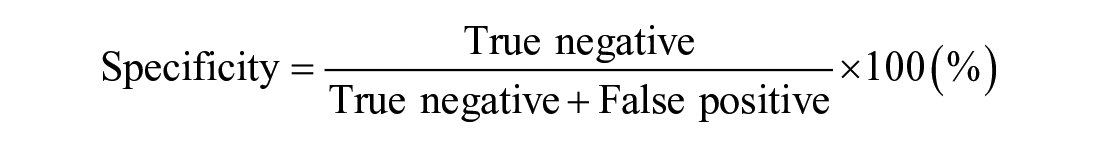
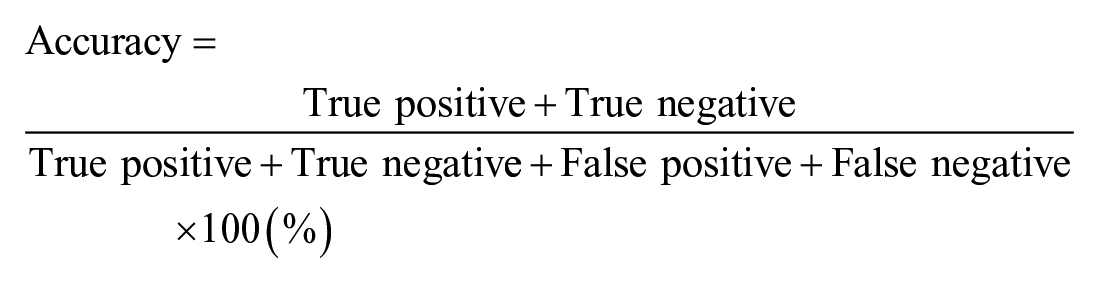
True positive indicates a measured time as “Boot On” on the smartwatch while a participant was wearing the boot (see Figure 2a). False negative indicates a measured time as “Boot Off” on the smartwatch while a participant was wearing the boot (see Figure 2a). True negative indicates a measured time as “Boot Off” on the smartwatch while a participant was not wearing the boot. False positive indicates a measured time as “Boot On” on the smartwatch while a participant was not wearing the boot. The value of the sensitivity, specificity, and accuracy ranges from 0% to 100%. Although there is no gold standard for a value of the sensitivity, specificity, and accuracy, a value close to 100% is ideal. In general, a high sensitivity of 80% or more indicates that the smart offloading boot system can measure the time of wearing the boot with more than 80% accuracy. A high specificity of 80% or more indicates that the smart offloading boot system can measure the time of not wearing the boot with more than 80% accuracy. A high accuracy of 80% or more indicates that the smart offloading boot system can measure the time of wearing and not wearing the boot with more than 80% accuracy. The value of the sensitivity, specificity, and accuracy was computed for each participant, and its mean, standard deviation, and 95% confidence interval (CI) were computed across all participants.
The COM sway area measured by the validated wearable system (BalanSens) was analyzed using SPSS (IBM Corp., Armonk, NY, USA). The Shapiro–Wilk test was used to assess the distribution of COM sway area data for eight balance tests (two stance conditions [normal and semi-tandem Romberg] × two boot conditions [wearing and not wearing the boot] × two eyes conditions [open and closed]). A one-way analysis of variance (ANOVA) was used for normally distributed COM sway area data, and a Mann–Whitney U test was used for non-normally distributed COM sway area data. Effect sizes were computed using Cohen’s d for eight balance tests, where Cohen’s d values < 0.20 indicate no noticeable effect, values between 0.20 and 0.49 indicate a small effect, values between 0.50 and 0.79 indicate a medium effect, and values above 0.80 indicate a large effect. 27 For all statistical analyses, the level of significance was set at the two-sided P < .05.
Using measured step counts with the SmartBoot (Step countsSB) and measured step counts with the validated wearable system (Step countsWS), the percentage error of step counts was computed as:
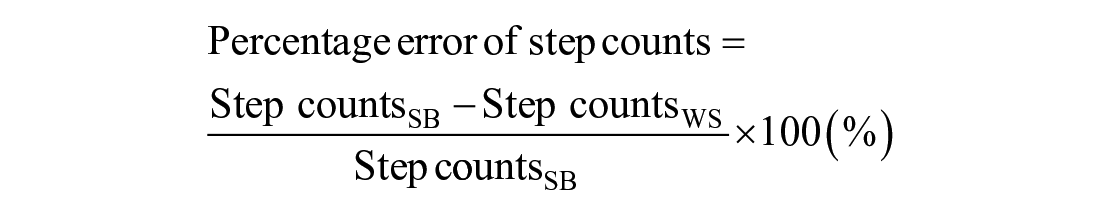
The average and standard deviation of the percentage error of step counts were calculated across all participants. In addition, the average and standard deviation of the gait speed measured by the validated wearable system were calculated across all participants for their slow, normal, and fast walking.
To assess user experience of the SmartBoot, the Likert scale responses were averaged over all participants for each survey question. The standard deviation of the corresponding average was also calculated.
Results
The average time spent in performing natural activities across all participants was 29.5 ± 11.7 minutes. Table 2 reports the results of the sensitivity, specificity, and accuracy. The average sensitivity, specificity, and accuracy values across all participants were 90.56%, 88.04%, and 89.29%, respectively. The 95% CI values ranged from 85.25% to 95.87% for sensitivity, 81.29% to 94.80% for sensitivity, and 85.08% to 93.50% for sensitivity. The 90.56% sensitivity rate (ie, true positive rate), 88.04% specificity rate (ie, true negative rate), and 89.29% accuracy rate indicate that the SmartBoot can detect adherence and non-adherence to wearing the boot accurately.
Results of the Accuracy Measure for Adherence to Wearing the Boot.
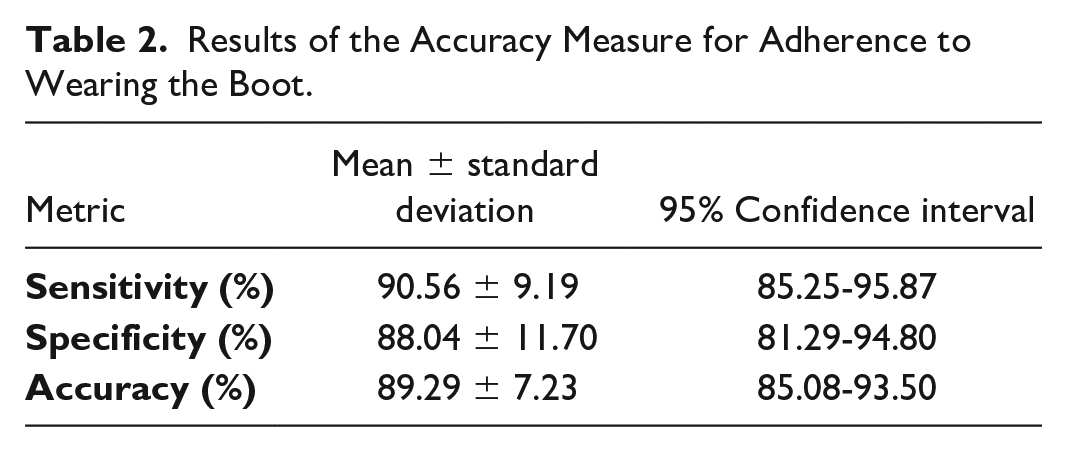
Figure 3 shows the results of the COM sway area as a function of the two eyes conditions (eyes open and closed) and two boot conditions (wearing and not wearing boot) during normal stance. With wearing the boot compared to not wearing the boot, participants showed a significant decrease in the COM sway area during eyes open (P = 0.002) and closed (P = 0.041). The Cohen’s d effect size was large for both eyes conditions (eyes open: 1.22 and eyes closed: 0.85). Figure 4 shows the results of the COM sway area as a function of the two eyes conditions (eyes open and closed) and two boot conditions (wearing and not wearing boot) during semi-tandem Romberg stance. With wearing the boot compared with not wearing the boot, participants showed a significant decrease in the COM sway area during eyes open (P = 0.012) and closed (P = 0.013). The Cohen’s d effect size was large for the eyes open condition (1.0) and medium for the eyes closed condition (0.67). The results of the COM sway area indicate that balance control of all participants was more stable with wearing the boot than with the boot off, regardless of whether participants had their eyes open and closed during normal and semi-tandem Romberg stance.
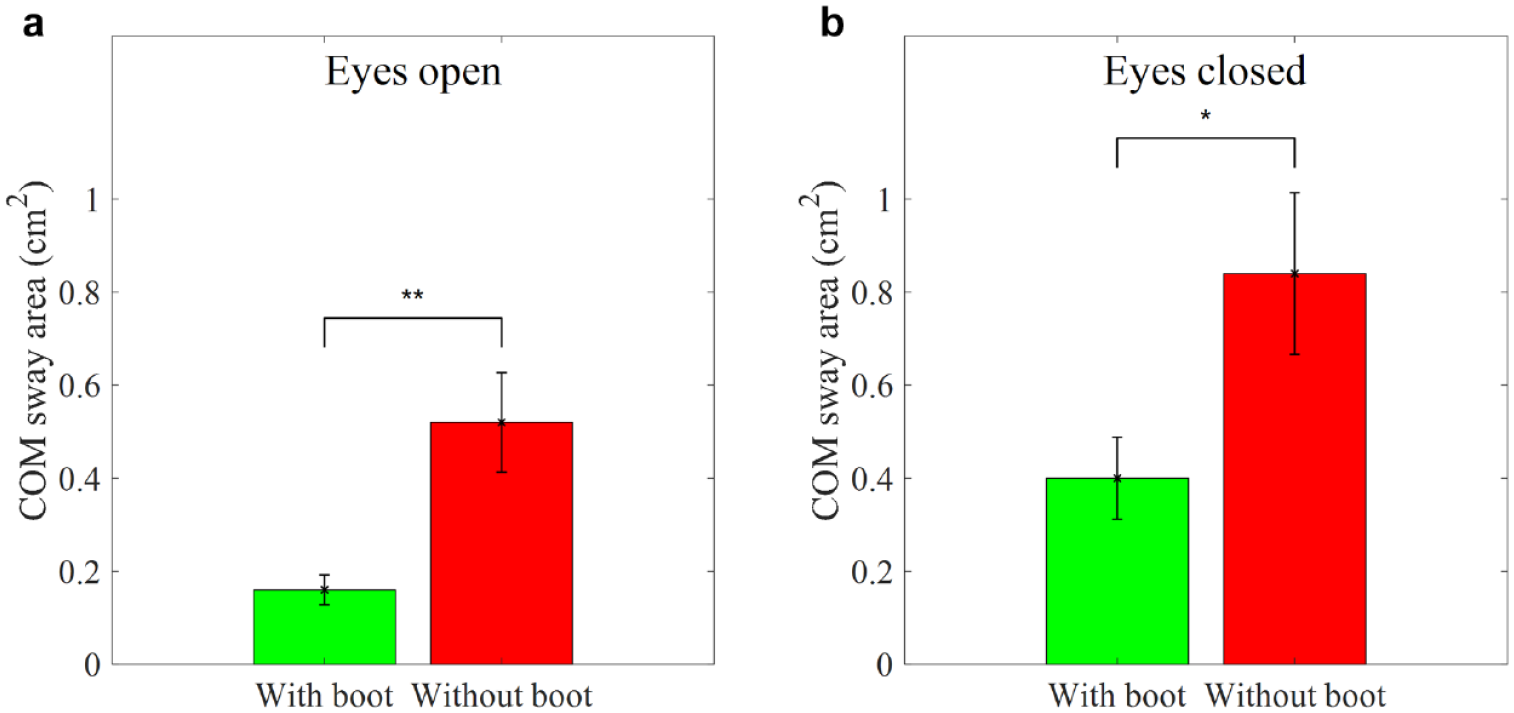
Average COM sway area while wearing and not wearing the SmartBoot in the normal stance. (a) Eyes open condition. (b) Eyes closed condition. Error bars indicate the standard error of the corresponding average (*P < .05 and **P < .01).
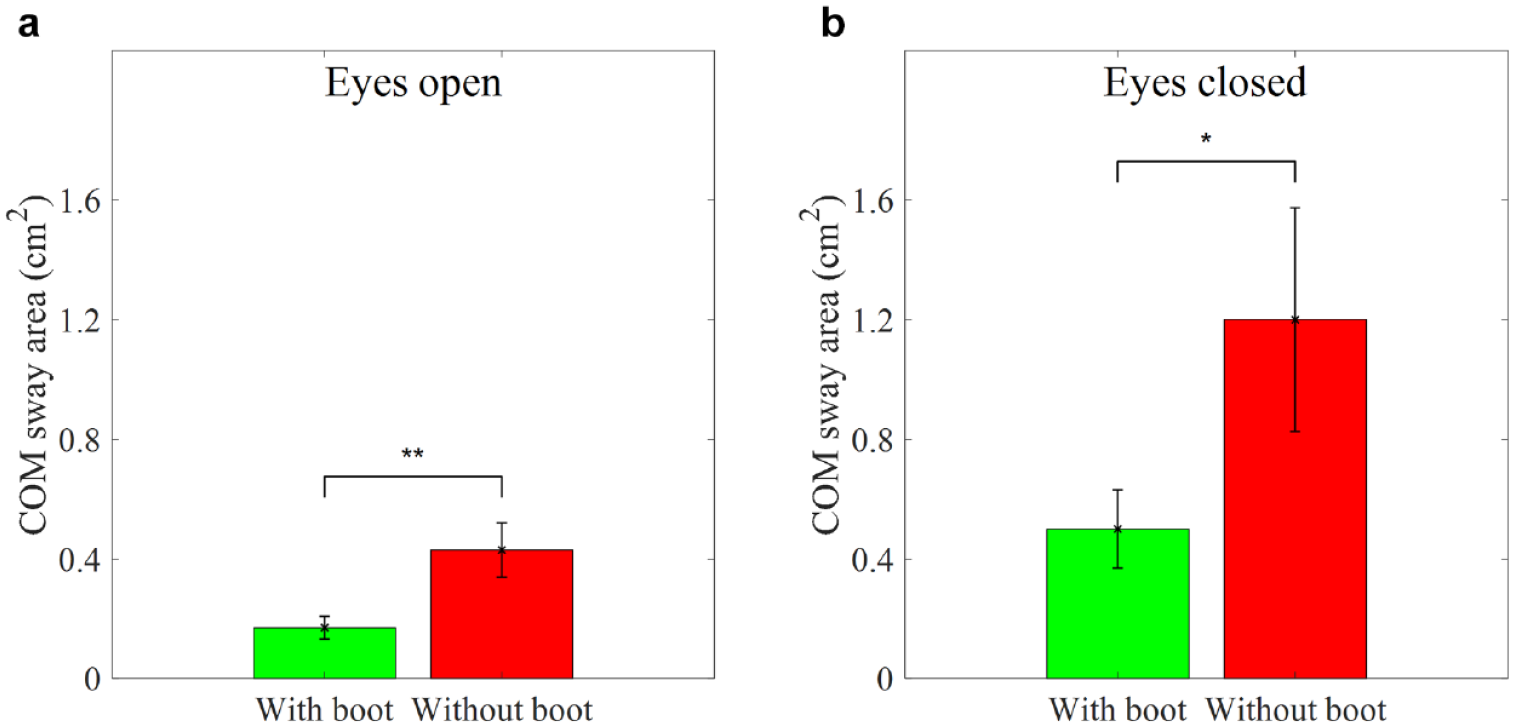
Average COM sway area while wearing and not wearing the SmartBoot in the semi-tandem Romberg stance. (a) Eyes open condition. (b) Eyes closed condition. Error bars indicate the standard error of the corresponding average (*P < .05 and **P < .01).
The results of gait assessments showed that the percentage error of step counts was 4.4 ± 31.5% for slow walking, 36.2 ± 48.7% for normal walking, and 16.0 ± 50.3% for fast walking. The gait speed measured by the validated wearable system (LegSys) for slow, normal, and fast walking was 0.6 ± 0.2, 0.9 ± 0.2, and 1.1 ± 0.2 m/s, respectively.
Table 3 reports the results of the TAM including nine questions. According to the results, the majority of the participants agreed that the SmartBoot was easy to use (Q1-Q3), comfortable (Q4 and Q5), not intrusive (Q6), and innovative (Q8). Participants agreed that they would recommend the SmartBoot to their friends (Q9). However, participants recommend improving the SmartBoot’s appearance (Q7).
Results of the Likert Scale Survey.
The Likert scale rank included strongly disagree (1 point), disagree (2 points), neither disagree nor agree (3 points), agree (4 points), or strongly agree (5 points) for each question.
Discussion
This study examined the accurate measure of adherence and non-adherence to wearing the SmartBoot and its influence on balance control, gait performance (ie, step count), and user experience in healthy adults. The results suggest that the SmartBoot is reliable and accurate in remote monitoring adherence and non-adherence to wearing the offloading boot at a high sensitivity rate of 90.56%, specificity rate of 88.04%, and accuracy rate of 89.29%. The SmartBoot offers innovative remote patient monitoring solutions to offloading interventions, enabling real-time monitoring of activity, adherence, and non-adherence to wearing the boot. In addition, it provides a real-time alert for non-adherence and facilitates remote monitoring through cloud storage and web services. Multiple systematic reviews stress the importance of adherence to offloading interventions to prevent and heal DFUs.11,15,17 With the advances in wearable sensors, the ability to measure multiple factors affecting plantar tissue stress has become more achievable.28,29 Wearable sensors could be embedded in insoles, shoes, or offloading devices to measure plantar pressure and its distribution as well as wearing time. Notably, Najafi et al have demonstrated the effectiveness of real-time alerting using a smart insole and watch to improve adherence to plantar pressure offloading over time in patients with diabetes who experience a loss of protective sensation. 30 The authors concluded that the real-time alert is a key factor to assist a user in improving the perception of the benefits of using the technology, which in turn may contribute to improving adherence to footwear over time. 30 Similarly, the SmartBoot provides a real-time alert via a smartwatch showing time associated with wearing and not wearing the offloading boot, but more importantly, the SmartBoot sends alert signals to cloud storage, allowing the care team to monitor patient adherence and reinforce adherence behaviors with the patients.
The results of balance assessments showed that the COM sway area was smaller with the boot than without the boot, regardless of whether participants had their eyes open or closed for two stance conditions (normal and semi-tandem Romberg). Functional balance control relies on controlling ankle joints and well-controlled contraction of dorsiflexors and plantarflexors. 31 Thus, it is reasonable to infer that the increased balance control (ie, decreased COM sway area) can be attributed to the improved ability to control an ankle joint in both anterior-posterior and medial-lateral directions by wearing the boot during both stance conditions. Specifically, the boot as an ankle support device provides additional somatosensory feedback32-34 and mechanical support against ankle motion,35,36 which contribute to maintaining stable balance.25,37,38 A study has demonstrated that durations of standing and walking were significantly higher with a removable offloading device than with an irremovable offloading device in patients with DFUs. 22 Therefore, we assume that the SmartBoot system could decrease the risk of falling and facilitate weight-bearing activities (eg, standing and walking) in patients with DFUs.
The results of gait assessments showed that the SmartBoot could estimate step counts. Early studies have highlighted the importance of the relationship between weight-bearing activities (eg, standing and walking) and the increased risk, healing, and prevention of DFUs (see Najafi et al 28 and Najafi and Mishra, 29 for review). However, monitoring weight-bearing activity in patients with DFUs commonly depends on the participant’s self-report. Therefore, some studies have investigated the use of wearable technologies to monitor weight-bearing activities. For example, Najafi et al invented a smart tag system incorporating a smartwatch and radio-frequency-identification (RFID) tag that detects standing and walking while wearing an offloading boot.29,39 Similarly, the SmartBoot detects weight-bearing activities (ie, standing and walking) as well as step counts. However, the results showed a high error rate of step counts. The errors could be attributed to a sample frequency, poor connectivity between the SmartBoot and smartwatch leading to missing protected steps reported on the smartwatch, or the signal processing used to estimate steps. These problems will be addressed in subsequent studies. Nevertheless, the ability to send information about weight-bearing activities to cloud storage is expected to improve future ulcer treatment regimens with offloading devices.
The collected survey results indicate that most participants feel that the SmartBoot could be used easily, intuitively, comfortably, and unobtrusively. Participants indicated a higher confidence level of technology innovation and its usefulness. However, they suggest improving the appearance of the SmartBoot.
With the majority of US carriers transitioning to 5G, wearable sensors, and smartwatch technology constantly evolving to be more powerful and miniaturized, the 4G LTE spectrum becomes more available and more affordable for this type of mobile Health (mHealth) IoT applications.29,40 The results of this study also suggest that wearing the SmartBoot can reduce the COM sway area while standing and estimate step counts during locomotion, potentially increasing the balance stability of patients with DFUs by providing proprioceptive feedback while standing and walking. Although the clinician dashboard was not used in this study, this functionality can offer the care team the opportunity to remotely monitor the amount of time spent wearing the SmartBoot, improve patient-clinician communication, and reinforce the critical importance of offloading and monitoring activity, which could potentially improve wound healing outcomes for those with DFUs. The system may also help clinicians identify patients that for multiple reasons (eg, peripheral neuropathy, dementia, or social and environmental barriers) are unable to adhere to the offloading protocol and make adjustments in their care plan. These patients may require escalation of care to make the boot non-removable. The SmartBoot system allows clinicians to remotely monitor patient adherence in a transparent, objective, nonintrusive manner. As mHealth and telemedicine are expanding rapidly, we expect that the SmartBoot with bidirectional feedback and interaction between patients and clinicians can potentially increase beneficial outcomes for patients with DFUs. 41
Subsequent studies will improve algorithms for detecting step counts and other weight-bearing activities (eg, standing and walking) and systematically evaluate the efficacy and effectiveness of the SmartBoot offloading system for monitoring adherence in patients with DFUs over weeks or months. We will also assess outcomes from DFU treatments with the smart offloading boot system and conventional offloading devices. Furthermore, we will explore the validity of the clinical dashboard to manage, encourage, and reinforce adherence to offloading remotely.
The main limitations of the present study are its small sample size, a participant cohort (ie, healthy adults), and short time period for adherence assessments. In future studies, we will increase the sample size, recruit patients with DFUs, and conduct the study over weeks or months. In addition, the step counting algorithm of the smartwatch misunderstood some arm movements as steps while the subject was sitting at the desk and triggered the boot removal algorithm. This issue will be addressed by improving the watch step detection algorithm with refined signal processing techniques. Furthermore, we will improve the step counting algorithm of the boot for having high accuracy and enhance the appearance of the SmartBoot.
Conclusions
This is the first study to evaluate the use of a smart offloading boot system incorporating an embedded sensor, a smartwatch app, and cloud infrastructure to remotely measure and monitor adherence to an offloading prescription. In addition, the SmartBoot system provides a real-time alert to the user when they are out of compliance with their prescribed offloading regimen, prompting improved adherence. The clinician dashboard allows the care team to remotely monitor and assess adherence, and provide feedback in near real time, which could assist in reinforcing adherence to offloading. However, these speculations should be validated in future studies. In addition, the SmartBoot influence on balance control, gait performance (ie, step count), and user experience was evaluated. The main findings of this study suggest that a wearable motion sensor as a part of the SmartBoot system, combined with a 4G LTE equipped smartwatch and cloud solution, can accurately measure adherence and non-adherence to offloading.
Footnotes
Acknowledgements
The authors thank Ms Maria Noun for supporting with data collection and communication with IRB.
Abbreviations
ANOVA, one-way analysis of variance; BTLE, Bluetooth Low Energy; COM, center of mass; CI, confidence interval; DFUs, diabetic foot ulcers; IMU, inertial measurement unit; IoT, Internet of things; mHealth, mobile health; RFID, radio-frequency-identification; SB, SmartBoot; SmartBoot, smart offloading boot; TAM, technology acceptance model; WS, wearable system.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.V., M.M., and S.R. are with Sensoria Health Inc., manufacturer of the sensor used in this study. Their contributions were limited to technical support and contribution in the discussion section to interpret the technical difficulties. They were not involved in study design, subject recruitment, and data collection and analysis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by a grant from the National Institute of Health, National Institute of Diabetes and Digestive and Kidney Diseases, NIDDK (Award# R01DK124789).
