Abstract

Centralization of labs causes delays on patients’ results while clinicians need immediate results of HbA1c to better educate patients 1 and to facilitate the therapeutic decision-making. Before implementation of point of care (POC) in clinical practice its analytical performances, given the accuracy requirements according to the NGSP (National Glycohemoglobin Standardization Program) recommendations, 2 are essential. The aim of our study was to evaluate the measure of HbA1c levels (NGSP units) on a Cobasb101© POC (Roche Diagnostics) against HPLC (Tosoh Bioscience HLC®-723G8), and DCA Vantage© (Siemens) used routinely in a pediatric department. Interferences from variants and carbamylated hemoglobin were investigated.
The POC showed imprecision values close the minimum goal acceptable of 3.5%. 3 At HbA1c levels of 4.5% and 7.0% the intra-assay CV was 2.7 and 2.0%, respectively. The interassay CV ranged from 3.5 to 4.0%. The correlation between POC and HPLC (Table1) on 110 routine samples ranging from 3.8 to 15.5% was excellent with a mean difference (±SD) of −0.09% ± 0.27%, lower than in previous studies,4–5 confirming the acceptable agreement for POC system. Unlike in a previous study, 4 the POC did not pass the NGSP criteria (clinically significant limits of ±6%) for 10 samples, may be due to a lot-to-lot variation as reagent number (720041) was different from NGSP certification data. The Kappa coefficient was higher than 0.83, and the strength of agreement could be considered to be very good. According to the classification of the American Diabetes Association, 6 a total of 9% results disagreed with the category assignment of HPLC, however without reaching the significance. All outliers were with HPLC HbA1c below 6.5% but overestimated with POC (2.8% relative increase). None patients were underestimated by the POC for HPLC HbA1c below 7% as the threshold for the management of diabetes in adults. As expected, the comparison between Cobasb101 and DCA Vantage exhibited a good correlation (Table 1), since both POC technologies were based on latex agglutination inhibition immunoassay methodology.
(A) Comparison Between TosohG8 (x) and Cobas b101 (y) According to Passing-Bablok Regression, Bland Altman Analysis, and Bias and (B) Comparison Between DCA Vantage (x) and Cobasb101 (y) on 20 Samples.
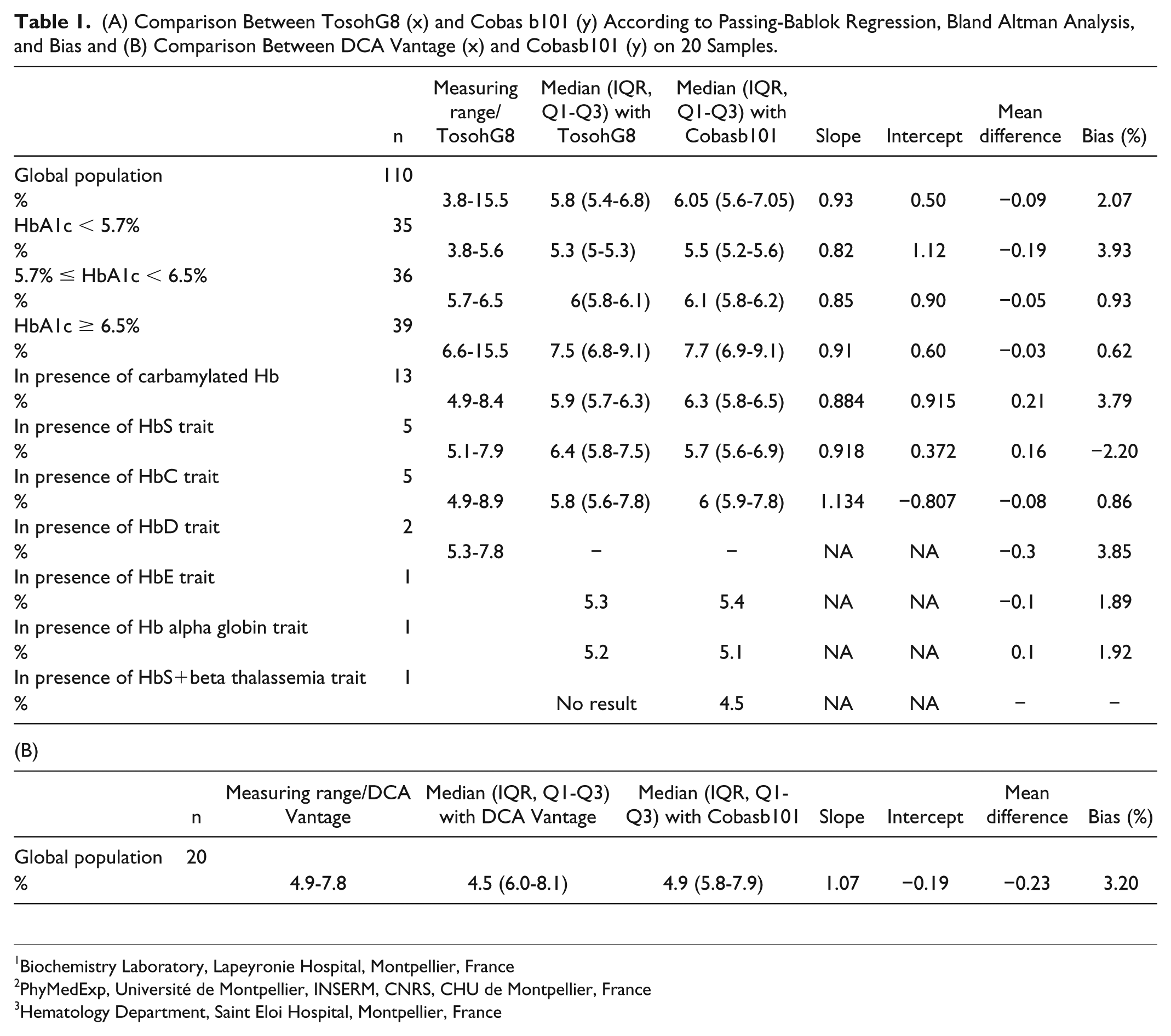
On 13 samples with uremia ranging from 22 to 40 mmol/l, the mean relative difference with HPLC results was 3.8%, higher than all others results suggesting more precautions to take in patients with renal failure. No result from Tosoh was provided in presence of HbS+beta thalassemia form while a value of 4.6% was provided by the POC. The mean relative difference between results from HPLC and POC in presence of HbAS, HbAC, HbAD, HbE, and alpha thalassemia, was close to previous study, 4 except for HbAE (Table 1). In presence of HbC or HbS, three results or one obtained by POC, disagreed with HPLC category assignment, respectively. The small number of samples did not allow us to report the bias by HbA1c levels. The evaluation of the POC by nurses confirmed its ease of use.
Our study highlighted necessary precautions before POC implementation, even if NGSP certified (1) not to use for the first-line measurement, in the presence of variants or in patients with urea levels >20 mmol/L, (2) use only for monitoring diabetes patients to facilitate immediate management, and (3) need to conduct periodic comparisons of POC test results with those from central laboratory.
Footnotes
Abbreviations
CV, coefficient of variation; HbA1c, hemoglobin A1c; HPLC, high-performance liquid chromatography; IQR, interquartile range; NGSP, National Glycohemoglobin Standardization Program; POC, point of care.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
