Abstract

Keywords
Insulin is a drug with a very narrow therapeutic index and a high incidence of dosing errors. 1 To add to that, it is administered more often by the patient himself at home than by a qualified nurse or doctor. We hereby report two cases of insulin dosing error that occurred due to the availability of two different insulin concentrations—40 IU/mL and 100 IU/mL—and a multitude of delivery devices.
A 16-year-old girl, on treatment for Type 1 diabetes mellitus (DM) for the last 6 years, presented in severe diabetic ketoacidosis (DKA). There was no history of any missed doses or any other risk factor for precipitating DKA. She was taking 30 units of premixed (regular and neutral Protamine Hagedorn [NPH]) 30/70 insulin in the morning and 20 units at night (around 1 unit/kg/day). Though not very well calibrated, this dose seemed adequate to keep her out of DKA. There was no identifiable precipitating cause for DKA. She was using an insulin pen with insulin cartridges to inject insulin. On inquiring, we found that the girl was taking insulin drawn from a 40 IU/mL vial and injected with a 40 IU/mL syringe till a few months back when she was introduced to the pen. She found the insulin pen less painful and more convenient to use, but the cost was high. However, the insulin cartridges were expensive, while the vials were available free from a government store. So she started refilling the cartridges with insulin drawn from a vial without knowing that the cartridge was calibrated for insulin of 100 IU/mL concentration (Figure 1 and Video 1). As a result, she got insulin only 0.4 times the required dose.
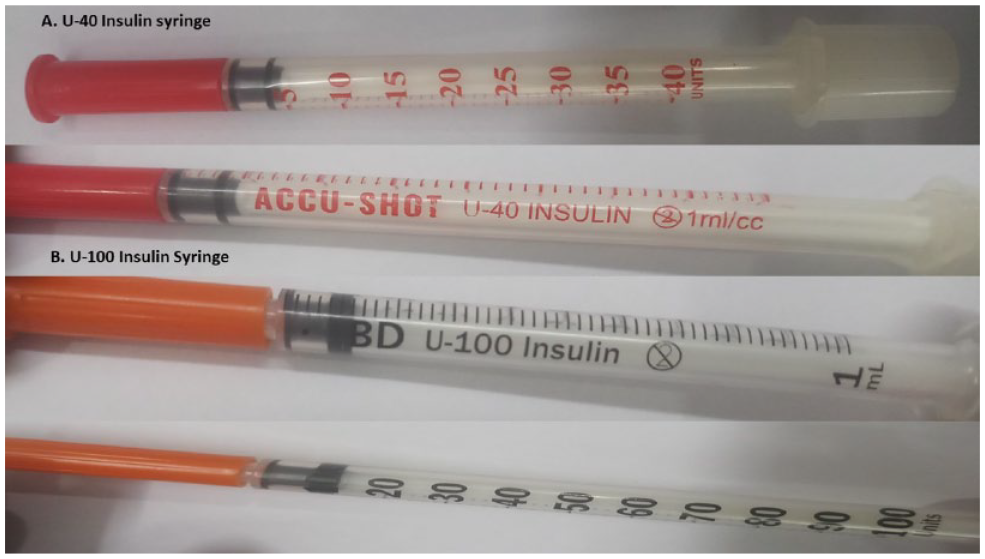
Image showing U-40 and U-100 insulin syringe, the orange and red caps can be confused to be shades of the same colour.
In another case, a 65-year-old man, known case of Type 2 DM on premixed insulin (regular and NPH) 30/70, got his fresh supply of insulin and new syringes from two different sources. While he got insulin vials of 40 IU/mL, he got syringes calibrated for 100 IU/mL which again led to underdosing. These are just two cases that came to us where insulin dosing errors occurred due to an incorrect delivery device. It is hence essential that insulin syringes and vials/cartridges have color-coded indicators that can help match insulin with the appropriate delivery device.
The insulin manufacturing companies and drug regulators should immediately take this into notice and do the needful. The problem of U-40 and U-100 insulin has been recognized in the past. 2 With the introduction of more concentrated insulins, such as U 300 and U 500, the problems of insulin dosing shall be further compounded. 3 In developed countries, insulin errors are known to occur even in inpatient settings. 4
In the opinion of the authors, the best solution to this would be to start color-coding insulin syringes/pens and their compatible insulin solutions, for example, insulin with a concentration of 100 IU/mL should have a band/sign of a particular color around the vial/cartridge and the insulin syringe/pen/delivery device calibrated to deliver 100 IU/mL should have the same.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
