Abstract
Background:
No current technology exists to ensure the dose of insulin administered in hospitals matches the physician order.
Objective:
Assess the feasibility of using computer vision to identify insulin syringe preparation errors.
Methods:
Twenty-two nurses prepared 50 insulin doses (n=1100) each. A computer vision device (CVD) measured the volume drawn up and identified air present. Syringes identified as inaccurate by the CVD were confirmed by two observers, and a random sample of 100 syringes identified as accurate was validated by two independent observers.
Results:
Ten syringes (1.0%) had the wrong volume prepared, and 68 syringes (6.5%) contained air sufficient to meet the definition of inaccuracy. All errors were confirmed by two independent observers.
Conclusion:
CVDs could reduce insulin administration errors in hospitalized patients.
Introduction
Insulin is a well-recognized high-alert medication, 1 and its misuse is associated with a high capacity for patient harm. Insulin-dosing schemes are uniquely complicated and require consideration of patient resistance, acute illness, nutrition status, and unique pharmacokinetics of various insulin types. Automated workflows, including Barcode Medication Administration (BCMA), have been implemented in inpatient settings to ensure correct product selection, but this technology does not validate the dose prepared for administration.
Dose preparation of insulin is a highly complex workflow, particularly in the inpatient setting. For rapid-acting and short-acting insulin administration, nurses must obtain a point-of-care (POC) glucose value, determine the indicated nutritional and correctional doses, and coordinate insulin administration with nutrition. Oftentimes, nurses must complete this workflow for multiple patients under their care in a narrow time window. BCMA is recognized as a best practice standard to ensure the selection of the correct insulin, but many health systems rely on manual processes for dose calculation, preparation, and timing of administration.
To mitigate potential “wrong dose” errors, some hospitals have implemented pharmacy preparation of long-acting insulin doses. However, removing insulin preparation from the bedside can contribute to missing doses, late administration, and waste associated with dose changes made following preparation and distribution. Additionally, pharmacy preparation of rapid-acting insulin is not feasible given the coordination required with POC glucose assessments and nutrition. In situations where doses are prepared in patient care areas, independent double-check workflows have been pursued but have failed to demonstrate significant error reduction. 2
Very little is known about insulin “wrong dose” errors related to inaccurate syringe preparation in patient care areas. Given the manual nature of dose preparation, facilities are reliant on voluntary reporting of errors, which does not provide an indication of overall error frequency and magnitude.3,4 Studies have documented poor accuracy for dose preparation of 2 units or less when accuracy was measured by gravimetric or scintigraphic methods.5-8 Nguyen et al 9 identified one incorrect dose out of 229 observed (0.4%) during an observational study.
The primary goal of this study was to explore the feasibility of using a computer vision device (CVD) to measure insulin doses prepared from a vial and syringe and to understand the frequency and magnitude of “wrong dose” insulin errors in a nonclinical setting.
Methods
This proof-of-concept observational study recruited 22 nurse volunteers to prepare 50 syringes of regular insulin (Humulin R vials, Eli Lilly, Indianapolis, Indiana) stored at room temperature using 50-unit insulin syringes (Becton-Dickinson, Franklin Lakes, NJ, USA). Study participants were asked to anonymously complete a survey documenting their years of clinical experience and the frequency with which they prepare insulin doses using vial and syringe (Table 1). The study protocol was evaluated by the hospital institutional review board and given exempt status.
Nursing Reported Frequency of Insulin Preparation from Vial and Syringe.
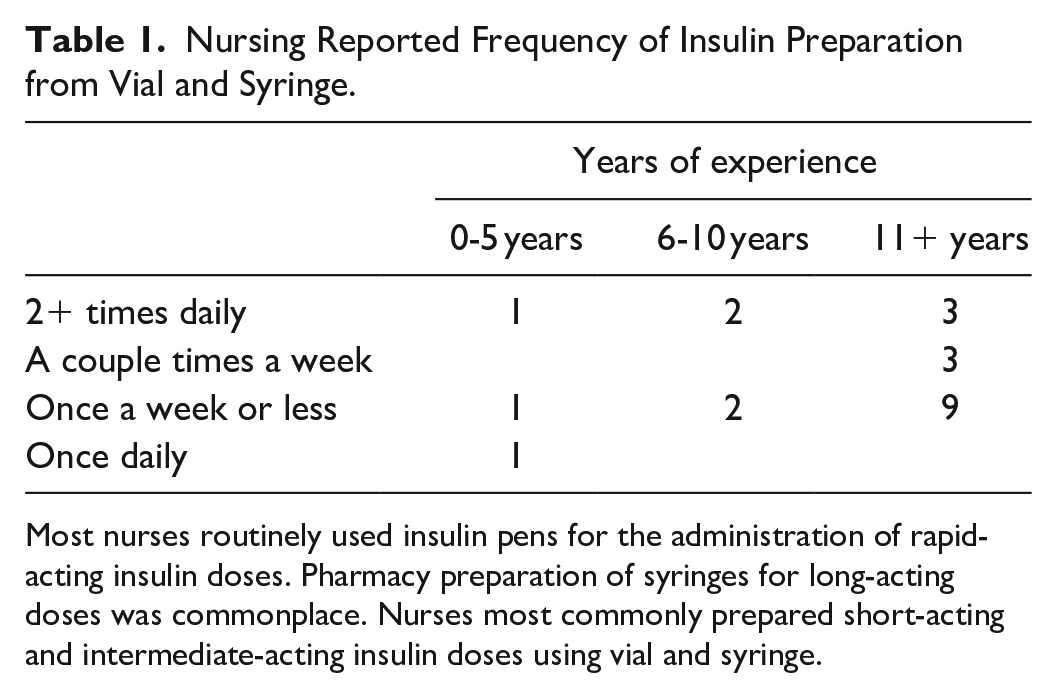
Most nurses routinely used insulin pens for the administration of rapid-acting insulin doses. Pharmacy preparation of syringes for long-acting doses was commonplace. Nurses most commonly prepared short-acting and intermediate-acting insulin doses using vial and syringe.
A random number generator provided the target dose for preparation. Target doses were integers ranging from 5 to 45 units. Doses were randomized 2:1 for doses of ≤25 units to more closely mimic doses commonly encountered in daily nursing practice. After the target dose was displayed, the syringe was prepared and placed on the syringe platform. The nurse then pressed the GO button on the CVD touch screen, which triggered image capture and automatically advanced to display the next target dose (Figure 1) regardless of preparation accuracy. The CVD also captured the date/time of preparation, target dose, and the dose measured by CVD. An additional algorithm was used to estimate the amount of air in each syringe.

The CVD instructed the nurses to prepare a randomized target dose ranging between 5 and 45 units. Following preparation, the nurse would place the syringe on the image capture platform and press “go”. Following image capture, the nurse would remove the syringe from the platform and place it in the “out” bin.
Error was defined as a deviation from the target dose by greater than 0.5 units for target doses from 5 to 10 units and greater than 1 unit for target doses from 11 to 45 units. Error definitions were established using criteria that could be validated by the human eye to align with current workflow practices.
Two observers independently confirmed the inaccuracy of all syringes identified by the CVD; the observed doses were recorded to the nearest 0.5 unit. To ensure all inaccuracies were identified, two independent observers validated a random sample of 100 syringes identified as accurate; the observed dose was recorded to the nearest 0.5 unit. If the two independent observers did not agree on the dose in a syringe, a third observer independently evaluated the dose.
Measurable outcomes included the frequency and magnitude of insulin syringe preparation errors, by preparation volume and the presence of air. Syringes determined to be unusable due to poor image quality were excluded from further analysis.
Descriptive statistics were used to characterize the inaccuracies noted.
Results
Of the 1100 syringes prepared, 1043 useable images were obtained. Reasons for exclusion are described in Table 2. Human errors while taking the picture accounted for 80.8% (n = 46), while 19.2% (n = 11) were attributed to overexposure to lighting.
Syringe Exclusion Criteria.

Error Frequency
Errors were observed in 78 (7.5%) syringes. Sources of inaccuracy included preparation of the wrong volume (10, 1%) and significant air (68, 6.5%). All CVD detected errors were validated by two independent observers.
Error Magnitude
Syringes prepared to the wrong volume line (n = 10) ranged from 5.5 units under the target to 3 units over the target dose (mean [SD] = 1.8 [2.3] units). The doses prepared ranged from 75% to 113% of the intended target (mean [SD] = 10.5 [12.7]%). Magnitude of error was also validated by two independent observers.
The CVD was unable to quantify the volume of air present beyond the thresholds defined for error. These syringes (n = 68) were measured by two independent observers to contain 1 to 15 units of air (mean [SD] = 3.8 [3.1] units). Doses in syringes with air ranged from 25% to 97% of the intended target dose (mean [SD] = 78.5 [15.6]%).
Discussion
This study evaluated syringe preparation errors in a nonclinical setting to understand the fundamental risk for error when distractors are removed.
The CVD accurately captured both errors due to the wrong volume and the presence of significant air bubbles in the syringe.
However, a significant number of syringes had to be excluded from analysis due to inconclusive images. Nurses were given one opportunity to prepare and take the image of the intended dose; the system was not configured to prompt the nurse for a second image in the event of an “inconclusive” result.
The study setting possibly contributed to the high frequency of syringes with significant air, if the nonclinical setting encouraged participants to expedite syringe preparation. Twelve nurse participants reported preparing insulin from a vial and syringe once a week or less, which also may have contributed to poor preparation techniques and a higher incidence of air pockets. However, it is possible that preparation errors are more common in a nursing unit where there are frequent distractions, and multitasking is inevitable. If the frequency of error observed is replicated in a clinical setting, improving dosing accuracy could be a significant opportunity for glycemic quality improvement.
Future studies will be designed to assess strategies for incorporation into nursing workflows in a simulation-type environment, involving time studies and workflow process mapping. System functionality will be revised to include real-time assessment of dose appropriateness and prompts for a second picture when results are inconclusive. Additionally, lighting requirements will be more clearly defined to ensure optimal image quality. Integration with electronic health care documentation and dose calculators will be pursued to further mitigate the risk of dosing errors.
Conclusions
Many complex workflows have been developed to promote safe administration, but technological solutions to ensure safety are lacking. The use of a CVD could be used to confirm the accuracy of doses prepared and minimize the risk of over or under dosing events.
Footnotes
Acknowledgements
The authors would like to thank Mr Robert Thai for providing the illustrations.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kevin Brown, PharmD, is Chairman of the Board of Digital Hospital, Inc., developer of an insulin dose confirmation device. The other authors have no potential conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Digital Hospital, Inc. provided funding for this research.
