Abstract
Background:
In this study, we evaluated the analytical performance of the second-generation factory-calibrated FreeStyle Libre Flash Glucose Monitoring (FreeStyle Libre 2) System compared to plasma venous blood glucose reference, Yellow Springs Instrument 2300 (YSI).
Methods:
The study enrolled participants aged four and above with type 1 or type 2 diabetes at seven sites in the United States. Adult participants (18+ years) participated in three in-clinic sessions and pediatric participants (4-17 years) participated in up to two in-clinic sessions stratified to provide data for days 1, 2, 3, 7, 8, 9, 12, 13, or 14 of sensor wear. Participants aged 11+ underwent supervised glycemic manipulation during in-clinic sessions to achieve glucose levels across the measurement range of the System. Performance evaluation included accuracy measures such as the proportion of continuous glucose monitoring (CGM) values that were within ±20% or ±20 mg/dL of reference glucose values, and bias measures such as the mean absolute relative difference (MARD) between CGM and reference values.
Results:
Data from the 144 adults and 129 pediatric participants were analyzed. Percent of sensor results within ±20%/20 mg/dL of YSI reference were 93.2% and 92.1%, and MARD was 9.2% and 9.7% for the adults and pediatric participants, respectively. The System performed well in the hypoglycemic range, with 94.3% of the results for the adult population and 96.1% of the data for pediatric population being within 15 mg/dL of the YSI reference. The time lag was 2.4 ± 4.6 minutes for adults and 2.1 ± 5.0 minutes for pediatrics.
Conclusions:
The System demonstrated improved analytical accuracy performance across the dynamic range during the 14-day sensor wear period as compared to the previous-generation device.
NCT#: NCT03607448 and NCT03820050
Introduction
The Abbott FreeStyle Libre Flash Glucose Monitoring System (Abbott Diabetes Care, Alameda, CA), introduced in 2014, was the first factory-calibrated continuous glucose monitoring (CGM) device available to people with both type 1 diabetes (T1D) and type 2 diabetes (T2D).1-3 Factory-calibrated CGM devices offer distinct advantages over the previous user-calibrated devices, including reduced burden, reduction in test-strip usage, and removal of inaccuracy imparted from user glucose testing.4-6 The improvement in device accuracy has also resulted in approval for insulin dosing from CGM values (non-adjunctive use), further reducing user burden.7,8 Direct dosing from CGM has since been demonstrated to be safe and effective in several randomized clinical trials.9-12 The expansion of CGM utility and reduction in technology burden has contributed to an increased adoption of CGM by people with both T1D and T2D.13-17
This first-generation product is used to guide diabetes management decisions, including insulin dosing. The Free-Style Libre 2 System (hereafter referred to as “System”) additionally provides optional hypoglycemia and hyperglycemia alarms. When configured, the device sends data to the receiver every minute to generate an alarm. This product secured the Conformitè Europëenne (CE) mark in 2018. 18 The product was updated in 2020 with a new algorithm, based on the analysis of additional reference paired sensor data, to further improve accuracy.
The study described here evaluated the accuracy of this System with the new glucose algorithm. There was no change to the sensor or insertion mechanism compared to the on-market device. The study enrolled participants aged four and above. Participants older than 11 years underwent glycemic challenges to obtain sufficient data for the evaluation of sensor performance at low and high glucose concentrations.
Methods
Study Design
The System was evaluated in two prospective multicenter studies enrolling 153 adults of ages 18 and above (at five clinical sites) and 144 pediatric participants of ages 4-17 (at four clinical sites) with T1D or T2D. Seven adult participants and five pediatric participants screen-failed or discontinued prior to the sensor insertion, and thus were not evaluable for the study effectiveness assessment. The studies were registered with clinicaltrials.gov (NCT# NCT03607448 and NCT03820050).
All participants were required to wear the sensor for up to 14 days. Participants aged 4-17 years were required to perform four self-monitoring of blood glucose (SMBG) measurements per day, while participants above 18 years were required to perform eight SMBG measurements. All SMBG measurements were made using the built-in blood glucose meter that uses FreeStyle Neo test strips. Participants received no training on the devices (but were trained on study procedures) and inserted one sensor on the back of each of their upper arms following the instructions for use. For pediatric participants, either the parent or the participant inserted the sensor. Sensors that failed within the first hour after insertion were replaced.
The participants aged 11 and above were required to have intensive insulin therapy (either multiple daily injections or continuous subcutaneous insulin infusion [CSII]) with known insulin sensitivity factor and were required to undergo controlled manipulation of glucose levels during at least one of the in-clinic sessions. Exclusion criteria included pregnancy, anemia, or any condition, per investigator discretion, that could place the subject at risk by glucose manipulation. No exclusions were made based on the type of medications or supplements.
To ensure that the CGM data adequately covered the System’s entire reportable range (40-500 mg/dL), glucose levels were manipulated to induce high or low blood glucose levels through carbohydrate consumption and insulin timing during the clinic sessions for participants aged 11 and above.
Adult participants were scheduled for up to three in-clinic sessions of ten hours on days 1-3, 7-9, and 12-14. Pediatric participants ages 6-17 years were scheduled for up to two in-clinic sessions of eight hours on days 1-2, 7-9, or 12-14 depending on their body weight. All sessions included comparison of sensor readings to plasma venous glucose concentrations using a laboratory reference method (Yellow Springs Instrument YSI 2300 [hereafter “YSI]; YSI, Inc., Yellow Springs, OH). Participants had venous blood drawn every 5-15 minutes for the duration of each clinic session for YSI measurement. The samples were centrifuged within 15 minutes of the blood draw and tested on YSI within 15 minutes afterwards. A heating pad was applied around the intravenous site to keep the insertion site open. The CGM devices were masked to participants and clinic staff for the entire sensor wear.
Participants or caregivers/study staff scanned both sensors with the paired reader immediately following reference sample collection. The first inserted sensor with the YSI-sensor pair was used for the analysis of the System performance, while the second applied sensor was used for the assessment of precision. Sensor values were paired with YSI values by choosing the sensor value that was closest in time to the corresponding YSI blood draw, but no more than five minutes before or after the YSI blood draw. Each blood draw sample was assayed on the YSI in duplicate and the average of the readings was used.
Device Description
The FreeStyle Libre family of products is unique among existing interstitial glucose monitoring technologies that uses a wired enzyme technology with glucose oxidase. The sensor is “factory only” calibrated and has a wear time of up to 14 days without any user calibration. This System has the same form factor (shape and size) as the first-generation product.
The user is required to scan the sensor to get the current glucose, glucose trend, and historic glucose up to eight hours. It also has optional hypoglycemic and hyperglycemic alarms and when configured, the Bluetooth-enabled sensor will send the glucose information to the receiver (either the reader or smartphone running the FreeStyle LibreLink app) every minute to generate the alarm. The glucose algorithm has been updated to improve accuracy at the low end of the dynamic range.
Data Analysis
Only those matched YSI-sensor pairs where the sensor results were within the reportable range (40-500 mg/dL) were used for the performance evaluation, as shown in the tables 3–7. The agreement levels were calculated relative to a glucose concentration difference (in mg/dL) when the reference glucose value was up to 80 mg/dL and relative to a normalized concentration difference (in %) otherwise, and were evaluated at four different ranges: ±15%/±15 mg/dL, ±20%/±20 mg/dL, ±30%/±30 mg/dL, and ±40%/±40 mg/dL. The mean absolute relative difference (MARD) was calculated as the absolute value of the average percent difference between the paired sensor and reference glucose values. The consensus error grid analysis was performed for paired glucose data. 19
Paired YSI-sensor data were grouped into four wear periods: beginning (days 1, 2, and 3 for adults, and days 1 and 2 for pediatric participants), early middle (days 7 and 8), late middle (days 9 and 12), and end (days 13 and 14). The total drift of the sensitivity over wear period was assessed by performing a linear regression on the paired readings of relative difference between the sensor and SMBG against sensor elapsed time. The between-sensor precision was calculated as the coefficient of variation (CV) from the paired glucose readings from two sensors worn simultaneously with matched wear time.
The ability of the System to alarm appropriately when alerts were set at different thresholds was assessed by comparing sensor results to YSI measurements within a 15-minute time window at low (60, 70, 80, and 90 mg/dL) and high (120, 140, 180, 200, 220, 240, and 300 mg/dL) glucose threshold levels and determining whether an alert would have been generated. At each threshold, true alarm rate (whether YSI is in agreement with the device when the device alerts) and detection rate (whether the device alerts when YSI is within the threshold) were calculated. The false alarm rate and missed detection rates were calculated as (100% ─ true alarm rate) and (100% ─ detection rate) respectively.
All analyses were conducted using SAS 9.4 Software.
Results
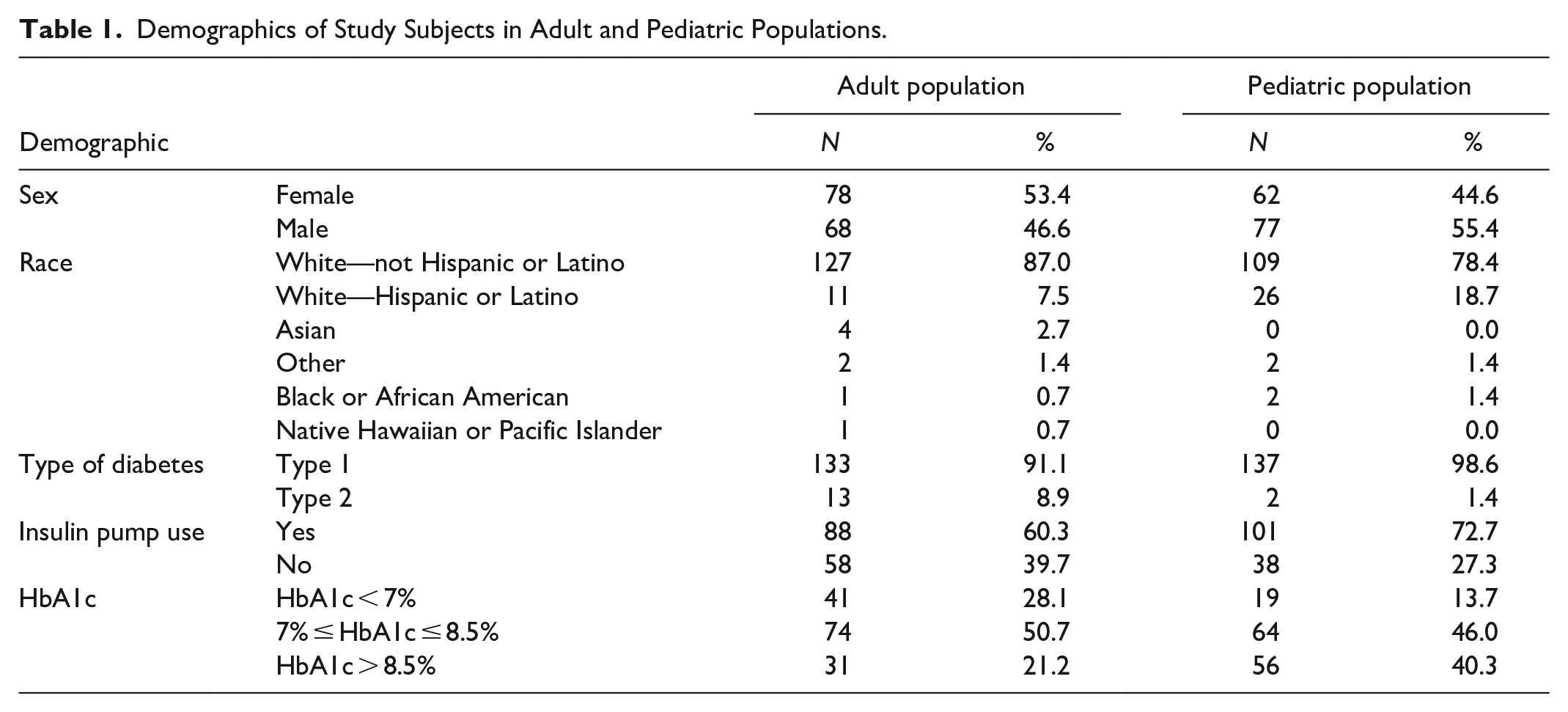
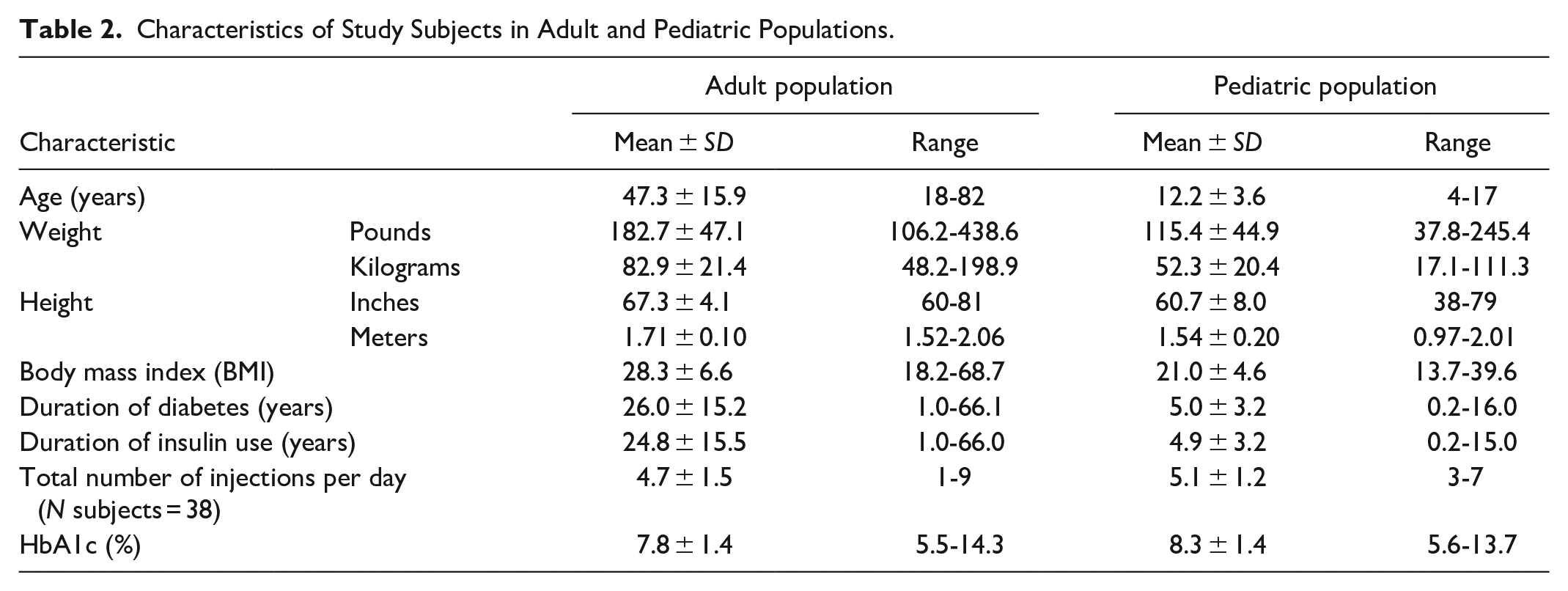
Demographics and baseline characteristics of the evaluable participants in the study are provided in Tables 1 and 2. The subject population had a good spread of HbA1c values covering a wide range and represented both CSII and multiple daily injection (MDI) users. The body mass index (BMI) ranged from 18.2 to 68.7 kg/m2 for adult population and from 13.7 to 39.6 kg/m2 for the pediatric population. Greater than 90% of the participants had T1D.
Demographics of Study Subjects in Adult and Pediatric Populations.
Characteristics of Study Subjects in Adult and Pediatric Populations.
Only data compared to the YSI reference are used for evaluating the performance of the System. One hundred and forty-six adults and 139 pediatric participants were evaluable. Two adult and ten pediatric (including eight aged between four and five years who did not have YSI reference measurements) participants did not have paired sensor to YSI data; thus, they were excluded from the accuracy assessments presented in Tables 3–7. This resulted in 144 adult and 129 pediatric participants with YSI reference measurements.
Overall Performance of the System by Subject Age Group.

Abbreviation: MARD, mean absolute relative difference.
Accuracy Performance at Different Reference Glucose Levels (YSIref): (a) Data From the Adult Population; (b) Data From the Pediatric Population; and (c) Data From the Adult and the Pediatric Populations in the Medically Relevant Glucose Ranges.
(a)
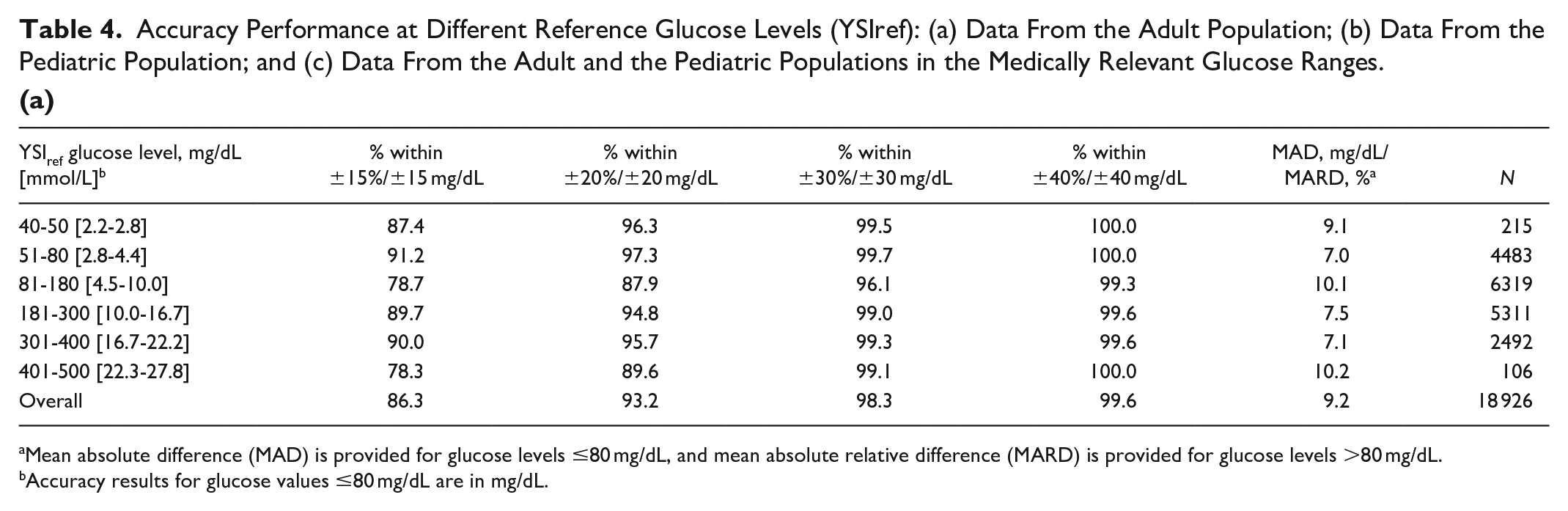
Mean absolute difference (MAD) is provided for glucose levels ≤80 mg/dL, and mean absolute relative difference (MARD) is provided for glucose levels >80 mg/dL.
Accuracy results for glucose values ≤80 mg/dL are in mg/dL.
(b)
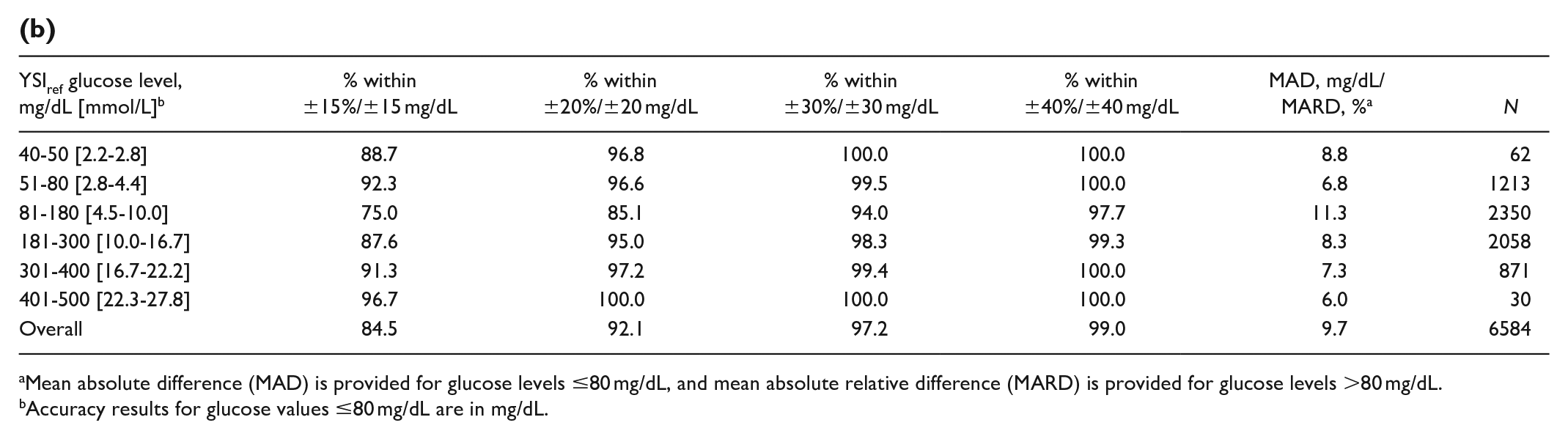
Mean absolute difference (MAD) is provided for glucose levels ≤80 mg/dL, and mean absolute relative difference (MARD) is provided for glucose levels >80 mg/dL.
Accuracy results for glucose values ≤80 mg/dL are in mg/dL.
(c)
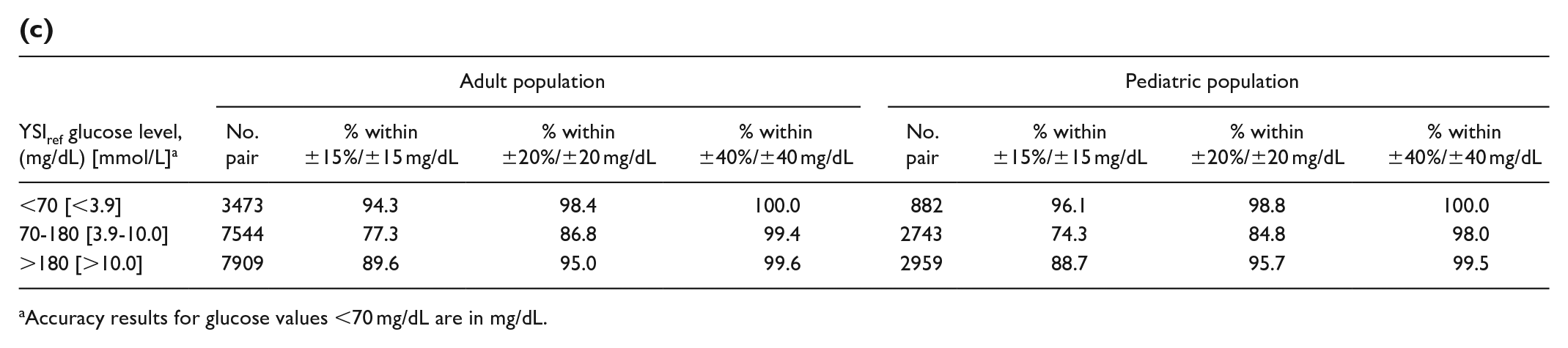
Accuracy results for glucose values <70 mg/dL are in mg/dL.
Percentages Within 20%/20 mg/dL of YSI reference Performance at Different Wear Periods for Adult and Pediatric Populations.
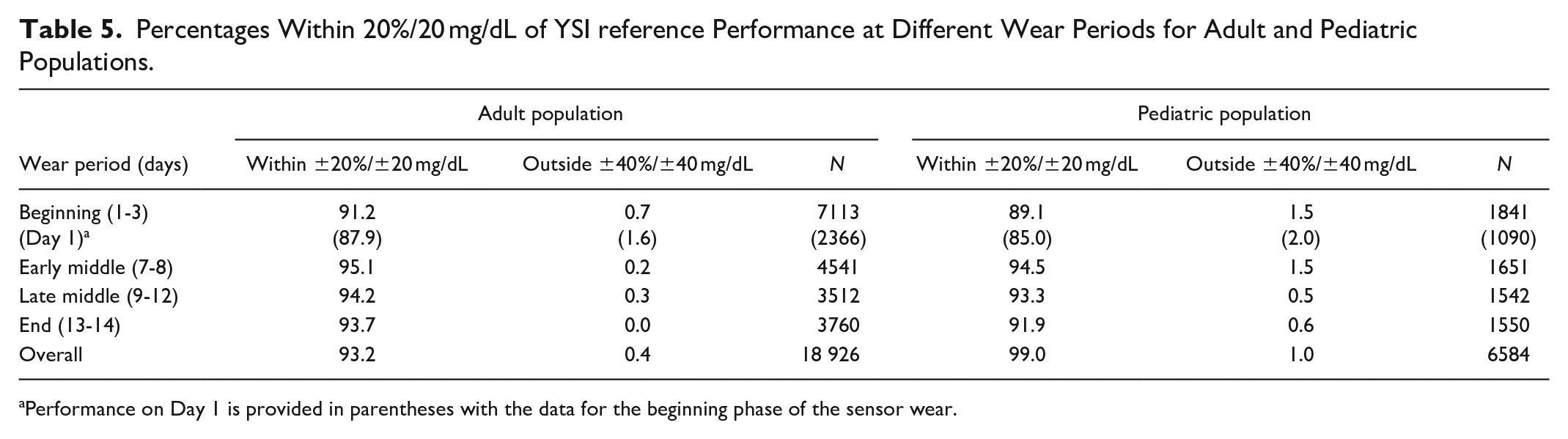
Performance on Day 1 is provided in parentheses with the data for the beginning phase of the sensor wear.
MARD Performance at Different Wear Periods for Adult and Pediatric Populations.
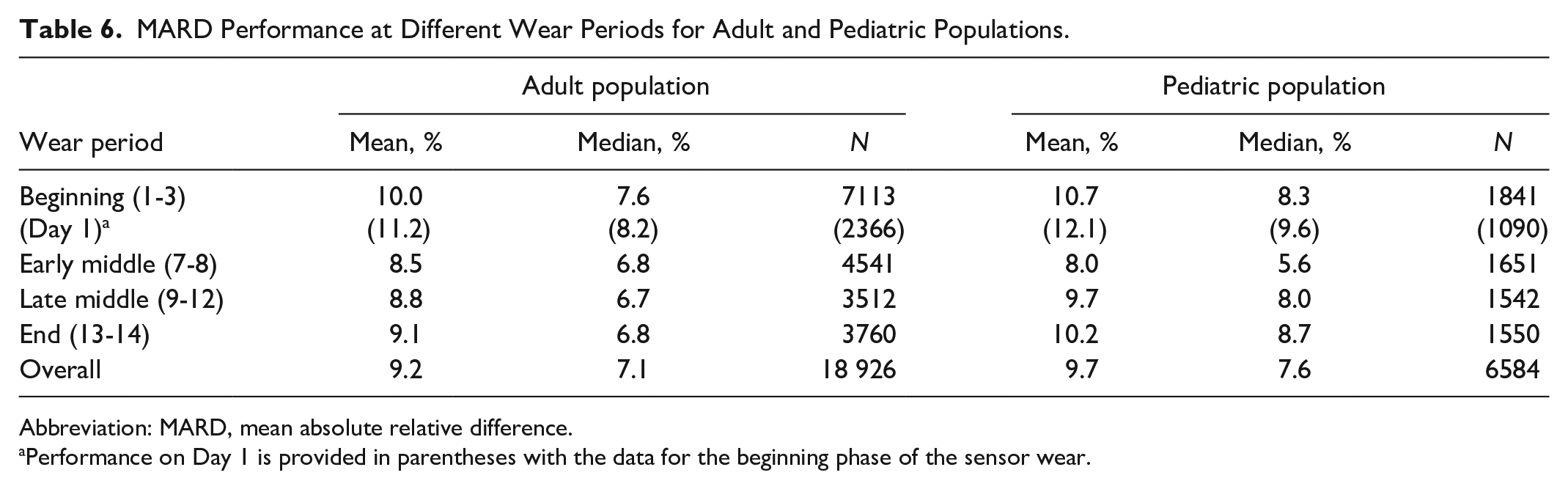
Abbreviation: MARD, mean absolute relative difference.
Performance on Day 1 is provided in parentheses with the data for the beginning phase of the sensor wear.
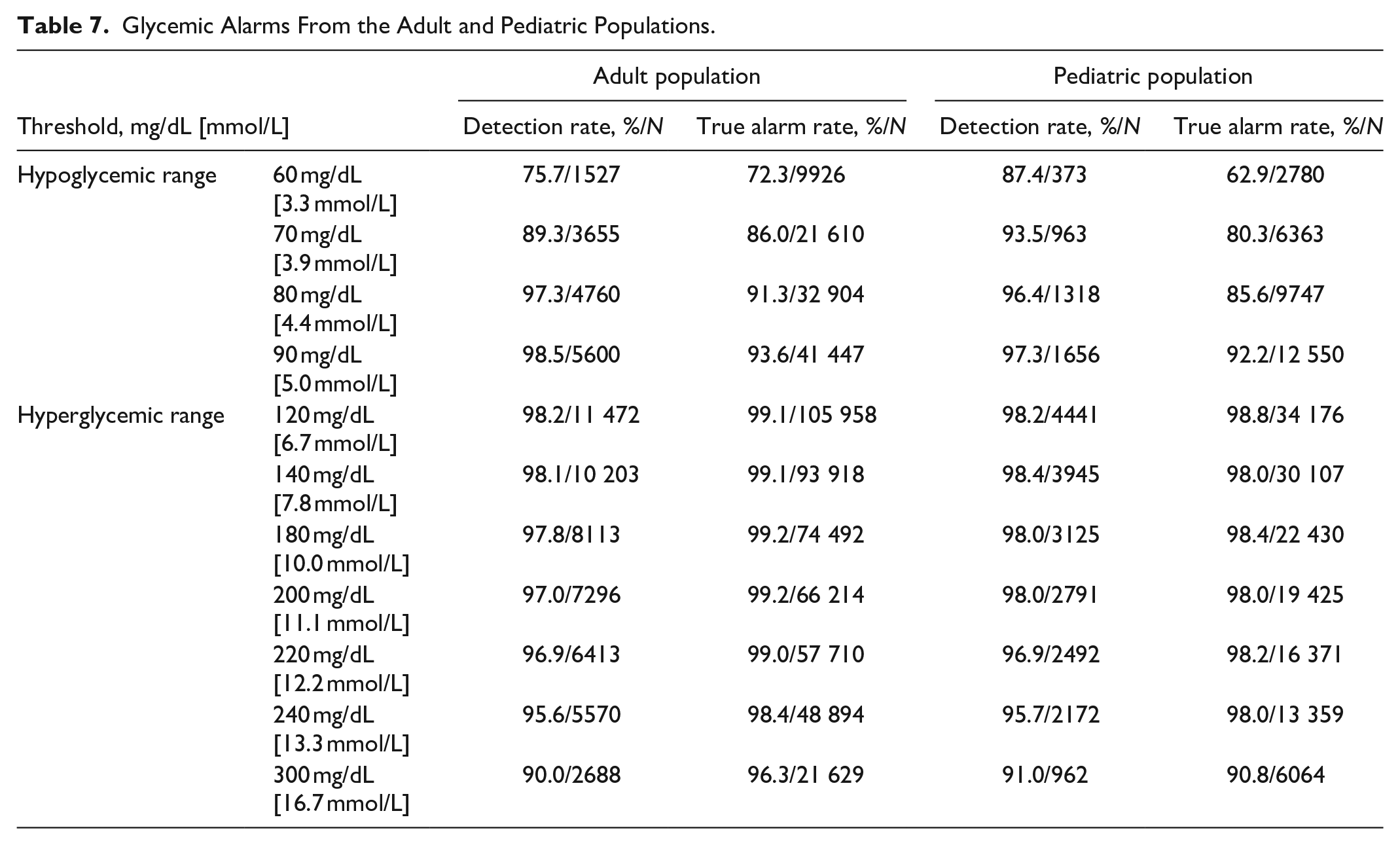
Glycemic Alarms From the Adult and Pediatric Populations.
The overall performance of the System as per age group is presented in Table 3. There were a total of 18 926 matched data pairs in the adult group and 6584 matched data pairs in the pediatric group. The MARD of the adult group was 9.2% (95% CI [8.7-9.9]) and of the pediatric group was 9.7% (95% CI [8.9-10.7]). (For the four- to five-year-old participants, the MARD against SMBG reference was 11.8%. For comparison, MARD for the adult population against SMBG was 10.7%.) The System showed that 93.2% and 92.1% of sensor readings were within 20% or 20 mg/dL of YSI reference for the adult and pediatric groups, respectively. The subgroup analysis did not show any marked difference in overall performance relative to age (P value = .6445), type of diabetes (P value = .2689), clinical site (P value = .3473), insulin administration (P value = .4216), or HbA1c (P value = .3554). The consensus error grid analysis 19 showed 93.2% and 92.6% of the data in the A zone and 99.9% and 100% of the data in the A+B zone of the error grid for the adult and pediatric groups, respectively.
Relative agreement at different YSI glucose ranges is presented in Table 4. Both the adult and pediatric studies exhibit similar agreement level across glucose ranges.
The percentage within 20% or 20 mg/dL of YSI reference and MARD performance throughout the wear duration was assessed for both adult and pediatric studies. The percentage within 20% or 20 mg/dL of YSI reference for both study populations (Table 5) remains consistent across the wear periods. Percentages of the data within 20% or 20 mg/dL of YSI reference for the adult and pediatric data on the first day of the sensor wear were 87.9% and 85.0%, respectively. The mean absolute difference (MAD) and MARD for both study populations (Table 6) remained consistently low across the wear periods. The MARD on the first day of the sensor wear for the adult and pediatric data was 11.2% and 12.1%, respectively.
The total drift estimated over the 14-day wear duration was 0.6% and -0.2% for the adult and pediatric populations, respectively. Estimated probability of sensor surviving 14 days was 71.1% for adults and 78.1% for the pediatric population. Of these, 4.1% for adults and 4.3% for pediatric population were due to the System terminating the sensor early, and the remaining failures were due to sensors getting knocked off during wear.
The alarm performance when alarms were set at different thresholds at low (60, 70, 80, and 90 mg/dL) and high (120, 140, 180, 200, 220, 240, and 300 mg/dL) glucose threshold levels is presented in Table 7.
The time lags between the venous reference and the sensor result for adults and pediatric population were 2.4 ± 4.6 minutes and 2.1 ± 5.0 minutes, respectively. The mean CVs between the sensors in adult and pediatric population were 5.7% and 5.8%, respectively.
Discussion
The FreeStyle Libre has allowed easy access to the sensing technology with over two million patients using the device to manage their diabetes. 20 The System with improved accuracy, specifically at the hypoglycemic ranges, also adds optional hypoglycemia and hyperglycemia alarms. The new System has the same form factor as the previous generation, and therefore the ease of use and comfort to wear have not changed. The System is factory-calibrated without the need for fingerstick calibration by the user.
Studies on the System with the new glucose algorithm have demonstrated a performance of 93.2% and 92.1% of the results within ±20% or ±20 mg/dL of YSI reference for adult and pediatric populations, respectively, with over 25 000 paired datapoints between the two studies—a performance that is comparable to that reported for the Dexcom G6 system (92.3%).21,22 The System performed well in the hypoglycemic range with 94.3% of the results for the adult population and 96.1% of the results for the pediatric population being within 15 mg/dL of the YSI reference. Accuracy of the sensor is stable over the sensor wear period and the performance on the first day of sensor wear (with the results within 20% or 20 mg/dL of the YSI reference was 87.9% for adults and 85.0% for pediatric populations) is comparable to the reported numbers for the G6 system (88.6%). 22 With this factory-calibrated system, the System did not show any marked difference in overall performance relative to age, type of diabetes, clinical site, insulin administration, or hemoglobin HbA1c. The overall MARD of the System is 9.2% (MARD for pediatric population is 9.7%) with the new algorithm, which compares favorably against the G6 system (MARD of the G6 for the adult and pediatric population are 9.9% and 10.1%, respectively, and the previous generation of the System had a MARD of 12.0% against YSI reference for the adult population).2,22
The new glucose algorithm reduces the time lag for the System to about two minutes (2.4 minutes for adults and 2.1 minutes for pediatric population) compared to the previous-generation the product (4.5 minutes, in a study without glucose manipulation). 2
These results have clinical implications for individuals with diabetes and for the clinicians who treat them. A sensor with a longer wear period that does not require fingerstick calibration with its associated burden supports more frequent sensor use with improved glycemic outcomes. Randomized controlled studies and real-world data have revealed better glycemic control with the use of the FreeStyle Libre System over a sustained period of time.9,11,23 This System with the optional alarm feature may benefit groups that have demonstrated poor adoption due to alarm fatigue with other CGM systems, wherein some of the alarms are not optional. 24
Conclusion
The performance of the System was demonstrated by the overall accuracy of sensor readings and the stability of accurate readings over 14 days of use. The System continues to provide the same easy-to-use and comfortable sensor wear experience for up to 14 days as its previous versions, without the need for fingerstick measurements. With the improved accuracy and the optional alarm feature, it is anticipated that the System will increase the adaption of sensor-based technology for management of diabetes by people with diabetes.
Footnotes
Acknowledgements
The authors acknowledge the study participants and the research staff at the study sites.
Abbreviations
BMI, body mass index; CGM, continuous glucose monitoring; CE, Conformit� Europ�enne; CSII, continuous subcutaneous insulin infusion; HbA1c, Glycated hemoglobin; MAD, mean absolute difference; MARD, mean absolute relative difference; MDI, multiple daily injection; T1D, type 1 diabetes; T2D, type 2 diabetes; SMBG, self monitoring of blood glucose; YSI, Yellow Spring Instruments.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SA, ESB, and HL are employees of Abbott Diabetes Care.
TB conducts research sponsored by Abbott, Capillary Biomedical, Dexcom, Diasome, Eli Lilly, Kowa, Lexicon, Medtronic, Medtrum, Novo Nordisk, REMD, Sanofi, Senseonics, Viacyte, vTv Therapeutics, Zealand Pharma, and has been a consultant, speaker, and/or advisory board member for Abbott, LifeScan, Metronom Health, Novo, Sanofi.
RB conducts research sponsored by Abbott, Medtronic, Roche, Novo, Lilly, Sanofi, and Senseonics.
KC conducts research sponsored by Medtronic, Dexcom, Abbott, Insulet, and Novo Nordisk, and has been a speaker for Dexcom and Abbott.
MPC conducts research sponsored by Abbott, Biolinq, Dexcom, Eli Lilly, Novo Nordisk, Medtronic, Insulet, Sanofi Aventis, Merck, and Senseonics.
GPF conducts research sponsored by Medtronic, Dexcom, Abbott, Tandem, Insulet, and Lilly, and has been a consultant, speaker, and/or advisory board member for Medtronic, Dexcom, Insulet, Tandem, and Lilly.
MK conducts research sponsored by Abbott, Dexcom, Eli Lilly, Medtronic, NGM Biopharmaceuticals, Tolerion, Pfizer, and Senseonics.
DRL conducts research sponsored by Abbott, Dexcom, Drawbridge Health, Eyenuk, Medtronic, Provention Bio, Senseonics, and Trial Net.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by Abbott Diabetes Care.
