Abstract
Background:
The hemoglobin A1c (HbA1c) is a gold-standard test to diagnose and monitor diabetes mellitus and has been incorporated into population health performance metrics for quality care. However, patients and practices remain challenged in completing timely HbA1c tests. Point-of-care testing (POCT) for HbA1c provides a quick, easy, reliable method for monitoring diabetes in the primary care office setting. The objectives of this quality improvement study were to evaluate the impact of HbA1c POCT on onsite HbA1c testing frequency as a component of population health performance, as well as to measure the utility of HbA1c POCT in identifying clinically meaningful change in disease.
Method:
Prospective quality improvement cohort study among sequentially scheduled adult patients with diabetes due for HbA1c testing across three primary care practices.
Results:
Practices with HbA1c POCT were 3.7 times less likely to miss HbA1c testing at the time of the visit compared with practices in which HbA1c POCT was not available (P < .001). Nearly one in four patients in each group were found to have clinically worsening diabetes (defined by an increase in HbA1c of ≥0.5% or 5.5 mmol/mol). Nearly half of those patients in the intervention group were identified by POCT.
Conclusions:
HbA1c POCT can improve population health-driven HbA1c testing adherence at office visits in primary care and may enable more timely intervention of diabetes management for patients with worsening disease.
Keywords
Background
The growing endemics of diabetes and prediabetes, estimated to affect 10% and 30% of the adult population, respectively, highlight the need for effective and reliable methods for disease monitoring and management. 1 Left unmanaged or undermanaged, diabetes can lead to serious and costly complications, including cardiovascular and neurovascular disease, kidney disease, amputation, and premature death. 1 The hemoglobin A1c (HbA1c) test is one of the gold-standard tests to both diagnose and monitor diabetes mellitus in the care of patients. Experts recommend that people with diabetes and stable glycemia have an HbA1c test at least twice per year, and those with unstable glycemia up to every three months. 2 In particular, the percentage of patients with diabetes who receive HbA1c testing during the measurement year has become one of the quality performance process measures among the National Quality Forum ambulatory diabetes metrics that are considered for Healthcare Effectiveness Data and Information Set (HEDIS) accreditation. 3 In the current healthcare model, healthcare organizations are often incentivized by payers for achieving diabetes care HEDIS-like metrics across their patient populations, which has required them to develop effective care delivery models. As such, HbA1c testing frequency targets may vary across regions or organizations. Population health management generally requires a number of simultaneous approaches to meeting metric targets—some of them occurring synchronously with (shortly before, during, or after) patient visits, while others occurring asynchronously or completely independent of a clinic visit (Figure 1).
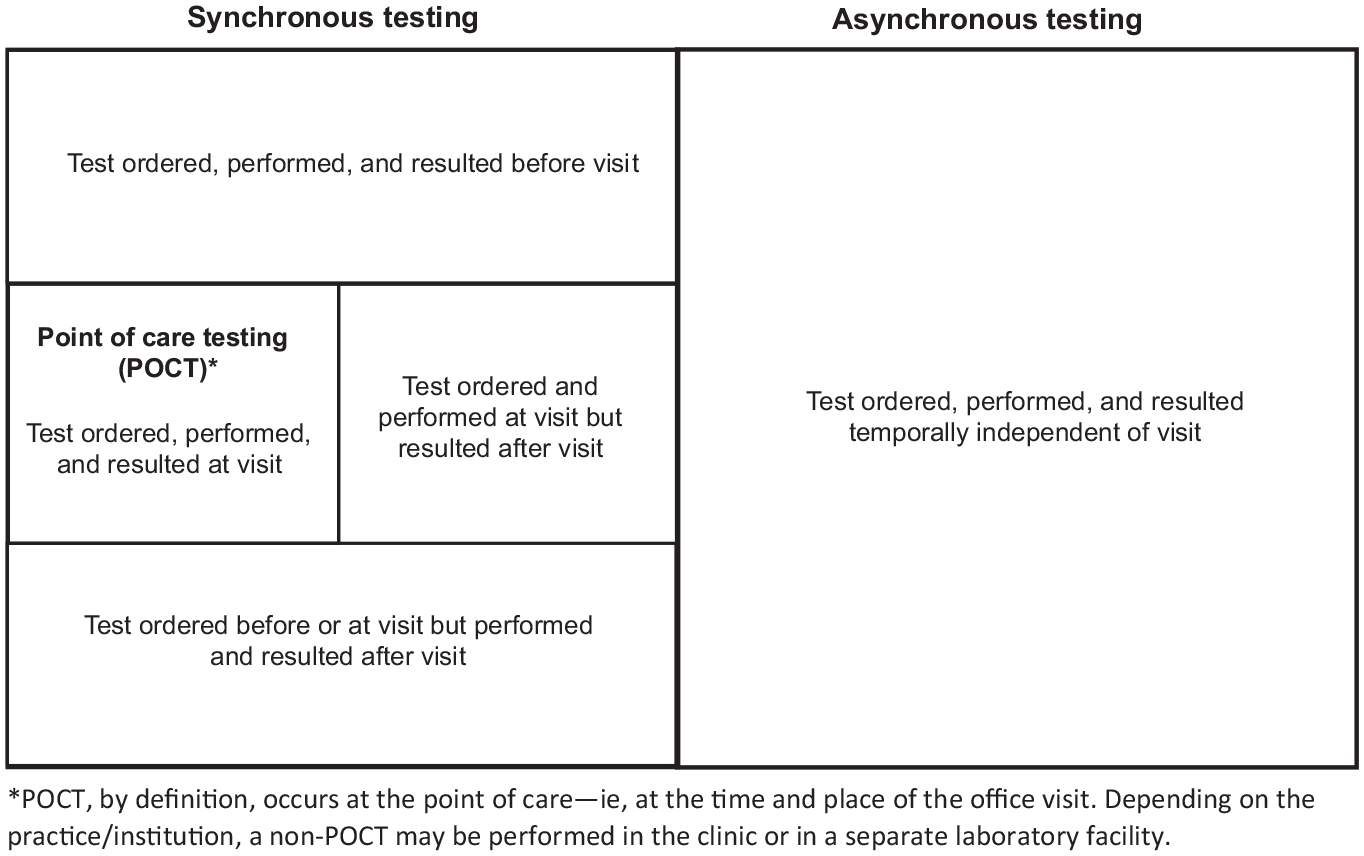
General lab testing scenarios in primary care.
Nonadherence to HbA1c laboratory requests may contribute to higher rates of poor glycemic control. Indeed, up to one in seven patients do not complete HbA1c testing within six months of the request, and the median elapsed time from order to lab completion is seven to ten days. 4 Point-of-care testing (POCT) for HbA1c provides a quick, easy, and reliable method for monitoring diabetes in primary care. Multiple studies have demonstrated comparable accuracy, improved workflow and office efficiency, strong patient satisfaction, and improved adherence to diabetes therapy with HbA1c POCT, compared with traditional HbA1c lab testing. 5 By nature of where it is performed (in the clinical office) HbA1c POCT is considered to be a synchronous testing effort. The objectives of this quality improvement study were to evaluate the impact of POCT on synchronous HbA1c testing frequency for population health performance metrics in the primary care setting, to measure the utility of HbA1c POCT in identifying clinically meaningful change in disease, and to measure staff satisfaction with POCT compared with use of traditional A1c testing.
Study Context
With institutional review board (IRB) approval, we conducted a six-month (January to July 2019) prospective cohort quality improvement study at three academic, primary care practices at the Massachusetts General Hospital (MGH) in Boston, MA. Adult patients 18 years or older were included in the study. Informed consent from patients was not required by the IRB because the study involved was based only on review of medical records.
Two practices were assigned to the intervention group and were provided HbA1c POCT in addition to conventional central lab testing. The first practice in the intervention group, MGH Back Bay Primary Care (Back Bay), employs 6.75 full-time equivalent (FTE) providers (MD and NP), 4 medical assistants (MAs) and 2 licensed practical nurses caring for approximately 7900 adult patients, 408 of them with diabetes. The second practice in the intervention group, MGH Internal Medicine Associates (IMA) Team 3, employs 9 FTE providers, 8 MAs, and 5 registered nurses (RNs), caring for over 10 100 patients, 1021 of them with diabetes. A third practice was assigned to the control group, which only had standard central laboratory HbA1c testing available. This control group, IMA Team 1, is staffed with 7.3 FTE providers, 6 FTE MAs and 5 RNs, caring for a total cohort of 11 796 adult patients, with 1292 patients with diabetes. As part of standard population health quality care performance assessment, all primary care practices at MGH are routinely measured by the rate at which their patients on the MGH Diabetes registry (as defined by having a diagnosis of diabetes or prior HbA1c ≥6.5% or 48 mmol/mol) receive every six-monthly HbA1c tests. This quality improvement study, therefore, specifically examined the completion rate of every six-monthly HbA1c testing in patients with diabetes arriving for scheduled appointments.
Methods
Onsite HbA1c POCT was implemented using the Siemens DCA Vantage Analyzer (Siemens Healthcare, Norwood, MA, USA). The DCA Vantage Analyzer performs and provides results for HbA1c from a fingerstick blood sample in six minutes and has been available for integrated electronic heath record (EHR) use in outpatient clinical practice at MGH since 2012. The devices were validated for accuracy,
Each intervention practice was given at least three months to incorporate HbA1c POCT workflows into their practice. Then, from January 14 through July 14, 2019, the medical records of 530 unique and sequentially scheduled patients in the practices with HbA1c POCT available (intervention) and 377 unique and sequentially scheduled patients in the practice without HbA1c POCT (control) were both prospectively and retrospectively reviewed. Clinic schedules were reviewed one week in advance of an appointment to identify patients on the MGH Diabetes Registry who were due or coming due within one month for a semi-annual HbA1c test. For each patient, we recorded age,
No-show rates, defined as the percentage of all scheduled visits that were cancelled within the prior week or unarrived, were calculated for each group. The nonadherence rate of synchronous HbA1c testing was defined by calculating the percentage of arrived eligible patients who did not complete an HbA1c test. Finally, the prevalence of a meaningful change in HbA1c, as defined in the literature by an increase or decrease of at least 0.5% (5.5 mmol/mol), 6 was also calculated for each group. Staff at each medical practice, including physicians, nurse practitioners, MAs, nurses, and practice administrators, were administered an anonymous staff satisfaction survey (Figure 2) at the end of the study period. The survey tabulated responses by role and solicited additional comments.

Staff satisfaction survey.
Data concerning patient demographics (age and
Results
Table 1 summarizes the demographic comparisons between the intervention and control groups. The combined intervention group had a total of 530 unique patients. The control group had 377 unique patients. There were no significant differences in sex prevalence or in mean HbA1c between the intervention and control groups, but there was a significant difference in age with the intervention group being three years younger.
Demographic Comparisons Across Study Groups.
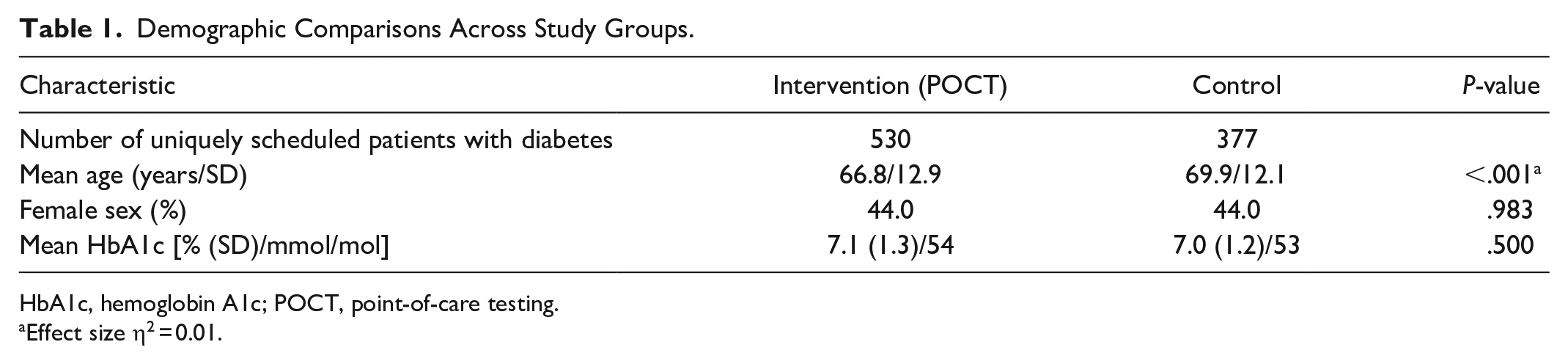
HbA1c, hemoglobin A1c; POCT, point-of-care testing.
Effect size η2 = 0.01.
Table 2 summarizes the effect of HbA1c POCT availability on synchronous HbA1c testing performance and the opportunity to respond to clinically meaningful changes in HbA1c across each study group. A total of 622 consecutively scheduled visits were recorded among the intervention group, representing 472 uniquely arrived patients. Among the control group, 462 consecutively scheduled visits were recorded, representing 350 uniquely arrived patients. There was no significant difference in no-show rates among the intervention or control groups for patients with diabetes who were eligible for HbA1c testing. Across 822 testing opportunities (uniquely arrived visits), practices without HbA1c POCT were 3.69 times less adherent to every six-monthly HbA1c testing (20.3% vs 5.5%, P < .001). A similar number of patients in each study group were found to have clinically worsening diabetes (≥0.5% or 5.5 mmol/mol rise in HbA1c). Nearly half (44.9%) of all clinically worsening glycemic control in the intervention group were identified by HbA1c POCT.
Visit Adherence and HbA1c Testing Performance Across Study Groups (January Through July 2019).
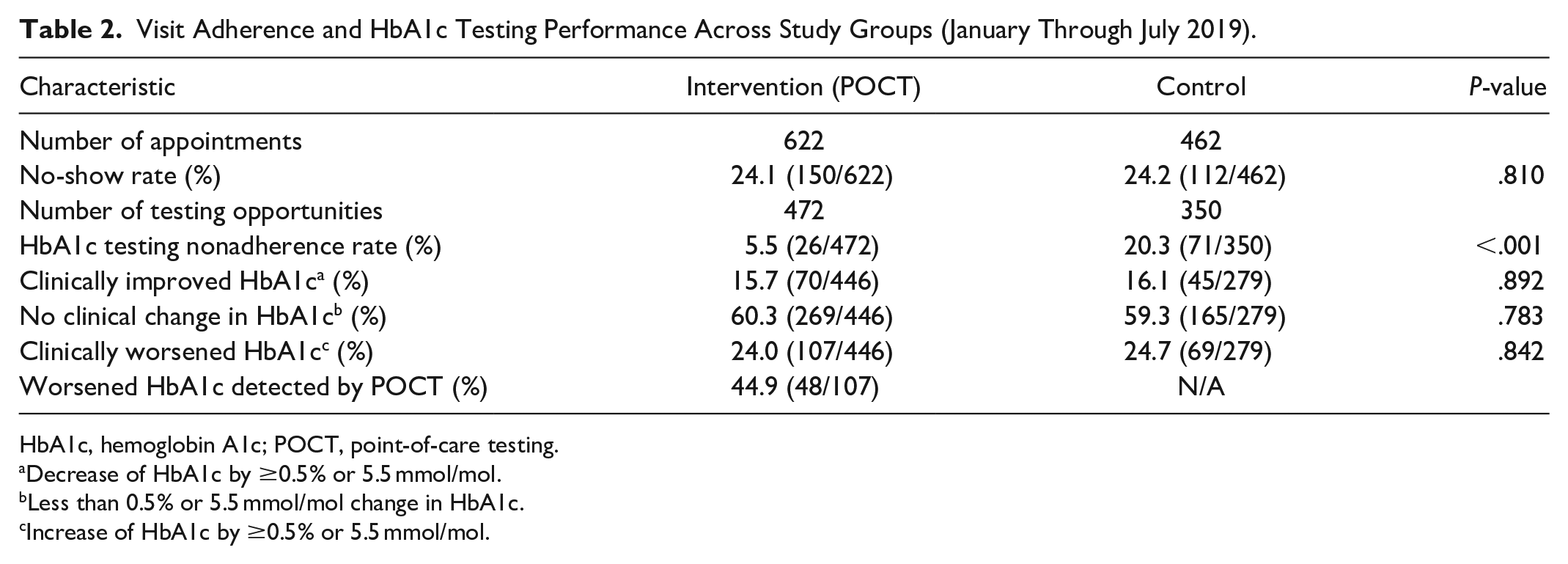
HbA1c, hemoglobin A1c; POCT, point-of-care testing.
Decrease of HbA1c by ≥0.5% or 5.5 mmol/mol.
Less than 0.5% or 5.5 mmol/mol change in HbA1c.
Increase of HbA1c by ≥0.5% or 5.5 mmol/mol.
Of 41 respondents to the staff satisfaction survey, 73% were providers. Seventy-five percent of respondents found HbA1c POCT A1c to be equal to or more favorable to administer compared to other point-of-care tests currently in use. Eighty-eight percent of respondents found HbA1c POCT to be equal or superior than central lab testing. Twenty-nine percent of respondents offered additional comments, which largely focused on the test’s superior operational utility.
Discussion
This study demonstrates that implementation of HbA1c POCT in primary care practices significantly improves HEDIS-based HbA1c testing frequency that occurs synchronously with an office visit. The findings highlight this technology’s advantage in providing an opportunity for more timely management for patients with clinically worsening disease. This study also suggests strong acceptance, by professional care teams, of HbA1c POCT when compared to other types of office-based point-of-care tests and when compared to standard central lab HbA1c testing.
The significant three-year age difference between the intervention and control groups does not achieve practical significance as evidenced by the extremely weak effect size (η2 = 0.01). Such a small difference in age is not likely to explain meaningful clinical differences in visit adherence, patient preference for POCT, or overall testing adherence.
The no-show rate in all groups was higher than the reported 19% national average rate for primary care, 7 suggesting that aggressive strategies to keep patients engaged with scheduled care may be particularly needed in patients with diabetes. A statistically similar no-show rate among the intervention and control groups suggests that the significant difference in HbA1c testing nonadherence is more likely explained by the availability of the HbA1c POCT itself rather than visit attendance rate. While each practice in the study used the same EHR, which displays built-in population health reminders, variations in site-specific workflows to identify patients due for HbA1c testing at or prior to their appointment could affect testing performance on arrived patients. Two practices that utilized similar workflows (IMA Team 1 and IMA Team 3) were specifically incorporated in the control and intervention arms of the study to address this potential bias.
HEDIS measures are used by more than 90% of health payers to assess and collect data on the performance of providers and healthcare institutions in an effort to incentivize higher-quality care and better patient outcomes. 8 Referring again to Figure 1, the synchronous nature of where HbA1c POCT is performed can only address testing in a subpopulation of patients with diabetes—namely, in those patients who arrive for an appointment. Additional testing strategies that are asynchronous and independent of the office visit are needed to help reach HbA1c testing targets across a clinic’s complete population of patients with diabetes. Nonetheless, HbA1c POCT may play a valuable role as one among an arsenal of strategies to improve population health-based testing adherence. We could imagine a future state of EHR-integrated satellite POCT facilities that could provide testing independent of an office visit, or in an expedited manner prior to an office visit. The inability to counsel with the patient about results at the point of testing, however, could pose challenges for treatment adherence and clinical outcome similar to those encountered with standard laboratory testing.
In general, each 1% reduction in HbA1c reduces the risk of microvascular complications (eye, kidney, nerve, cardiovascular) by up to 30%.9-11 Among more than 1500 primary care physicians surveyed in both the United States and United Kingdom, diabetes was among the most frequently listed condition for which respondents felt POCT could improve disease monitoring and management.12,13 This may have to do with this particular condition’s multiple risks and comorbidities, a relatively higher frequency of lab testing compared with other chronic conditions (such as hypercholesterolemia and hypertension), and the inherent and time-consuming need to engage and support patients along their “decision journey” 14 around behavioral components. While these types of shared decision-making processes in primary care have been shown to yield better health outcomes and satisfaction,15-17 they happen less than half of the time in the clinical setting.18,19 It has long been appreciated that time is “of the essence” during a primary care office visit, with clinicians having to multitask a number of responsibilities with the goal of the delivery and coordination of comprehensive care. 20 In this study, POCT not only improved office-based HbA1c testing adherence, but also identified nearly half of all patients in the intervention group who experienced clinically worsening diabetes (ie, increase of at least 0.5% or 5.5 mmol/mol in HbA1c), suggesting that without HbA1c POCT, there is a major missed opportunity to leverage clinical decision-making such as counseling, education, and pharmaceutical therapy, to enhance glycemic control. The components of clinical care that can be affected by the limited time of the office visit include patient satisfaction, compliance, medical outcomes, prescribing practices, and physician/care team satisfaction.21-25 POCT can reliably mitigate these risks as a result of precious time being returned to the physician during the visit to engage in these higher-level tasks. At a time of declining use of primary care among commercially insured adults in the United States, 26 the ability to test and consult at the point of care may provide a critical advantage in care delivery.
A prior study used a three- to six-month quasi-experimental, pretest and posttest design method among randomly sampled patients with type 2 diabetes in the primary care setting to infer the impact of HbA1c POCT on documented HbA1c levels and found a weakly significantly improved overall testing adherence. 27 The study also found a statistically significant but less clinically meaningful decrease of 0.4% (4.4 mmol/mol) in mean HbA1c six months post-implementation among those who received POCT. In this present study in sequentially scheduled and arrived patients, we found no significant difference in the proportion of patients whose HbA1c had meaningfully changed. Because HbA1c is generally measured no more than once every three months, a more robust study over a much longer time period (beyond the six months period of this study) would be needed to confidently measure change in HbA1c attributable to the testing modality itself (POCT vs traditional central lab testing). Such a study could also evaluate the impact of HbA1c POCT on a clinic’s entire diabetes population HbA1c testing adherence (including both synchronous and asynchronous testing).
The similar staffing ratios among both groups suggest that differences in HbA1c testing adherence are likely not explained by variation in the availability of staff to perform POCT. Staff satisfaction survey results support prior findings that HbA1c POCT can have a positive impact when employed in primary care, including reduction of clinical team workflow burden, improved patient–provider communication and faster overall clinical decision-making. 28 Survey comments also echo commonly perceived barriers to POCT—namely perceived inaccuracy, overall mixed attitudes, and uncertainty about their cost-effectiveness. 29 Though a formal financial feasibility study was outside the scope of this particular study and costs for the testing equipment, supplies and reagents, and reimbursement for HbA1c POCT may vary among region and payers, prior publications have demonstrated financial feasibility of POCT in the primary care setting. 22
Several limitations are worth considering in this study. First, this study evaluated HbA1c POCT at an urban academic medical center. Second, the comparison of demographics includes all scheduled patients, not just those who arrived for their visit. As a quality improvement study, we felt it important to compare no-show rates in a live clinical environment to rule out testing adherence bias. Other potential biases such as prevalence of diabetes-associated comorbidities, race, insurance status, daily visit volume, or visit length between groups were not evaluated. We suspect these factors could more strongly affect disease outcomes rather than operational impact on testing adherence. Prior publications have demonstrated historically strong preference and acceptability of fingerstick POCT over venipuncture among a variety of patient populations, races, in multiple clinical settings.30-34 Alternatively, clinician bias in whom to administer POCT could be a meaningful investigative pursuit. Third, POCT results in this study were automatically and fully integrated into the EHR. Depending on the geographic location of the POCT device itself, other clinics without full integration could experience delays in transmission of results to the provider that might negatively impact timeliness and availability of data and, therefore, clinical utility during the visit.
Conclusion
This study demonstrates that HbA1c POCT can improve population health-based HbA1c testing adherence in the primary care clinic and is a satisfactory tool among clinical staff.
Footnotes
Acknowledgements
We thank Karen E. Lynch, Director of Analytics, Applied Informatics, Laboratory of Computer Science, MGH and Megan Williams, Senior Population Health Coordinator, MGH for electronic health record chart review for group identification, visit status, and testing status and results;
Kimberly Gregory, Associate Director, Point of Care Testing, Department of Pathology, MGH for device installation, validation, and training; and Steve Condon, Senior Data Analyst, Mass General Physicians Organization for population health data support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Siemens Point of Care to cover costs for project management, research assistance, and device installation and validation. Siemens had no role in either the design and implementation of the study, data collection and analysis, or manuscript preparation.
