Abstract
Background:
Glucommander is an eGlycemic management system (eGMS) for intravenous (IV) and subcutaneous (SQ) insulin therapy in hospitalized patients. The purpose of this study was to evaluate the efficacy and safety of Glucommander compared to previously utilized nomograms in the community hospital setting.
Methods:
This study was a retrospective, single-center cohort study comparing measures of efficacy and safety of IV and SQ insulin therapy via eGMS versus nomogram-driven IV insulin therapy followed by provider-ordered basal-bolus SQ insulin. The primary efficacy endpoint was percent of blood glucose (BG) readings per patient in target glycemic range. Safety objectives were percent of hyperglycemic events, hypoglycemic events, and severe hypoglycemic events after achieving target blood glucose range, and mean number of each event per patient.
Results:
The percentage of BG readings in range was significantly higher for eGMS patients (n = 110) than comparison cohort patients (n = 108, 84.6% vs 76.8%, P < .001). Hyperglycemic events occurred for significantly fewer patients in the eGMS cohort relative to the comparison cohort (81.8% vs 92.6%, P = .03). Overall, there was no significant difference between cohorts in rate of hypoglycemic events, but hypoglycemic events while on IV insulin occurred in a significantly higher percentage of eGMS cohort patients than comparison cohort patients (30.9% vs 15.7%, P < .01). There were no significant differences in incidence of severe hypoglycemic events.
Conclusions:
Our study found that Glucommander maintained a higher percentage of BG readings in target BG range per patient compared to previously utilized nomograms. This result was driven by an improvement in hyperglycemia, but not hypoglycemia.
Introduction
Patients admitted to community hospitals may need intravenous (IV) insulin therapy for a variety of conditions, including diabetic ketoacidosis (DKA), non-DKA hyperglycemia, or postoperative glucose management for cardiothoracic (CT) surgery. Glucommander (Glytec, Greenville, SC) is a computer-based, nurse-driven algorithm for the management of IV and subcutaneous (SQ) insulin therapy for hospitalized patients. 1 This eGlycemic management system (eGMS) directs nurses and providers on insulin dose adjustments based on rate of change in blood glucose (BG) and when to check next due BG for patients on IV or SQ insulin. It has previously been studied separately for the management of IV insulin in CT surgery patients, IV insulin in DKA patients, and SQ basal-bolus insulin regimens in hospitalized patients. These prior studies have shown statistically significant improvement in glycemic control in hospitalized patients compared to previous nomograms and provider-driven insulin regimens.2-4
Glucommander for IV and SQ insulin management was piloted in two units in our 369-bed community hospital. The two pilot units were the critical care unit and the telemetry unit, where patients are managed after CT surgery or treated for DKA or non-DKA hyperglycemia. The launch date for the eGMS program on the pilot units was February 26, 2019. Prior to implementation on these units, most patients requiring IV insulin were managed using the Lien-Spratt nomogram (or a modified nomogram for CT surgery patients called the Bell-Bass nomogram) 5 ; these nomograms are still utilized for the care of patients in the emergency department and all floors other than the two pilot units at our institution.
Previous studies have examined the use of eGMS for specific indications or patient populations such as critically ill patients or patients with DKA, but this study evaluated its use for multiple indications as utilized in one hospital. The purpose of this study is to evaluate the efficacy and safety of Glucommander compared to the previously utilized nomograms in the community hospital setting.
Methods
This study was a retrospective, single-center cohort study comparing measures of efficacy and safety of IV and SQ insulin therapy via eGMS versus nomogram-driven IV insulin therapy followed by provider-managed basal-bolus SQ insulin.
Patients were selected for inclusion in the eGMS cohort based on the following criteria: age 18 and older; admitted between March 1, 2019 and August 31, 2019; treated on one of the two pilot floors; and received IV insulin followed immediately by SQ insulin through the eGMS.
Patients were selected for inclusion in the non-eGMS comparison cohort based on the following criteria: age 18 and older; admitted between March 1, 2018 and August 31, 2018; admitted initially to one of the two floors that became pilot floors (but could transfer to another floor during their hospital course); and received IV insulin drip according to previously utilized nomogram followed immediately by a provider-managed SQ insulin regimen. This admission time frame was chosen to capture a similar patient population in the year prior to program implementation.
Patients were excluded from the study for either group if: (1) they were given IV insulin but not immediately transitioned to a SQ insulin regimen when IV insulin was discontinued; (2) they were ordered to receive IV insulin but never received the ordered medication. The intent of the study is to determine the efficacy and safety of the eGMS when used exclusively from start of therapy with IV insulin through the transition to SQ insulin. Therefore, in the eGMS cohort only, patients were excluded if: (1) they were given IV insulin using the eGMS program but were then transitioned to SQ insulin outside of the eGMS program (i.e., provider-managed SQ regimen); (2) they received only one dose of SQ insulin via the eGMS program and were then managed outside of the system. Due to limitations of the data collection system used, patients included in either group may have received one-time insulin doses ordered by providers outside of intended nomogram/management system.
The primary efficacy endpoint was percent of BG readings per patient in target glycemic range. Only point-of-care (POC) BG readings were included, as the eGMS utilizes POC BG readings for therapy management. BG readings were done by nurses at intervals directed either by the eGMS or when indicated by the Lien-Spratt nomogram. The target range for CT surgery patients was 70-150 mg/dL and the target range for non-CT surgery patients (DKA or hyperglycemia patients) was 70-200 mg/dL. BG readings for both groups were recorded only after the patient had initially reached the target glycemic range. For the eGMS cohort, any BG readings in the target glycemic range before initiation of the eGMS were not included and data collection stopped at the time the eGMS program was discontinued.
Time to reach target BG range was not assessed as an efficacy endpoint because the eGMS was not used in the emergency department during the study period. Because of this, patients were started on IV insulin using the Lien-Spratt nomogram in the emergency department and then transitioned to the eGMS once they were admitted to their floor unit. Thus, the time to target BG would not be reflective of the eGMS alone.
The safety objectives assessed in this study were the percent of patients experiencing hyperglycemic events, hypoglycemic events, or severe hypoglycemic events. Hyperglycemic events were defined as BG readings >150 mg/dL for CT surgery patients or >200 mg/dL for DKA or hyperglycemia patients after reaching target glycemic range. Hypoglycemic events were defined as BG readings between 50 and 69 mg/dL. Severe hypoglycemic events were defined as BG readings <50 mg/dL. Mean number of each event type per patient was also assessed. For patients with hyperglycemic events, rate of rebound hyperglycemia was also assessed, with rebound hyperglycemia defined as a hyperglycemic event preceded within one hour by a treated hypoglycemic event.
This retrospective study was approved by the Duke University Health System institutional review board.
Statistical Analysis
Demographic and baseline characteristics were compared using chi-square/Fisher’s exact tests or independent samples t tests. Efficacy endpoints were compared between cohorts using independent samples t tests and safety endpoints were examined using Fisher’s exact tests. Analyses were conducted using the full sample, and a second set of analyses was then conducted wherein patients were grouped according to indication for IV insulin to determine if that impacted the overall results. A priori power calculations were performed with alpha of 0.05 and power of 0.80. A projected sample size of 96 patients per group was needed to detect a moderate effect size between groups. A P value of <.05 was considered significant. Statistical analysis was performed using IBM SPSS version 26.
Results
A total of 460 patients were identified for inclusion using DEDUCE, a web-based clinical research query tool: 145 for the eGMS cohort and 315 for the comparison cohort. Thirty-five patients were excluded from the eGMS cohort and 207 patients were excluded from the comparison cohort, as shown in Figure 1. The most common reasons for exclusion from the eGMS cohort were issues related to being managed with one module (either IV or SQ) but not both—as deemed necessary in the inclusion criteria. Most excluded patients in the comparison cohort did not have IV insulin ordered during a qualifying admission, meaning that the patients were treated with IV insulin outside of the planned study time frame.
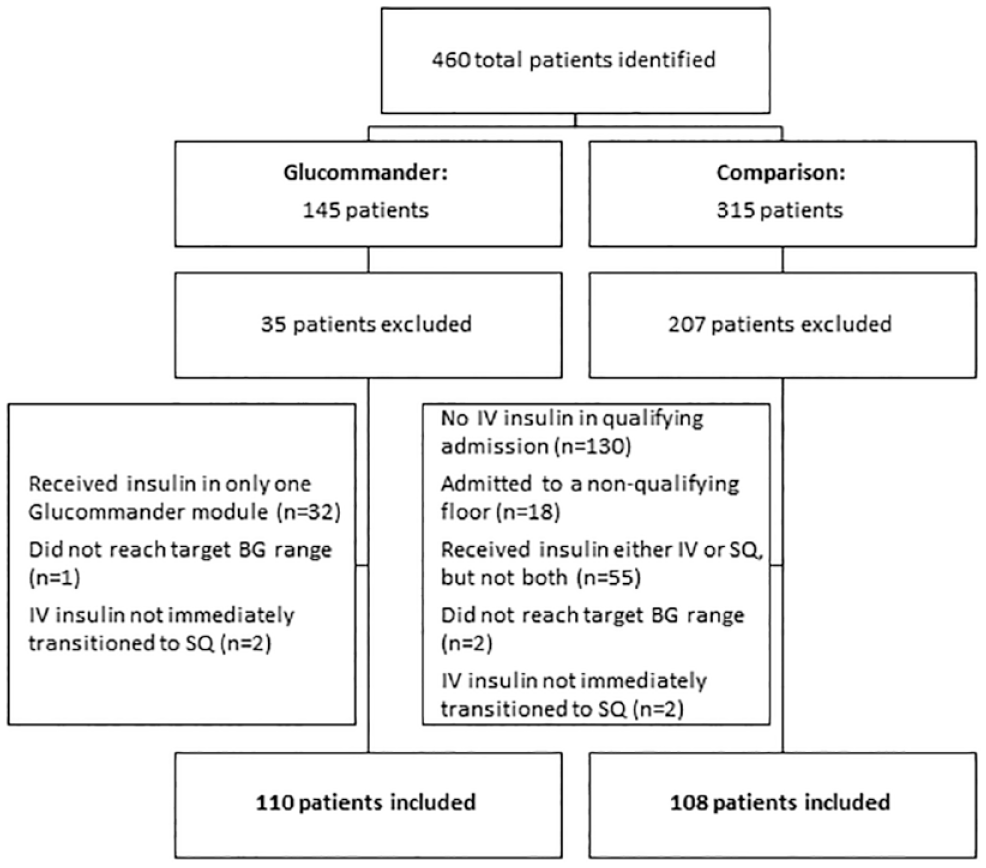
Patient enrollment.
Patient Characteristics
A total of 218 patients were included in the analyses: 110 in the eGMS cohort and 108 in the comparison cohort. Demographics are displayed in Table 1. Cohorts were similar in age, gender, and weight. There was a significantly higher percentage of patients with a history of diabetes in the comparison cohort than in the eGMS cohort (88.9% vs 76.4%, P = .02). The indication for IV insulin significantly differed between cohorts, with more patients in the eGMS cohort having CT surgery as their indication for IV insulin (57.3% vs 38% in comparison cohort).
Demographics and Baseline Characteristics.
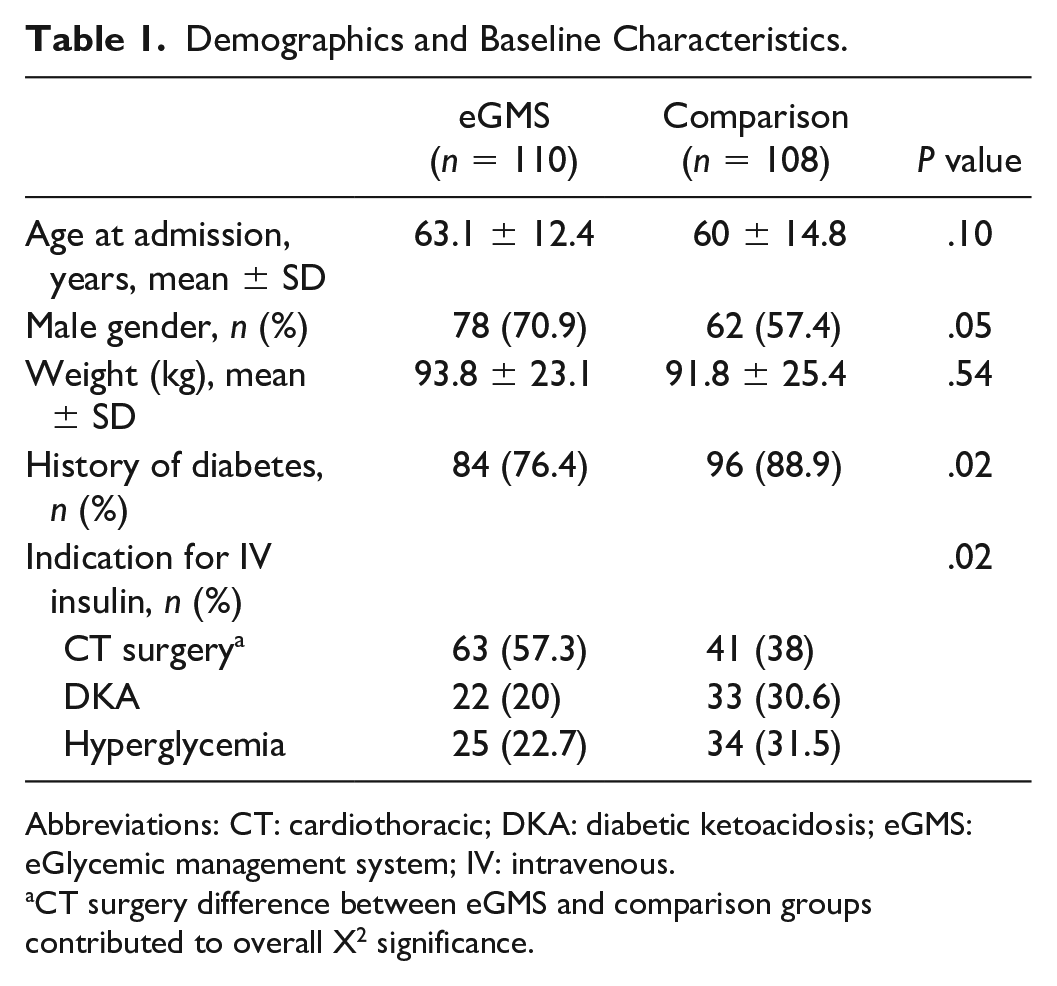
Abbreviations: CT: cardiothoracic; DKA: diabetic ketoacidosis; eGMS: eGlycemic management system; IV: intravenous.
CT surgery difference between eGMS and comparison groups contributed to overall X2 significance.
Efficacy and Safety Endpoints
Table 2 displays the results for efficacy and safety endpoints for the full sample analysis and according to indication for IV Insulin. The percentage of POC BG readings in range was significantly higher for the eGMS patients than comparison cohort patients (84.6% vs 76.8%, P < .001).
Efficacy and Safety Endpoints.
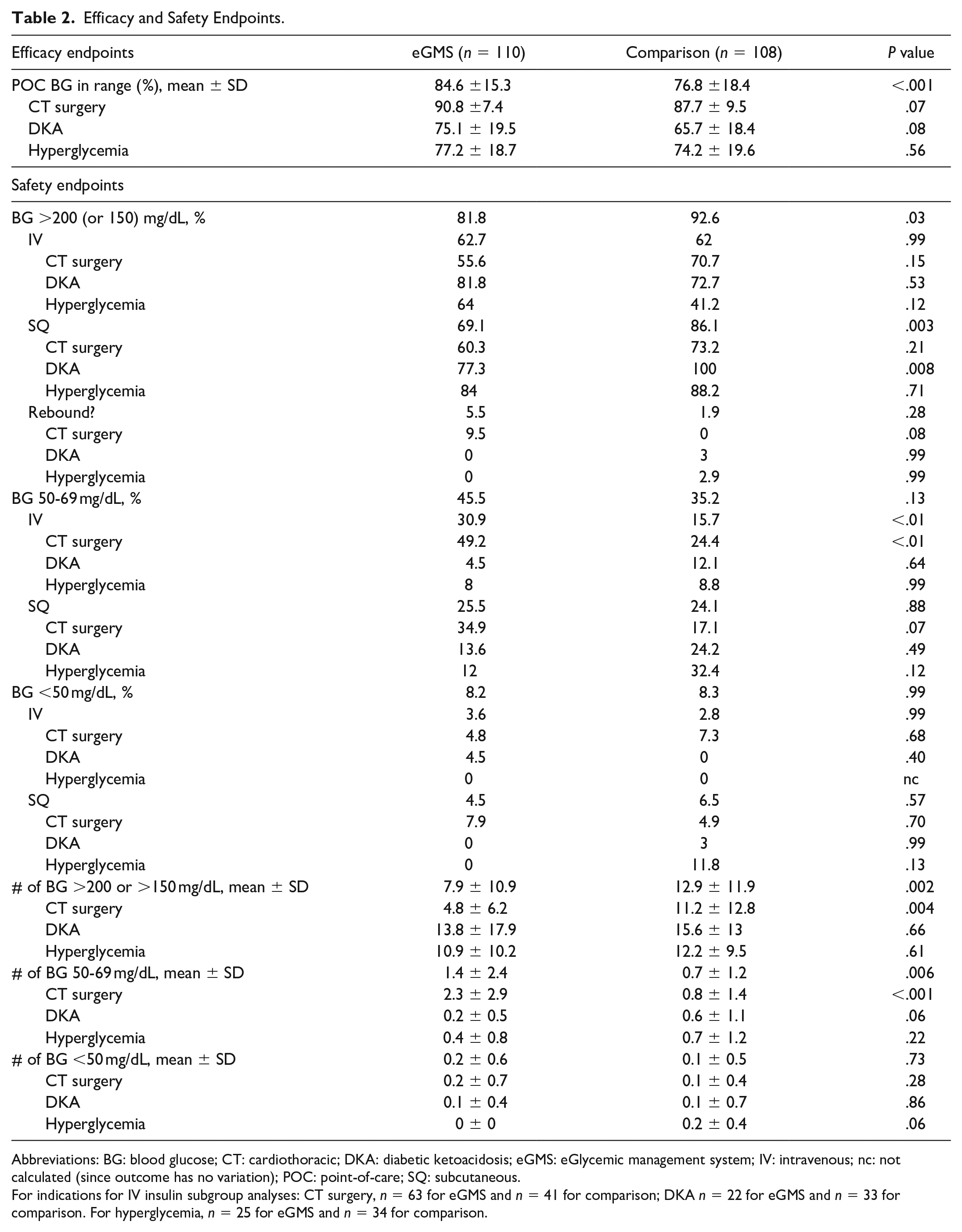
Abbreviations: BG: blood glucose; CT: cardiothoracic; DKA: diabetic ketoacidosis; eGMS: eGlycemic management system; IV: intravenous; nc: not calculated (since outcome has no variation); POC: point-of-care; SQ: subcutaneous.
For indications for IV insulin subgroup analyses: CT surgery, n = 63 for eGMS and n = 41 for comparison; DKA n = 22 for eGMS and n = 33 for comparison. For hyperglycemia, n = 25 for eGMS and n = 34 for comparison.
Hyperglycemic events occurred for significantly fewer patients in the eGMS cohort relative to the comparison cohort (81.8% vs 92.6%, P = .03). Hyperglycemia while on SQ insulin was less likely to occur in the eGMS cohort relative to the comparison cohort (69.1% vs 86.1%, P = .003), and this was likely due to the DKA subgroup, with 77.3% of the eGMS patients and 100% of comparison patients having at least one hyperglycemic episode (P = .008).
Overall, there was no significant difference between cohorts in rate of hypoglycemic events (BG 50-69 mg/dL). However, hypoglycemic events while on IV insulin occurred in a significantly higher percentage of eGMS cohort patients than comparison cohort patients (30.9% vs 15.7%, P < .01); the main contributor to this difference was the subgroup of CT surgery patients (49.2% for eGMS vs 24.4% for comparison, P < .01). Other insulin indication groups (DKA and hyperglycemia treatment) did not differ significantly for hypoglycemic events while on IV insulin.
There were no significant differences overall or by route/indication for insulin between cohorts on incidence of severe hypoglycemic events (BG <50 mg/dL).
The mean number of hyperglycemic events per patient was significantly higher for the comparison cohort than the eGMS cohort (12.9 vs 7.9, P = .002), and the insulin indication subgroup analysis showed that this overall difference can be attributed to the CT surgery patient differences (4.8 for eGMS vs 11.2 for comparison, P = .004). At the same time, the average number of hypoglycemic events was higher overall for the eGMS cohort (1.4 vs 0.7, P = .006) with the main contributor being the CT surgery group (2.3 for eGMS vs 0.8 for comparison, P < .001). The average number of severe hypoglycemic events did not differ significantly between the two cohorts.
Discussion
In this study, we found that there was a significantly higher percentage of BG readings in the target range in the eGMS cohort than in the comparison cohort. For the safety objectives, there were significantly fewer hyperglycemic events in the eGMS cohort, driven by the SQ insulin subgroup. There was no significant difference overall in incidence of hypoglycemia between cohorts, but the eGMS IV insulin subgroup had significantly more hypoglycemic events (driven by the CT surgery population who had a lower target BG range). There was no significant difference in rates of severe hypoglycemic events between cohorts.
The results of this study indicate a favorable efficacy profile for the eGMS when used across multiple disease states in a community hospital. A similar study of another eGMS in the community hospital setting found no significant difference in efficacy as measured by BG values within target range or safety as measured by rates of hyper- or hypoglycemia. 6 Clinically, a higher percent of BG readings in range means better overall glycemic control during the hospitalization. This may translate into lower risk of postoperative complications for CT surgery patients and better glycemic control at the time of discharge for patients with DKA or non-DKA hyperglycemia. Future prospective studies should be done to assess correlation between glycemic management with an eGMS and clinical outcomes in these patient populations.
Our study found lower rates of hyperglycemia for SQ insulin in DKA patients, which is clinically meaningful as it means that patients were achieving more stable control of BG during the hospitalization. However, our institution elected to stop using the eGMS SQ insulin module after the study period due to provider perceptions that patients were not achieving adequate BG control, as well as technical difficulties faced by nurses and providers in the transition from IV to SQ in the program. Our institution has continued use of the IV insulin module with expansion of the program to patients in four total units.
Higher rates of hypoglycemia were anticipated for the CT surgery patients in this study based on the narrower target BG range of 70-150 mg/dL. This narrow BG range was chosen due to provider preference for tight BG control in the perioperative period at our institution. In order to achieve the tight BG control desired, a lower BG target was chosen in the eGMS than the target BG used in our CT surgery Bell-Bass nomogram. A significantly higher proportion of patients in the eGMS group were CT surgery patients, and a significantly lower portion had a history of diabetes. These two factors combined likely indicate that more CT surgery patients did not have diabetes at baseline in the eGMS cohort than in the comparison cohort and may have been more prone to hypoglycemia. This lower target likely contributed to the higher rates of hypoglycemia (BG 50-69 mg/dL) among CT surgery patients treated with IV insulin with the eGMS.
Though retrospective in design, the study included ample patients to assure adequate power to detect a difference between the eGMS and comparison cohorts for the primary endpoint. Another strength of the study was the inclusion of patients with a variety of conditions and mixed level of care; most previous studies of eGMS have focused utilization of the program in a single patient population.
The study does have a few limitations to consider. It was retrospective in design, so there was no active intervention. The two cohorts differed in composition, with the eGMS cohort having a significantly higher percent of patients undergoing CT surgery than the comparison group. Data collection for the eGMS cohort stopped at the time that the program was stopped, either due to discharge or transfer to a floor that did not utilize the program; therefore, there may be more data points per patient for the comparison cohort since data collection continued for this cohort until discharge or discontinuation of insulin. The study did not account for steroid use in critically ill patients, which may have influenced rates of hyperglycemia.
In conclusion, our study found that Glucommander maintained a higher percentage of BG readings in target BG range per patient compared to previously utilized nomograms. Therefore, we would recommend eGlycemic managements systems be considered for management of IV and SQ insulin for patients in a community hospital setting.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
