Abstract
The price of insulin has increased dramatically over the past two decades. Medicare reimbursement for insulin is based on the route of administration via syringe (Part D) or via pump (Part B). Recently, the Centers for Medicare & Medicaid Services (CMS) announced a voluntary model for Part D enhanced plans that lower out-of-pocket costs to a co-pay of ≤$35/month. Meanwhile, the 21st Century Cures Act and manufacturer price increases raised the cost of insulin for pump users by 304% in less than 2 years. Because insulin is a life-saving necessity and unlike other Part B infused drugs, we call on CMS to determine drug reimbursement on a drug-by-drug basis and to develop a payment model that lowers out-of-pocket costs for patients using insulin delivered via pumps.
Keywords
Background
The price of insulin has increased dramatically over the past two decades. For example, the average list price (often referred to as the wholesale acquisition cost [WAC]) of one vial of insulin soared from $9.61 to $25.38 between January 2013 and July 2018, a 164% increase in less than 6 years (see Table 1 for the definitions of common terms used in drug pricing). Many issues related to the pharmaceutical supply chain have contributed to insulin’s high cost. First, all of the insulin purchased in the United States is sold by only three manufacturers, limiting market competition. Second, wholesalers, health insurance plans, pharmacy benefit managers, and other intermediaries drive up costs by negotiating undisclosed rebates and price concessions with one another. Third, generic insulin options are not widely available, although insulin was patented in 1923 and two generic products entered the market in May 2019. In addition, insulin reimbursement is determined by how the insulin is administered—via syringe (Medicare Part D) or via pump (considered durable medical equipment [DME] under Medicare Part B). (The Part D “syringe-injected” description as used here includes but is not limited to syringes and pens.)
The Difference Between AWP, ASP, and WAC.
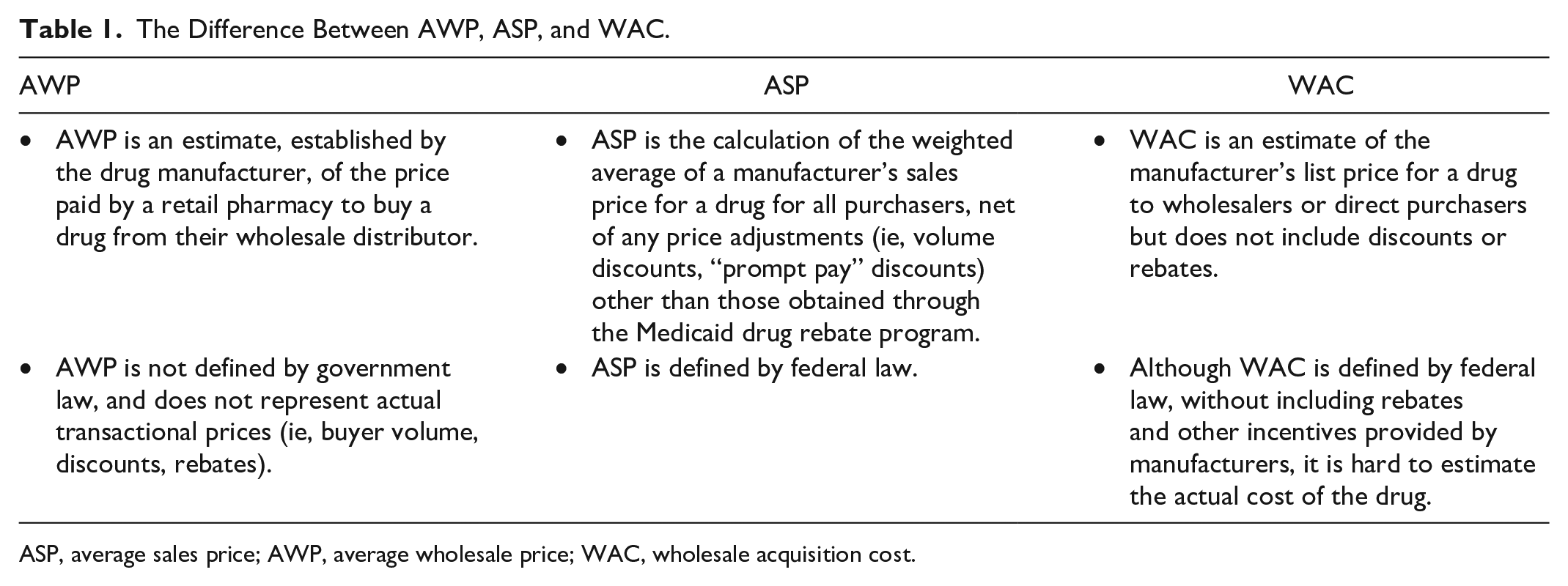
ASP, average sales price; AWP, average wholesale price; WAC, wholesale acquisition cost.
Medicare reimbursement levels are determined in part both by legislation, such as the 21st Century Cures Act, 3 and by the Centers for Medicare & Medicaid Services’ (CMS) administrative oversight. On March 11, 2020, CMS announced plans to lower out-of-pocket costs for syringe-injected insulin to a maximum of $35 copay per month using a voluntary model for Part D payments. Savings may or may not be passed along to patients as the model is voluntary, and beneficiaries must be enrolled in participating plans to benefit. Although this initiative may lower insulin costs for some patients who inject insulin, it does nothing to alleviate the financial burden for insulin pump users. They see an average copay of $54.26/month, >50% more than the model’s cap.
As for Medicare Part B, Section 5004 of the 21st Century Cures Act (“Reducing Overpayments of Infusion Drugs,” effective January 1, 2017) inadvertently contributed to insulin’s high cost by changing the reimbursement methodology for the approximately 30 DME-infused drugs, including pump-delivered insulin. 3 (The number of covered drugs typically fluctuates each quarter depending on the set of DME-infused drugs for which there were claims.)
Medicare Reimbursement for Pump-Delivered Insulin Before 2017
Before 2017, overall Medicare payments for DME-infused drugs exceeded average sales price (ASP). 1 The US Department of Health and Human Service’s Office of Inspector General (OIG) reported that Medicare overpaid $282 million for 21 of the approximately 30 DME-infused drugs to pharmacies, due to the use of 2003 average wholesale price (AWP) in reimbursement formulas. Figure 1 illustrates the difference in 2015 between using ASPs versus AWPs to calculate reimbursement for DME-infused drugs.
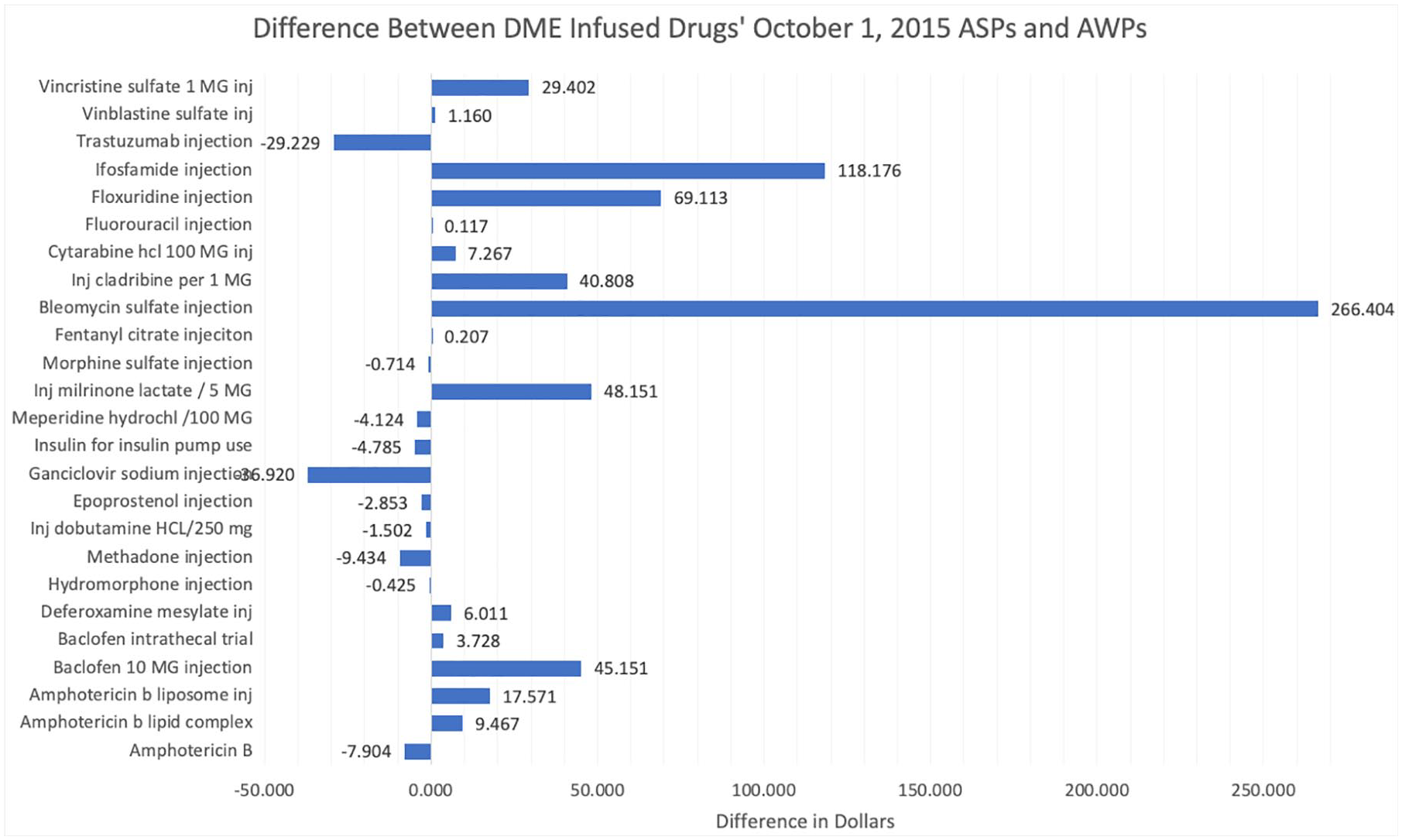
Difference between durable medical equipment (DME)-infused drugs’ average sales price (ASP) and average wholesale price (AWP). For example, pump-delivered insulin’s AWP is $4.785 less than its ASP.
A pharmacy acquires drugs from wholesalers at the AWP, then sells these drugs to patients where they receive a copay from the patient (where applicable) and a payment from the patient’s insurance (eg, Medicare). Before 2017, Medicare payments to pharmacies for Part B DME-infused drugs were set based on 95% of a drug’s October 2003 AWP.1,2 Thus, in 2013/2014, the Medicare payment for 25% of the approximately 30 DME-infused drugs covered under Part B was less than a pharmacy’s acquisition cost, and the Medicare payment for 75% of drugs covered under Part B was more than a pharmacy’s acquisition cost (Table 2). 2
Medicare Reimbursement in Q4 2015. 5

Because Medicare was underpaying for pump-delivered insulin, large suppliers stopped providing it to Medicare Part B beneficiaries, making it difficult for patients to fill their prescriptions. 5 In contrast, Medicare’s overpayment for other infused drugs created potential incentives for overutilization and improper billing. 5
Given the cost and reimbursement discrepancies, OIG recommended a legislative change requiring Medicare Part B payments for DME-infused drugs to be brought in line with other Part B drugs, which are based on 106% of current ASP,1,2 instead of using 2003 AWPs. 2 This recommendation was enacted with the 21st Century Cures Act. 3 However, simply adjusting payments for the two most highly overpaid DME-infused drugs (milrinone lactate and immune globulin), instead of revising the reimbursement for all DME-infused drugs, would have addressed 95% of the identified overpayments. Total savings would have been $267 million during the same time period, and insulin cost would not have been affected. 2
After the 21st Century Cures Act
For Medicare Part B beneficiaries using insulin pumps, the 21st Century Cures Act effectively raised the cost of insulin by 251% overnight (between December 31, 2016 and January 1, 2017; see Figure 2) without any notification to patients and providers. As the reimbursement rate of DME-infused drugs stayed steady at 2003 AWP, OIG found that the number of Medicare suppliers submitting claims for pump-delivered insulin decreased annually year after year from 2003 to 2016, creating an access issue for patients who needed to fill their prescriptions for pump-delivered insulin. Therefore, fewer and fewer patients were able to receive pump-delivered insulin at the 2003 AWP price and thus were indirectly affected by the sudden increase of 251% overnight in January 2017. Seventeen months later, manufacturers increased the price of insulin by another 53%. 4 Thus, the cost to patients increased by 304% in less than 2 years.
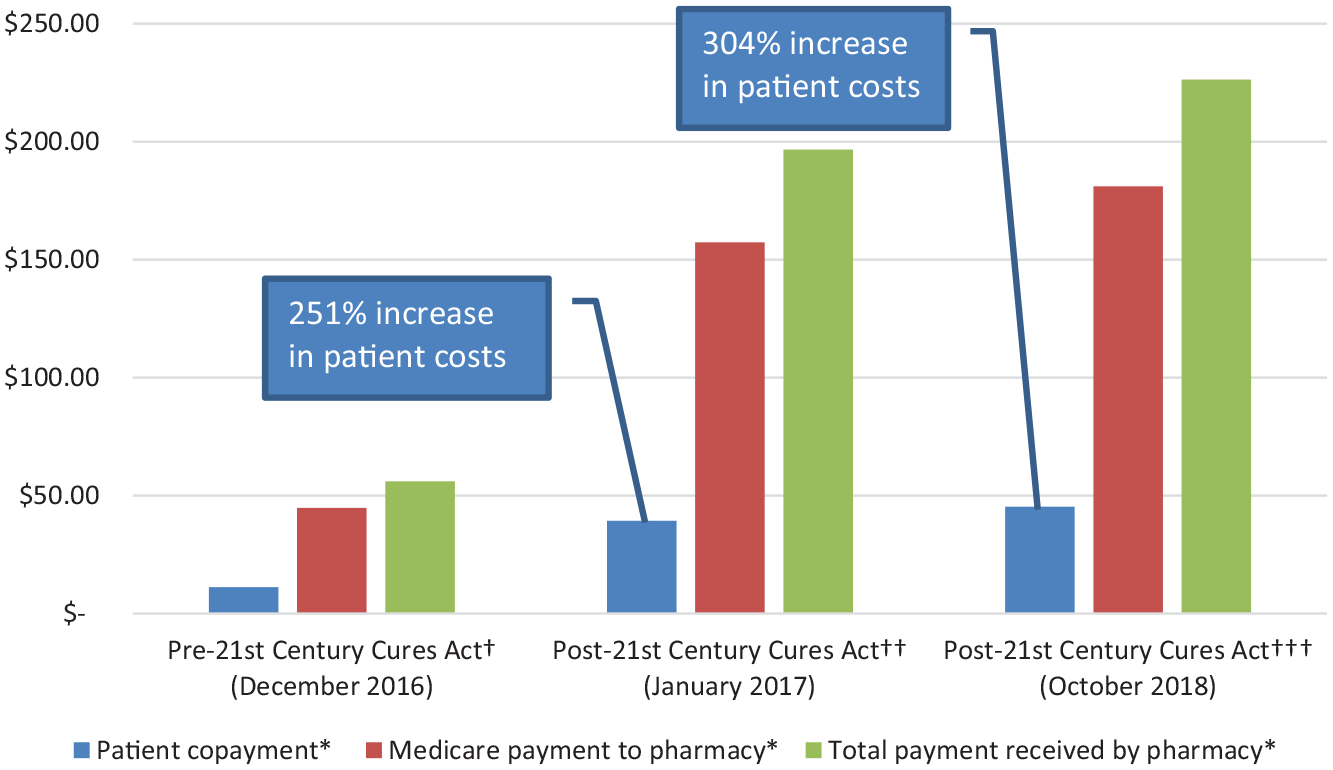
Economic impact of the 21st Century Cures Act on pump-delivered insulin. 4
One might argue that most Medicare Part B beneficiaries have additional coverage—Medi-gap or Medicaid—that lowers actual out-of-pocket costs. Yet, only 29% of Medicare beneficiaries have Medi-gap coverage. 6 A total of 6.1 million people have no supplemental coverage (19%) and an additional 21.5% of Medicare beneficiaries are on Medicaid, which leaves them vulnerable to even modest price increases.
Policy Changes Affected Patients Negatively
It is alarming that the high cost of insulin has been associated with subsequent underuse. For example, 25% of patients at an urban diabetes center reported that they used less insulin than prescribed, stopped using insulin, or did not fill an insulin prescription, actions associated with poor glycemic control. 7 More than one-third of these patients did not discuss the issue with their physician. Although the patients in this study were not identified as syringe versus pump users, 17.5% of the patients receiving Medicare Part D reimbursement reported cost-related underuse.
A survey of patients who started pump use after transitioning to Medicare found that 57.5% reported issues with obtaining supplies, and 29% were challenged by the cost of supplies; 64% said they left the site in place >3 days or reused pump supplies (34%) as a result. 8 These behaviors led to more erratic (48%) or higher (42%) blood glucose levels.
Call to Action
Because insulin is a life-saving, essential medicine for the 7.5 million Americans with insulin-dependent diabetes, we believe that it must be affordable for everyone who needs it. We call on CMS, the US Congress, and the medical community to help patients properly manage their diabetes by addressing the increasing cost of insulin. Therefore, we recommend that CMS:
Develop a payment model for Medicare Part B patients using insulin delivered via pumps that lowers their out-of-pocket copays ($54.26), in line with the voluntary Part D monthly copay ($35.00).
Determine drug reimbursement rates on a drug-by-drug basis and in consultation with specialists who prescribe the drug.
Test pricing methodologies before implementation.
Ask OIG to reanalyze costs to see if savings targets are being met.
Notify patients and their health care providers in advance of a change, so that they can better prepare for increased costs.
The current pricing methodology for a life-sustaining medicine, such as insulin, results in a significant economic impact over the lifetime of a person with diabetes, compared with other DME-infused drugs that are used at the end of life (eg, morphine sulfate) or for a shorter duration of time (eg, trastuzumab). Exorbitant insulin pricing resulting in nonadherence can negatively impact glycemic control and lead to poorer health and economic outcomes, which can lead to increased healthcare costs as a result of more hospitalizations and emergency room visits, and increased mortality rates.7,9 Insulin pump use results in the reduced risk of short-term diabetes complications and better glycemic control compared with injections,8,10 and yet, insulin pump users are penalized financially. The existing policy may drive pump users toward injection therapy and away from the option that helps them better manage their condition.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HC and RAV are employees of Medtronic Diabetes (Northridge, CA, USA).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The underlying analyses by JB-G were underwritten by Medtronic Diabetes (Northridge, CA, USA). Sarah Staples, MA, ELS, assisted with manuscript preparation.
