Abstract
Background:
Conventional home blood glucose measurements require a sample of blood that is obtained by puncturing the skin at the fingertip. To avoid the pain associated with this procedure, there is high demand for medical products that allow glucose monitoring without blood sampling. In this review article, all such products are presented.
Methods:
In order to identify such products, four different sources were used: (1) PubMed, (2) Google Patents, (3) Diabetes Technology Meeting Startup Showcase participants, and (4) experts in the field of glucose monitoring. The information obtained were filtered by using two inclusion criteria: (1) regulatory clearance, and/or (2) significant coverage in Google News starting in the year 2016, unless the article indicated that the product had been discontinued. The identified bloodless monitoring products were classified into three categories: (1) noninvasive optical, (2) noninvasive fluid sampling, and (3) minimally invasive devices.
Results:
In total, 28 noninvasive optical, 6 noninvasive fluid sampling, and 31 minimally invasive glucose monitoring products were identified. Subsequently, these products were characterized according to their regulatory, technological, and consumer features. Products with regulatory clearance are described in greater detail according to their advantages and disadvantages, and with design images.
Conclusions:
Based on favorable technological features, consumer features, and other advantages, several bloodless products are commercially available and promise to enhance diabetes management. Paths for future products are discussed with an emphasis on understanding existing barriers related to both technical and non-technical issues.
Introduction
Optimal diabetes management requires glucose monitoring at regular intervals or continuously. 1 In the 1970s, the commercial sector responded by developing analytical systems that provided accurate readings of glucose concentrations in capillary blood samples. This technology is commonly referred to as either self-monitoring of blood glucose (SMBG) or assisted monitoring of blood glucose (AMBG) and is recognized as a major advance in managing diabetes. This technology permitted, for the first time, the ability for people with diabetes to monitor their individual glycemia on a daily basis. This technology also enabled the clinical studies that established the benefits of tight glycemic control in delaying the onset of diabetes complications.2,3
Despite improvements in sample volume requirements, analysis times, and measurement accuracy, the pain, cost, and inconvenience of SMBG technologies are driving the commercial sector to develop new analytical devices that are not based on individual blood measurements, but rather on continuous, or near continuous, glucose measurements in non-blood samples. Non-blood glucose sensing technologies have been under development since the mid-1980s, yet relatively few have realized commercialization and received regulatory approval.
This review reports the results of a literature search to uncover a listing of established and nascent commercial products that are based on non-blood glucose sensing technologies. Each product is classified as either of the following:
(1) Noninvasive optical glucose monitor (NIO-GM),
(2) Noninvasive fluid sampling glucose monitor (NIFS-GM), and
(3) Minimally invasive glucose monitor (MI-GM).
These technologies are generally characterized as providing continuous or intermittent glucose measurements with minimal or no pain.
Definitions of Glucose Monitoring Devices
The discovered products are classified according to the following set of definitions.
Invasive glucose monitors
The most recent published definition of an invasive procedure, based on an analysis of almost 400 articles from the medical literature, is “Where purposeful/deliberate access to the body is gained via an incision, percutaneous puncture, where instrumentation is used in addition to the puncture needle, or instrumentation.”
4
By this definition, when a product requires puncturing of the skin with a lancing device, this product is defined as an
Home IGM devices involve collecting a sample of capillary blood by breaking the skin barrier at the subject’s finger with a sharp lancing device.5,6 Such IGM systems have been marketed for almost half a century and have undergone continual improvements to require less blood volume and shorter measurement times. Unfortunately, the cost, pain, blood waste, and finger calluses related to this method reduce patient enthusiasm for frequent glucose testing. 7
Noninvasive optical glucose monitors
Using the above definition of an invasive procedure, a technology where the concentration of glucose is measured without inserting a device into the body is considered noninvasive. When a noninvasive measurement involves passing a type of radiation into a vascular region of the body, then the instrumentation is defined as a
Noninvasive fluid sampling glucose monitoring
A second type of noninvasive measurement involves analysis of a fluid sample that is collected without an invasive procedure. In this case, the body fluid, such as tears, sweat, saliva, and urine, is available without puncturing skin and the analytical information is determined ex vivo. Once the processes involved in collecting and analyzing a sample are packaged into an integrated product, the system is defined as a
The above definition differs from those used in previous reviews that categorize glucose sensing technologies.9-11 In other reviews, analysis of fluids other than blood are grouped under either the terms minimally invasive or noninvasive, as illustrated in Figure 1A and 1B. The rationale for treating these methods as minimally invasive is based on the fact that these NIFS-GM methods require collection of a body fluid and the analytical measurement does not involve passing radiation through the body. On the other hand, other reviews place an emphasis on the lack of puncturing skin for NIFS-GM systems, thereby categorized these devices as noninvasive. Developers of such products prefer the term noninvasive over minimally invasive because of marketing advantages.
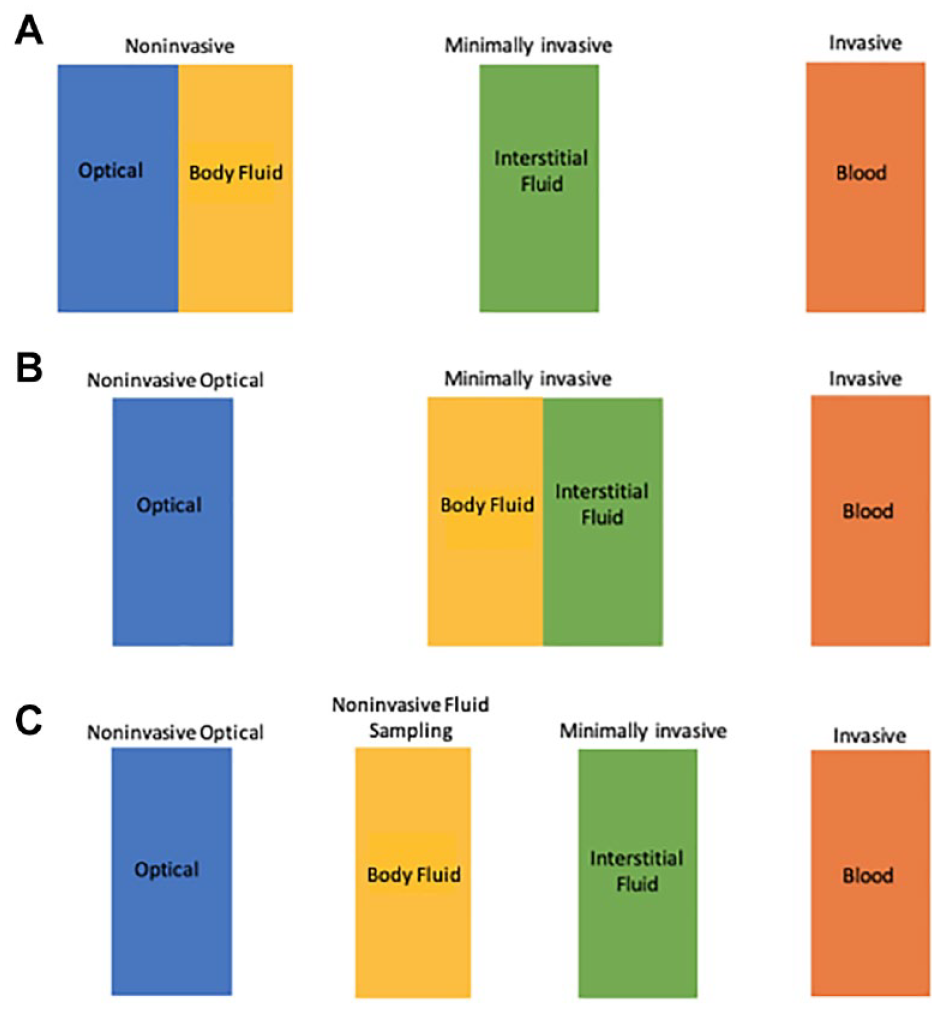
How NIFS-GM technologies have been classified. (A) Previously classified by some experts as a noninvasive technology; (B) Previously classified by other experts as a minimally invasive technology; (C) Classified per our definition in this article as a distinct technology separate from NIO-GM and MI-GM.
In our assessment, NIFS-GM systems are distinctive and require a separate designation, as illustrated in Figure 1C. These technologies are uniquely characterized by a lack of puncturing skin, instrumentation applied to the body, a potential for skin trauma, and the need for an established clinically valid correlation between the concentrations of glucose in the fluid and in blood.
Minimally invasive glucose monitoring
The insertion of a sensor into the subcutaneous tissue is an invasive procedure, but this procedure is characterized as involving minimal pain and providing glucose concentrations over an extended period with a single insertion. For these reasons, such an approach is defined as a minimally invasive glucose monitor (MI-GM). These technologies are capable of staying in place for periods of days, weeks, or even months while performing glucose measurements repeatedly.
MI-GM devices have been marketed worldwide for the past two decades and serve as the basis for state-of-the-art wearable continuous glucose monitor (CGM) systems. The commercial success of these devices is illustrated by the data presented in Figure 2. For the industry, total revenues exceeded $1.5 billion USD as of the fourth quarter of 2020. 12
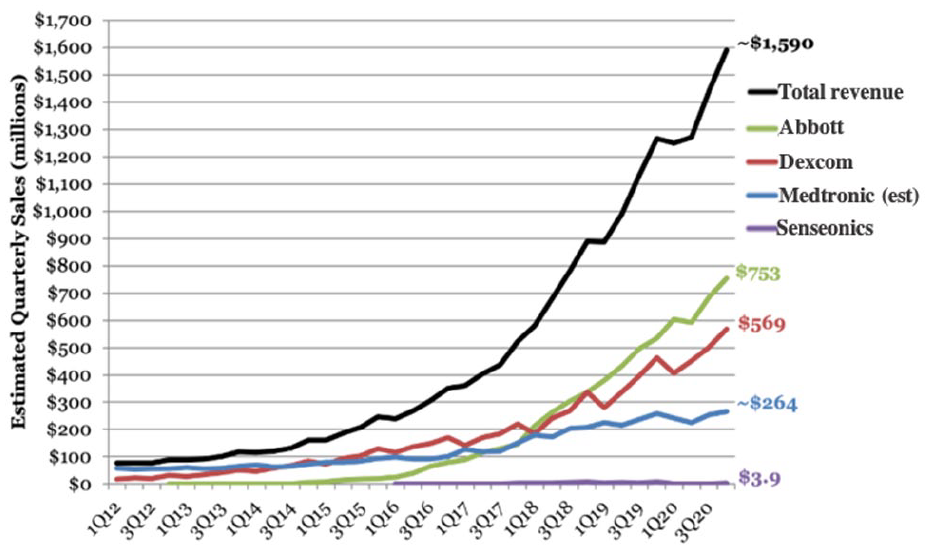
The total quarterly sales for MI-GMs produced by the world’s four largest manufacturers as of the fourth quarter of 2020.
Technological Approaches for Glucose Monitoring Products
The various glucose monitoring products rely on different technological approaches, as described below.
Technology for noninvasive optical glucose monitoring
NIO-GM involves sending harmless, low-energy radiation through a vascular body site and extracting information about the glucose concentration from the collected signal. In many cases, a selected band of electromagnetic radiation is applied to skin and the diffusely scattered photons are collected. Glucose concentrations are estimated by a multivariate analysis of the resulting spectrum. Several recent review articles provide detailed descriptions of the different NIO-GM technologies currently under development.5,13-15 These technologies include near-infrared spectroscopy, mid-infrared spectroscopy, Raman scattering spectroscopy, optical polarimetry, approaches based on microwave and radio wave sensing, and others.5,13-15
Technical hurdles for success of NIO-GM products include issues related to detection limits and selectivity of glucose measurements. Glucose concentrations in blood and ISF are at milli-molar concentrations, which places them at an intermediate level – meaning they are lower than the principal components of skin and higher than many clinical biomarkers. Still, spectroscopic signals originating from glucose molecules are weak, which challenges the signal-to-noise ratio (SNR) of the instrumentation.16-19 Fundamentally, the SNR for the instrumentation must be sufficient to enable measurement of the weak signals from glucose over background noise and other sources of instrumental variation. In addition, the measurement signal must be selective relative to all other components of skin, including membranes, glycosylated structures, and soluble compounds within the ISF matrix, such as albumin, urea, amino acids, and ascorbic acid. Ideally, such a selectivity is derived from the chemical structure of glucose, thereby providing a robust basis for measurement accuracy. Depending on the radiation used, a viable NIO-GM has to take into account skin pigmentation, surface roughness, skin thickness, breathing artifacts, blood flow, body movements, and ambient temperature. 20 Individual calibration can reduce the impact of the skin’s contribution to the results of measurement.
NIO-GM devices have been proposed based on primary or direct glucose sensing as well as secondary, or indirect glucose sensing. Primary measurements involve collecting a signal derived directly from the glucose molecule, while secondary measurements involve measuring a parameter impacted by the concentration of glucose, such as (1) heart rate changes with electrocardiography, 21 (2) rate of red blood cell aggregation with ultrasound, 22 (3) blood volume dynamics with photoplethysmographic measurement of blood,23,24 (4) dielectric properties of the skin matrix with diffuse scattering or temperature-modulated localized reflectance, 25 or (5) sudomotor dysfunction with electrochemical skin conductance and sweating asymmetry. 26
No NIO-GM product has received clearance by the United States Food and Drug Administration (FDA). Although both direct and indirect noninvasive glucose measurements bear the burden of establishing measurement accuracy, this burden can be greater for indirect methods owing to the lack of a selective signature originating from the glucose molecule. Indirect methods are based on correlations of physiological signals that may be impacted by parameters other than glucose, thereby confounding selectivity and measurement accuracy to a considerable extent. However, machine learning and neural network approaches might provide a successful path for selective indirect glucose measurements. 21 Successful approaches might require a combination of direct and indirect sources of analytical information. It is possible to quantify glucose in standard solutions and tissue phantoms under idealized conditions by such approaches; however, in the real world, the accuracy and precision of glucose measurements could be inadequate for clinical use. Measurement accuracy over time represents the major analytical challenge of all NIO-GM technologies.
The FDA recently distributed a warning concerning the impact of skin pigmentation on the accuracy of clinical pulse oximetry measurements. 27 NIO-GM are potentially subject to the same concerns depending on the wavelength of the probing radiation. Absorption of the incident radiation by melanin, or other skin pigments, will certainly impact signal measurements. 28 Substances that may affect skin pigmentation, skin structure, and reflectance properties of the probing radiation include topical medications, cosmetics, cosmeceuticals, and estrogen, as well as tobacco, and alcohol. 29 Radio waves and microwaves are not expected to be impacted and infrared wavelengths are also unlikely to be affected, although visible and near infrared wavelengths near the visible spectrum (ie, less than 1 micron can be impacted). The impact of skin pigmentation must be experimentally assessed for each system.
The only approach to developing an NIO-GM product that does not involve measurements in skin, involves direct measurements in the aqueous humor of the eye. 30 This approach eliminates the confounding effects presented by the skin barrier, however, none of the products covered in this review is based on this approach.
Although no NIO-GM device has been approved by the FDA, interest in noninvasive measurements is high. In 2020, 343 articles were referenced in PubMed with the search term “noninvasive glucose.” Also, the number of such articles in PubMed has increased progressively over the past 45 years. 31 Initially, interest in this field was mostly academic, but since the 1990s, a number of companies have been attempting to develop NIO-GM products.32-35
Technology for noninvasive fluid sampling glucose monitoring
NIFS-GM products center on technologies capable of collecting and analyzing samples of ISF, tears, sweat, or saliva. Electrochemical glucose oxidase biosensors are available to determine the concentration of glucose in the collected fluid. Sample collection technologies are specific for the measurement fluid. Reverse iontophoresis enables the collection of a representative sample of ISF without puncturing the skin. Other technologies are reported for measurements in lacrimal, 36 and tear fluids. 37
Two NIFS-GM technologies have either received clearance from the FDA or have been granted a Conformité Européenne (CE) Mark. The first is the measurement of glucose in ISF samples extracted through the skin surface by reverse iontophoresis. 38 This is an active transport system stimulated by an electrical current applied to the skin. The concentration of glucose in the resulting fluid is measured with a sensor patch placed on the skin surface. Glucose concentrations in such samples are two to three orders of magnitude below those in the original ISF, which presents an analytical challenge. 39 The SugarBEAT from Nemaura (Loughborough, England) is a NIFS-GM device that uses this method and has received a CE Mark. A comparable approach received FDA clearance in 2001 and was marketed as the GlucoWatch Biographer by Cygnus, Inc. (Redwood City, California).40,41 The resulting glucose concentration measurements lacked sufficient clinical accuracy and the device suffered from poor user experience. 42 A skin permeation process using ultrasound was briefly used more than ten years ago to collect ISF with a sensor placed over the site of skin permeation for an investigational needle-free product, 43 and this type of product is still under development. 44
Technically, urine test strips fall under the definition of a NIFS-GM device. These test strips are ineffective in estimating real-time blood glucose concentrations and are rarely used for making treatment decisions. Urine glucose testing is characterized by the following three characteristics:
(1) a long and unpredictable lag time between dynamic blood glucose concentrations and urine glucose concentrations,
(2) a high blood glucose concentration threshold for urine glucose excretion, below which glucose does not appear in the urine, and
(3) a wide person-to-person variability in this blood glucose threshold concentration where glucose spills into the urine. 45
For NIFS-GM technologies designed to measure glucose in samples of sweat, 37 saliva,46,47 and tears, 48 the actual analytical measurements might be accurate; however, the correlation is poor for glucose levels in these fluids compared to those in blood, and this correlation might be influenced by regionalized physiological stressors or stimuli. 49 Such correlations have proven difficult to control, thereby raising the possibility that physiology will render such an approach to be impractical for commercialization.
Technology for minimally invasive glucose monitoring
The majority of MI-GM products currently on the market are devices that require the user to insert the sensing element in the subcutaneous space. The sensing element is an electrochemical glucose biosensor which reports a glucose concentration every 1-15 minutes to a receiver located outside the body. The sensor is limited to 10-14 days of operation.
Three generations of biosensor technology are reported for MI-GM devices. Each generation differs by the internal path of electrons associated with the enzyme-catalyzed reaction and the electrochemical detection. In all generations, the enzyme catalyzes the oxidation of glucose to form gluconic acid, thereby producing the reduced form of the enzyme cofactor. The electron generated from the oxidation of glucose is ultimately transferred to either oxygen (first generation), an electron mediator (second generation), or the electrode directly (third generation). 50 Accuracy of glucose measurements and their resistance to interferences by drugs or other substances have improved with each successive generation of glucose sensors. Third generation sensors are not yet commercially available. 51
As an alternative to electrochemical MI-GM devices, the principles of reversible affinity sensing are available. For this purpose, two molecules, one of which is a glucose binding protein (GBP), are brought together with a dextran derivative. At low glucose concentrations, the GBP forms a macromolecular complex with the dextran-derivative. As the concentration of glucose increases, the GBP will prefer glucose, thereby dissociating the macromolecular complex. The measurement of the glucose concentration is carried out by determining the change in a physical parameter related to this dissociation of the macromolecular complex. Examples of measurements include a change in viscosity, 51 osmotic pressure against a membrane, 52 or fluorescence.53,54 Depending on the measuring principle, the change in the measured quantity is detected with either pressure or optical tranducers and the signal is correlated to a change in glucose concentration by a calibration curve.
The Eversense CGM, offered by Senseonics (Germantown, Maryland) is an FDA approved MI-GM device that measures the concentration of glucose by a fluorescence signal generated from a reversible affinity sensing mechanism involving a selective glucose binding protein. 53 This device has demonstrated clinical sufficient glucose measurement accuracy for 180 days,55,56 although it was initially cleared by the FDA for 90 days of use.57,58 This device must be implanted under the skin and removed by a healthcare professional and requires twice daily calibration.
Technological Challenges across Platforms
Regardless of the sensing technology, NIO-GM, NIFS-GM, or MI-GM, the following technical challenges must be considered for each clinically viable product.
Time delays in glucose concentrations between blood and measurement fluids
When blood glucose concentrations change, glucose levels in other physiological fluids, such as ISF, do not show similar changes instantaneously. As a result, differences in glucose concentrations reported from NIO-GM, NIFS-GM and MI-GM devices compared to blood measurements may not reflect inaccuracy in the measurement technology but may have a physiological basis. The rate of transporting glucose between the different fluids is not instantaneous and time is required for glucose to diffuse between compartments during periods of disequilibrium. Relevant body fluids include ISF within the subcutaneous matrix,59-61 extracted samples of sweat, 62 saliva, 63 tears, 64 or ISF harvested using reverse iontophoresis, 10 and aqueous humor measured from outside the body. 65
Time lags for glucose concentration measurements have three components: First, a physiological lag occurs as glucose equilibrates by diffusion (a passive transport to or from blood and the body fluid). The impact of this source of time delay depends on the rate of change in blood glucose levels. This physiological lag time can vary between individuals; but tends to be consistent over time for a specific individual for a given fluctuation in the blood glucose pattern. Second, a measurement lag can occur if the sensor’s response is slow. Third, a delay can occur owing to the data-smoothing algorithm. Such algorithms can introduce a time delay between the current glucose sensor reading and previous readings to smooth fluctuations, which leads to a delay in the measurement output. This third element of lag time is only relevant for CGM technology and not for spot glucose measurements.66,67
Reported total lag times vary depending on the measurement conditions; however, they generally range between 5 and 20 minutes. Lag times tend to be shorter with NIO-GM and MI-GM sensor methods and tend to be longer with NIFS-GM. In the latter case external glucose sensors measure fluids that must be extracted before the analytical measurement. If a manufacturer provides a single number to represent the lag time in minutes for ISF glucose compared to blood glucose for a glucose sensor, then this number cannot necessarily be relied upon because much of the lag time is dependent on the individual whose lag is being measured. 60 Furthermore, the lag time can vary depending on activities being performed 68 or whether the blood glucose concentration is rising or falling. 69
Skin irritation
SMBG and AMBG capillary blood measurements can lead to calluses and scarring on the punctured finger tips. 70 MI-GMs and the two categories of noninvasive glucose monitors (of the skin only), NIO-GMs and NIFS-GMs, have shown varying levels of skin irritation that could cause discomfort, scarring, or local skin infections. These skin reactions can be induced by the adhesives used to affix the glucose sensing element to the skin. Such adhesives have been reported to cause skin irritation and even allergic reactions. 71 Stronger adhesives used to facilitate longer wear times can lead to even more pronounced skin problems. 72 Acrylates being in the adhesives (and also in the plastic material of the sensor housing) are known to induce skin reactions.73,74
Short term studies with MI-GMs that use transcutaneous microneedles to sample ISF have demonstrated minor edema and erythema skin irritation that resolve without treatment.75,76 The use of a powerful laser for NIO-GM measurements poses a risk of pronounced skin damage. NIFS-GM methods sampling from the skin using no needles may cause dermatitis after prolonged collection (e.g., by reverse iontophoresis, 10 skin permeation, or sweat collection 77 ). NIFS-GMs that collect fluids from sites other than the skin as well as optical noninvasive glucose monitors have not been reported to cause skin irritation.
Toxicity
The issue of toxicity is more of an issue for MI-GM technologies compared to the other less invasive approaches. 51 For IGM, NIFS-GM, and MI-GM products, there is direct contact between the sensing element and a physiological fluid containing glucose. However, operation of MI-GM devices calls for the sensing element to be inserted into the body for periods of multiple days, which demands safety assurance. For IGM and NIFS-GM devices, the glucose measurements occur outside the body, thereby permitting the use of materials that would be unsuitable for indwelling MI-GM sensors. NIO-GM devices might involve direct contact between the physical glucose sensing element and skin. If so, then toxicity effects of the sensing materials must be evaluated. Manufacturers of NIO-GM systems must also be concerned with adverse effects caused by the energy and power of the probing radiation applied to the skin.
User involvement
Glucose monitoring methods that rely on user action are not well suited for automatic and continuous glucose monitoring. User involvement can be extensive for IGM and some NIFS-GM devices as well as for any bulky NIO-GM devices. Either a minimally invasive implanted sensor or a wearable noninvasive sensor would be suitable for measuring continuous and automatic glucose measurements. In comparison to intermittent measurements, CGM devices can provide the following important information and functions with minimal user inputs or adjustments:
(1) real-time glucose concentrations including the timing and magnitude of peak responses to food intake, physical activities, and medications, 78
(2) a 24-hour recording of changes of glucose levels over time, 79
(3) short-term and long-term glycemic trends, 80
(4) warnings of impending or surpassed clinically relevant low or high glucose concentrations, 81
(5) control of an automated insulin delivery (AID) system, 82 and
(6) interface with advanced digital systems for diabetes management. 83
Methods
Overview
We conducted a database review to survey products for monitoring glucose that did not use invasive blood glucose sampling. We searched for products using optical noninvasive, fluid sampling noninvasive, and minimally invasive glucose monitoring technologies. We performed four types of searches to find candidate NIO-GM, NIFS-GM, and MI-GM products and their manufacturing companies to be included in our list for this article, and these searches are updated as of December 17, 2020. We listed an NIO-GM, NIFS-GM, or MI-GM product if the product met at least one of two inclusion criteria: (1) clearance or approval by FDA or CE, and/or (2) significant coverage in a news story or press release from a search of the product (or, if the product did not turn up, then a search of the manufacturer) on Google News, indicating the product was reported as viable since 2016. When we discovered a product that would possibly qualify for our list from one of our four sources of information and designated these as “candidate products.” We then performed two types of, what we called, “Inclusion Searches.” The product was defined to meet our first criterion if its manufacturer’s website or a Google search indicated that the product was cleared by FDA or had a CE Mark. The product was defined to meet our second criterion if a search of Google News for an article published since 2016 provided significant coverage, which we defined as at least one paragraph of this product. In summary, we searched for information about candidate products for NIO-GM, NIFS-GM and MI-GM technologies through four information sources. For each discovered candidate product, we applied the two inclusion criteria to determine whether the product would be included on our “identified products” list. Figure 3 presents a PRISMA diagram of how we performed our database reviews to create a list of identified NIO-GM, NIFS-GM, and MI-GM products.
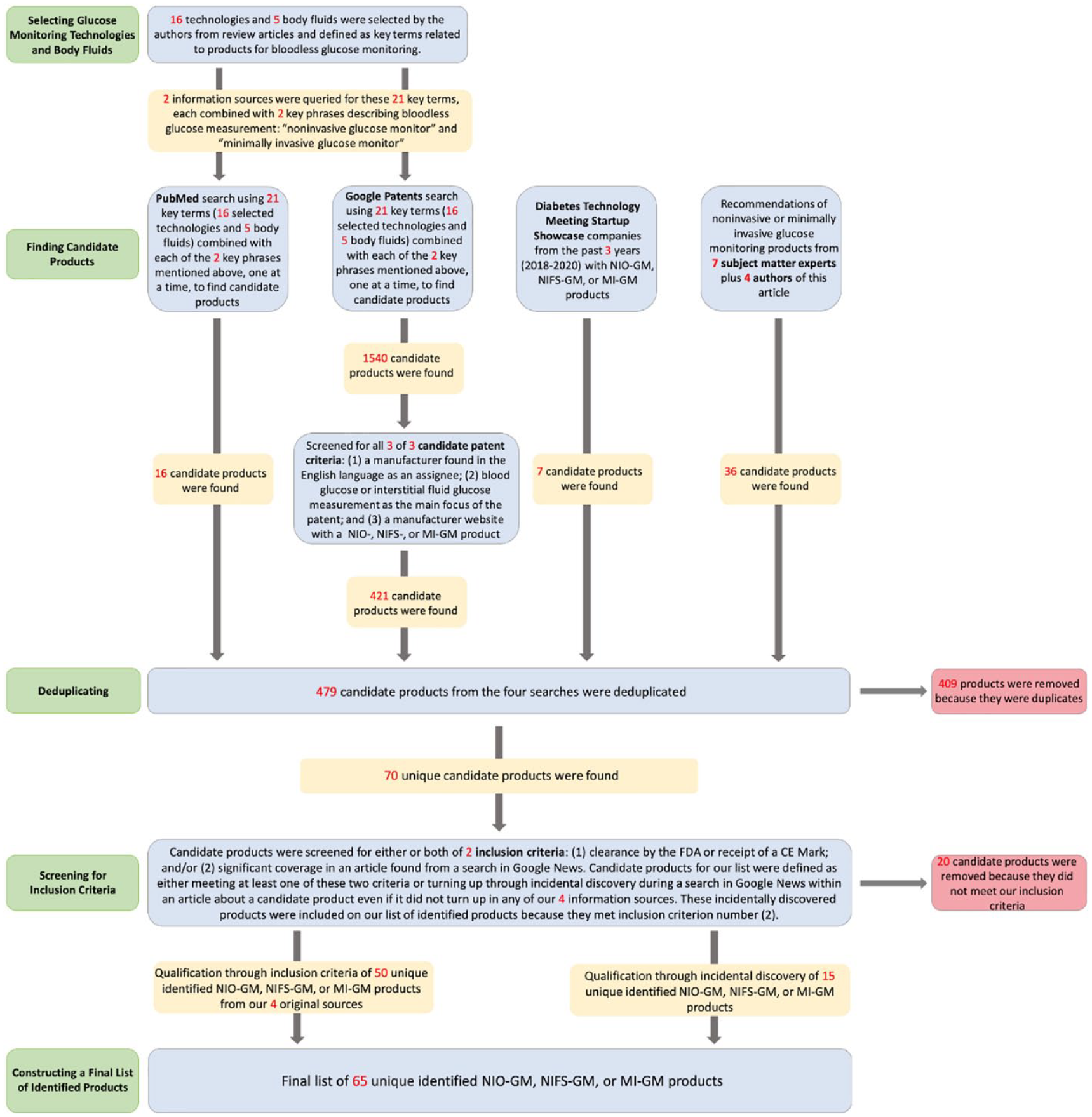
A PRISMA diagram of how we performed our database reviews to create a list of identified NIO-GM, NIFS-GM, and MI-GM products.
Information Sources for Finding Candidate Products
PubMed
Our first source of information for finding candidate products came from two types of searches in PubMed-referenced journals for articles describing technologies for measuring glucose noninvasively (either optically or via fluid sampling) and minimally invasively. We selected 16 technologies that have been reported to be used for NIO-GM, NIFS-GM, or MI-GM products in review articles about bloodless glucose monitors.5,13-15 These 16 technologies included: (1) spectroscopy, (2) Raman scattering spectroscopy, (3) fluorescence, (4) iontophoresis, (5) polarimetry (optical rotation of polarized light), (6) impedance, (7) osmotic pressure, (8) electrochemistry, (9) absorption, (10) scattering, (11) photoacoustics, (12) quantum cascade laser, (13) photothermal detection, (14) photoplethysmography, (15) quartz crystal microbalance, and (16) DNAzyme.5,13-15 We performed a set of 16 PubMed searches of technologies with two search terms: one of these 16 technologies + “glucose monitor.” From the articles that we found, we searched each article for a glucose monitoring product and its manufacturer. We also searched for glucose monitoring products that measured any of five body fluids that we selected from review articles about bloodless glucose monitors.5,13-15,37 These body fluids included: (1) ISF, (2) sweat, (3) tears, (4) saliva, and (5) breath vapor. We performed a set of five PubMed searches of body fluids with two search terms: one of these five body fluids + “glucose monitor.” We sorted searches by “best match” and terminated each search after finding no additional relevant articles about glucose monitoring after ten consecutive additional articles. From the articles that we found, we searched each article for a glucose monitoring product and its manufacturer. We upgraded a candidate product to our identified products list if it met at least one of the two inclusion criteria.
Google Patents
Our second source of information for finding candidate products came from a series of 42 searches in the Google Patents database, each using one of the same 16 key technology terms and five body fluid terms we used in our PubMed search plus either the phrase “diabetes minimally invasive glucose monitor” or the phrase “diabetes noninvasive glucose monitor.” We limited this search to active or pending patents published starting from January 1, 2010 using the Google Patents “Date” filter. The Google Patents search engine automatically generated the most relevant patents first and progressively less relevant patents afterward. Each page contained ten patent applications. We elevated a patent to the stage of assessing whether it met our two inclusion criteria for our list of identified products if it turned up in one of our 38 searches and also met three criteria for a candidate patent that we established for our patent search. These criteria specified that a candidate patent had: (1) a manufacturer named in the English language as an assignee, (2) a main focus on measuring glucose in blood or a body fluid, and (3) a manufacturer’s website with an NIO-GM, NIFS-GM, or MI-GM listed as a product. For each of our 42 searches, we reviewed successive pages for candidate patents. We continued reviewing page after page as long as we found at least one new candidate patent per page. If a page contained no such patents, then we reviewed one subsequent page and if that page also contained no candidate patents, then this search was terminated. If a candidate patent was found on one of those two pages, then we resumed searching page after page. For the purpose of termination, if a manufacturer generated more than one patent application during a search, then each subsequent patent after the first candidate patent was not counted as a new candidate patent. However, subsequent patents to a first patent were counted for the total number of candidate patents found, and they were later deduplicated if they were the same product. We upgraded a candidate product to our identified products list if it met at least one of the two inclusion criteria.
Diabetes technology meeting startup showcase
Our third source of information for finding candidate products came from a review of any NIO-GM, NIFS-GM, or MI-GM product presented by startup companies that participated in Diabetes Technology Society’s three Startup Showcases during the 2018, 2019, and 2020 Diabetes Technology Meetings. We used a proprietary database provided by Diabetes Technology Society to find the companies. We upgraded a candidate product to our identified products list if it met at least one of the two inclusion criteria.
Experts in glucose monitoring
Our fourth source of information for finding candidate products came from seven experts in glucose monitoring: (1) Guido Freckmann, MD (Institute for Diabetes Technology GmbH, Ulm, Germany), (2) Avner Gal, MBA, MSCEE, MSc (Iridium Consultancy and Technologies, Ltd., Herzliya, Israel), (3) H. Michael Heise, PhD (South-Westphalia University of Applied Science, Iserlohn, Germany), (4) Jeffrey La Belle, PhD, MS (Grand Canyon University, Phoenix, Arizona, United States), (5) Jeffrey Joseph, DO (Thomas Jefferson University, Philadelphia, Pennsylvania, United States), (6) John Pickup, MD, PhD (King’s College of London), and (7) Mark Prausnitz, PhD (Georgia Institute of Technology, Atlanta, Georgia, United States) and asked them to name a product and/or a manufacturer developing novel NIO-GM, NIFS-GM and MI-GM technology. We also used information provided by five of the authors (AT, MA, BV, LH, DK) to name similar products and/or manufacturers. We searched every named manufacturer’s website for a product name. We upgraded a candidate product to our identified products list if it met at least one of the two inclusion criteria.
Inclusion Criteria for Upgrading a Candidate Product to an Identified Product
FDA clearance or CE Mark
Our first inclusion criterion for upgrading a candidate product to an identified product was regulatory clearance of a candidate product. We searched the product name in the “Device Name” search box, or the manufacturer’s name in the “Applicant Name” search box of the 510(k) Pre-Market Notification Database of the FDA 84 to determine FDA clearance. If we were unable to find information on this database, then we also searched the manufacturer’s website for a claim of FDA clearance and/or a CE mark. If a manufacturer’s website did not indicate regulatory clearance, then we also searched the first page on Google using the search terms of the product name or manufacturer’s name + “glucose monitor” + “FDA” and the product name or manufacturer’s name + “glucose monitor” + “CE Mark.” If a product had FDA clearance and/or a CE mark, then we included this information in Table 1 in a column entitled “Which Inclusion Criteria Were Met.”
Regulatory Features of NIO-GM, NIFS-GM, or MI-GM Identified Products (in Alphabetical Order Within Each Category of Invasiveness).
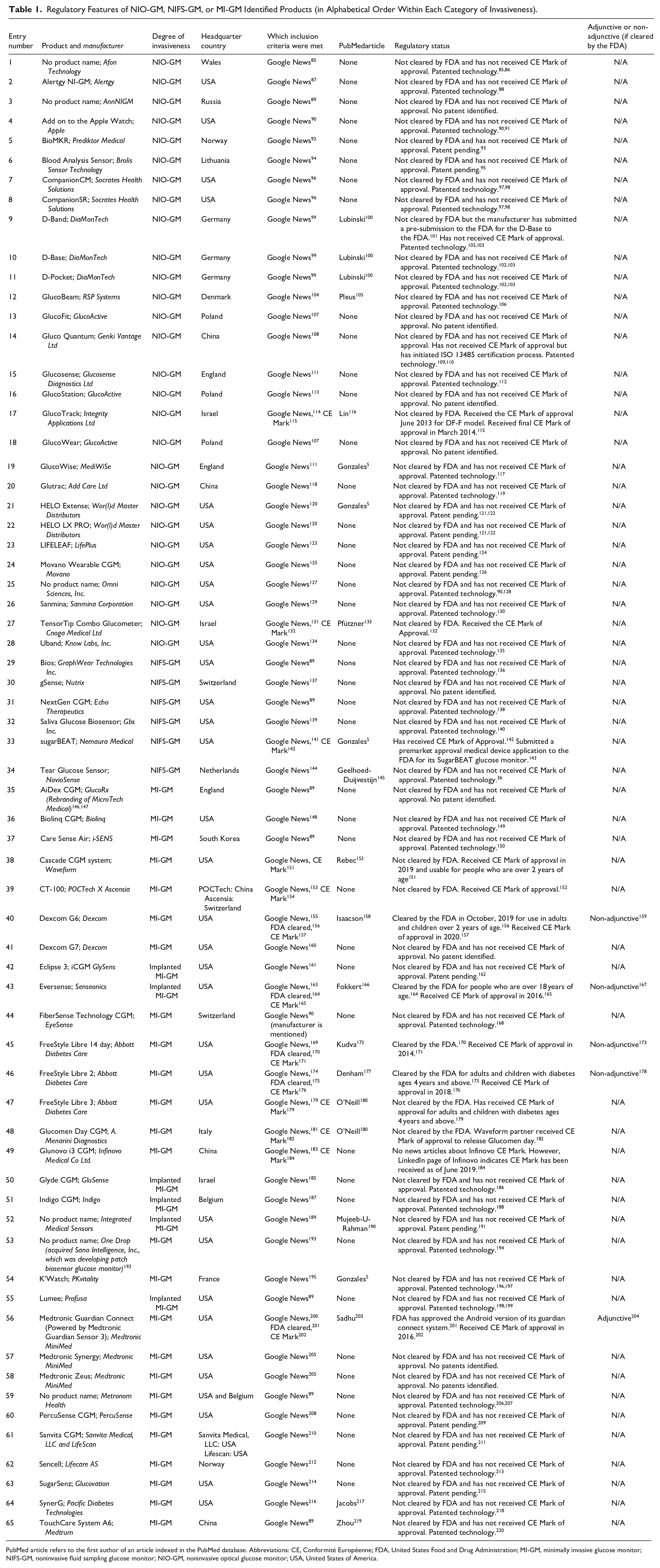
PubMed article refers to the first author of an article indexed in the PubMed database. Abbreviations: CE, Conformité Européenne; FDA, United States Food and Drug Administration; MI-GM, minimally invasive glucose monitor; NIFS-GM, noninvasive fluid sampling glucose monitor; NIO-GM, noninvasive optical glucose monitor; USA, United States of America.
Google News
Our second inclusion criterion for upgrading a candidate product to an identified product was a mention in a news article from a search on Google News of a candidate product. Such a search followed a termination process similar to the Google Patents search under the “Information Sources for Finding Candidate Products” section. The Google News search generated the most relevant articles first and progressively less relevant articles afterward. In the Google search box, we searched for the product name + “glucose monitor,” then clicked the “News” tab on Google to find news articles. Each page contained ten articles. For each candidate product we discovered from our four sources, we reviewed Google News pages for candidate articles that were published any time on or after January 1, 2016. If the first Google News page did not contain any articles that described the product in at least one paragraph, then we would continue searching the next four pages for an article containing at least one paragraph of information. If the product name + “glucose monitor” did not show any candidate articles, then we would also search the manufacturer’s name + “glucose monitor.” If a news article was found about a product, then we included the reference in Table 1 in a column entitled “Which Inclusion Criteria Were Met.” We included no more than one news article per product in this column as an example, even if more than one article was found on Google News. If no article was found about a product that lacked regulatory clearance, then we did not include this candidate product on our identified product list.
If an article found on Google News described the product we were searching for within a longer list of products, then we read through the article to see if any of the other products mentioned were already included in our list of candidate products. If any products were not included in the list of candidate products, then we added them to our candidate products. We then passed these incidentally discovered products (not originally found within our four original sources) through our inclusion criteria.
An Additional Scenario for Finding a Candidate Product and Upgrading it to an Identified Product
We upgraded one candidate product to an identified product using different inclusion criteria than the other identified products. An expert suggested the EyeSense (Basel, Switzerland) MI-GM called “FiberSense CGM,” so we then passed the product name + “glucose monitor” and manufacturer’s name + “glucose monitor” through Google News to determine if it fit one of our inclusion criteria. We found an article on Google News describing an NIFS-GM using tears being developed by EyeSense, 221 but we were unable to find specific mention of the MI-GM “FiberSense CGM.” The manufacturer’s website did not mention the NIFS-GM product using tears; however, the manufacturer’s website did mention the MI-GM “FiberSense CGM.” We decided to include this MI-GM “FiberSense CGM” in our identified products list because of the article we found in Google News mentioning the manufacturer.
Product Classification
We classified every product on our identified product list of glucose monitors, that did not use invasive sampling, into one of three categories: (1) noninvasive optical (measuring glucose with electromagnetic radiation from a blend of tissue sources including blood, ISF, and cells); (2) noninvasive fluid sampling (measuring a body fluid other than blood or urine extracted without the use of an implanted needle employing a technology requiring contact of the sensor with the fluid, including sweat, and ISF); and (3) minimally invasive (measuring ISF with a sensor inserted or implanted into the skin requiring contact of the sensor with the ISF). All three of these methods are distinct from traditional invasive blood glucose testing, which refers to measuring glucose with a needle that is inserted into the body to puncture a blood vessel so that blood can be in contact with a sensor.
Product Features
We considered 11 features for each NIO-GM, NIFS-GM, or MI-GM product, which were: (1) which inclusion criteria were met, (2) PubMed article, (3) regulatory status, (4) adjunctive/non-adjunctive status (if cleared by FDA), (5) mechanism of glucose sensing, (6) matrix of glucose sensing, (7) degree of accuracy, (8) interferences, (9) measurement cycle, that is, continuous or intermittent, (10) pricing model, and (11) size/shape. We divided these features into regulatory features (Feature 1-4), technological features (Features 5-8), and consumer features (Features 9-11). We then summarized the glucose sensing technologies and the products that belonged to each category of glucose sensing.
Finding information for product features
Information about product features was obtained from: (1) manufacturer websites, (2) articles found in a PubMed search for identified products, (3) patents found in a Google Patents search, (4) news stories found in a Google News search, (5) results from a Google search. Not every product was described on all five of these outlets (ie, a manufacturer’s website, PubMed, Google Patents, Google News, the first page of results on Google) and in many cases, no information was available about specific features of the listed glucose monitors. For products that have been cleared by the FDA or have a CE Mark, we asked an official from the manufacturer about what were their interfering substances. While we considered lag time between fluctuations in concentrations of blood glucose and NIO-GM, NIFS-GM, or MI-GM system glucose measurements, as well as the presence of skin irritation from contact by the sensor or adhesive around the sensor with the body, to be important technological features, there was not enough reliable published data on the lag times or the status of the skin problems of chronic users for a high percentage of the various products to include more than general statements for all of these products.
Manufacturer websites
To fill information about product features, we first looked through manufacturer websites. In order to find a manufacturer’s website, we performed a search on Google using the search terms: (product name OR manufacturer’s name) + “glucose monitor.” We would look through the first page of Google results to find the manufacturer’s website, which featured details about the product.
PubMed articles
When we discovered a product using the second (Google Patents), third (Startup Showcase), or fourth (experts in the field) initial information sources and the product met our inclusion criteria, we then conducted a PubMed search for articles mentioning identified products. We used the search terms product name + “glucose.” If we found no relevant product-related articles, then we conducted a second PubMed search of the manufacturer’s name + “glucose.” If a PubMed article was found about a product, then we included the reference in Table 1 in a column entitled “PubMed Article.” We included no more than one article per product in this column as an example, even if more than one article was found on PubMed. This search was not an inclusion search, but rather was conducted to find published references to be used if the product met at least one of our two inclusion criteria. Any PubMed article we found through both the information source search and this search may also have been used to provide information for columns in other tables.
Patents on Google Patents
We searched Google Patents by putting the manufacturer’s name under the “Assignee” and using the term “glucose monitor” in the search, which was a different type of search than we initially conducted using Google Patents as a source of candidate products (as described in the third paragraph of the “Methods” section). We used the information found on these patents to fill out information about product features.
News stories found in Google News
We used the same method, as was used under the “Google News” subsection of the “Inclusion Criteria” section, to find news stories in Google News to fill out details regarding product features. We summarized the most important product features in tables.
Results from a Google Search
We performed a Google Search with the (product name OR manufacturer’s name) + “glucose monitor” + “the product feature we are looking for (e.g., matrix)” on Google and looked through the first page of results for mention of a patent. We looked through each result to find details about the product feature.
Detailed Explanation for Specific Columns in Tables 1 and 2
Technological Features of NIO-GM, NIFS-GM, or MI-GM Identified Products (in Alphabetical Order Within Each Category of Invasiveness).
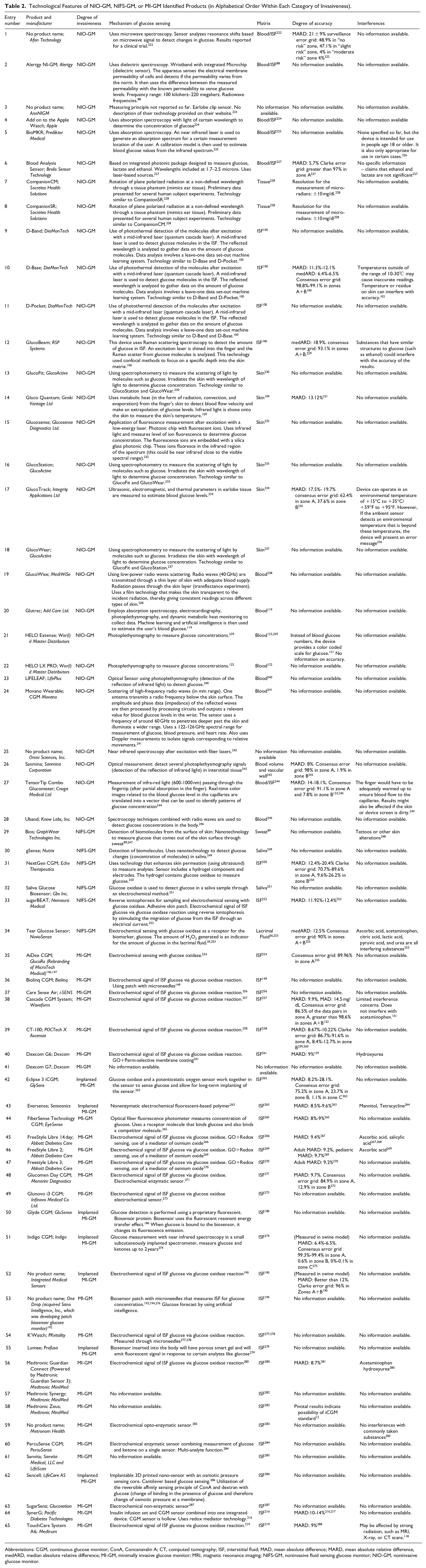
Abbreviations: CGM, continuous glucose monitor; ConA, Concanavalin A; CT, computed tomography; ISF, interstitial fluid; MAD, mean absolute difference; MARD, mean absolute relative difference, medARD, median absolute relative difference; MI-GM, minimally invasive glucose monitor; MRI, magnetic resonance imaging; NIFS-GM, noninvasive fluid sensing glucose monitor; NIO-GM, noninvasive glucose monitor.
Patents under the “Regulatory Status” column in Table 1
The Table 1 column on “Regulatory Status” specifies either clearance by the FDA, clearance by CE, or (if there was no clearance) whether we found a patent or any mention of having a patent. If a product was cleared by a regulatory agency, then we assumed that the product had a patent and we did not specifically search for one. If a product was not cleared by a regulatory agency, then we attempted to identify whether the product had a patent by using the five methods described under the “Finding Information for Product Features” section to identify a patent or mention of a patent. For the “First Page of Google Results” method, we used “patent” as our term for “the product feature we were looking for.” If we did not identify a patent through these five methods, then we stated “No patent identified.” If a manufacturer reported only patent(s) that could pertain to both a cleared product and a non-cleared product, then for the non-cleared product, we stated “No patent identified.”
“Mechanism of glucose sensing” column in Table 2
Just as with the other product features, the mechanism of glucose sensing presented in Table 2 was found using the five methods described under the “Finding Information for Product Features” section. Some manufacturers have developed both cleared and non-cleared products intended for bloodless glucose monitoring, but we were unable to find details about the mechanism of glucose sensing for their non-cleared products in manufacturer websites, articles on PubMed, articles on Google News, or results on the first page of a Google search. In this case, our source of information would theoretically be the patent if it was available. However, if the patents we reviewed did not call out the product by name, then it was unclear which product the patent was associated with. Therefore, we stated “No information available.”
Product Designs
We included examples of product design if the product website included an image and if a manufacturer’s official approved our request to publish an image of their product design. We reached out to every manufacturer that had a product design on their website if contact information was available. Representatives from manufacturers of the following products provided a separate image, that was not on the manufacturer’s website, for us to use for this article: Dexcom G6, Freestyle Libre 14-day, Freestyle Libre 2, Freestyle Libre 3, GlucoBeam, Gluco Quantum, LifeLeaf, SynerG, and UBand. Every other product image came from the product website.
Advantages and Disadvantages
From the manufacturer’s website and the first page of Google results (as per the search method defined in the “Overview”), we determined which identified product had FDA and/or CE clearance. For each cleared product, we evaluated its features and created a list of advantages and disadvantages based on our experience with glucose monitoring technologies and treating people with diabetes. Advantages and disadvantages were not presented for any product which has not received regulatory clearance, because that product might eventually contain different features than what we are currently able to determine.
Results
Summary of Identified Products
We listed 65 identified products based on three categories for the degrees of invasiveness of the sensor relative to the body: (1) NIO-GM, (2) NIFS-GM, and (3) MI-GM technology. The products included 28 NIO-GMs, 6 NIFS-GMs, and 31 MI-GMs. We recorded available information on 12 different features for each glucose monitoring product. We classified these identified products according to (1) headquarter country, (2) which inclusion criteria were met, (3) PubMed article, (4) regulatory status, (5) adjunctive/non-adjunctive status (if cleared by FDA), (6) mechanism of glucose sensing, (7) matrix, (8) degree of accuracy, (9) interferences, (10) measurement cycle, that is, continuous or intermittent, (11) pricing model, and (12) size/form factor. We divided these features of the 65 identified products along with their manufacturer and degree of invasiveness into (1) regulatory features (Feature 1-5, Table 1), (2) technological features (Features 6-9, Table 2), and (3) consumer features (Features 10-12, Table 3). We presented a summary of the underlying technologies used by each identified product in Table 4. We presented images of product designs for selected identified products in Table 5A (FDA cleared and/or CE marked) and Table 5B (not FDA cleared or CE marked). We presented advantages and disadvantages of regulatory cleared products in Table 6.
Consumer features of NIO-GM, NIFS-GM, or MI-GM Identified Products (in Alphabetical Order Within Each Category of Invasiveness).
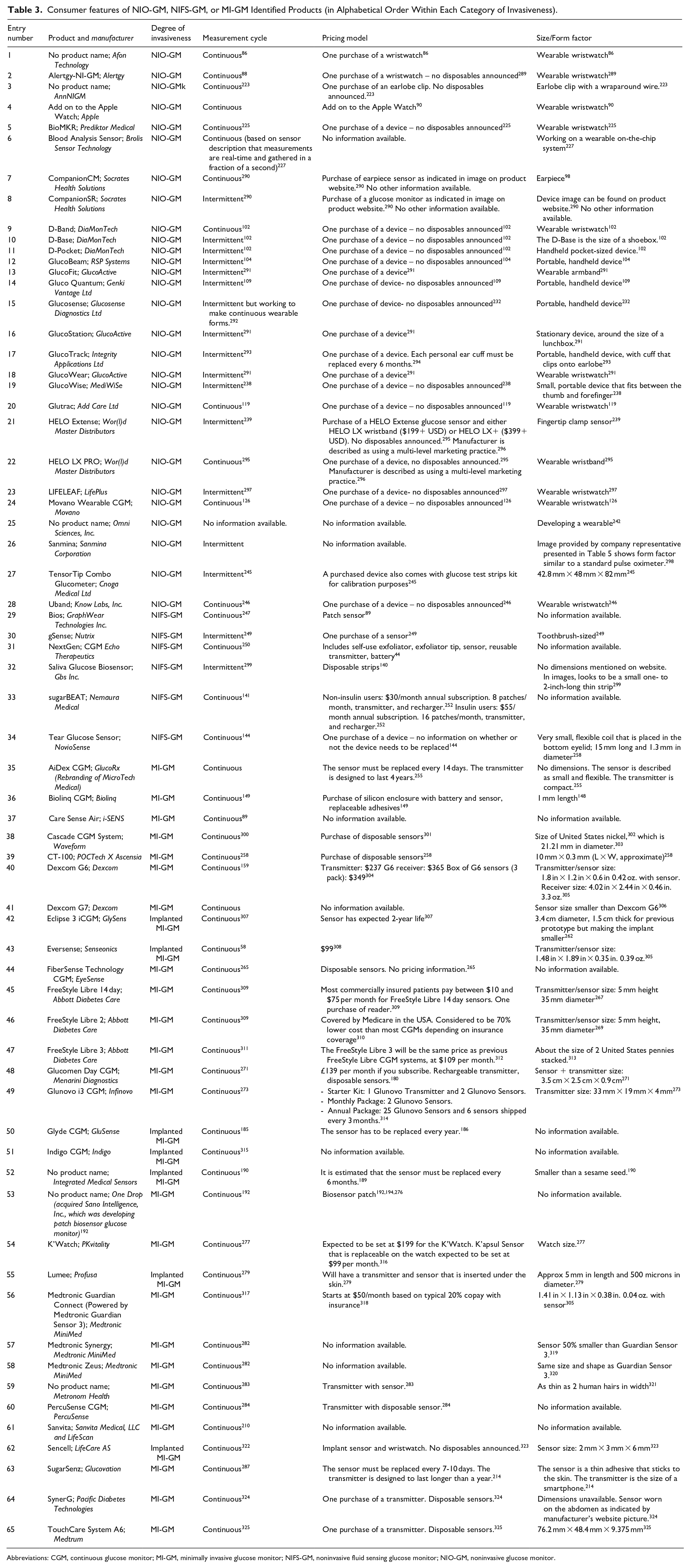
Abbreviations: CGM, continuous glucose monitor; MI-GM, minimally invasive glucose monitor; NIFS-GM, noninvasive fluid sensing glucose monitor; NIO-GM, noninvasive glucose monitor.
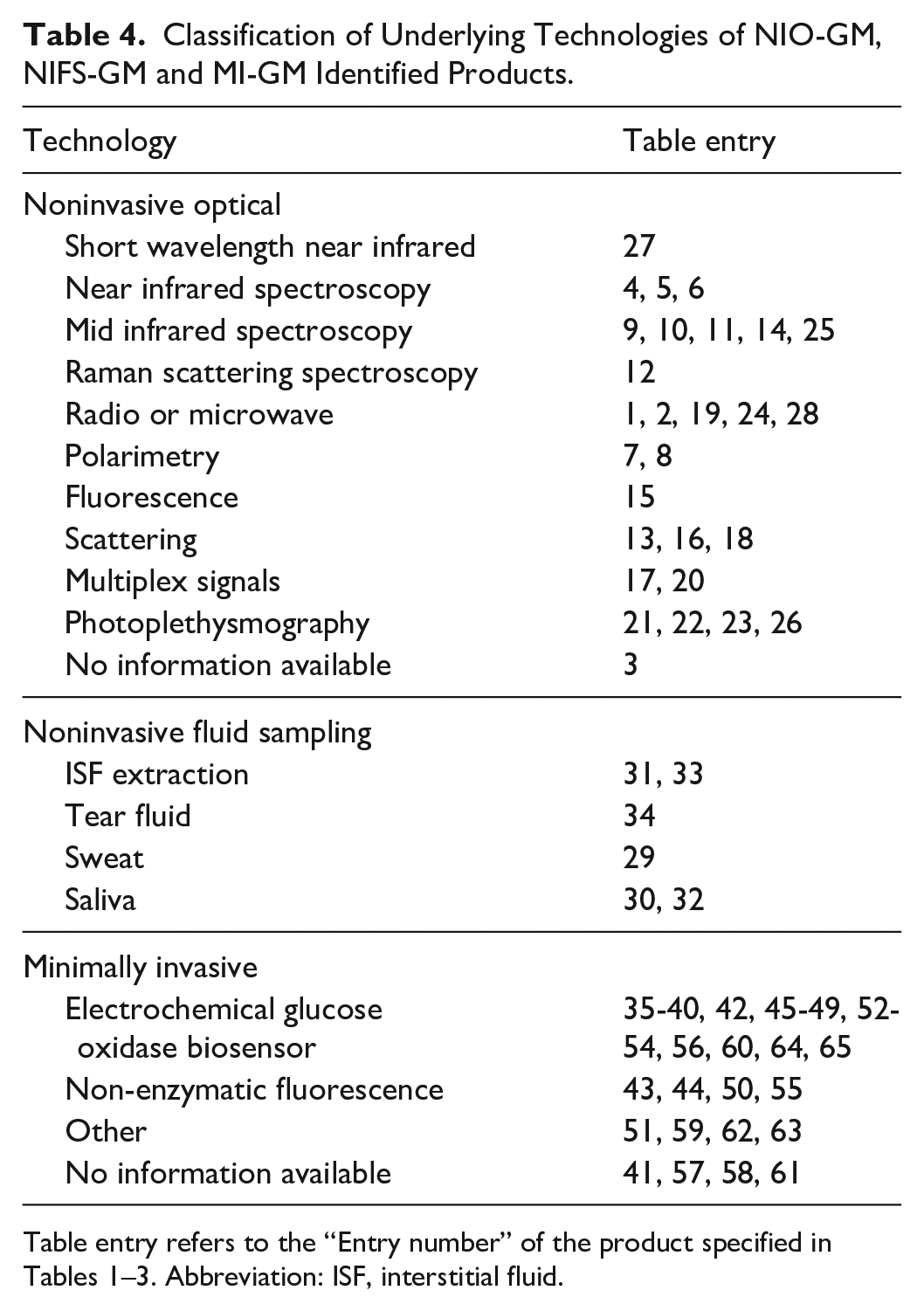
Classification of Underlying Technologies of NIO-GM, NIFS-GM and MI-GM Identified Products.
Designs of NIO-GM, NIFS-GM, and MI-GM Identified Products with Regulatory Clearance (in Alphabetical Order Within Each Category of Invasiveness).
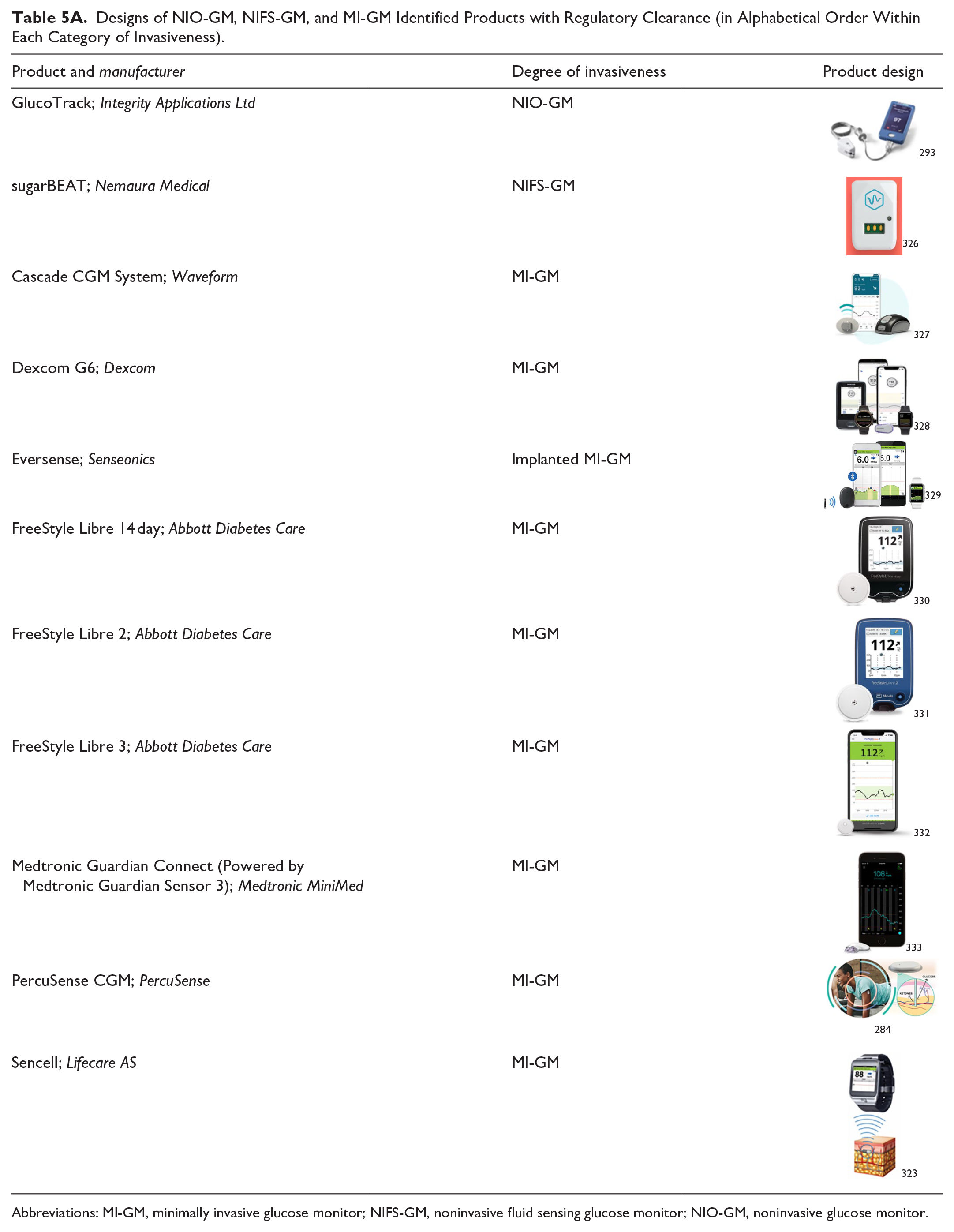
Abbreviations: MI-GM, minimally invasive glucose monitor; NIFS-GM, noninvasive fluid sensing glucose monitor; NIO-GM, noninvasive glucose monitor.
Designs of NIO-GM, NIFS-GM, and MI-GM Identified Products without Regulatory Clearance (in Alphabetical Order Within Each Category of Invasiveness).
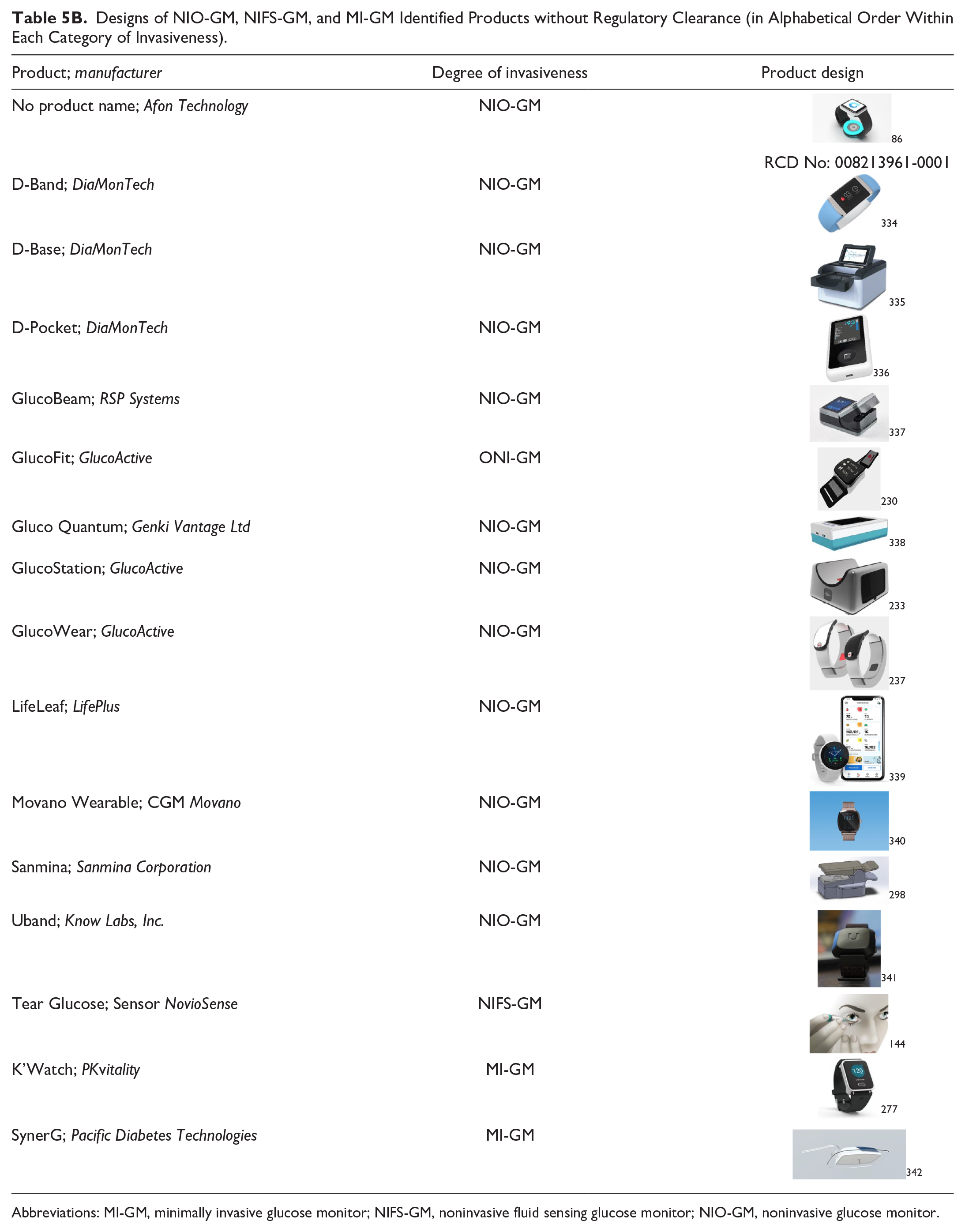
Abbreviations: MI-GM, minimally invasive glucose monitor; NIFS-GM, noninvasive fluid sensing glucose monitor; NIO-GM, noninvasive glucose monitor.
Advantages and Disadvantages of NIO-GM, NIFS-GM, or MI-GM Identified Products that were Cleared by the FDA or CE Marked (in Alphabetical Order Within Each Category of Invasiveness).
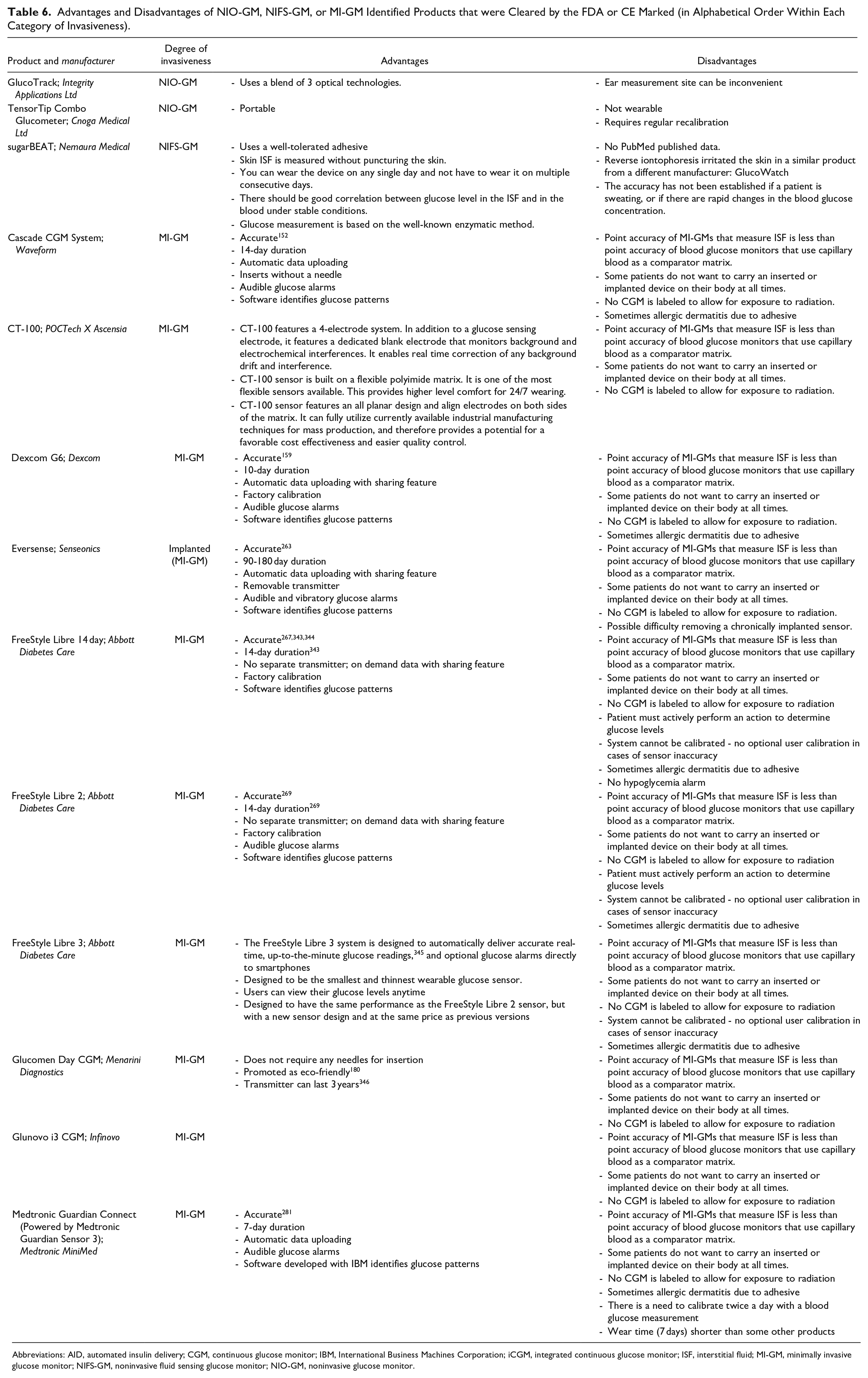
Abbreviations: AID, automated insulin delivery; CGM, continuous glucose monitor; IBM, International Business Machines Corporation; iCGM, integrated continuous glucose monitor; ISF, interstitial fluid; MI-GM, minimally invasive glucose monitor; NIFS-GM, noninvasive fluid sensing glucose monitor; NIO-GM, noninvasive glucose monitor.
Regulatory Features
Table 1 presents five regulatory features of the 65 identified products, including whether they received FDA clearance or a CE mark. Obtaining a CE mark requires proof of safety of the device, however, obtaining FDA clearance, requires proof of both the safety and efficacy of the device. 347 Although FDA clearance is more difficult to obtain, it is a more powerful certification. 348
Technological Features
Table 2 presents four technological features of the 65 identified products. The next four paragraphs discuss these features.
The first technological feature we considered is the mechanism of glucose sensing, which is the process through which the sensor detects and interacts with glucose. These processes were based on a variety of chemical and physical properties of glucose.
The second technological feature we considered is the matrix where sensing occurs. For each glucose monitor, the sensing of glucose is carried out within a specific matrix. The matrix can be ISF with MI-GMs, ISF, sweat, tears, or saliva with NIFS-GMs, or a combination of ISF, cells, and blood with NIO-GMs.
The third technological feature we considered is accuracy. The accuracy of glucose monitors that are not invasive is typically reported in one of two ways. First, accuracy can be reported as the percentage of data pairs of glucose monitor values compared to reference values falling within various accuracy ranges, such as within 5%, 10%, 15%, 20%, 30%, and 40%. Second, accuracy can be presented as the mean absolute relative difference (MARD) or median absolute relative difference (medARD) of the measured glucose value compared to the reference glucose value. The two values for normally distributed datasets are linked by an approximate conversion factor of 2.5. For example, if a glucose monitor has a MARD of 10% and a set of glucose values is distributed normally, then approximately 95% of its measurements will fall within +/−25% of the reference glucose value because 2.5 × 10% equals 25%. Clinical accuracy is typically reported using an error grid analysis which presents the risk to a user based on actions taken from a measured glucose value compared to the correct action that would have been based on a reference glucose value. Three widely used error grids for blood glucose have been proposed, and over time they have reflected increasingly more modern expectations of analytical accuracy, understanding of risks of hypoglycemia, and prescribing patterns for modern insulins. The most recent error grid and that which reflects input from the greatest number of diabetes clinicians (compared to earlier iterations of error grids known as the Clarke Error Grid 349 and the Parkes or Consensus Error Grid 350 ) is the Surveillance Error Grid. 351 Each grid presents data pairs in five zones of risk, although the Surveillance Error Grid is constructed so that it is possible to also generate more than five risk zones. Neither analytical accuracy data nor clinical accuracy data was available for every product because not every product has conducted clinical trials or published performance data.
The fourth technological feature we considered was the device’s possible interferences or interfering substances that might affect the glucose monitor’s accuracy. The accuracy of a glucose monitoring device might be affected by interferences or interfering substances, which are molecules that can interfere with sensing, often because of a similar structure as glucose. These substances are important to identify and characterize to ensure that users understand why or when their readings might be inaccurate.
Consumer Features
Table 3 presents three consumer features of the 65 identified products. The first consumer feature we considered is the measurement cycle, or whether the device makes automatic continuous or intermittent measurements. Any automatic measurement device must be engineered to be small enough to be a wearable device, but an intermittent measurement device does not necessarily need to be built small. The second consumer feature we considered is the pricing model. There are two main models that are used. In the first model, the user purchases the sensor and can make as many measurements as desired during the lifetime of the sensor. In the second model, the patient is charged for each measurement. Combinations of these two models are also possible. One device might require a single large payment for a device that could last for years, while another device might require periodic purchases of consumable parts or else payment of a toll charge each time the monitor is used. The third consumer feature we considered is the size/form factor of the product. A small sized sensor, compared to a large sized sensor, is usually more convenient and preferred by most users. However, a small minimally invasive implanted sensor could break off from its transmitter and be lost under the skin. A small, wearable noninvasive sensor with a streamlined form factor, compared to a large, bulky, free-standing noninvasive sensor, will usually be more costly because component miniaturization and assembly are expensive.
Underlying Glucose Sensing Technologies used in NIO-GM, NIFS-GM, or MI-GM Identified Products
Table 4 classifies the underlying glucose sensing technologies of the 65 identified products. Under NIO-GM, the glucose sensing technologies included: short wavelength near infrared, near infrared spectroscopy, mid infrared spectroscopy, Raman spectroscopy, radio or microwave devices, polarimetry, fluorescence, scattering, multiplex signals, and photoplethysmography. A few products were NIO-GM, but their exact glucose sensing technologies were not specified because this information was not available. Under NIFS-GM, the glucose sensing technologies included: saliva, tear fluid, sweat, and ISF extraction. Finally, under MI-GM, the glucose sensing technologies included: electrochemical glucose oxidase biosensor and non-enzymatic fluorescence sensors. Some MI-GM did not belong to either of these technologies, and some do not have publicly available information on what technology they use.
Images of Product Designs of Selected Identified Products
Table 5 presents images of product designs of identified products. We included images only if we were able to receive permission from manufacturer representatives. Images are not to scale relative to each other. Table 5A is for products that are FDA cleared and/or CE marked, and Table 5B is for products that are neither FDA cleared nor CE marked.
Advantages and Disadvantages of Identified Products
Table 6 presents advantages and disadvantages of the identified products that were cleared by the FDA or CE marked. The features that we present in Table 6 for specific products are important highlights of each product, rather than a comprehensive list of advantages and disadvantages. In general, there are advantages and disadvantages for each of the three categories of glucose monitors employing bloodless sampling. All three categories of glucose monitors are less accurate than IGMs. (1) As a class, NIO-GM products are painless and do not generate biological waste, but they are less accurate than NIFS-GM, MI-GM, and IGM products. (2) As a class, NIFS-GM products require the sensor to be in direct contact with a body fluid that is not blood, and these systems generally produce less pain and less biological waste than IGMs. However, there can be a significant lag-time in their measurements, thereby rendering them less accurate than IGMs or MI-GMs. (3) As a class, MI-GM products have been studied the most of the three types of bloodless glucose monitors. More products in this category, compared to the other two categories, have received regulatory clearance. Currently, many MI-GMs are more accurate, more robust, and more conveniently calibrated than most NIO-GM and NIFS-GM products. The Abbott (Chicago, Illinois) and Dexcom (San Diego, California) MI-GMs are factory calibrated. All MI-GMs, however, require insertion or implantation of the glucose sensor and must be worn, which in rare cases creates an unpleasant sense of being tethered to a machine. 352 Many MI-GMs and some NIO-GMs and NIFS-GMs require adhesives for stabilization and allergic dermatitis has been reported with some adhesives. 73 The convenience, performance, and connectivity of MI-GMs have been advancing rapidly over the past few years and product manufacturers have predicted further advances in these areas. 353 The worldwide market for CGMs was valued at $1.8 billion in 2019 and with a projected compound annual growth rate of 22%, the market is expected to reach $8.8 billion by 2027. 354 This market has likely already surpassed four million CGM users. 355
Discussion
Evolution of Bloodless Glucose Monitoring Products
In the past, one NIFS-GM 356 and one NIO-GM 357 were introduced to the market but were eventually discontinued, and two NIO-GMs358,359 were cleared but never introduced to the market.356-359 These products were all highly anticipated before their release but failed to live up to expectations. A particular problem was that such devices were touted in the lay press, raising expectations among people with diabetes and their families. Unfortunately, their performance repeatedly failed to meet these expectations. The NIFS-GM GlucoWatch Biographer from Cygnus Inc. was cleared by the FDA, marketed, and then discontinued. 360 Three NIO-GM products were cleared with a CE Mark and then discontinued either soon after entering the market (Pendra Device from Pendragon Medical, Zürich, Switzerland), or without entering the market (NBM-200G from OrSense, Tel Aviv-Yafo, Israel and Optical Glucose Monitor from C8 MediSensors, San Jose, California). Two NIO-GM products that were cleared with a CE mark are currently on the market. They are the GlucoTrack (Integrity Applications Ltd, Ashdod, Israel) and TensorTip Combo Glucometer (Cnoga Medical Ltd, Caesarea, Israel).
The original GlucoWatch Biographer 41 was an NIFS-GM device. This device was cleared by the FDA in 2001 and marketed. 40 An updated version (the GlucoWatch G2 Biographer 42 ) was also cleared in 2002 for use by individuals under 18 years of age. 356 Because these devices used reverse iontophoresis, a low-level current was passed through the skin as part of the glucose sensing method. These products were discontinued after users complained of a burning sensation while using the device. 360
The Pendra Device, which measured the change in the frequency-dependent resistance (impedance spectroscopy) in the interstitial flow when the glucose concentration changed, 361 was initially cleared with a CE Mark of Approval in 2003. However, this device was discontinued because it had readings that were considered dangerously inaccurate and it was incompatible with 30% of people considered for using the device. In 2005, Pendragon Medical went out of business. 357
The NBM-200G, which used occlusion spectroscopy, received the CE Mark of Approval in 2007 358 but was discontinued by OrSense for unspecified reasons. The C8 Optical Glucose Monitor received the CE Mark of Approval in 2012, 359 but is no longer on the market officially because of a lack of funding. 362 Other NIO-GM products have been announced to be in development and then have subsequently disappeared from the public eye. These products include the ALIRA Infrared Biosensing sensor (Princeton University, Princeton, New Jersey), Glucoband (Calisto Medical, Plano, Texas), and a Glucose-Sensing Contact Lens (Novartis, Basel, Switzerland working with Verily, South San Francisco, California).
These shortcomings are discussed to emphasize the importance of recognizing, addressing, and avoiding past mistakes. These previous failures to create a usable product also highlight the barriers that must be overcome before a viable NIO-GM can be widely distributed, including (1) large device size, (2) potential skin reactions, (3) poor accuracy, (4) high power requirements, (5) interfering substances, and (6) the lack of a clear regulatory pathway. Although the majority of the NIO-GM products discovered in the search presented in the article are neither cleared nor publicly available, these products have potential, but one must guard against being overly optimistic for the sake of potential users.
Trends in Technology – A Path for Better Products
Many technologies are now being pursued to achieve the objective of noninvasive glucose measurements in people with diabetes. At this time, few NIO-GM or NIFS-GM systems are sufficiently mature for commercialization. The MI-GM market, however, is strong in many countries and growing worldwide. Many of these technologies not using IGM that are in development appear to have poor accuracy. Still, noninvasive approaches are attracting more attention by both researchers and investors owing to the tremendous need for managing diabetes optimally in an ever-growing number of patients and also because of commercial applications beyond medicine.
The commercial potential differs for the products identified in this article. The NIO-GMs are becoming more of a priority for medical device researchers. NIFS-GM sensors seem to be the least likely to satisfy user needs because of the requirement to harvest a representative fluid. Such approaches are prone to issues related to skin irritation as well as to the complexities of collecting, handling, and disposing of a representative clinical fluid. These issues render the noninvasive fluid sampling approach inconvenient in comparison to both the noninvasive optical and minimally invasive approaches. The former eliminates fluid handling and the later provides continuous measurements. The commercial attractiveness of NIO-GM technologies is driven by the potential to achieve either continuous glucose measurements or repeated spot-measurements, both with minimal user interactions. Commercial success has already been realized for MI-GM technologies and will likely grow as future innovations lead to longer periods of operation and the quantitation of multiple analytes.
Success of NIO-GM, NIFS-GM, and MI-GM devices has started to decrease the market size of invasive blood glucose measurement systems considerably. Just as urine glucose testing was replaced by blood glucose testing, eventually these three bloodless technologies will replace blood glucose monitoring. This trend will continue within the next 10-20 years as new sensing strategies that do not require blood become increasingly accurate, practical, and affordable. Today many of the leading blood glucose monitor manufacturers are developing NIO-GM, NIFS-GM, or MI-GM technologies or partnering with companies with these types of products. The need for invasive blood glucose measurements will also decrease as factory-calibrated MI-GM devices become more accepted by people with type 1 diabetes mellitus (T1DM) or type 2 diabetes mellitus (T2DM). A CE marked MI-GM was recently introduced for athletes without diabetes. 363 As the accuracy and reliability of factory-calibrated MI-GM devices improve, the need to check an IGM reading will decrease.
Commercial success of any CGM system, regardless of whether it is noninvasive or minimally invasive, must recognize the demands placed on the user. Proper training is paramount for CGM users, particularly as it relates to the continuous information provided by these devices. 364 The users must gain confidence in viewing the large amount of data, interpreting the recorded glucose profiles, and using the data to guide therapeutic decisions. Trained users value the added analytical information provided by CGM and find that it benefits their therapy. 365 However, improved data management software will be helpful for the users who are overwhelmed by the data output.
Noninvasive Technology for Diagnosing Diabetes
The current standard for diagnosing diabetes specifies invasive blood sampling, followed by measuring either plasma glucose concentration or hemoglobin A1c concentration. 366 While several noninvasive and minimally invasive methods have been developed for monitoring glucose levels, as outlined throughout this paper, there have been few attempts to validate the use of such devices for making a diagnosis of diabetes. There is, however, an incipient technology using photoplethysmography that is intended to make this diagnosis.
Photoplethysmography measures the interaction of light with the vascular bed to determine the changes in blood volume with each heartbeat. 367 This technique is currently used in four NIO-GM products under development, including Wor(l)d Global Network’s (Miami, Florida) HELO Extense 239 and HELO LX Pro, 122 LifePlus’ (San Jose, California) LifeLeaf, 240 and Sanmina’s (San Jose, California) NIO-GM. 243 Furthermore, Avram et al. have reported using a smartphone-based photoplethysmography technology with a deep neural network scoring system to prospectively evaluate people with diabetes, by correlating the score with plasma glucose levels. 368 This study’s results suggest that this method could be potentially useful as a digital biomarker for diabetes diagnosis, 368 but further work is needed. 369
Miniaturization and Nanotechnology
Miniaturization is critical for future applications and user adoption of the glucose sensing technologies under development worldwide. Wearable devices demand small sensing packages that can be worn for prolonged periods with minimal user discomfort. Miniaturization will also facilitate comfortable systems that combine the sensing element with an insulin infusion system, perhaps in a single package, for novel AID systems. 82 As the sensing technology advances to the stage of reliable, calibration-free operation for extended periods, miniature solid-state sensing systems will be necessary to serve as the glucose sensing element of an implantable autonomous AID systems.
Nanotechnology is a materials approach with the potential to facilitate device miniaturization while enhancing sensor performance. 370 Nanostructures are capable of unique properties not available in bulk materials. The surface area to volume ratio is large for nano-dimensional materials which offers unique physical and chemical effects, such as novel interfacial phenomena, altered reactivity, and charge carrier and quantum mechanical effects. 371 Such nanoscale properties can advance miniaturization and sensor operation through higher surface areas, thereby yielding larger sensitivity, quicker responses, and higher catalytic loadings. These enhancements promise superior overall analytical performance.
Nanofabrication techniques can also generate glucose sensors with small dimensions. 372 Such small dimensions can facilitate implantation or injection of glucose sensing “tattoos.”373,374 Nanofabricated devices could potentially diminish or avoid troubling foreign body responses, resulting in simpler calibration processes and longer operational lifetimes. Solid-state photonic systems hold promise for miniaturizing spectroscopic sensors for application as NIO-GM devices. The system under development by Brolis (Vilnius, Lithuania), for example, uses a novel integrated photonic package for solid-state measurements of multiple analytes, including glucose, lactate, and ethanol. Finally, micro- and nano-electronic technologies offer the possibility of low-cost mass production, which can drive down user’s costs and expand adoption.
Overcoming Technical Barriers to Widespread Adoption
Adoption of any bloodless glucose sensing technology will depend on a host of technical and nontechnical issues. First, and foremost, the technology must be accurate and robust in operation while adding value to the quality of life for the user. Advances in engineering, physics, clinical chemistry, and medicine will also be required to overcome the technical barriers that impede widespread adoption of NIO-GM, NIFS-GM, and MI-GM products.
From the engineering perspective, more sensitive sensing technologies are needed, particularly for ex-vivo NIO-GM and NIFS-GM devices. The ability to identify and detect a robust selective signature for glucose is paramount along with instrumentation able to collect such a signature reliably under non-laboratory conditions. Measurement accuracy in the low glucose range is critical to achieve tight glycemic control while avoiding potentially dangerous hypoglycemic episodes.
From the physics perspective, algorithms are needed to convert raw sensor signals into correct glucose concentration values. For NIO-GM technologies, a principal challenge is to effectively filter out variations in the measured signal, both the noise of the instrumentation as well as uncontrolled changes in the background matrix against which the glucose signature is determined. Success calls for understanding the source of such background variances and developing multivariate data analysis methods to overcome the adverse impact of the background variance. For NIFS-GM devices, the major barrier is to find a correlation between the concentration of glucose in blood and the sampled fluid. For MI-GM systems, reliable analytical measurements are needed for periods longer than 14 days.
From the clinical chemistry perspective, a better understanding is needed about how to assure the accuracy of bloodless measurements by establishing traceability of their readings to a reference standard. 375 This challenge is currently unresolved because of the difficulty of obtaining comparator ISF samples for testing against blood. Also, the controversy about whether capillary blood or venous blood is the best comparator matrix for ISF measurements must be resolved. 375 Overall, a convenient and reliable method for collecting representative samples of ISF would have a large positive impact on the advancements of these and future NIO-GM, NIFS-GM, and MI-GM products.
From the medicine perspective, physicians need a better understanding of the clinical values of glucose measurements in fluids other than blood, that is, how to adjust the insulin dose of a patient in case of exercise, is the glucose signal from blood the more reliable signal or that from ISF? The utility of ISF glucose concentrations must be established and might actually be preferable over blood glucose concentrations for treatment decisions because the glucose concentration in ISF compared to that in blood might more closely track the glucose concentration in the brain. 54 Others have suggested that ISF glucose concentrations can be superior to blood glucose concentrations during periods of rapidly fluctuating glucose concentrations or hypoglycemia. Another clinical dimension for physicians to determine is the level of accuracy needed for various types of patients, such as those with: T1DM on multiple dose insulin therapy, T1DM on sensor augmented pump therapy, T1DM on AID therapy, T2DM using insulin, T2DM using other diabetes drugs known to cause hypoglycemia, T2DM using diabetes drugs not known to cause hypoglycemia, and T2DM using only diet and exercise for control.
Overcoming Non-Technical Barriers to Widespread Adoption
Overcoming nontechnical barriers will be required for the widespread adoption of bloodless glucose sensing technologies:
(1) The cost of these products must be reduced to make adoption possible for large populations, especially in resource-poor regions of the world.
(2) The interface between the user and instrumentation and the way in which the user interacts with the instrumentation must translate into an easily deployed wearable device that does not cause embarrassment or inconvenience, particularly in social settings.
(3) Patients must accept the concept of wearing a continuously functioning device without concern that they are losing their humanity by being dependent on a mechanical device. 376
(4) A motivational factor for many potential users can be an understanding of the present and future environmental risks caused by the large amounts of medical and plastic waste generated by today’s invasive blood glucose monitoring technologies.
The overall strategy is to provide a bloodless glucose sensing technology that simultaneously reduces waste, increases convenience, and saves money.
Regulatory agencies guided by solid clinical evidence will play an important role in determining the level of accuracy that each of these new bloodless devices must provide to achieve non-adjunctive status. This is the level of clearance whereby manufacturers can indicate on the product label that their glucose monitoring product can be used to make a treatment decision without the need for an accompanying blood glucose reading as a confirmatory test. Accuracy and robustness of the sensing product are required for non-adjunctive clearance, which will undoubtedly boost the appeal of these products. As shown in Table 3, four MI-GM products have a non-adjunctive indication per the FDA and it is likely that many future bloodless products will demonstrate adequate accuracy to also gain this distinction.
Conclusions
Commercial products for bloodless monitoring of glucose were identified and reviewed. These products are based on three categories of emerging technologies: (1) NIO-GM, (2) NIFS-GM, and (3) MI-GM. Many of these products are based on spectroscopic, electrochemical, and affinity sensing strategies for quantifying glucose molecules in ISF. A multiple database search was used to identify a total of 65 bloodless glucose monitoring products, of which 13 have received regulatory clearance and are currently on the market. The remaining products are under development. Further refinement of these technologies is anticipated over the upcoming decade to enhance both analytical performance and adoption of these products by people with diabetes. Future developments in bloodless glucose monitoring will require technical and nontechnical advances related to (1) superior analytical performance, (2) robust algorithm development, (3) traceability testing for glucose-containing matrices besides blood, such as ISF, and (4) appreciation of the clinical value of ISF glucose measurements. Bloodless glucose monitoring products, such as the ones discussed in this article, are expected to become key components of novel wearable digital health tools for monitoring glucose concentrations in the diabetes market and the fitness market.
Addendum
In May 2021, after this article was in press, Rockley Photonics disclosed a program to develop a chip size laser-based technology suitable for implementation in a smartwatch format. Health monitoring is its principal application and Apple is featured as a major partner in the development of this technology. Rockley stated that Apple is the largest of six customers with which it has entered into contracts with or engaged with, developing health and wellness devices. These companies are developing smartwatches and medical devices for biomarker detection. 377 Rockley’s sensors are designed to quantify glucose, alcohol, and oxygen levels non-invasively with infrared spectroscopy. Rockley will deliver its chipsets in 2022. 378
Footnotes
Acknowledgements
We thank Annamarie Sucher-Jones for her expert editorial assistance. We thank Guido Freckmann, MD, Avner Gal, MBA, MSCEE, MSc, H. Michael Heise, PhD, Jeffrey La Belle, PhD, MS, Jeffrey Joseph, DO, John Pickup, MD, PhD, and Mark Prausnitz, PhD for naming candidate products and manufacturers.
Abbreviations
AID, Automated insulin delivery; AMBG, assisted monitoring of blood glucose; CE, Conformité Européenne; CGM, continuous glucose monitor; ConA, Concanavalin A; DF, fluorescently labeled dextran; FDA, United States Food and Drug Administration; GBP, glucose-binding protein; IGM, invasive glucose monitor; ISF, interstitial fluid; MARD, mean absolute relative difference; medARD, median absolute relative difference; MI-GM, minimally invasive glucose monitor; NIFS-GM, noninvasive fluid sampling glucose monitor; NIO-GM, noninvasive optical glucose monitor; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses; SMBG, self-monitoring of blood glucose; SNR, signal-to-noise ratio; USA, United States of America
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TS, JYZ, and BNV have nothing to disclose. AT was Scientific Manager at Medtronic until April 2020. Currently he has nothing to disclose. MAA discloses a consulting arrangement with LifePlus, Inc. LH is a partner of Profil Institut für Stoffwechselforschung in Neuss, Germany. He is a member of advisory boards for Roche Diagnostics, Zense, and Medtronic. He is also on the Board of Directors for Lifecare. DCK is a consultant for EOFlow, Fractyl, Lifecare, Novo, Roche Diagnostics, Samsung, and Thirdwayv.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Foundation for Innovative New Diagnostics through support from the Swiss Agency for Development and Cooperation (SDC).
