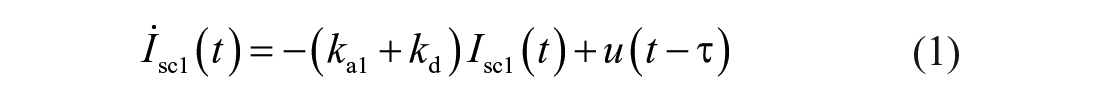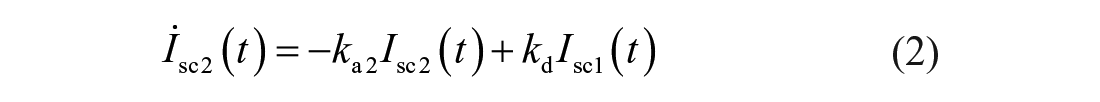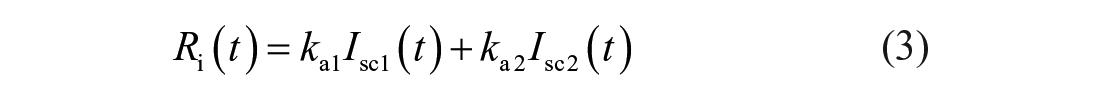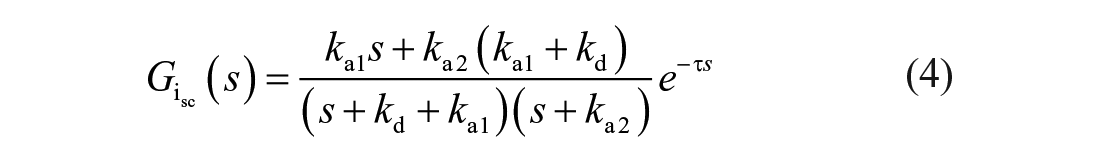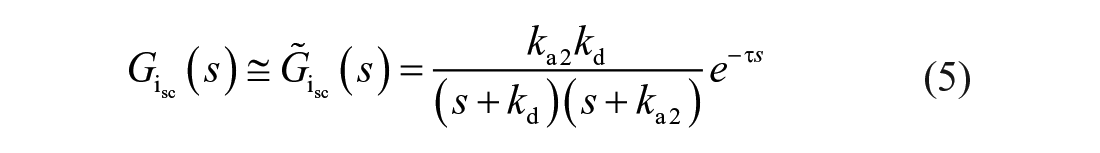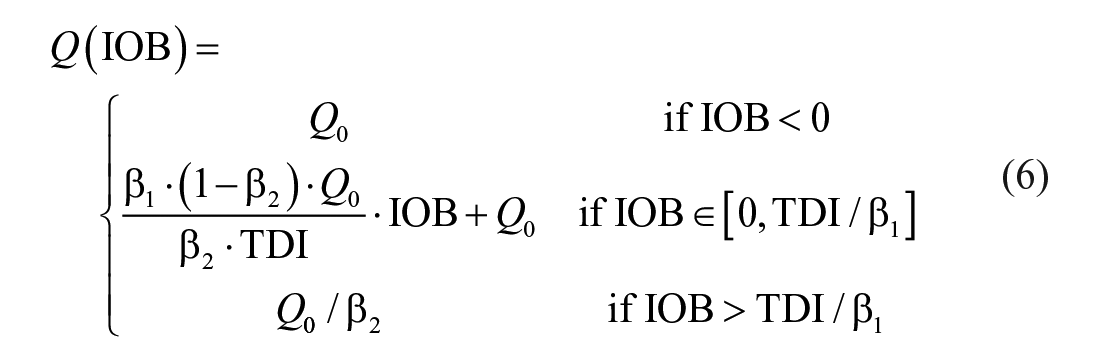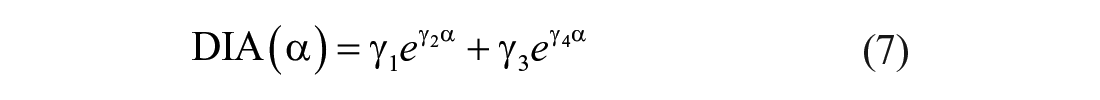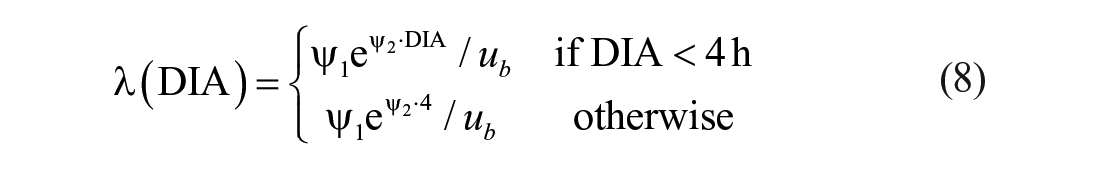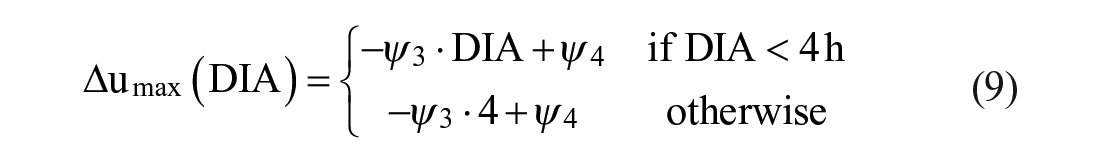Abstract
Background:
Controlling postprandial blood glucose without the benefit of an appropriately sized premeal insulin bolus has been challenging given the delays in absorption and action of subcutaneously injected insulin during conventional and artificial pancreas (AP) system diabetes treatment. We aim to understand the impact of accelerating insulin and increasing aggressiveness of the AP controller as potential solutions to address the postprandial hyperglycemia challenge posed by unannounced meals through a simulation study.
Methods:
Accelerated rapid-acting insulin analogue is modeled within the UVA/Padova simulation platform by uniformly reducing its pharmacokinetic time constants (α multiplier) and used with a model predictive control, where the controller’s aggressiveness depends on α. Two sets of single-meal simulations were performed: (1) where we only tune the controller’s aggressiveness and (2) where we also accelerate insulin absorption and action to assess postprandial glycemic control during each intervention.
Results:
Mean percent of time spent within the 70 to 180 mg/dL postprandial glycemic range is significantly higher in set (2) than in set (1): 79.9, 95% confidence interval [77.0, 82.7] vs 88.8 [86.8, 90.9] ([Note to typesetter: Set all unnecessary math in text format and insert appropriate spaces between operators.] P < .05) for α = 2, and 81.4 [78.6, 84.3] vs 94.1 [92.6, 95.6] (P < .05) for α = 3. A decrease in percent of time below 70 mg/dL is also detected: 0.9 [0.4, 2.2] vs 0.6 [0.2, 1.4] (P = .23) for α = 2 and 1.4 [0.7, 2.8] vs 0.4 [0.1, 1.4] (P < .05) for α = 3.
Conclusion:
These proof-of-concept simulations suggest that an AP without prandial insulin boluses combined with significantly faster insulin analogues could match the glycemic performance obtained with an optimal hybrid AP.
Keywords
Introduction
Postprandial glycemia makes a substantial contribution to overall glycemic control in diabetes treatment. Unfortunately, meeting postprandial glycemic target values has been challenging due to slow absorption and action of subcutaneously injected insulins. Insulin secretion from a healthy β-cell is a highly dynamic process, where glucose is the main stimulator of insulin release, leading to the characteristic biphasic pattern consisting of a brief first phase of insulin secretion (~10 minutes), followed by a sustained second phase. The earliest secreted insulin is a necessary element to offset the rapid rise in postprandial blood glucose. Unlike the rapid physiologic action of insulin after its release from a healthy β-cell, the maximum glucose lowering action from a subcutaneously injected insulin could be observed as late as 90 minutes to two hours after its injection.1,2 The underlying reasons for delay in insulin action are multifactorial, with chemical properties of insulin and factors concerning subcutaneous (SC) tissue being the principal contributors. 3 Moreover, subcutaneously delivered insulin may pose additional glycemic risks due to its prolonged action (up to six hours), potentially increasing the risk of late postprandial hypoglycemia.
A single-hormonal artificial pancreas (AP) system optimizes insulin delivery in real time, every five minutes, based on changes in sensor glucose levels. While most current systems function best with a premeal insulin bolus (hybrid AP), a fully automated system would not benefit from this sharp and early increase in circulating insulin. Consequently, a fully automated AP insulin controller reacts to meals only after sensor glucose levels begin to rise. Besides, there is no insulin depot delivered in to the SC area as the insulin delivery is spread over hours in mini boluses; therefore, the delay in insulin absorption and action is further exacerbated during fully automated AP, representing one of the main barriers to its implementation.4,5 Thus, the most common strategy is to define a single- or dual-hormone system with a hybrid controller, where feedforward insulin boluses are manually delivered at mealtimes, and the control law takes over the basal rate.6-11 The drawback associated with this design is that manual priming requires user assessment of the total amount of carbohydrates for every meal, which is a burdensome and potentially inaccurate task for patients.12,13
Other insulin delivery routes than SC have been explored to generate more physiological plasma insulin profiles. For example, inhaled human insulin has shown tangible benefits with respect to SC insulin injections. 14 However, this scheme also depends on prandial manual doses. Another alternative is to deliver insulin into the intraperitoneal (IP) space to minimize delays. 15 For instance, fully automated AP combined with IP insulin delivery provided superior glucose control to that with SC insulin delivery in a short demonstration study. 16 Nevertheless, this approach’s clinical application is still limited by its inherent costs and risk profile. 17
Although fully automated AP has been successfully deployed in clinical studies,18-24 there is an undeniable compromise between the controller’s aggressiveness and insulin stacking due to the extended duration of insulin action (DIA). An ideal insulin analogue should mimic the pharmacokinetic (PK) and pharmacodynamic (PD) profiles of endogenous insulin to optimize exogenous insulin treatment. Rapid-acting insulin analogs with faster PK/PD profiles have been introduced recently toward this goal,25-27 but a significant unmet need for more rapid insulin absorption that provides superior postprandial glucose control remains, particularly as new AP technology enters clinical care.1,28
In this work, we delve into how much the analogue insulin lispro (LIS) glucodynamic action could be accelerated to safely increase the controller’s aggressiveness in a SC AP with a model predictive control (MPC) law. To this end, we leverage the UVA/Padova simulator 29 to test the performance of the proposed controller in scenarios that include both announced and unannounced meals and different synthetic insulins.
Methods
Model of Insulin Pharmacokinetics
In this article, we consider the two-compartment PK model of SC fast-acting insulin that was presented in Ref. 30 and later updated in Ref. 31 :
where
In Silico Generation of Faster Insulin Analogues
The model described by Equations (1) to (3) is a second-order time-delay linear time-invariant system with the following transfer function:
As shown,
In order to define faster insulin analogues, we accelerate the insulin absorption from the SC tissue by manipulating only the poles of
The bandwidth of a system is commonly defined as the lowest frequency satisfying −3 dB from its gain at zero frequency. Note that if the average bandwidth of the PK model for LIS is
In order to determine the PK/PD properties of the
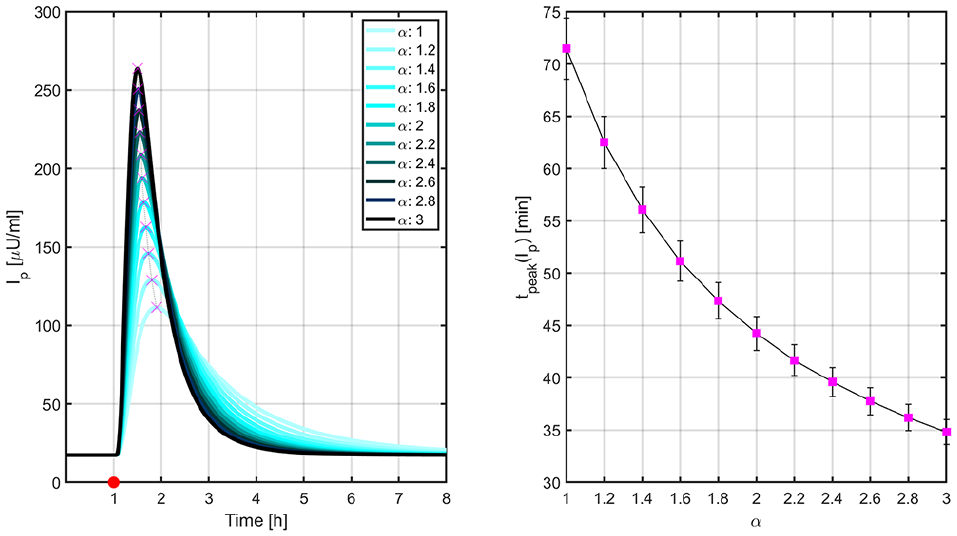
Mean PK profiles (left) and times to peak insulin levels (right) for different values of
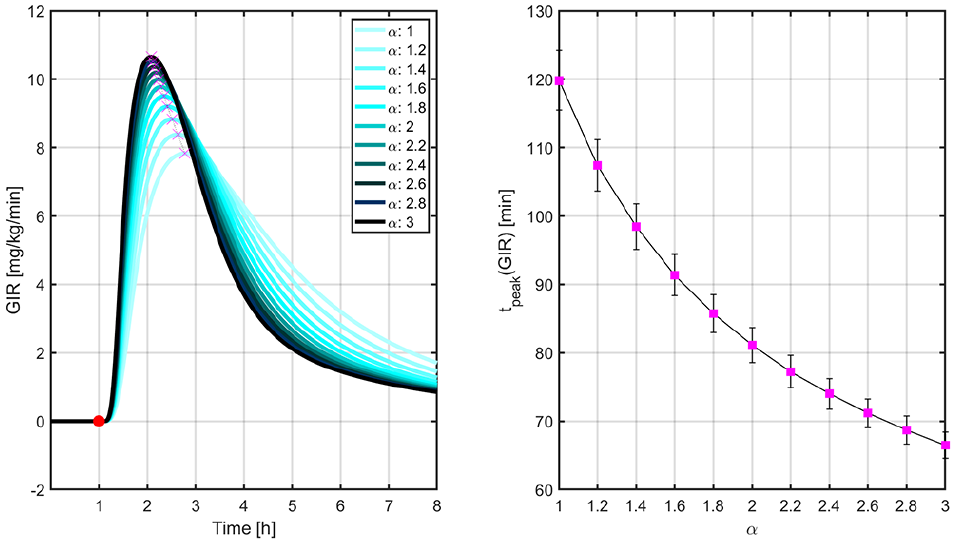
Mean GIR profiles (left) and times to peak GIR levels (right) for different values of
In Ref.
32
, this euglycemic glucose clamp was carried out on 38 adult patients with T1D to compare the PK/PD properties of LIS and ultra-rapid BioChaperone LIS
27
(BC-LIS). Results demonstrated that times to maximum insulin levels and GIR occur 20 and 30 min earlier, respectively, with BC-LIS. Bearing this in mind, and for merely illustrative purposes, we can associate BC-LIS with
MPC for Regulating the Blood Glucose Level
To assess the impact of faster insulins on the performance of an AP, we consider an originally hybrid MPC law as a baseline. This control strategy has been published by the authors elsewhere, 33 and a summary of its formulation is provided in the Appendix.
Detuning of the MPC controller aggressiveness (
)
In a hybrid AP approach, it is assumed that meal disturbances are mostly mitigated by feedforward insulin boluses that are delivered at mealtimes. In this case, the user needs to calculate the prandial dose based on, among other factors, the meal size in grams of carbohydrates (gCHO) and his/her insulin-to-carbohydrate ratio (CR) in gCHO/U. In order to avoid a controller overreaction to postprandial glucose excursions, the scalar weight
where
Detuning of
and
Long delays in insulin peak and duration substantially limit the achievable sensitivity of an AP to glucose deviations, because an aggressive control law can lead to late hypoglycemia due to insulin stacking. Here, we propose to re-tune the controller’s aggressiveness based on the dynamics of the insulin analogue: the faster the insulin analogue is, the more aggressive the controller can be. To this end, the average DIA was calculated for several
with
Numerical values of the tuning parameters
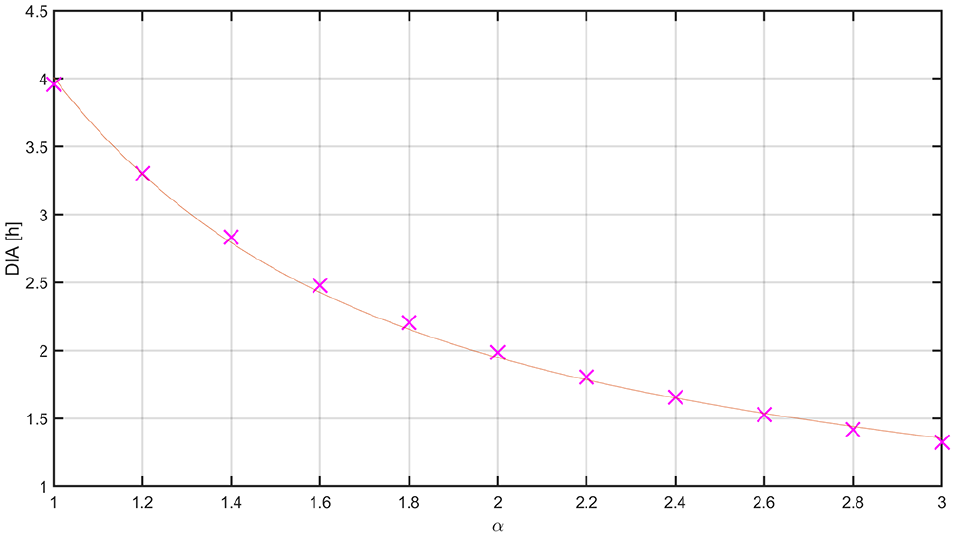
Mean DIA for different values of
Tuning Parameters of the MPC Law.
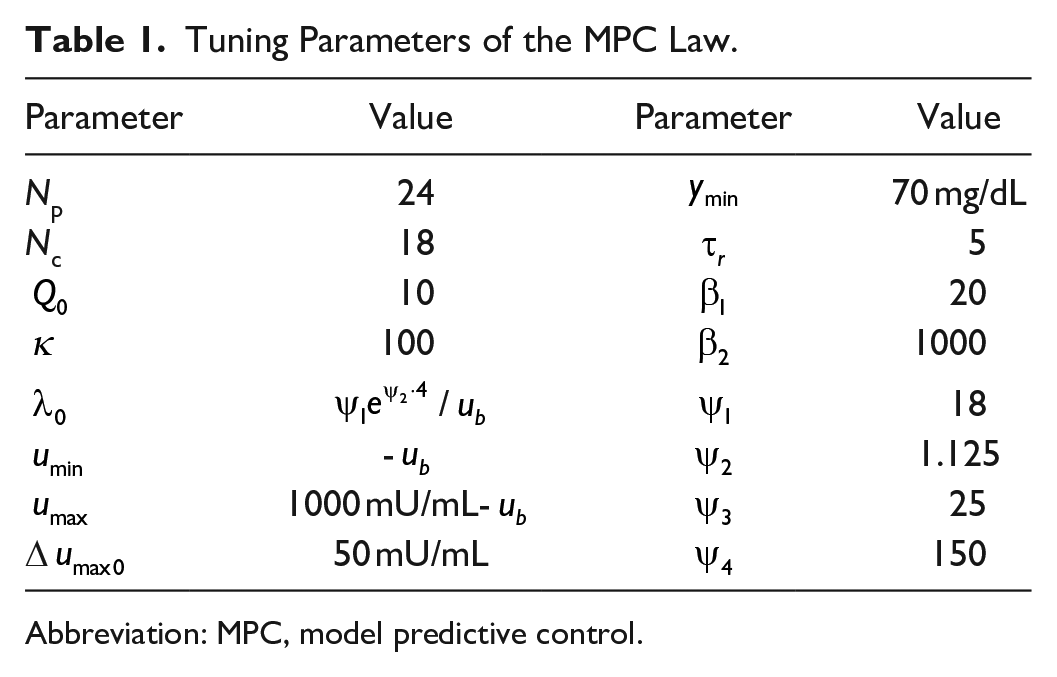
Abbreviation: MPC, model predictive control.
Results
In this section, a battery of tests is presented to evidence the impact of accelerating insulin absorption and action on postmeal hyperglycemia mitigation with a fully automated AP controller. To this end, 12-hour simulations that include different
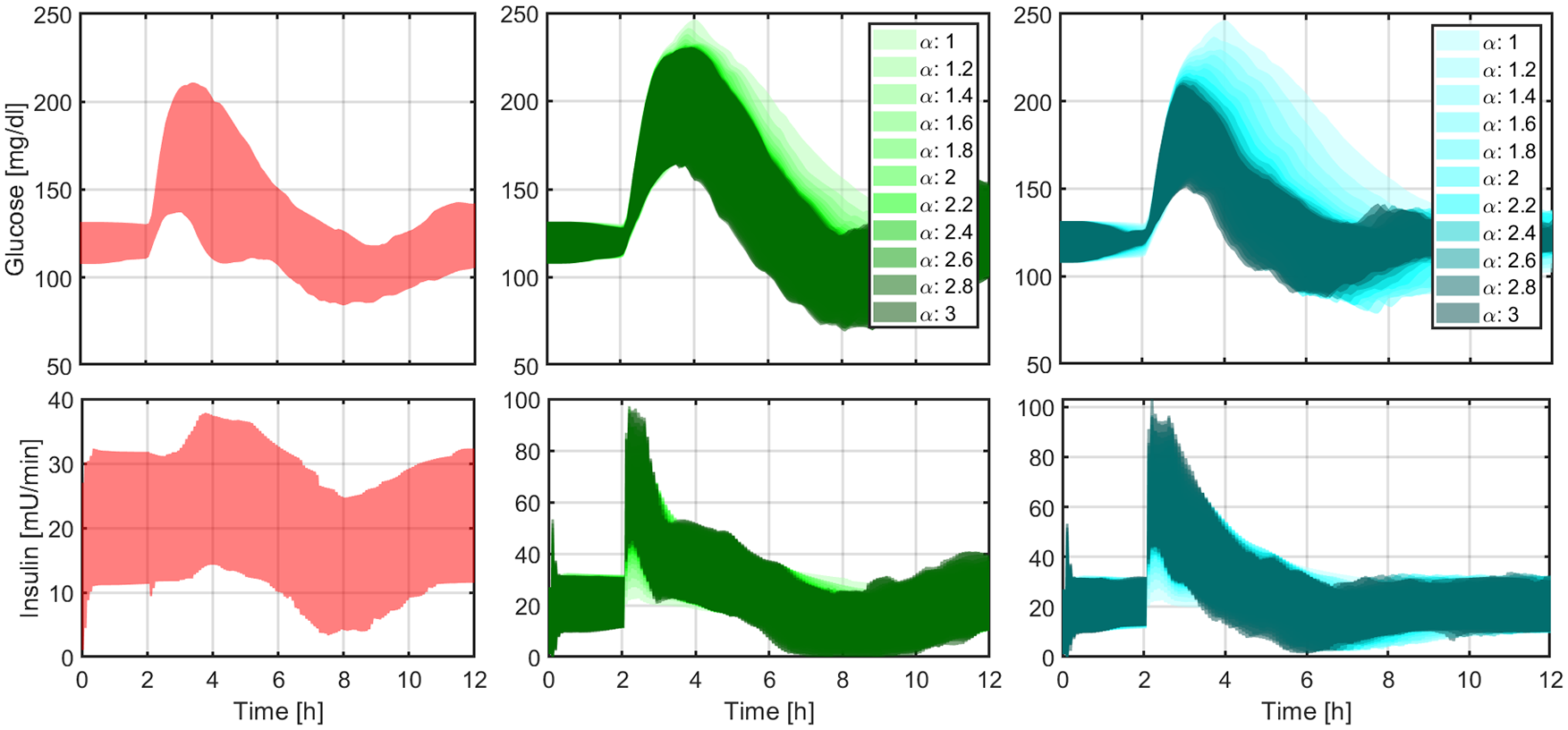
Closed-loop responses obtained with LIS to an announced meal (left panel), with LIS and different levels of controller’s aggressiveness to an unannounced meal (middle panel), and with
Comparison Between Numerical Results Related to Each Set of Closed-Loop Simulations.
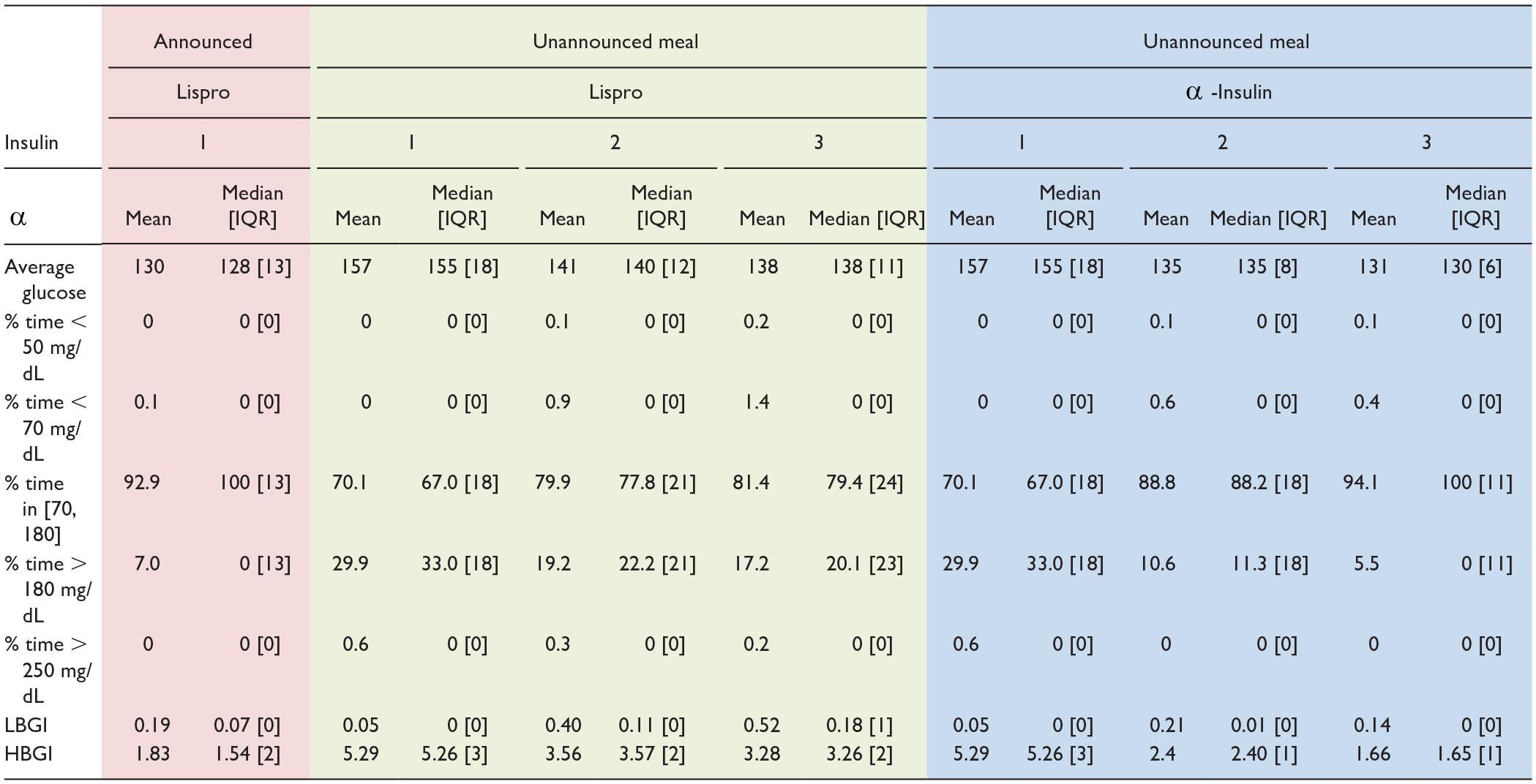
Abbreviations: HBGI, high blood glucose index; IQR, interquartile range; LBGI, low blood glucose index.
Glycemic Control Using a Hybrid Approach
To define a baseline of hybrid glucose control, a first set of simulations is carried out with LIS and meal announcement, that is, delivering feedforward meal boluses at mealtimes. Given the meal size
Glycemic Control With Unannounced Meals
Here, we eliminate the prandial bolus and gradually increase the controller’s aggressiveness. To this end, we tune the MPC using Equations (8) and (9) but keep using LIS in the simulations, that is,
The final step is to repeat these simulations but switching from LIS to the corresponding accelerated
Figure 5 indicates how the percentages of time <70 mg/dL and >180 mg/dL evolve with LIS and the
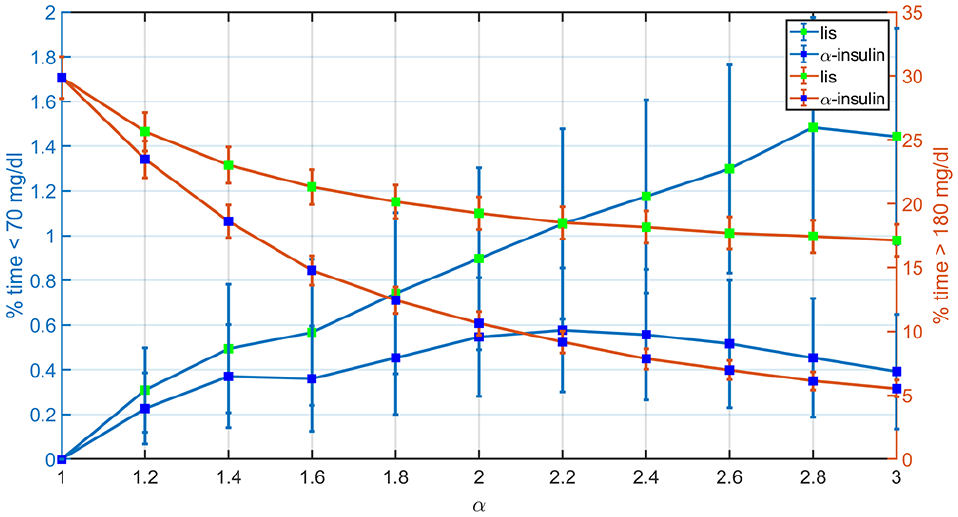
Comparison between the mean percentages of time <70 mg/dL and >180 mg/dL achieved with LIS and
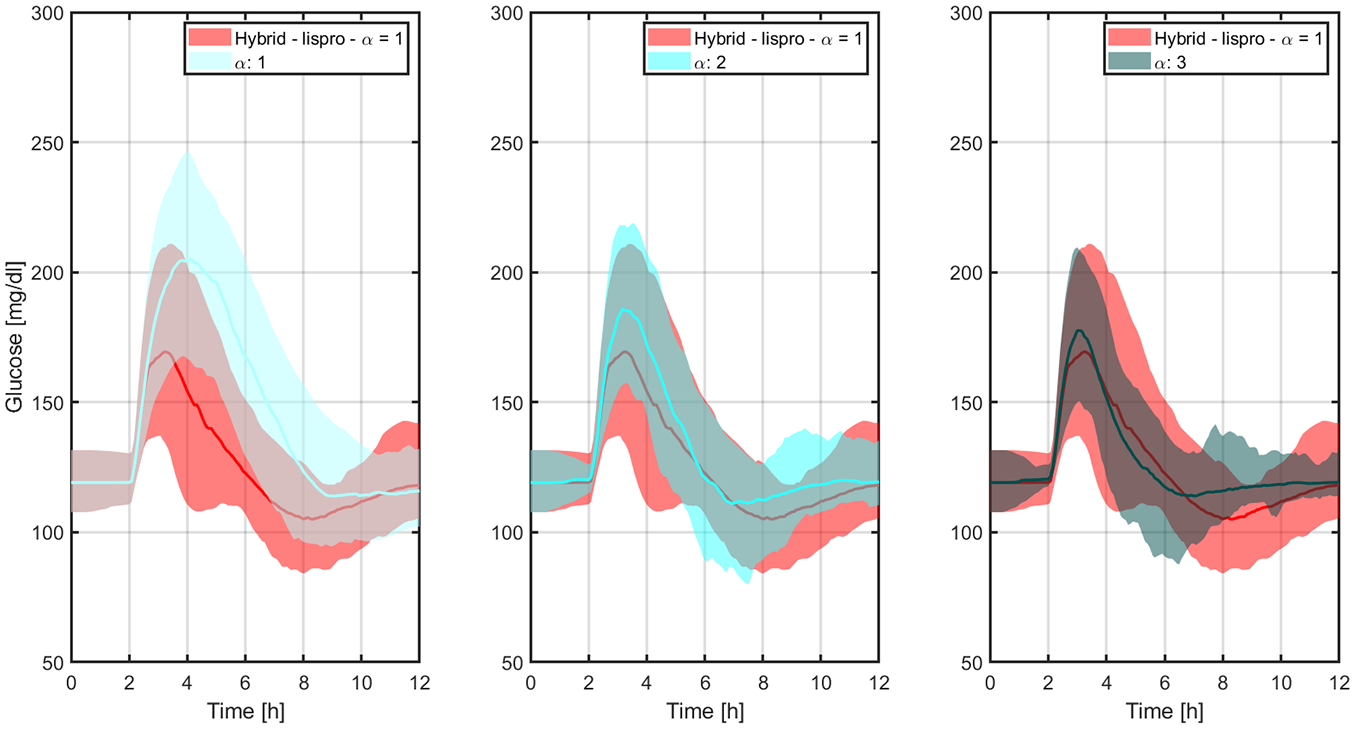
Comparison between the glucose trajectories obtained with LIS and the baseline hybrid controller, and the ones obtained with
It is important to emphasize that if the acceleration of the insulin analogue is not accompanied by an increase in the controller’s aggressiveness, then the benefits of faster insulins in glucose control are less noticeable. For instance, if
Discussion
Hybrid AP systems rely on feedforward insulin boluses to manage postprandial glucose excursions and on the glucose controller to maintain normoglycemia by modulating the basal insulin delivery. Users play a key role in this scheme since carbohydrate counting is cornerstone for meal insulin bolus calculation. Although this method reduces the stress on the controller, it is burdensome for patients and prone to human errors that may affect the achievable glucose control performance. One alternative is to eliminate the meal announcement from the control structure and tune the controller to be more reactive to glucose deviations. The longer the time to peak, the more sensitive the controller needs to be to alleviate postprandial hyperglycemia. As evidenced in Figure 4, if the baseline hybrid MPC is used without meal boluses (
Finally, it is worth remarking that in this article, we have explored the impact of accelerating the insulin analogue on a predefined AP system, and consequently, results depend, to some extent, on the proposed baseline control scheme. In addition, further simulations should be carried out to test the robustness of the control strategy against, for example, variations in insulin sensitivity. Nevertheless, since this article discusses about fundamental limitations on control system performance, the same procedure could be applied to different control strategies, and general conclusions with respect to the benefits of using faster insulins in automatically controlling the blood glucose level in T1D will remain unaffected.
Conclusion
In this article, a methodological tuning of an MPC law for an AP was proposed to accommodate the controller’s aggressiveness according to the velocity of the insulin analogue glucodynamic action. In these proof-of-concept simulations, we evidenced that current fast-acting insulin analogues need to be further accelerated concerning their glucodynamic action for fully automated insulin treatment approaches to reach safely the glycemic performance of optimal hybrid strategies.
Footnotes
Appendix
The proposed MPC is based on the so-called Subcutaneous Oral Glucose Minimal Model.
35
To embed this model into the MPC formulation, it is first linearized at the steady state given by the subject-specific insulin basal rate ub (mU/min) and a blood glucose setpoint of 120 mg/dL, and later discretized with a sampling period Ts = 5 min. In this way, a triplet
Let
with cost function
subject to
Predictions of the insulin-glucose dynamics are made using the obtained state-space realization
Sequence
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PC, JG-T, and KK have nothing to declare. EC is Scientific Advisory Board Member/Consultant of Novo Nordisk, Adocia, MannKind, and Arecor, and holds a Research Grant from Dexcom. MDB consults for Roche Diagnostics, Sanofi-Aventis, and Ascensia Diabetes Care; receives research support from Dexcom, Senseonics, Tandem, Roche Diagnostics, Sanofi-Aventis, and Ascensia Diabetes Care; and holds equity in TypeZero Technologies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health (NIH) [Grant 1DP3DK106826-01], JDRF [Grants 2-APF-2019-737-A-N and 3-SRA-2016-244-M-R], and the National Center for Research Resources (NCRR) and the National Center for Advancing Translational Science (NCATS) [CTSA Grant Number UL1 RR024139].