Abstract
Background:
My Dose Coach (MDC) is a mobile application combined with a web portal that can suggest optimized basal insulin (BI) injection doses using Self-Measured Plasma Glucose (SMPG) and hypoglycemia data. This study aimed to evaluate its efficacy on patients reaching SMPG and Fasting blood glucose (FBG) target range 90-130 mg/dl (5-7.2 mmol/L) goals without severe hypoglycemic episodes. We also addressed the mean reduction in glycated hemoglobin (A1C), FBG, and SMPG and the improvement in the WHO’s Five Well Being Index (WBI).
Methods:
This prospective pilot study involved the use of MDC in outpatients with type 2 diabetes (T2DM) from a Hospital in Northern Mexico. Patients on treatment with any BI were included in the study. The follow-up was of 16 weeks. Student t-tests or McNemar test were used for effect comparisons.
Results:
We included 158 patients (46.8% women), mean (SD) age 51 (10.3) years. We achieved SMPG target range in 58.9% [mean (95CI) reduction of 30.9 mg/dl (22.5-37.7; P < .001)] of the patients [66(28) days], with no severe hypoglycemia events. FBG goal was reached in 55.7% [mean (95CI) reduction of 63.4 mg/dl (49.6-77.2; P < .001)]. The mean (95CI) reduction of A1C was 1.78% (1.47-2, P < .01) with the last observation carried forward. There was a mean (95CI) increase of 2.23 (−3, −1.4, P < .01) points in WBI scale.
Conclusions:
MDC successfully helped to achieve FBG and SMPG goals, reduced A1C, and increased WBI with no severe hypoglycemia events.
Keywords
Introduction
Type 2 diabetes (T2DM) is a complex, chronic illness requiring continuous medical care to reduce macro and microvascular complications. The International Diabetes Federation and the American Diabetes Association recommend the start of basal insulin (BI) when a person with T2DM does not achieve glucose control after 3 months of use of a maximal dose of oral antidiabetic medication and lifestyle interventions.1,2 Once insulin management is started, the effectiveness requires time and an appropriate assessment of insulin dosing. Usually, BI is titrated at the physician’s discretion during routine clinic visits. Even though the patient receives support and advice on simple treatment algorithms, the visits may be infrequent (eg, 3-months intervals or longer) and there is a risk on the delay in insulin optimization.
With appropriate training, several studies and meta-analyses with and without digital tools have shown that patient self-titration of insulin could lead to faster glycemic control than physician lead insulin titration.3,4 A study that used self-managed BI every 3 days rather than weekly by a medical physician, using a hardware tool (Self-measured blood glucose meter with in-build titration algorithm), showed better glycemic control. 5 However, people with T2DM often worry about making mistakes when they titrate themselves due to poor or incomplete medical information, challenges with arithmetic skills, and have concerns about hypoglycemia.6-8
Various approaches have been developed to improve the experience of insulin titration for people with diabetes such as informational brochures and educational programs. These strategies have good results in achieving glycemic control and decrease providers’ excessive caseloads.9,10
Digital tools may empower individuals with T2DM by allowing them to make informed treatment decisions and reduce the waiting time for doctors’ feedback through medical appointments. My Dose CoachTM (MDC) is a mobile application (app) combined with a web portal that can suggest optimized BI injection doses using fasting Self-Measured Plasma Glucose (SMPG) and hypoglycemia data. A pilot study conducted in India demonstrated efficacy in reducing fasting blood glucose (FBG). 11 The present study is the first one conducted in Mexican Population. The primary aim was to evaluate the efficacy of MDC in reaching SMPG target range 90-130 mg/dl (5-7.2 mmol/L) without severe hypoglycemic episodes. Secondary aims were to evaluate the efficacy of MDC in reaching FBG target range 90-130 mg/dl (5-7.2 mmol/L) without severe hypoglycemic episodes, mean reduction in glycated hemoglobin (A1C), FBG, and SMPG. In addition, we addressed the improvement in the WHO’s Five Well Being Index (WBI).
Methods
Design
This prospective, single-group interventional, open-label study included patients with T2DM from Hospital Clínica Nova (HCN), located in Northern Mexico. The recruitment and follow-up period were from September 1, 2018 to August 31, 2019. The study followed CONSORT guidelines, 12 was performed in accordance with Good Clinical Practice and the Declaration of Helsinki. Local IRB approved the protocol, and all subjects signed consent forms. The Clinical Trial.gov Identifier is NCT03775057.
Procedure
We selected patients from a database of HCN. The inclusion criteria of the sample were individuals between 18 and 75 years old, all genders, diagnosed with T2DM, treated with any BI, A1C > 7%, and a smartphone compatible with the app. Subjects could be included if they were on oral agents for T2DM. Patients were excluded if they presented type 1 diabetes, gestational diabetes, MODY, or other types; also pregnant or nursing women. Other exclusion criteria were subjects with glomerular filtration rate < 30 ml/min, loss of vision, diseases that made the use of a smartphone difficult (cognitive, motor, or terminal illness), NYHA grade III or IV heart failure, and history of cardiovascular disease or stroke in the last 3 months or history of hospitalizations in the last 30 days. In addition, we excluded patients in treatment with systemic glucocorticoids, with no access to local medical service, lack of family support (assessed by social workers), or who were unable to meet specific protocol requirements (eg, inability to perform blood glucose measurements or administer insulin dose).
The app worked according to a titration scheme previously programed by the medical provider and suggested optimized BI injection doses using SMPG and hypoglycemia data. The patients used Glargine-U300 or Glargine-U100, according to physician discretion. The latter established the schedule for BI administration according to the daily activities of the patient. BI dose was determined according to the following scheme: (1) insulin-naïve patients: 0.2 IU × kg body weight dose. (2) Patients on previously BI treatment: same initial dose. The established titration scheme in this study followed the next parameters: 10% increase if SMPG > 180 mg/dl; 5% increase if SMPG 140-180 mg/dl; no change if SMPG 80-140 mg/dl to secure target range in the middle); 5% decrease if SMPG 70-79 mg/dl and 10% decrease if SMPG < 70 mg/dl. A health provider uploaded the titration scheme in the healthcare professional portal and could modify it according to his criteria. After the patient downloaded the app, the diabetes educator activated the titration scheme. The patient performed fasting SMPG each morning with daily registration in the app. Insulin adjustments were performed according to app suggestions after 3 days of monitorization. Figure 1 shows the functioning of the app. A glucometer (FreeStyle Neo by Abbott USA) was provided for glucose monitoring free of charge.
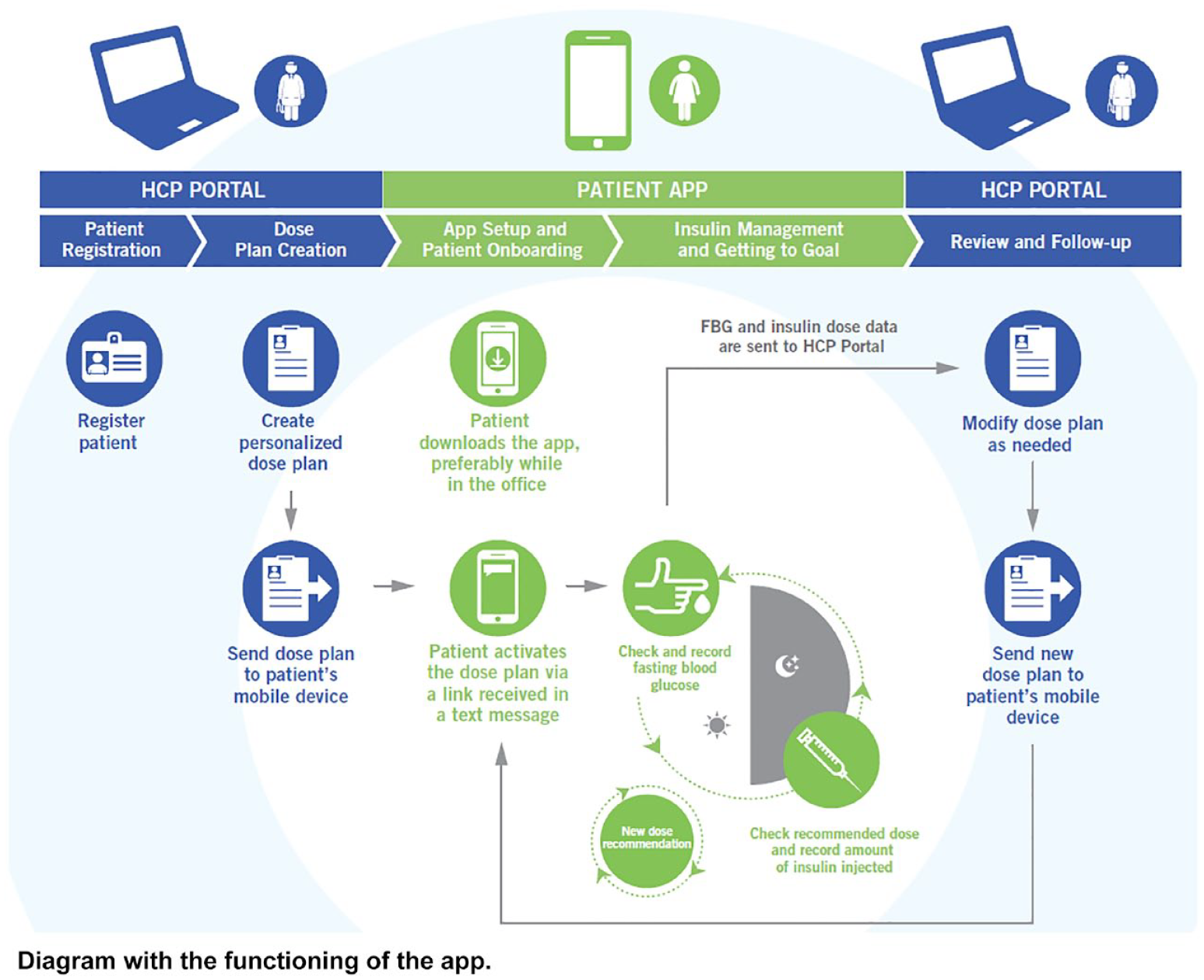
Functioning of the app diagram. Figure shows Health care professional Portal (HCP Portal) and Patient app.
The patients were followed up for 16 weeks, and the procedure flow chart is presented in Figure 2. If the patient achieved the target range for 2 subsequent weeks, reduction of glucose monitoring decreased to 2 times a week. Also, a 7/24 emergency number was provided for patients’ attention at the beginning of the trial.
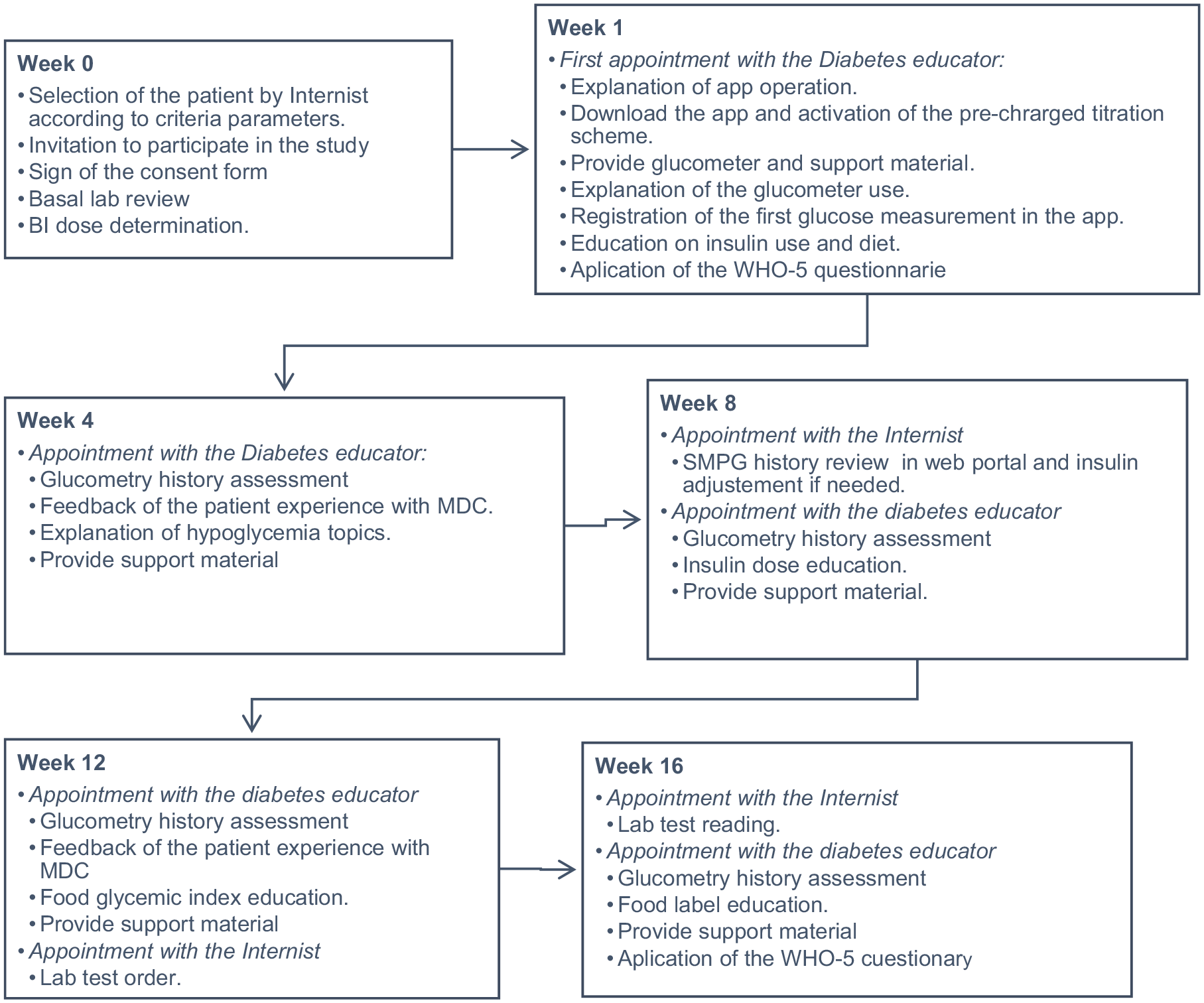
Procedure flow chart. Figure shows follow-up from week 0 to week 16. MDC, My Dose Coach.
The main variables assessed in the study were age, gender, A1C (percentage), FBG (mg/dl), SMPG (mg/dl), basal insulin units (IU), WBI score (created in 1998 by the World Health Organization, validated in Spanish), the proportion of patients achieving SMPG and FBG goals, mean days to reach goals. Hypoglycemia events were defined according to the International Hypoglycemia Study Group as follows: mild (Level 1) when a glucose value of 70 mg/dl (3.9 mmol/L) or less was found; moderate (Level 2) as a glucose level <54 mg/dl (<3 mmol/L) and severe (Level 3) when the patient experienced severe cognitive impairment requiring external assistance for recovery. 13 We registered these variables at the beginning and end of the follow-up in each patient.
Statistics Analysis
The researchers reviewed the quality control and the anonymization of the database. We explored normality of the variables through the Shapiro Wilk tests and frequency histograms for the statistical analyses. We used descriptive statistics and reported results in mean and standard deviation for quantitative variables, frequencies, and percentages for categorical variables. We used paired t-tests and McNemar-test for effect comparisons before and after the intervention. We considered a significant change in the WBI scale if it was higher than 10%. The handling of missing data was done by analyzing complete cases and multiple imputations when they were below the 20%, with the last value carried forward.
We calculated the sample size through G Power version 3.1.9.4 program using paired t-test formula proving the hypothesis that the MDC program will have an effect size of the 25% reducing SMPG during the time of the study. With an alpha of 0.05 and power of 80%, the minimal sample size required was 127 patients. We used the statistical program SPSS version 25 for other statistical analysis a P-value ≤.05 was considered significant.
Results
We included 158 patients, of which 74 (46.8%) were women. The mean (SD) age was 51 (10.3) years, the duration of diabetes was 9.4 (6.5) years. Seventy (44.3%) were professionals working in the steel industry; the rest were their families. Shift workers were also included. One hundred and forty-one patients completed the study (89%), the drop out was 14 (9%) patients and 3 patients suspended insulin use due to lack of need. Before the study 120 (76%) were on basal insulin, 6 (5%) on Human mix 70/30, 8 (7%) on lispro/protamine lispro 25; 35 (29%) on glargine U-300, 69 (57%) were on glargine U-100, 1(1%) on insulin NPH, and 1 (1%) on Regular insulin. The compliance to the app’s suggestion was 90%, and the median (IQR) usage of the app was 91 (36) days. The mean (SD) body mass index (BMI) was 31.5 (4.8). The majority of the patients had 2 comorbidities; the most frequent (%) were dyslipidemia 75(46.8), arterial hypertension 63 (39.9), obesity 26 (16.5), primary hypothyroidism 19(12), depression 13(8.2), and fatty liver disease 6(3.8). Sixty-five patients (41%) used glargine U-100 during the study and the rest (n = 93, 59%) glargine-U 300.
We achieved the SMPG target of 90-130 mgl/dl in 58.9% of the patients in 66 (28) days; with a mean (95CI) reduction of 30.9 mg/dl (22.5-37.7; P < .001). FBG goals were achieved in 55.7% with a mean (95CI) reduction of 63.4 mg/dl (49.6-77.2; P < .001). In the subgroup analyses, patients that were previously using BI decreased FBG 44.71 mg/dl (95CI 30.4-59.02, P < .001), while the group that was not, decreased 100.44 mg/dl (95CI 67.81-133.08, P < .001). Patients that used glargine U-100 during the study decreased FBG 46.26 mg/dl (95CI 12.67-72.85, P < .001), while those using glargine U-300 decreased 67 mg/dl (95CI 52-81.96, P < .001).
The mean (95CI) reduction of A1C was 1.97% (1.6-2.3, P < .001) with multiple imputation model and 1.78% (1.5-2.1, P < .001) with last observation carried forward, respectively. In the subgroup analyses, patients that were previously using BI decreased A1C 1.53% (95CI 1.18-1.88, p < 0.001) with multiple imputation model and 1.42% (1.09-1.75) with the last observation carried forward. The group that was not on insulin decreased their A1C by 2.89% (95CI 2.3-3.55, P < .001) in both models. Patients that used glargine U-100 during the study decreased A1C 1.44% (95CI 0.94-1.93, P < .001) with multiple imputation model and 1.36% (95CI 0.91-1.84, P < .001) with the last observation carried forward. The group using glargine U-300 decreased 2.18% (95 CI 1.76-2.6) with multiple imputation model and 2.04% (95 CI 1.63-2.45, P < .001) with the last observation carried forward.
The WBI is a 5-item questionnaire that rates well-being from 0-5 (less to high), a 10% change is considered clinically important. In our study, the score presented a significant increase of 2.24 (-3, -1.4, P < .001) points, which equals a 11.6%. Table 1 shows the mean change of SMPG, FBG, A1C, and WBI.
Change in SMPG, FBG, Insulin Units, WBI, and A1C.
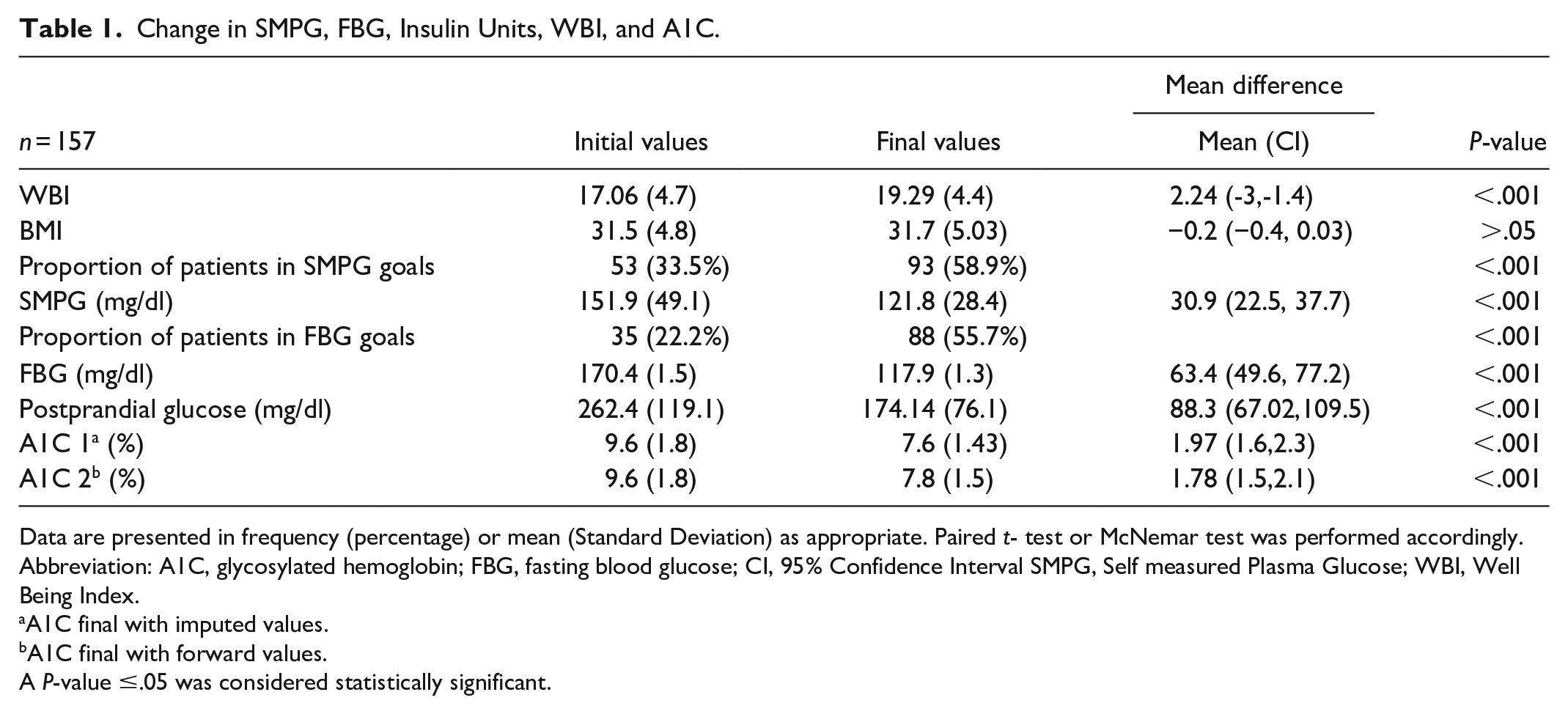
Data are presented in frequency (percentage) or mean (Standard Deviation) as appropriate. Paired t- test or McNemar test was performed accordingly.
Abbreviation: A1C, glycosylated hemoglobin; FBG, fasting blood glucose; CI, 95% Confidence Interval SMPG, Self measured Plasma Glucose; WBI, Well Being Index.
A1C final with imputed values.
A1C final with forward values.
A P-value ≤.05 was considered statistically significant.
The proportion of patients on oral drugs during the study was 96.2% in the beginning and 94.2% at the end of the study. The number of patients on sitagliptin and saxagliptin decreased, while alogliptin prescription increased during the follow-up in relation to the hospital’s changed DPPIV brand provider. However, the dose of oral drugs was stable during the time. There was no significant change in global BI requirements (P > .05); however, 81(51%) of the patients increased (change 10.09 Units), 23 (14%) maintained and 54(34%) reduced their dose during the study (change 10.8 Units). Table 2 shows the oral drug and insulin prescription during the study.
Oral Antidiabetic Drugs and Insulin.
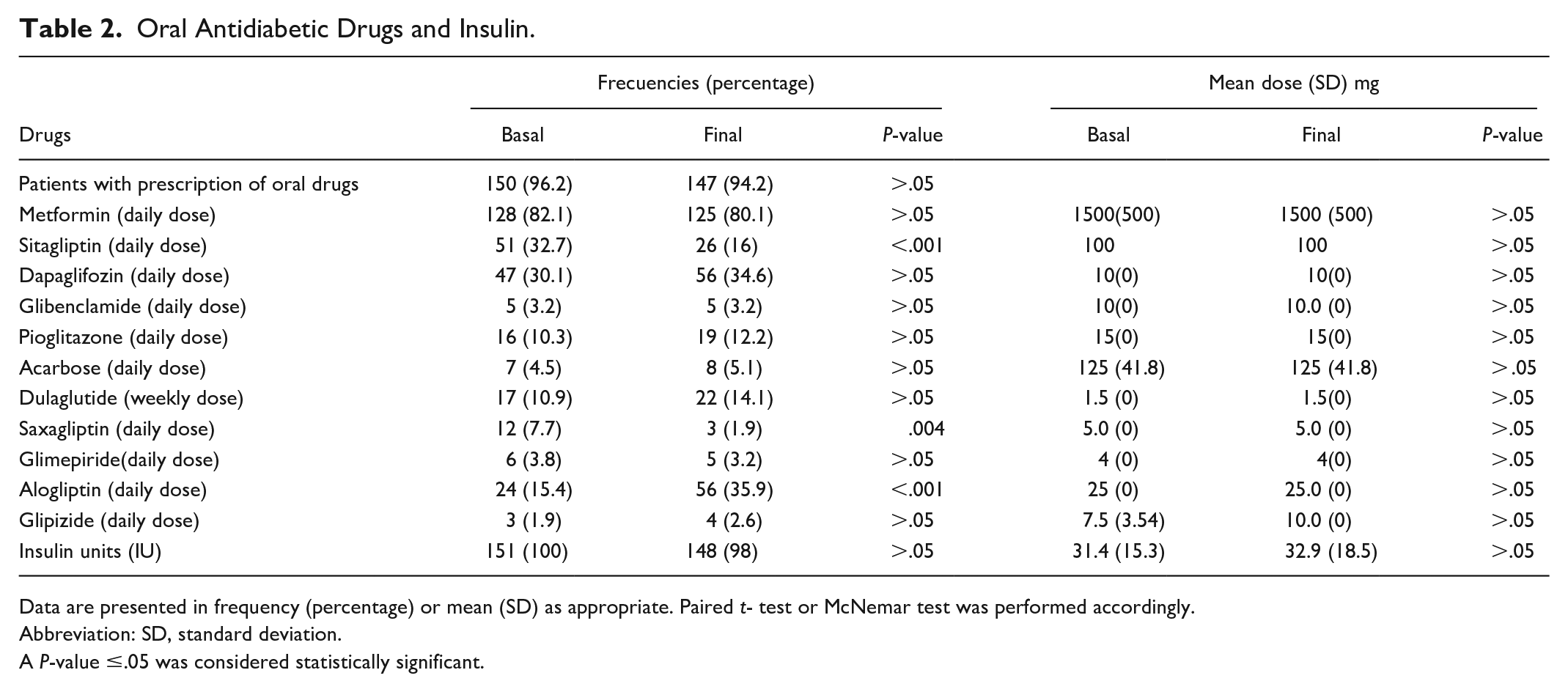
Data are presented in frequency (percentage) or mean (SD) as appropriate. Paired t- test or McNemar test was performed accordingly.
Abbreviation: SD, standard deviation.
A P-value ≤.05 was considered statistically significant.
A 40.5% of the patients had 1-9 events of hypoglycemia; the mean (SD) value was 58(8) mg/dl. There was a total of 226 events, 171(76%) mild (70-54 mg/dl) and 54 (24%) moderate (<54 mg/dl); all of them were successfully treated at home. There were no events of severe hypoglycemia, and no other adverse events (eg, death) in the patients reported during the study.
Discussion
This study showed that MDC effectively achieved an increase in the proportion of patients with SMPG and FBG target range without events of severe hypoglycemia. In addition, there was a decrease in the SMPG, FBG, and A1C values. In the subgroup analysis, patients who were not on BI before the study and those who used glargine U-300 had a higher decrease in FBG and A1C. Additionally, MDC showed an increase in the WBI score. Our sample size was powered enough for the calculation of our results. These tools can be useful in populations where frequent follow-up visits are difficult and where access to other technologies is difficult for economic reasons.
Studies have shown that digital tools lead to significant improvement in glycemic control, greater patient satisfaction, reduced emotional load, fear of hypoglycemia and distress of the regimen, fewer miscalculations, and fewer additional visits to the medical office.14,15 Smartphones and apps offer a unique platform for communication. The mobile health literature indicates that individuals using mobile apps for self-management achieve positive health outcomes. 16 Possible functions in mobile apps include interaction functions such as messages and chatting with health care personnel; health-monitoring functions such as tracking blood glucose and medication; lifestyle-monitoring functions like physical activity and dietary habits; educational functions supplying information, and medical care decision-making through algorithms.17,18 The AUTOMATIX, INNOVATE, and a study conducted by Pfützer Andreas et al. evaluated titration algorithms using insulin glargine.5,14,19
The study conducted by Pfützer Andreas et al., 19 collected data in relation to different titration algorithms for optimization or initiation of BI in patients with T2DM. On an individual bases the researchers applied 4 options: (1) standard (S: FBG target 90-130 mg/dL, n = 39), (2) standard-fast titration (S-FBG target: 90-130 mg/dL, larger dose increments at FBG >180 mg/dl, n = 1), less tight (LT: 110-150 mg/dL, n = 5), and tight (T: 70-100 mg/dL, n = 5). A1C improved from 7.9 (SD 0.8) to 7.5% (SD 0.7) with 14 mild and 9 moderate events, showing that an individualized algorithm could be effective and safe for glucose control. 19
The INNOVATE was a pilot trail using a web-based tool (LTHome, commercial name MyStar Web Coach) or a specialist (EUT) for insulin titration. The target range was 90-130 mg/dl. The titration algorithm consisted of an increase of 2 units if SMPG was >130 mg/dl and 4 units in case of a value >180 mg/dl. The decrease in insulin units could be made by percentage or standard value such as −2 units in case of 90-70 mg/dl and −4 units in case of <70 mg/dl. The primary outcome was a composite of 3 parameters: (1) at least 4 out of 7 FBG within the 10-day period in range 90-130 mg/dl, (2) Mean FBG for 3 consecutive prior FBG within the 10-day period in the range of 90-130 mg/dl, and (3) no severe hypoglycemia during the 7-10-day period. One hundred thirty-nine subjects were randomized. The results were 15% vs. 41% LTHome vs. EUT, P > .05 for primary outcome, 44% vs. 54% for secondary outcome, P > .05 and 47 vs. 51% for third outcome, P > .05. However, the satisfaction score was high in the LTHome, proving clinical utility. 14
The AUTOMATIX trail was a 16-week, randomized, parallel-group of patients with T2DM that used MyStar Dose Coach for BI titration with glargine U-300. The titration algorithm consisted of an increase of 2 units if SMPG was >130 mg/dl and 4 units in case of a value >180 mg/dl. If SMPG was on target range of 90-130 mg/dl no changes were made, and if SMPG was between 90-70 mg/dl, a decrease of 2 units was performed. Finally, if SMPG <70 mg/dl a reduction of 4 units was made. Their results demonstrated a good safety profile and no-inferiority to routine titration (led by diabetes specialists), with a trend toward shorter times being needed to reach fasting plasma monitoring glucose target. 5 Also, 34.3% of the patients of the device-supported titration group and 14.5% of the routine titration group obtained SMPG target range 90-130 mg/dl without confirmed [≤70 mg/dL (≤3.9 mmol/L)] or severe hypoglycemia. 5
Our algorithm had similar target ranges (90-130mg/dl) compared to the studies mentioned above and had a personalized insulin unit change based on a percentage. However, the SMPG target range was successful in more patients, and we had a higher decrease in A1C.
A real-world pilot study of patients with T2DM that used MDC for BI titration conducted in India, reported that 34% (n = 113) of their patients reached the FBG target range. 11 If we compare our results with this study, we had 58.9% in target, which means 24.9% more patients in range.
Even though we had a group of patients that increased their BI dose, another proportion maintained and reduced their dose. The global requirements of insulin units did not present a statically significant change during the trial. It has been reported that when the patients are involved in treatment decision-making, their understanding of diabetes care increases and positively impacts upon their self-management. 20 Patient personalized management, the increase in diabetes education, learning to use a glucometer, empowerment, and confrontation helped these results.
The reduction of A1C has been classified as an international standard of medical care in T2DM. 21 In this study, the mean reduction of A1C was statically significant. A systematic review that evaluated the effect of Technology-Enabling diabetes Self-management education using interventions that incorporated feedback loop components to connect people with diabetes and their health care team has shown similar results. 22
There was a drop out of 9% during the trial. In comparison, the AUTOMATIX had a drop out of 23% and 58% in the Indian study.5,11 When compared with our study, this is an important difference. We believe that the low drop out in our study is because our population is stable within a single company; both the patients and their families are easy to reach since medical providers are closely related to the head of the workers department. In addition, patients are engaged in their health following company policies.
As limitation to our study, we did not include a control group during follow-up; since we use a before and after design where the patients were the case and control after the intervention, this design has its own bias and confusion factors. Another limitation was the lack of randomization and a short follow-up time, 16 weeks. Finally, the patients used only one type of insulin (glargine).
We should conduct future studies concerning the performance of pragmatic studies that include daily life practice with incorporation of a control group. A larger sample size is required for other subgroup analyses. We must conduct further studies to test other BI used in the daily practice such as detemir, NPH, degludec, and so on. It could be of interest to test MDC in other types of diabetes such as gestational diabetes or LADA, Type 1 Diabetes, etc. A larger follow-up study (more than 16 weeks) must be conducted to evaluate long-term benefits. In addition, an economic study could be of great value. Additional studies should be conducted to compare algorithms reaching target, time to reaching target, number of hypoglycemia, and so on.
Conclusions
This is the first study of MDC app conducted in Latin America. The results showed that it successfully reduced and achieved goals of SMPG and FBG, in patients with T2DM being treated with BI. There was a statistically significant reduction of A1C with no events of severe hypoglycemia. These tools can be useful in patients with type 2 diabetes where access to frequent follow-up visits is difficult.
Footnotes
Acknowledgements
We are grateful to Lorenza Legarreta for language polishing and editing, Dr. David Soni Duque and Dr. Florisa Rodríguez Valadez for general collaboration in the research project.
Abbreviations
A1C, glycated hemoglobin; FBG, Fasting blood glucose; SMPG, Self-Measured Plasma Glucose; T2DM, Type 2 Diabetes; WBI, Well-Being Index.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sanofi Mexico provided My Dose Coach app and the glucometers for the patients. The rest of the research project was funded by the hospital’s private fundings. Sanofi Mexico suggested language polishing editor but had no role in data collection and analysis, decision to publish, or preparation of the manuscript.
