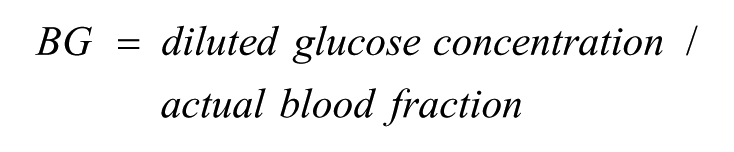Abstract
Background:
One major advantage of automated over manual clamps are continuous measurements of blood glucose concentrations (BG) allowing frequent adaptations in glucose infusion rates (GIR). However, BG measurements might be affected by changes in blood dilution. ClampArt®, a modern automated clamp device, corrects BG measurements for blood dilution, but the impact of this correction is unclear.
Methods:
The authors performed a retrospective analysis of BG during glucose clamps comparing values with a fixed dilution factor with those corrected for the actual blood dilution.
Results:
Clamp quality substantially improved with the consideration of blood dilution: Mean accuracy fell from 8.1% ± 2.9% to 4.1% ± 0.8%, precision improved from 9.6 ± 3.6 mg/dl to 3.7 ± 1.3 mg/dl and control deviation from −2.6 ± 4.2 mg/dl to 0.2 ± 0.2 mg/dl.
Conclusions:
Correcting continuous BG measurements for blood dilution significantly increases BG measurement and clamp quality.
The pharmacokinetic and pharmacodynamic effects of antidiabetic drugs are usually characterized with the glucose clamp technique: Blood glucose concentrations (BG) are kept as closely as possible (“clamped”) at a predefined target level by means of variable glucose infusion rates (GIR) which reflect the metabolic effects of the drug under investigation. 1 In automated glucose clamps with devices like the Biostator (Life Sciences Instruments, Elkhart, IN, USA), 2 the STG-22/STG-55 (Nikkiso Co, Tokyo, Japan), 3 or ClampArt® (Profil, Neuss, Germany), 4 BG is measured continuously and the GIR is calculated with an implemented algorithm every minute.
Frequent adaptations of GIR should allow higher clamp quality than with manual clamps where BG is only measured every 3-10 minutes; however, BG measurements of high quality might be more difficult to achieve with continuous measurements requiring a high blood dilution to reduce blood loss. Usually, in automated clamps arterialized blood is diluted with buffer and then transferred to the device’s glucose sensor. This is similar to the approach used in professional laboratory glucose analyzers (eg, YSI 2300 STAT plus blood glucose analyzer, AYI Life Science, Yellow Springs, Ohio, or SuperGL glucose analyzer, Hitado, Möhnesee-Delecke, Germany) which also use a high sample dilution but ensure that the dilution is exactly the same for every single measurement. However, for continuous BG measurements it is often difficult to ensure a constant dilution over the whole duration of a glucose clamp because of the very small flow rates and the pulsatile nature of the used tubing pumps. Obviously, changing blood dilution factors, if unconsidered, will impair the quality of BG measurements. Because of the high dilution factor (most commonly a factor of about 50), even small changes will have an impact on the displayed glucose value (eg, a true glucose concentration of 100 mg/dl [meaning that 2 mg/dl were measured by the sensor with a 50-fold dilution] will change to 111 mg/dl if the dilution is “only” 45-fold [translating into a measured sensor value of 2.22 mg/dl] but using the fixed factor of 50 for calculation).
To prevent impairments by changes in blood dilution, the actual dilution factor is measured by the glucose sensor using a second measurement channel without glucose oxidase. Considering the measured (“true”) dilution rather than using a fixed dilution factor should allow a higher quality of BG measurements and thereby improve glucose clamp quality; however, the impact of the consideration of blood dilution on BG measurements and clamp quality has never been investigated. We therefore compared accuracy and precision of BG measurements and clamp quality in automated glucose clamps with and without the consideration of blood dilution.
Methods
Data for this analysis were taken from a glucose clamp study that comprised two cohorts of people with T1DM who received single doses of either 0.4 or 0.6 U/kg/day of insulin glargine U300 and insulin degludec in a crossover fashion. Details of the study are described elsewhere. 5
To measure BG continuously, the device draws whole (arterialized) blood continuously from a subject’s vein. The blood is then diluted with buffer by a factor of about 50 and transported to the glucose sensor. The sensor (Dr Müller, Freital, Germany) has two channels one equipped with the glucose oxidase enzyme, the other without (see Figure 1). The current measured at the electrodes serves as the raw signal for glucose concentration and blood fraction (about 2%), respectively. The usual performance includes the correction of the measured BG concentration for the actual blood fraction:
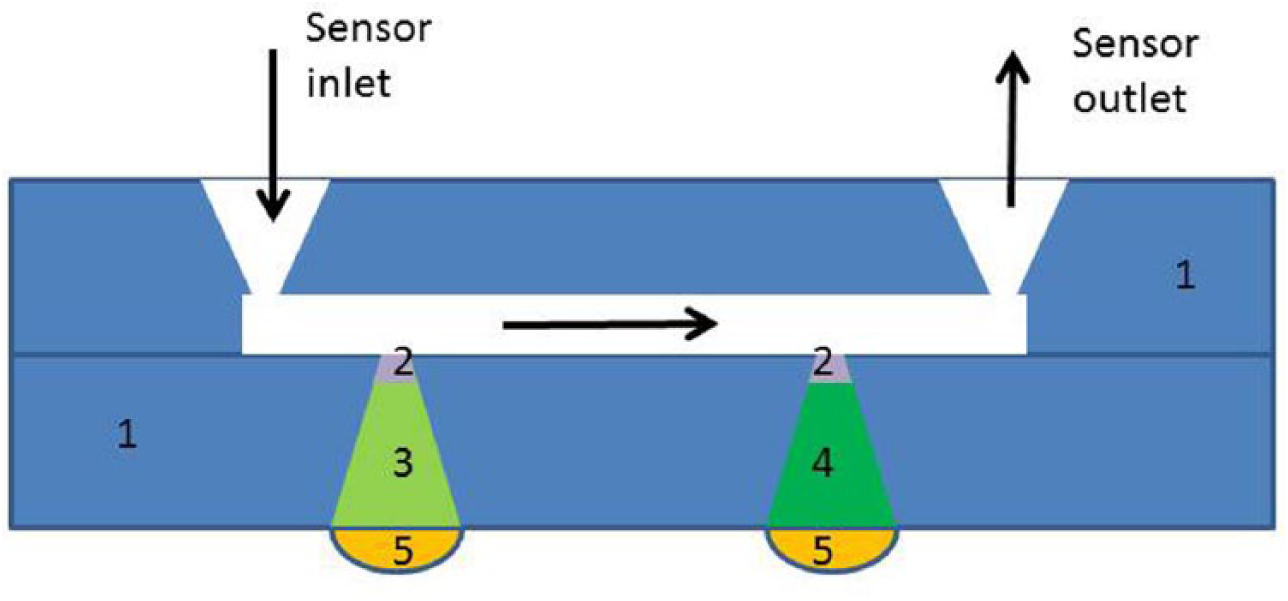
Schematic setup of the sensor: (1) housing, (2) membrane, (3) layer with glucose oxidase enzyme, (4) layer without glucose oxidase enzyme, (5) electrode.
Thus, if the diluted glucose concentrations at the sensor is measured as 2.22 mg/dl as in the previous example and the actual blood dilution is 45-fold, the blood fraction is 1/45 = 0.022 (= 2.2%) and the quotient (2.22/0.022) will result in the “true” blood glucose concentration of 100 mg/dl.
As the BG values of ClampArt® are always corrected for the actual blood fraction a retrospective analysis had to be done to evaluate clamp quality parameters 4 without considering the actual blood fraction. This retrospective analysis recalculated the BG values during the study using a fixed blood fraction (= BGfixed). For this purpose the blood fraction measured at the beginning of the clamp was used for all BG values during the clamp with the formula below:
Clamp quality parameters were compared between continuous BG concentrations measured during the clamps (taking blood dilution into account) and those of the retrospective analysis (using a fixed blood fraction). Altogether, the study comprised 94 glucose clamp experiments with in total 152 503 BG values which (with minute-by-minute BG measurements) translates into 2542 hours of glucose clamp data. The two data sets were compared with a two-sided paired t-test with a significance level of α < 0.05. The Super GL glucose analyzer using a glucose oxidase based sensor was used as a reference method. Blood samples were collected every 15 to 30 minutes at the contralateral arm.
In addition, to illustrate the impact of body movements and experimental procedures on blood glucose values corrected and uncorrected for blood dilution, data from clamp experiments used for the evaluation and improvement of the device are shown. These unpublished experiments, while done under Good Clinical Practice conditions and a study protocol, were aimed at investigating different challenges and further improving the device, for example, with respect to the performance of the tubing set, the glucose sensor, the infusion pumps, the algorithm, and so on, and therefore had different designs. As they were not part of the other study, 5 the data of these evaluation experiments were only used for Figures 2c and 2d and not included in the statistical analysis.
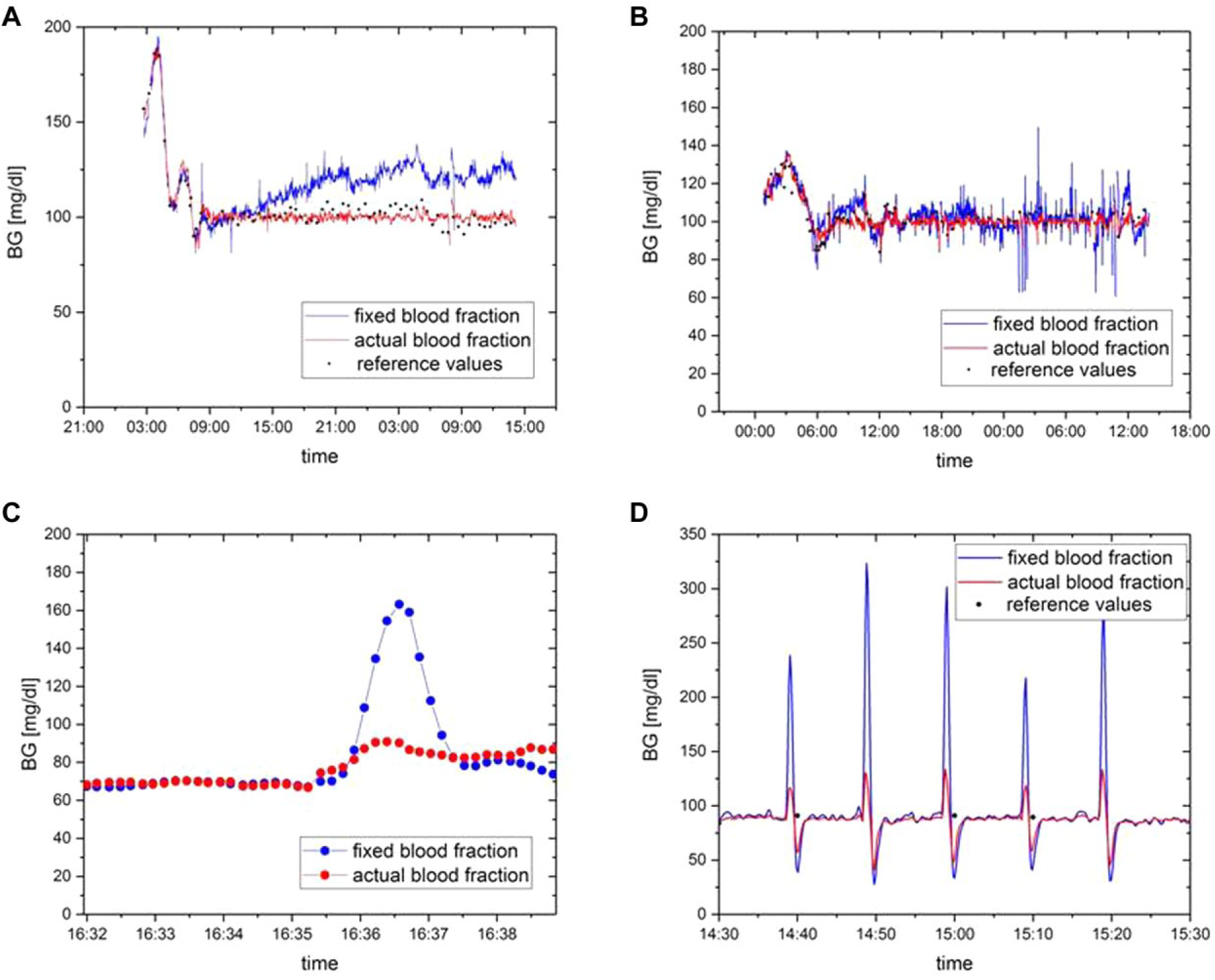
Examples for the influence of the use of the actual blood fraction when calculating BG values for a glucose clamp caused (a) by changes in tubing material characteristics, (b) by pulsation of pumps and/or small oscillations in pump rate of different pumps and/or random fluctuations at the point of mixing/diluting, (c) by body movement of the patient, and (d) by application of a tourniquet for manual blood sampling.
Results
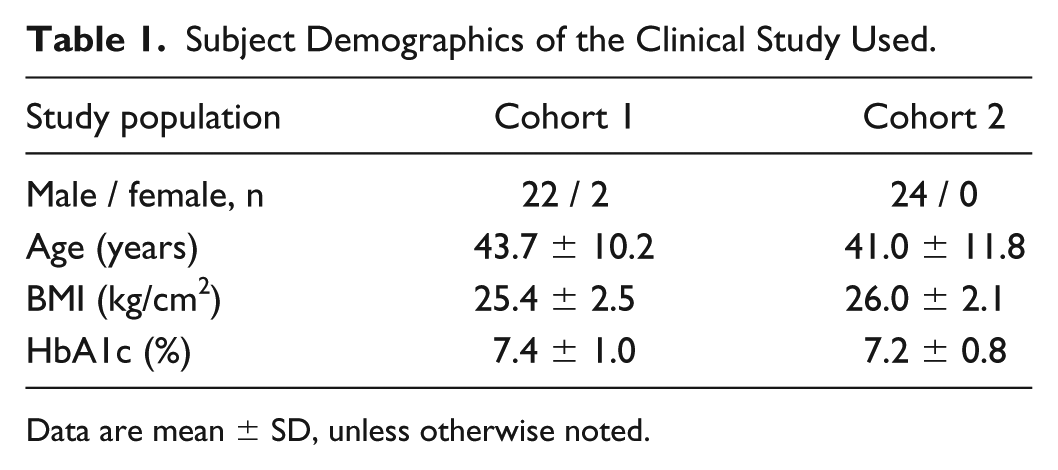
The main demographic characteristics of the patients included in the study are summarized in Table 1. The following clamp quality parameters were calculated from both the original BG and the recalculated BG with fixed blood fraction for each glucose clamp experiment:
a. Precision, ie, the standard deviation of BG values, as a parameter for BG variability during the clamp
b. Accuracy, ie, the difference between the obtained BG values and a reference method expressed as mean absolute relative difference (MARD)
c. Control deviation, ie, the mean difference of BG values from the clamp target level (100 mg/dl, 5.5 mmol/l)
All clamp quality parameters were improved when the actual blood dilution was taken into account (Table 2). Precision was improved by nearly 3-fold, accuracy by nearly 2-fold, and control deviation by a factor of 13, with P < .001.
Subject Demographics of the Clinical Study Used.
Data are mean ± SD, unless otherwise noted.
Clamp Quality Parameters Calculated With and Without the Use of the Actual Blood Fraction.
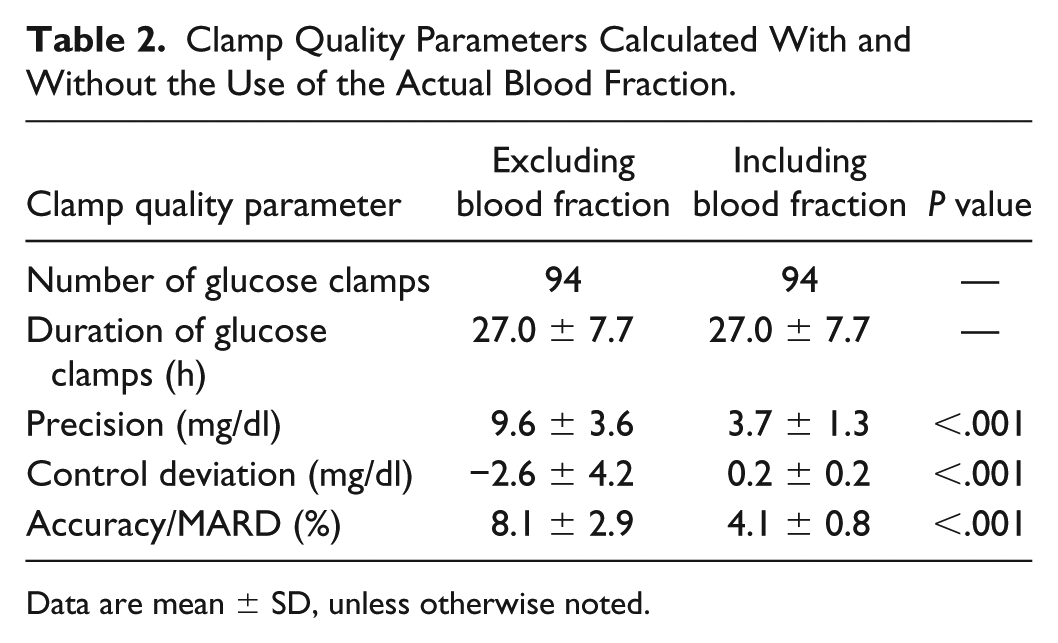
Data are mean ± SD, unless otherwise noted.
Discussion
The results of this investigation show that the consideration of blood dilution substantially improves clamp quality. We therefore investigated potential reasons for changes in blood dilution during automated glucose clamps and identified four main factors:
Tubing
The tubing material characteristics might change over time due to the constant pressure in the roller pump transporting blood from the patient to the glucose sensor and/or changes in temperature. An example of this slow, persisting drift in BG values (if blood dilution is not considered) is shown in Figure 2a, in which the drift lasts for about 10 hours and leads to a difference of up to 25 mg/dl between measured BG and BGfixed. Data of Figures 2a and 2b are examples from the study described in Bailey et al. 5 showing the usual performance (red) of the device.
Pump
Small changes in blood dilution occur within seconds due to the pulsation nature of the roller pumps as illustrated in Figure 2b. Similar changes can also occur when two different pumps are used for transportation of blood and buffer. If these pumps are not coupled, for example, by using a two-channel pump head at the same axis, the pulsatile character of the pumps will cause small oscillations on a short time scale. Finally, BG oscillations might occur because of random fluctuations when mixing small amounts of blood and buffer.
Patient
Body movements of the patient, for example, moving from supine position to upright position or raising the arm with the catheter, can cause pressure changes at the tip of the catheter and thereby short-term changes in blood dilution. These confounders were investigated in the evaluation experiments. One example is given in Figure 2c, showing that raising the arm with the catheter has a huge effect on the measured BG concentration (apparent BGfixed rise of about 100 mg/dl), but the effect is of short duration (less than 100s). After the results of the evaluation experiments, standard procedures at the site were therefore updated, so that patients (including the study participants of the study published in Bailey et al. 5 ) were advised to avoid major body movements during glucose clamps. Therefore, major arm/body movements were not observed in the investigated glucose clamp study although patients were still allowed to do minor movements.
Investigator
Changes of local blood pressure at the tip of the catheter can also be induced by the investigator for example by applying a tourniquet for manual blood sampling (the cannula for blood sampling is placed proximal at the same arm used for the BG measurements). This is illustrated in Figure 2d showing short, but large changes in BGfixed. While changes in BG concentrations are also seen with the correction for blood fraction, they are considerably larger without this correction. The data from Figure 2d also come from the evaluation experiments. Because of these data, investigators were trained to avoid disturbances such as applications of tourniquets, so these interferences were not observed in the investigated glucose clamp study. 5
Subject- and investigator-initiated factors can easily be eliminated in automated glucose clamps by training of both patients and investigators (eg, to avoid arm movements or blood collection using a tourniquet). The other factors, such as the pulsatile character of the tubing pumps and the characteristics of the tubing, can only be reduced but cannot be eliminated completely.
The influence of the roller pumps and the tubing characteristics on blood dilution is even more pronounced when tubes with a small inner diameter are used. The tubing set used for blood sampling for BG measurements has a small inner diameter to minimize the blood loss of the patient. This small inner diameter was one of the major reasons for implementing the measurement of blood fraction and the possibility to correct the measured BG for the actual blood dilution.
The exact method for the determination of blood dilution is proprietary information of the manufacturer. In principle, however, the dilution is measured by determining a signal that is independent of the glucose concentration, such as electrical conductivity/bioimpedance, 6 ionic current, 7 optical absorption. 8
This investigation has several limitations. First of all, this is a retrospective analysis. Second, the calculation of the GIR was based on BG values corrected for actual blood fraction. Therefore, it is not really surprising that the BGfixed values show a larger control deviation as GIR could not be adapted to actual BGfixed values. Nevertheless, the magnitude of the improvement in both clamp quality and, even more important, BG measurement quality does clearly indicate the advantage of BG values taking blood dilution into account.
In conclusion, this retrospective analysis clearly shows that correcting BG measurements for blood dilution in automated clamps significantly enhances BG measurement and thereby glucose clamp quality.
Footnotes
Acknowledgements
We are very grateful to the clinical staff at Profil for performing the clamp experiments.
Abbreviations
BG, blood glucose concentration; BGfixed, blood glucose concentration obtained by using a fixed blood fraction; BMI, body mass index; CV, coefficient of variation; GIR, glucose infusion rate; MARD, mean absolute relative difference.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CB, MK, and TH are employees and TH is shareholder of Profil, Neuss, Germany, a private research institute where ClampArt® was developed and is being used.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.