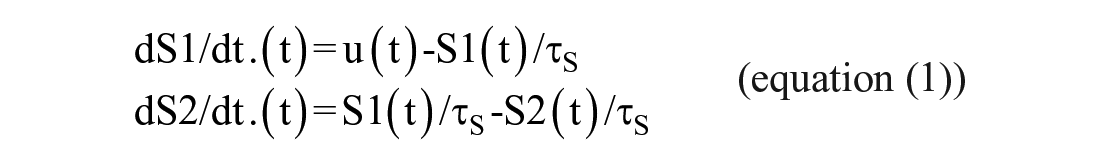Abstract
Objective:
There is room for improvement in the performance of closed-loop regulation algorithms during the prandial period. This in silico study evaluated the efficiency and safety of ultrarapid lispro insulin using the Diabeloop DBLG1® algorithm.
Methods:
We modeled the insulin profile of URLi according to literature data and integrated it to the model used within a simulation platform built from a 60 patients’ virtual cohort. We then ran the DBLG1® algorithm in silico with various meal intakes using modeled URLi, Aspart and Faster Aspart. The primary endpoints were glucose metrics (time in 70-180 mg/dL range and time below range).
Results:
When insulin time constant values were tuned, time in 70-180 mg/dL range was 69.4 [61.1-75.6] (Aspart) vs 74.7 [65.5-81.5] (URLi). Glucose coefficient of variation was reduced from 34.1 [29.7-37.8] to 28.4 [25.7-34.6]. Time below 70 mg/dL and 54 mg/dL were significantly reduced with URLi, whether or not DBLG1 was specifically tuned to this insulin. Metrics with Faster Aspart were intermediate and did not significantly differ from URLi.
Conclusions:
This simulation study performed on a virtual T1D population suggests that the use of URLi within an unmodified closed-loop DBLG1 regulation algorithm is safe and, with DBLG1 being tuned to this specific insulin type, improved the regulation performances as compared with Aspart. This fact supports the use of such an insulin in clinical investigations.
Keywords
Introduction
Several closed-loop insulin therapy systems are now being used in routine care by patients with type 1 diabetes following completion of pivotal trials.1-4 All these systems have shown a significant improvement in overnight and interprandial glucose profiles. Yet there is room for refinement during the prandial and post-prandial periods, since these hybrid systems rely upon declaration of meal and anticipated carbohydrate intake by the user. Inappropriate delivery commands by the algorithm may result in post-prandial hyperglycemia, or expose patients to a risk of hypoglycemic episodes. The inertia of these closed-loop systems, due to the absorption time for subcutaneously administered insulin, is one aspect of the problem. The availability of new, fast-acting insulin analogs, may be an option to overcome this inertia.
The first available of these new analogs, fast-acting insulin aspart (Fiasp® or Faster Aspart), was shown to be superior to insulin aspart regarding changes from baseline within 1-hour post-prandial glucose increment, as well as at 30 minutes and 2 hours, when used with continuous subcutaneous insulin infusion in open-loop. 5 When used in a closed-loop setting, Faster Aspart yielded ambiguous results. In an 8-week multinational crossover trial, the use of Faster Aspart in the CamAPS FX closed-loop system in 25 patients was associated with a slight reduction in time in hypoglycemia without difference regarding the time in target range. 6 Postprandial glucose control was improved with Faster Aspart in a 6-week open-label study using the Medtronic 780G system without impacting overall time in target range. 7 On the other hand, an inpatient crossover study of 27 hours in 20 patients involving unannounced meals using the Glucositter system showed greater prandial glucose increments with Faster Aspart, 8 whereas a pilot 2-week crossover trial of 19 patients using the MiniMed 670G system did not show any difference in glucose metrics. 9
Ultrarapid lispro (URLi or Lyumjev®) is a more recent rapid-acting insulin analog that was developed by the addition of citrate and treprostinil in order to accelerate the absorption of insulin lispro. Pharmacokinetic and pharmacodynamic studies have shown that continuous subcutaneous infusion of URLi is associated with earlier insulin action and earlier offset of exposure, when compared with Faster Aspart, Humalog® (Lispro) or Novorapid® (Aspart). 10 To our knowledge, only one study on the use of URLi in a closed-loop setting has been reported so far, and it did not reveal any difference in glucose control in a 2-week pilot trial with the MiniMed 670G system. 11
Our aim was thus to evaluate the safety as well as the efficacy and impact of URLi in the DBLG1 hybrid closed-loop system in an in silico study using a Virtual Patient simulator.
Methods
General Strategy
First, the URLi insulin transfer from interstitial to distant compartment was modeled through a model identification using data measurements of plasma insulin concentration after an impulse response test (ie, a bolus injection). Secondly, this identified model was integrated to the Hovorka model modeling the insulin-to-glucose metabolism. 12 Finally, this augmented Hovorka model was used to run the DBLG1 regulation algorithm on a 60 patients’ virtual cohort involving various meal intakes.
Literature Review
In order to identify the URLi insulin pharmacokinetic and pharmacodynamic characteristics, we used data obtained during a randomized crossover clinical trial involving 68 patients with T1D, receiving the same insulin dose prior to a liquid test meal, where insulin concentration measurements of URLi, Faster Aspart, Aspart, and Lispro were compared. 10 This study showed that early half-maximal drug concentration was reached as early as 13 minutes after administration of URLi, which was 6 minutes faster than Faster Aspart, 13 minutes faster than Lispro, and 14 minutes faster than Aspart.
We also used data from a clinical comparison between URLi and Lispro performed during a randomized crossover study including 41 young T1D subjects and 39 ederly subjects. 13 Patients received a 15U insulin dose followed by a 10-h automated euglycemic clamp maintaining the glucose level at a normal level while insulin and blood glucose concentrations were measured.
Similar studies were performed in a population with T2D and in a healthy population, this latter study showing that URLi was 2-5 min faster during the first 15 min after injection and onset of insulin action was 7-9 min faster and insulin action was 3-fold greater in the first 30 min with URLi versus Lispro across all tested doses.14,15
Other methods to identify insulin absorption models rely on model parameters estimation for both intra- and inter-subject variability based on a large dataset of real-patient insulin concentrations measurements. One recent study identified a fast-acting insulin absorption by applying a two-stage method consisting of first estimating the model parameters in each subject of the population and, then, assessing the mean vector and the covariance matrix providing a measure of the population variability. 16 Covariates, that is, dependencies of model parameters on subject characteristics such as the body mass index, the height or the age of the patient, as well as correlation between model parameters are also estimated. The approach used is the nonlinear mixed effects (NLME) modeling. In this framework, the insulin absorption of each is defined by a pharmacokinetic model shaped by parameters defined around population parameters, the so-called fixed effects.
Inter subject variability is then modeled by a random effects model around population parameters, including possible covariates and parameters inter-dependencies.
Model identification first relies on a structure model choice based on the Bayesian Information Criterion joining data fit accuracy and model parsimony. Different structures were tested involving various covariates of patient characteristics on model parameters. Fixed effects and random effects model parameters are then estimated using the Stochastic Approximation of Expectation Maximization (SAEM) approach involving a Monte Carlo Markov Chains (MCMC) method to estimate the maximum likelihood of the NLME model parameters. A Metropolis-Hastings (a MCMC Bayesian method) is also employed to estimate the Fisher Information Matrix useful to derive the covariance matrix of model parameters.
Urli Model Identification
Insulin could be modeled according to 2 linear models of order 2 and 3. Both structures were evaluated in the identification process and the one presenting the best fit with respect to measurement data were retained. The identification process aims at finding the best model parameter τS leading to the smallest distance between the measurement data and the model output of insulin concentration in the distant compartment. Measurement data were taken from Heise et al. 10
Results of model identification suggested that a linear order 2 model best fits the measurement data, with an optimal τS (insulin time constant) parameter of 49 min. Therefore this structure as well as this parameter value were used in the Hovorka model to build a virtual patient population using the URLi insulin. The 2nd order insulin model structure is recalled hereafter (equation (1)):
Equation (1) is the original insulin model contained in the Hovorka model 12 where u(t) [mu/min] represents the amount of insulin injected and S1(t), S2(t) represent the insulin absorption [mu] in the 2 distant compartments.
A similar approach was used to identify the Faster Aspart model, using data from Heise et al 10 (see details in appendix).
DBLG1 Algorithm Summary
The DBLG1 Closed Loop system used to conduct this study was composed of the following algorithmic modules:
A Hypominimizer is intended to cut off basal rate if forecast glycemia goes below a certain glycemia threshold. The glycemia forecast is intendly simple and based on a straightforward linear regression to make it robust and conservative. If current glycemia gets below another threshold, then the basal cut off is accompanied by a rescue carb recommendation to help glycemia reach the target again.
During meal periods a Meal Manager is in charge of computing meal boluses. These meal boluses can be standard single-shot boluses, or split boluses, depending on the physiological conditions. Immediate post-meal insulin management is also dynamic, based on observed changes (for example if glycemia rises faster than expected during post-meal phase then more insulin is sent), and managed primarily with micro-boluses.
During out-of-meal periods a Machine Learning algorithm, within a physiological framework, makes predictions and thus is used to make insulin decisions. The physiological framework is a proprietary framework initially derived from the Hovorka model. Patented, proprietary technology evaluates the reliability of this machine learning, and hands over the decision to an “expert system” if the machine learning’s reliability is too low.
The “Expert system” is a rule-based module co-designed with clinicians, diabetologists, expert patients and engineers. Insulin recommendations are computed according to patient glycemic state involving both current and forecast glycemia, patient insulin sensitivity factor and patient IOB.
More details about the DBLG1 algorithm and its clinical results can be found in Benhamou et al, 2 Amadou et al 17 and in the Appendix.
Simulation Tool
The implementation of URLi insulin in the DBLG1 regulation algorithm (v1.1.1) was made on the Diabeloop Virtual Patient Simulator. This simulator was built from a virtual population composed of 60 adult T1D patients from the WP7 clinical trial (NCT02987556). 2 The DBLG1 algorithm linked to the simulator then enabled the assessment of the glycemic response to insulin commands while the simulator contained the URLi insulin subsystem.
The 2 limitations of the Virtual Patient simulator were: (a) the absence of exercise modeling, and therefore the exercise management of the DBLG1 was not solicited; and (b) CGM communication loss and pump communication loss were not modeled which could not allow to assess the algorithm behavior in front of such missing data situations.
Finally a major difference between the simulation and the real life situation was the lack of rescue carbohydrates intake to prevent a hypoglycemic episode. Due to the lack of a sufficiently accurate model of rescue carbohydrates intake, it has been decided to disable this intake by the virtual patient.
It is also worth noting that our Virtual Patient Simulator was not certified by a notified body at the time of the study, therefore the results presented in the sequel should be taken with care.
Simulation Scenario
The simulation scenario was run on the whole Virtual Patient population composed of 60 patients during 4 days for each patient. The meal scenario on each day was derived from meal distributions obtained on data from WP7 clinical trial (NCT02987556). 2 All meals were announced to the system, with 3.4 meals per day on average. Mean CHO values for breakfast, lunch and dinner were 37, 52, and 49 g, respectively. The meals communicated to the algorithms contained declaration errors drawn from a uniform law ranging from [m - 24%; m + 80%] where m represents the actual meal value. This interval contains the average errors of 40% for overestimation and 12% for underestimation as reported in Meade and Rushton. 18 Meal intakes were declared to the algorithm at meal time. The noise applied to the observed and state variables was a gaussian white noise whose standard deviation was defined as 0.012 × x0 where x0 (which is peculiar to each virtual patient) defines the equilibrium value of a state variable.
We studied the behavior of DBLG1 regulation algorithm running on a virtual patient population using the URLi insulin compared to the same population using Aspart insulin, which is the insulin the DBLG1 system was clinically validated on Benhamou et al. 2 We also included a comparative run using Faster Aspart insulin in the process.
All simulations were run under different scenarios, distinguishing themselves with respect to the adjustment of the internal 2nd order Insulin On Board model time constant to the average physiological insulin time constant. This algorithm Insulin Time Constant parameter (also referred to as Tau_S or τS) was set to the following values: for Aspart: Tau_S = 69 min; for Faster Aspart: Tau_S = 56 min; for URLi: Tau_S = 49 min.
When specified, we also used a 65 min setting which is the default value implemented in the commercial DBLG1 device, designed to be used with fast acting insulins such as Aspart, in order to test for its safety if casually used with Faster Aspart or URLi.
Endpoints
The primary endpoints of our study were the following glucose metrics:
– Time In Range 70-180 mg/dL (%)
– Time below range (<70 mg/dL) (%)
– Time below range (<54 mg/dL) (%)
– Time above range (>180 mg/dL) (%)
– Average glucose (mg/dL)
– Coefficient of variation (CV) (%).
Results are presented as mean (+/− standard error) and median [Q1-Q3]. Performances were established on the overall simulation period as well as on night time periods (from 22:00 to 7:00) and meal time periods (from tm to tm + 4 hours, where tm designates meal start times) to better assess the benefits of the URLi on prandial management.
Results
Scenario 1. Adjusted Insulin Time Constant
In this scenario, the Insulin Time Constant was respectively adjusted to 69 min for Aspart, 56 min for Faster Aspart and 49 min for URLi .
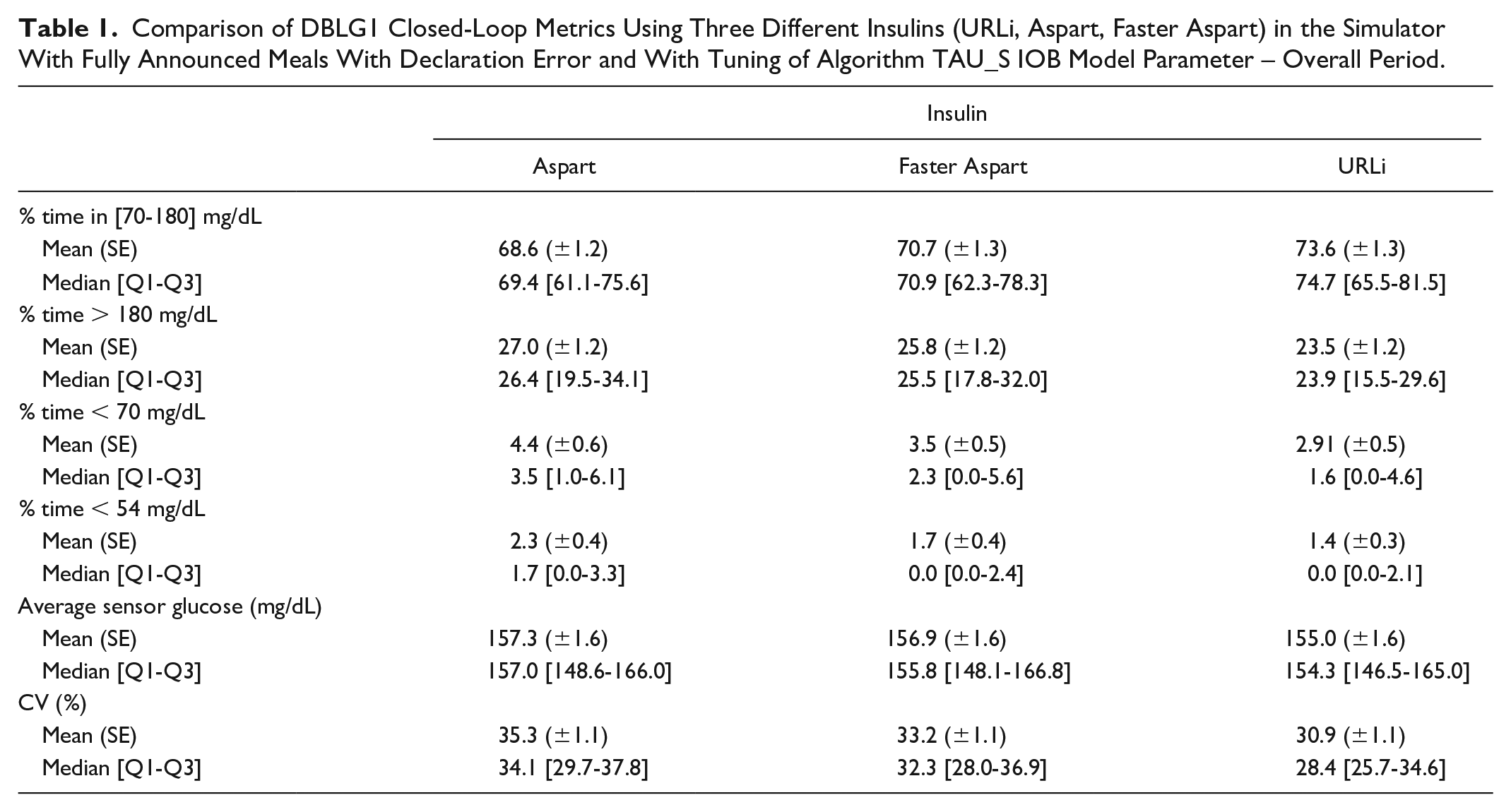
On the overall simulation period of 4 days, we observed that our virtual population exhibited an absolute increase of nearly 5% in time in range 70-180 mg/dL when exposed to URLi (74.7 [65.5-81.5]) as compared with Aspart (69.4 [61.1-75.6]). We also observed a significant decrease in time below 70 mg/dL as well as time below 54 mg/dL with URLi. Additionally, an absolute improvement of nearly 5% in coefficient of variation with URLi (28.4 [25.7-34.6]) vs. Aspart (34.1 [29.7-37.8]) was observed, without difference regarding mean glucose value. Metrics observed with Faster Aspart stood at intermediate levels between the two other insulins. Faster Aspart improved time below 70 mg/dL as well as CV versus Aspart. These data are summarized in Table 1 and Figure 1.
Comparison of DBLG1 Closed-Loop Metrics Using Three Different Insulins (URLi, Aspart, Faster Aspart) in the Simulator With Fully Announced Meals With Declaration Error and With Tuning of Algorithm TAU_S IOB Model Parameter – Overall Period.
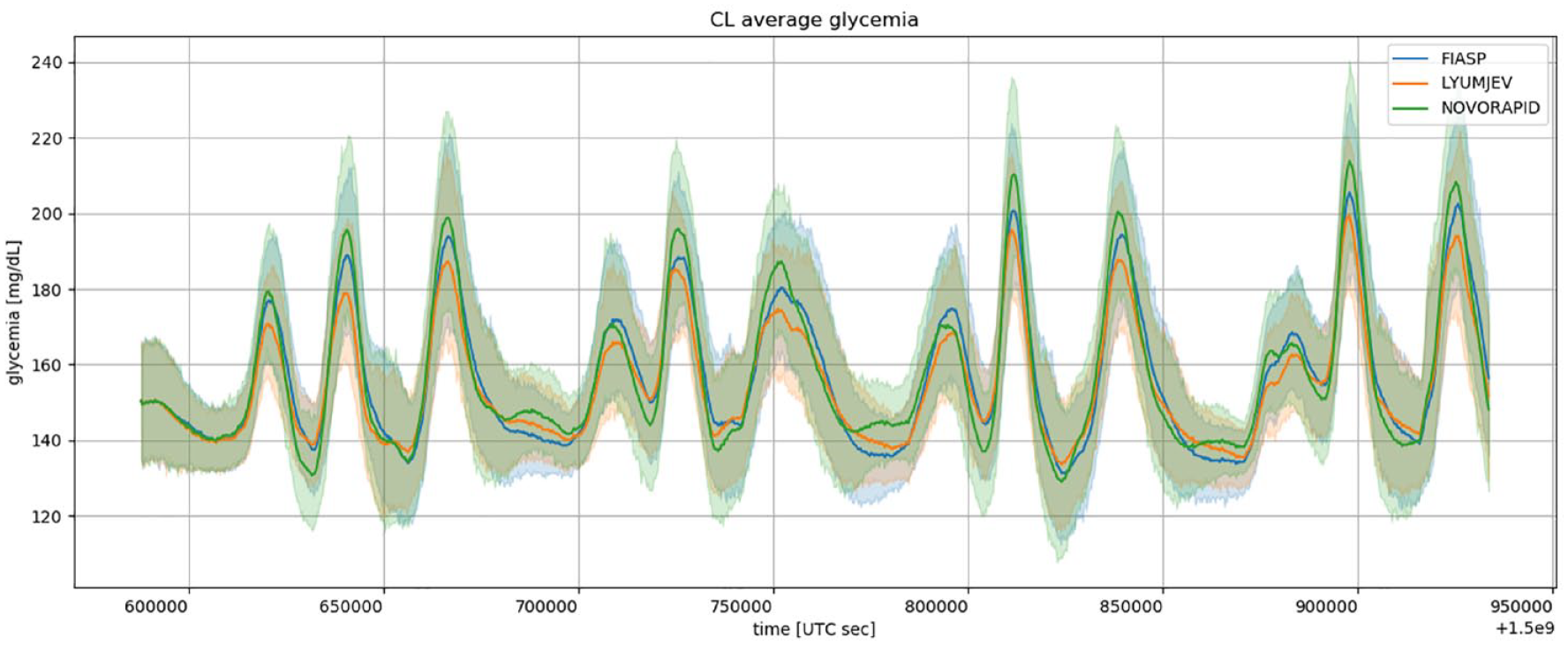
4 days glucose profile obtained with URLi, Aspart or Faster Aspart under the DBLG1 closed-loop regulation in a virtual-patient population. Insulin Time Constant was adjusted.
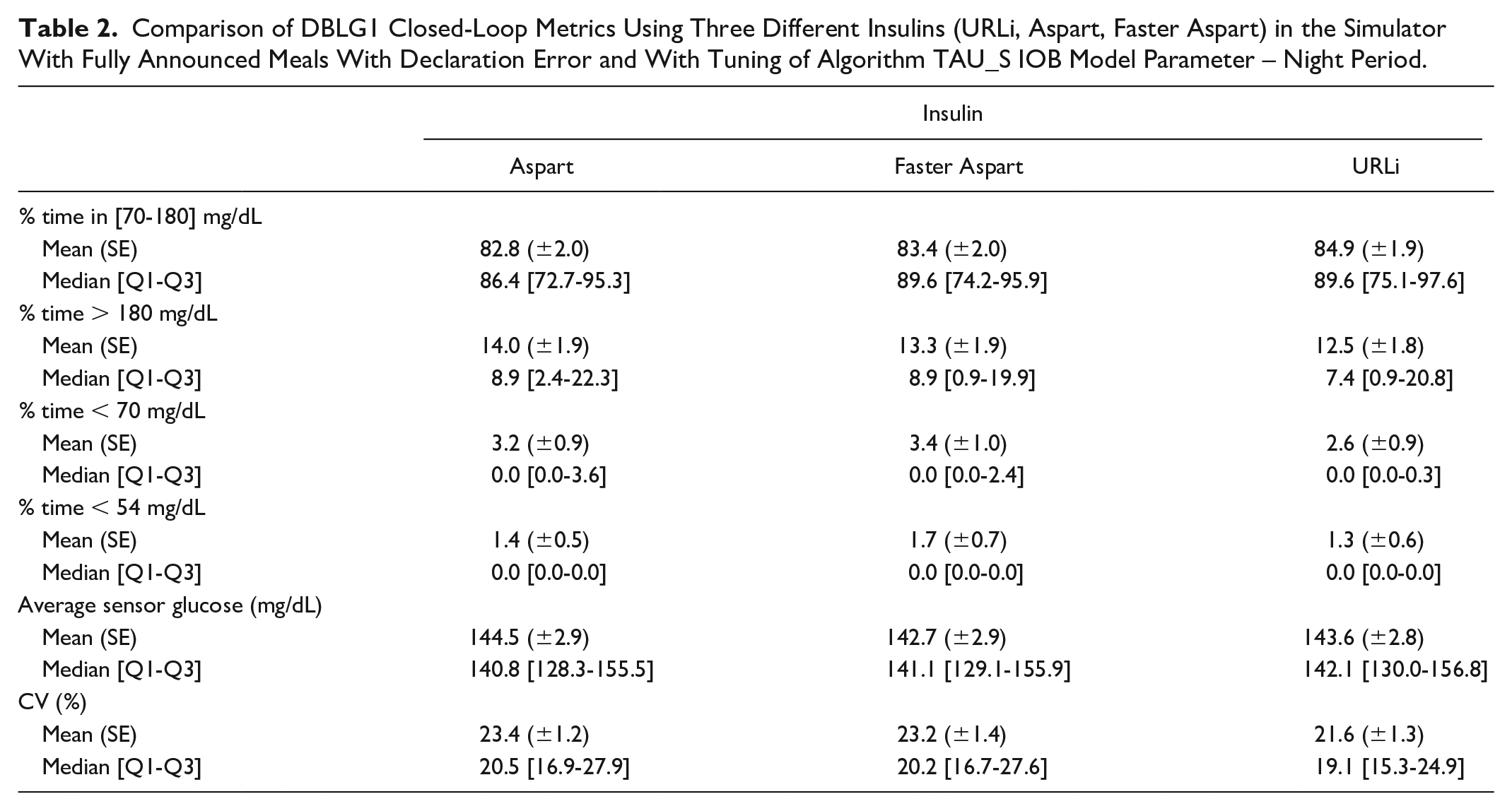
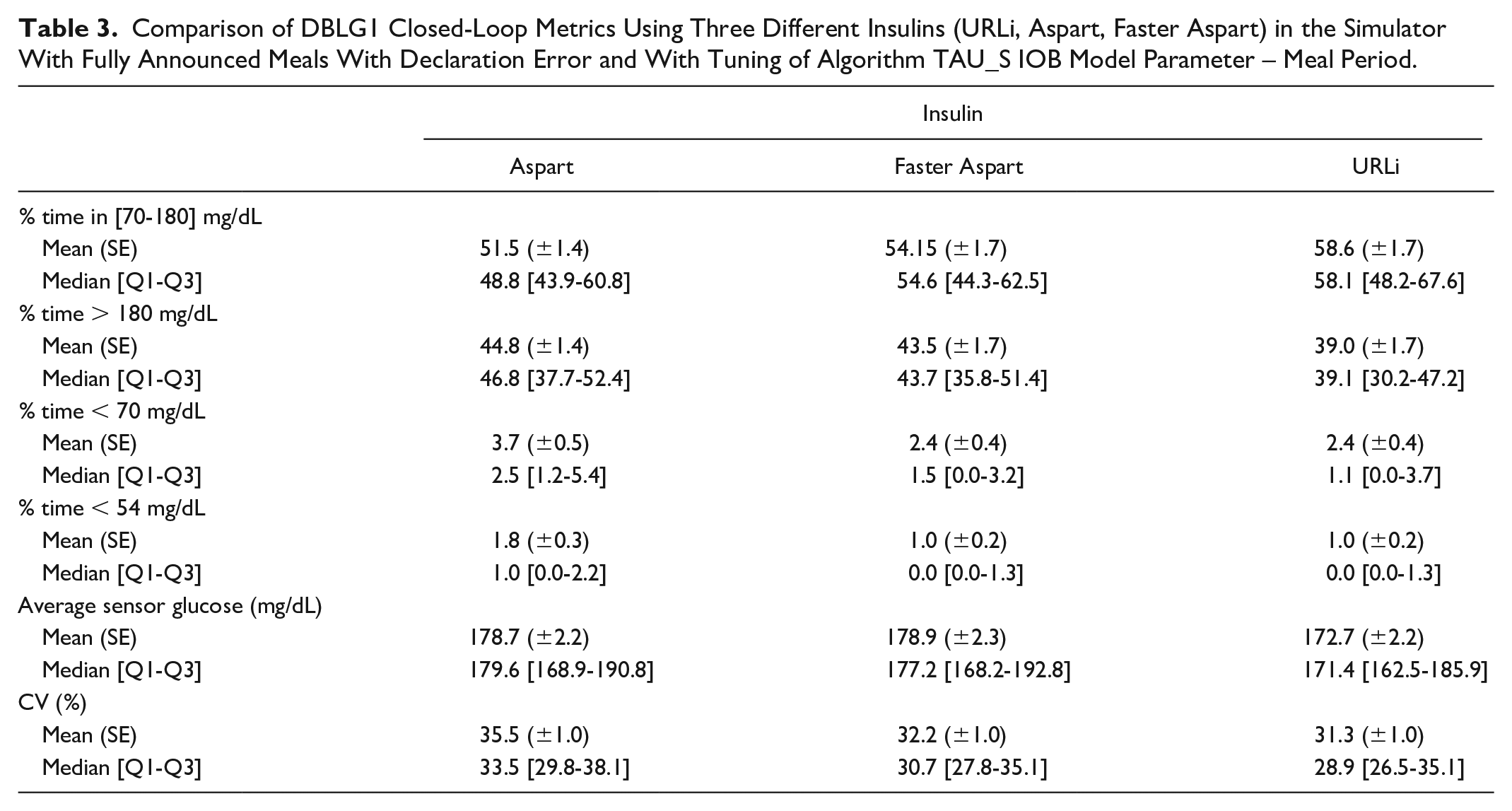
The night time and meal time performances available in Tables 2 and 3, although exhibiting similar trends as in the overall results (Table 1), suggests that URLi, and Faster Aspart to a lesser extent, are more beneficial to glucose control during prandial periods. Indeed, the results report a much higher TIR gain between URLi and Aspart (+ 9% in absolute: 48.8 [43.9-60.8] for Aspart vs. 58.1 [48.2-67.6] for URLi) during meal periods when compared with night periods (+3% in absolute: 86.4 [72.7-95.3] for Aspart vs 89.6 [75.1-97.6] for URLi). Same analysis can be conducted on the coefficient of variation which can be explained by the absence of strong disturbances during night periods.
Comparison of DBLG1 Closed-Loop Metrics Using Three Different Insulins (URLi, Aspart, Faster Aspart) in the Simulator With Fully Announced Meals With Declaration Error and With Tuning of Algorithm TAU_S IOB Model Parameter – Night Period.
Comparison of DBLG1 Closed-Loop Metrics Using Three Different Insulins (URLi, Aspart, Faster Aspart) in the Simulator With Fully Announced Meals With Declaration Error and With Tuning of Algorithm TAU_S IOB Model Parameter – Meal Period.
Scenario 2. Insulin Time Constant Set at Default Value of 65 min
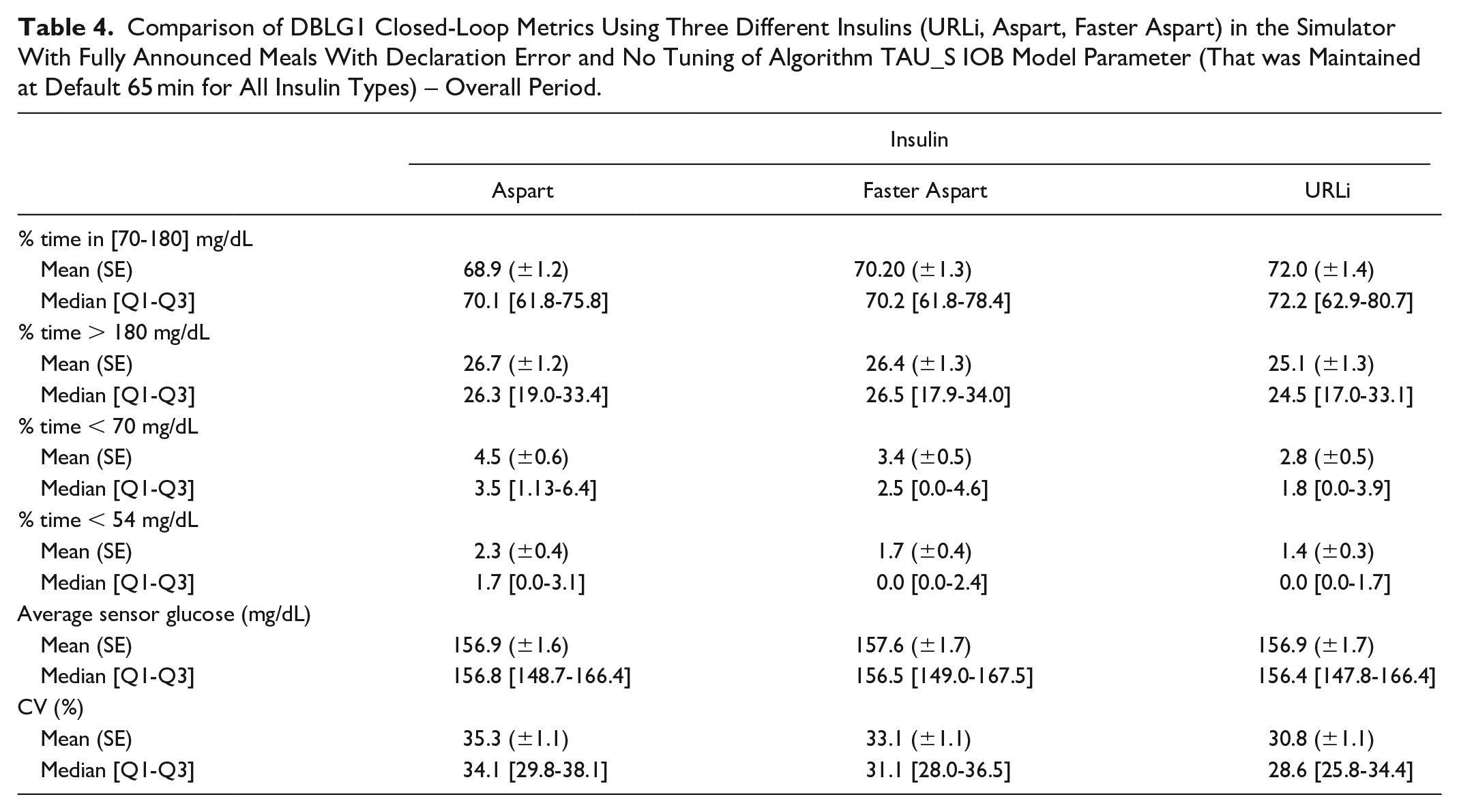
In a second phase, we studied a scenario where the DBLG1 algorithm was set at its default value regarding insulin time constant. The performances of all 3 insulins on the overall period (Table 4 and Figure 2) behave similarly as in scenario 1: performances of TIR 70-180 mg/dL improved by 2% when comparing URLi (72.2 [63.0-80.7]) to Aspart (70.1 [61.8-75.8]), hypoglycemia safety was still highly improved between Aspart (TBR 70 mg/dL: 3.5 [1.1-6.4] ; TBR 54 mg/dL: 1.7 [0.0-3.1]) and URLi (TBR 70 mg/dL: 1.8 [0.0-3.9]; TBR 54 mg/dL: 0.0 [0.0-1.7]) and stability was also reinforced through a reduced coefficient of variation: 34.1 [29.8-38.1] for Aspart vs 28.6 [25.8-34.4] for URLi.
Comparison of DBLG1 Closed-Loop Metrics Using Three Different Insulins (URLi, Aspart, Faster Aspart) in the Simulator With Fully Announced Meals With Declaration Error and No Tuning of Algorithm TAU_S IOB Model Parameter (That was Maintained at Default 65 min for All Insulin Types) – Overall Period.
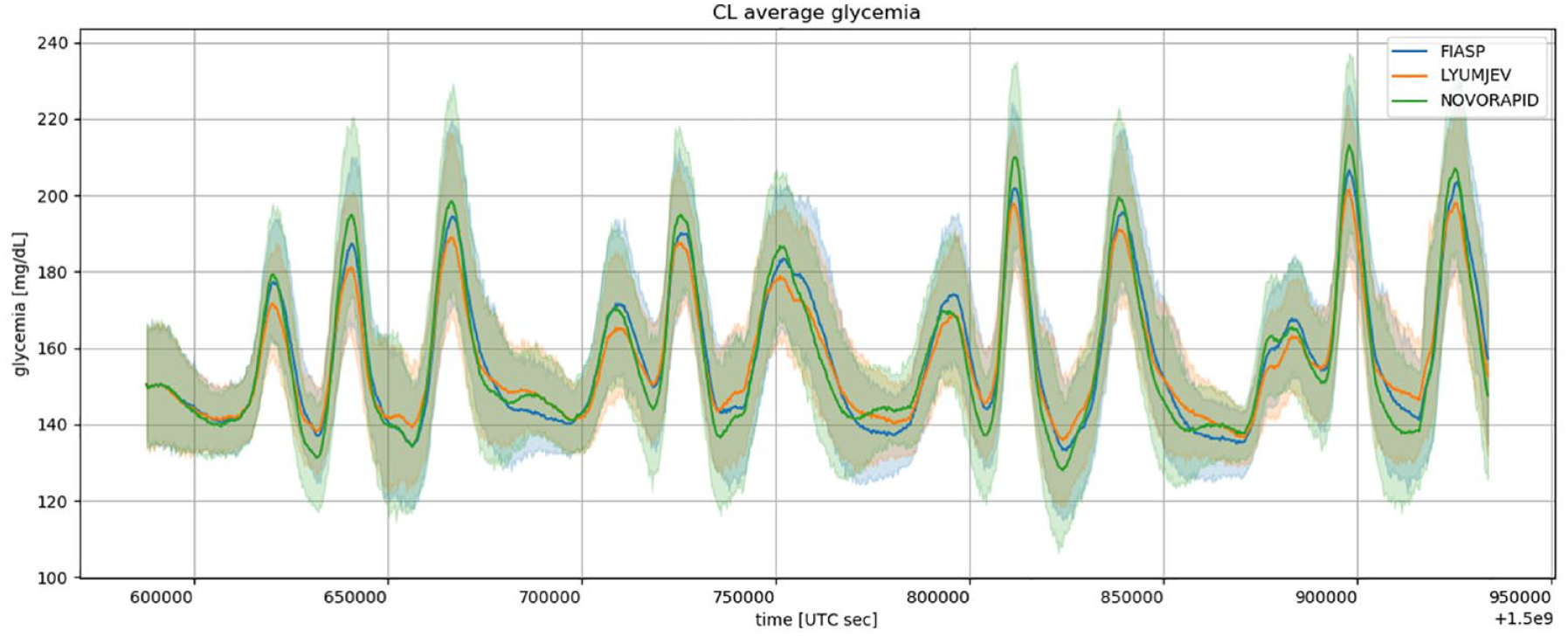
4 days glucose profile obtained with URLi, Aspart or Faster Aspart under the DBLG1 closed-loop regulation in a virtual-patient population. Insulin Time Constant was not adjusted but set at default 65 min.
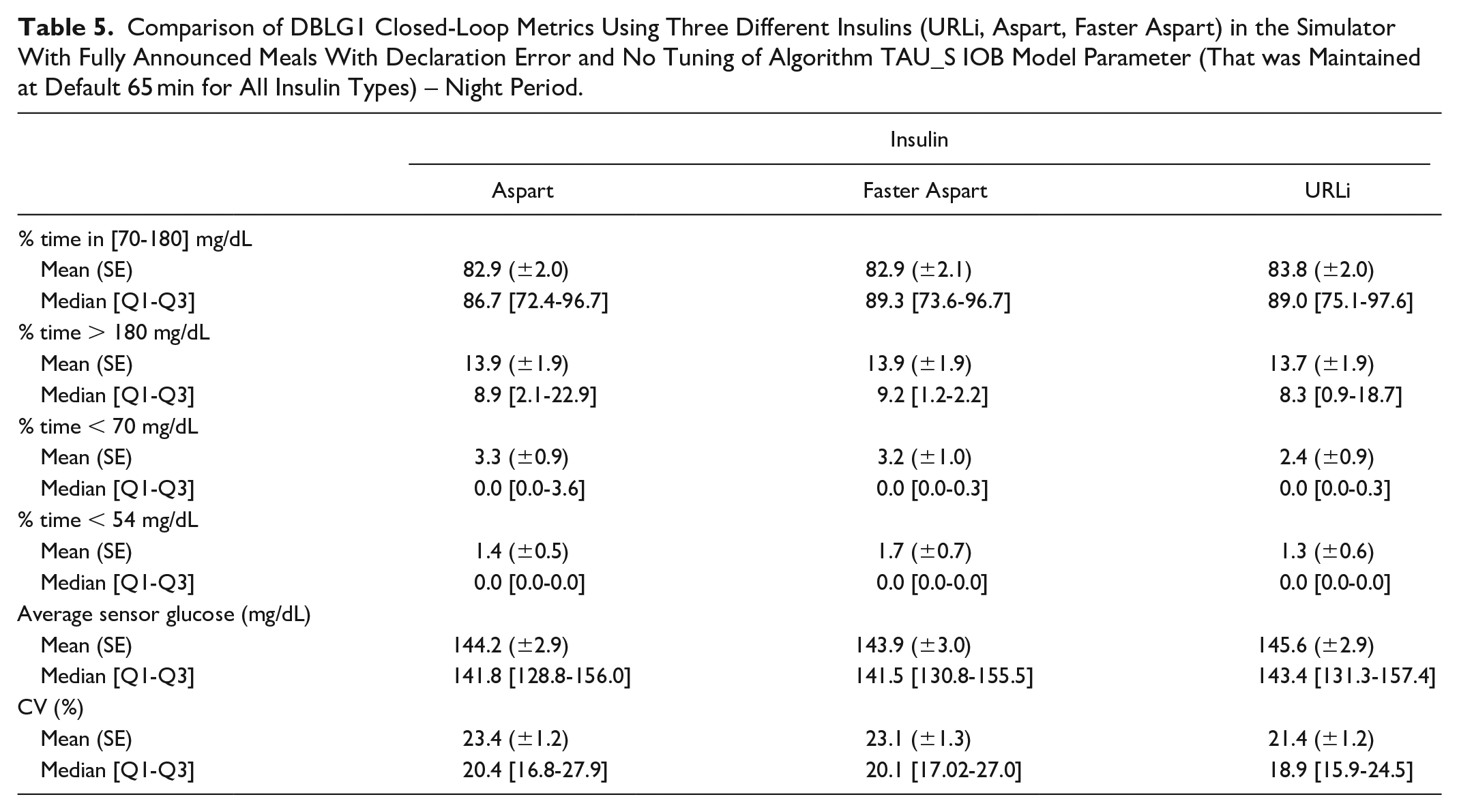
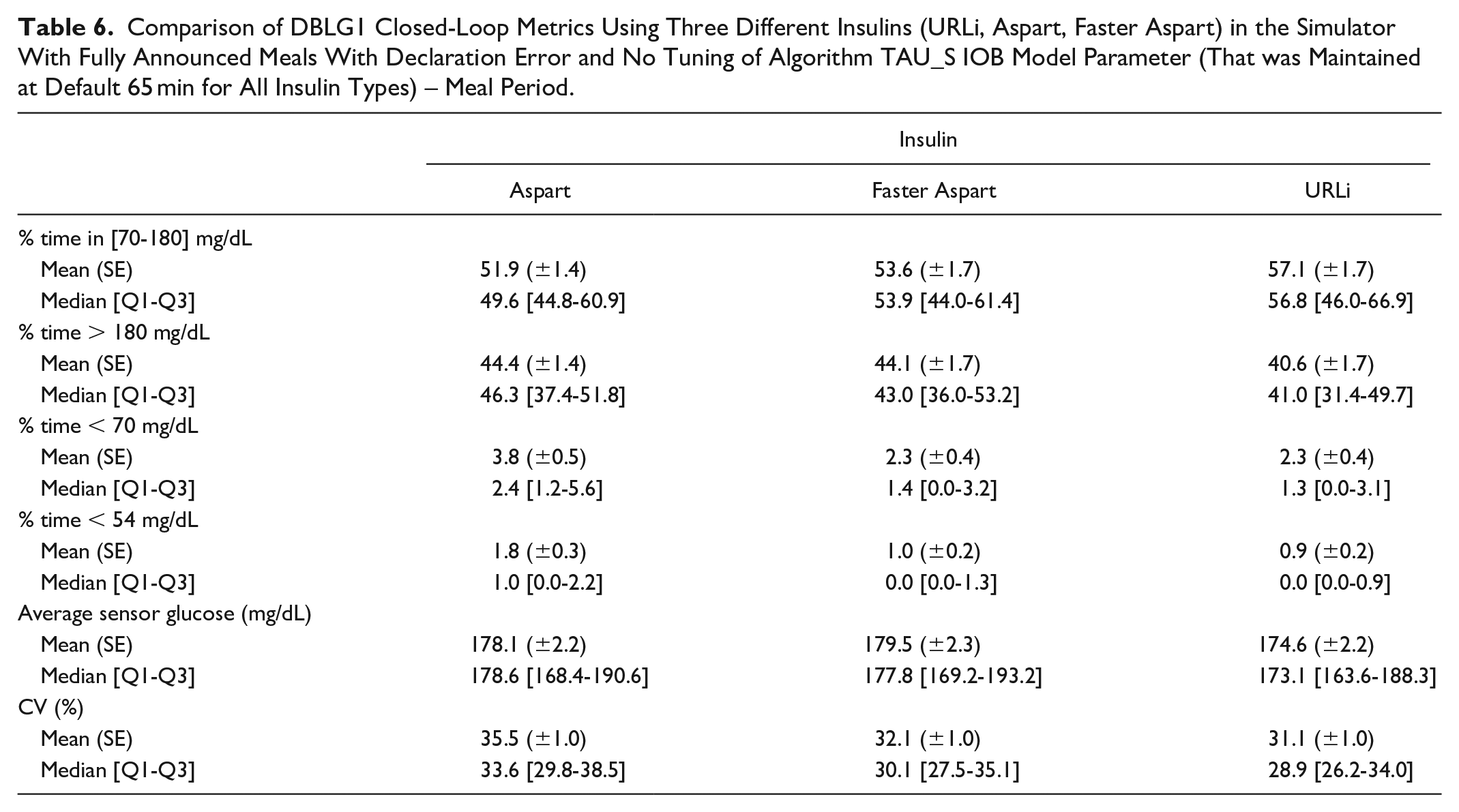
Meal period and night period metrics analysis leads to similar conclusion as in scenario 1: URLi and Faster Aspart especially improved prandial management performances, although night time safety was also improved with reduced TBR outcomes (Tables 5 and 6).
Comparison of DBLG1 Closed-Loop Metrics Using Three Different Insulins (URLi, Aspart, Faster Aspart) in the Simulator With Fully Announced Meals With Declaration Error and No Tuning of Algorithm TAU_S IOB Model Parameter (That was Maintained at Default 65 min for All Insulin Types) – Night Period.
Comparison of DBLG1 Closed-Loop Metrics Using Three Different Insulins (URLi, Aspart, Faster Aspart) in the Simulator With Fully Announced Meals With Declaration Error and No Tuning of Algorithm TAU_S IOB Model Parameter (That was Maintained at Default 65 min for All Insulin Types) – Meal Period.
Discussion
This study allowed us to model the insulin profile of URLi and to integrate it to the Hovorka model used within our virtual patient simulator. This insulin model was identified using the literature insulin concentration data from Heise et al. 10 We then evaluated, in silico, the DBLG1 closed-loop regulation algorithm with modeled URLi insulin within the simulator and compared it to Aspart (the insulin with which the DBLG1 system was validated) and to Faster Aspart. Closed-loop performance results showed that the use of URLi was safe when combined with DBLG1, as time below range (<70 and <54 mg/dL) were significantly reduced as compared to Aspart in all studied scenarios. We also observed that the use of URLi was more efficient, as time in range (70-180 mg/dL) improved by 2% to 5% (in absolute values) when compared with Aspart, when adapting the Insulin Time Constant of the algorithm. Finally, a 5% gain (in absolute) in coefficient of variation suggested an improved glucose stability. The metrics measured with Faster Aspart were at intermediate levels between Aspart and URLi.
Of note, even when used with predefined DBLG1 settings that were initially adapted for fast acting insulin profiles such as Aspart’s (ie, without tuning insulin time constant to the characteristics of used insulin), URLi still yielded better performances regarding hypoglycemia, glucose variability and TIR 70-180 mg/dL.
The meals communicated to the algorithm contained declaration errors uniformly distributed within a range [-24%; 80%] of the true values, as in real life, the exact CHO amount ingested is rarely correctly estimated by the patient.
Furthermore, the rescue carbohydrate intakes ingested by the virtual patient were disabled in our model. Indeed, in real life, patients often do not ingest their rescue carbs immediately and in the required quantity. Moreover, this discrepancy was impossible to measure and therefore an accurate model could not be derived easily. Therefore, one should keep in mind that the hypoglycemia rates displayed all along this study reflects the lack of rescue carbohydrate intake and that they would most likely be lower in real life situations.
To our knowledge, no clinical report has been made about the use of URLi in any closed-loop system, with the exception of a 2-week pilot trial with the MiniMed 670G system which did not show any difference. 11 Overall, our data support the use of this insulin in clinical investigations.
This study is a contribution to the field of prandial management applied to automated insulin delivery. One alternative would be to deliver insulin into the intraperitoneal space. 19 This route has been shown to provide better glucose control as opposed to SC insulin delivery. 20 However, there is no short-term perspective for the development of such pumps by the industrial world. Another alternative to improve prandial management, from a usability point of view, would be to get rid of meal announcement, which is a current limitation of all hybrid closed-loop systems. Managing unannounced meals could go through algorithmic refinements such as analysing the patient’s hourly meal probability, 21 but also through the usage of new, short-acting insulin analogs. 22
Finally, it is worth mentioning that our Virtual Patient Simulator is neither certified nor validated by a notified body. Therefore the simulation results presented throughout this paper should be taken with care. Nevertheless, the authors believe in the global trend exhibited in the obtained simulation results as a first step to a clinical trial to confirm those results.
Conclusion
We report results of a proof of concept, simulation study performed on a virtual T1D population, suggesting that the use of URLi within an unmodified closed-loop DBLG1 regulation algorithm was safer, whereas an adjustment of the settings of this algorithm could further improve the regulation performances as compared with Aspart. This suggests that clinical investigations are warranted.
Supplemental Material
sj-pdf-1-dst-10.1177_19322968211046021 – Supplemental material for Hybrid Closed-Loop Control with Ultrarapid Lispro Compared with Standard Insulin Aspart and Faster Insulin Aspart: An In silico Study
Supplemental material, sj-pdf-1-dst-10.1177_19322968211046021 for Hybrid Closed-Loop Control with Ultrarapid Lispro Compared with Standard Insulin Aspart and Faster Insulin Aspart: An In silico Study by Sylvain Lachal, Yousra Tourki, Sylvia Franc, Erik Huneker, Guillaume Charpentier and Pierre Yves Benhamou in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
CGM, continuous glucose monitoring; IOB, Insulin On Board; T1D, Type 1 Diabetes; T2D, Type 2 Diabetes; URLi, Ultrarapid Lispro; CV, Coefficient of Variation; TIR, Time In Range; TBR, Time Below Range; RSME, Root Mean Square Error; PK-PD, PharmacoKinetics-PharmacoDynamics; OL, Open Loop; CL, Closed Loop; SC, Subcutaneous.
Author Contributions
SL, YT, EH performed the simulation studies. PYB, SF, GC wrote and edited the manuscript.
Declaration of Conflicting Interests
SL, YT, EH are employees of Diabeloop SA.
SF declares congress invitations from Sanofi, Eli Lilly, MSD, Novo Nordisk, Roche, Abbott and Boehringer Ingelheim, and has received speaker honoraria from Eli Lilly and Novo Nordisk, and served on advisory board panels for Novo Nordisk, Roche, Sanofi, Janssen and LifeScan. She owns shares in Diabeloop SA.
GC has received congress invitations, honoraria and consultancy fees from Abbott, Dexcom and Medtronic, and owns shares in Diabeloop SA.
PYB has received speaker honoraria from Abbott, Roche, Eli Lilly, Novo Nordisk and Sanofi, and served on advisory board panels for Abbott, Diabeloop, Roche, Medtronic, Dexcom, Insulet, LifeScan, Eli Lilly, Novo Nordisk and Sanofi.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support of the study was provided by Diabeloop SA.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.