Abstract
Background:
As evidence emerges that artificial pancreas systems improve clinical outcomes for patients with type 1 diabetes, the burden of this disease will hopefully begin to be alleviated for many patients and caregivers. However, reliance on automated insulin delivery potentially means patients will be slower to act when devices stop functioning appropriately. One such scenario involves an insulin infusion site failure, where the insulin that is recorded as delivered fails to affect the patient’s glucose as expected. Alerting patients to these events in real time would potentially reduce hyperglycemia and ketosis associated with infusion site failures.
Methods:
An infusion site failure detection algorithm was deployed in a randomized crossover study with artificial pancreas and sensor-augmented pump arms in an outpatient setting. Each arm lasted two weeks. Nineteen participants wore infusion sets for up to 7 days. Clinicians contacted patients to confirm infusion site failures detected by the algorithm and instructed on set replacement if failure was confirmed.
Results:
In real time and under zone model predictive control, the infusion site failure detection algorithm achieved a sensitivity of 88.0% (n = 25) while issuing only 0.22 false positives per day, compared with a sensitivity of 73.3% (n = 15) and 0.27 false positives per day in the SAP arm (as indicated by retrospective analysis). No association between intervention strategy and duration of infusion sets was observed (P = .58).
Conclusions:
As patient burden is reduced by each generation of advanced diabetes technology, fault detection algorithms will help ensure that patients are alerted when they need to manually intervene.
Clinical Trial Identifier:
Keywords
Advanced device technology, including continuous glucose monitors (CGMs) and continuous subcutaneous insulin infusion (CSII) pumps, improves care for patients living with type 1 diabetes and has recently moved to closed-loop artificial pancreas (AP) systems.1-4 Advancements in automated systems, such as the removal of calibration requirements from the CGM 5 or elimination of required meal announcements,6-10 will reduce the frequency of patient interaction required, freeing patients from constantly worrying about their disease. 11 However, as patients place increasing trust in these devices, they could potentially be less aware of adverse events. Similar deskilling has been seen in type 1 diabetes patients reliant on automated bolus calculators, 11 nurses reliant on electronic patient records for planning care, 12 and physicians reliant on ultrasound-guidance for central venous cannulation. 13 Figure 1 presents a motivating example from a previous study showcasing a well-controlled patient under closed-loop insulin delivery that is disrupted by an infusion site failure (SF) associated with hyperglycemia. Only manual intervention from the patient in replacing the infusion set can restore satisfactory glycemic control and prevent eventual development of diabetic ketoacidosis. Alerting patients to SFs causing hyperglycemia would provide them with an alert they currently do not have, since most SF episodes fail to trigger a pump occlusion alarm. 14 During AP system use, algorithms track current glucose levels, active insulin, and the expected effect of delivered insulin. When there is a persistent mismatch of expected insulin action and sustained hyperglycemia, an SF is likely. Detecting SFs would increase trust in an AP, unless there was a high incidence of false alarms.
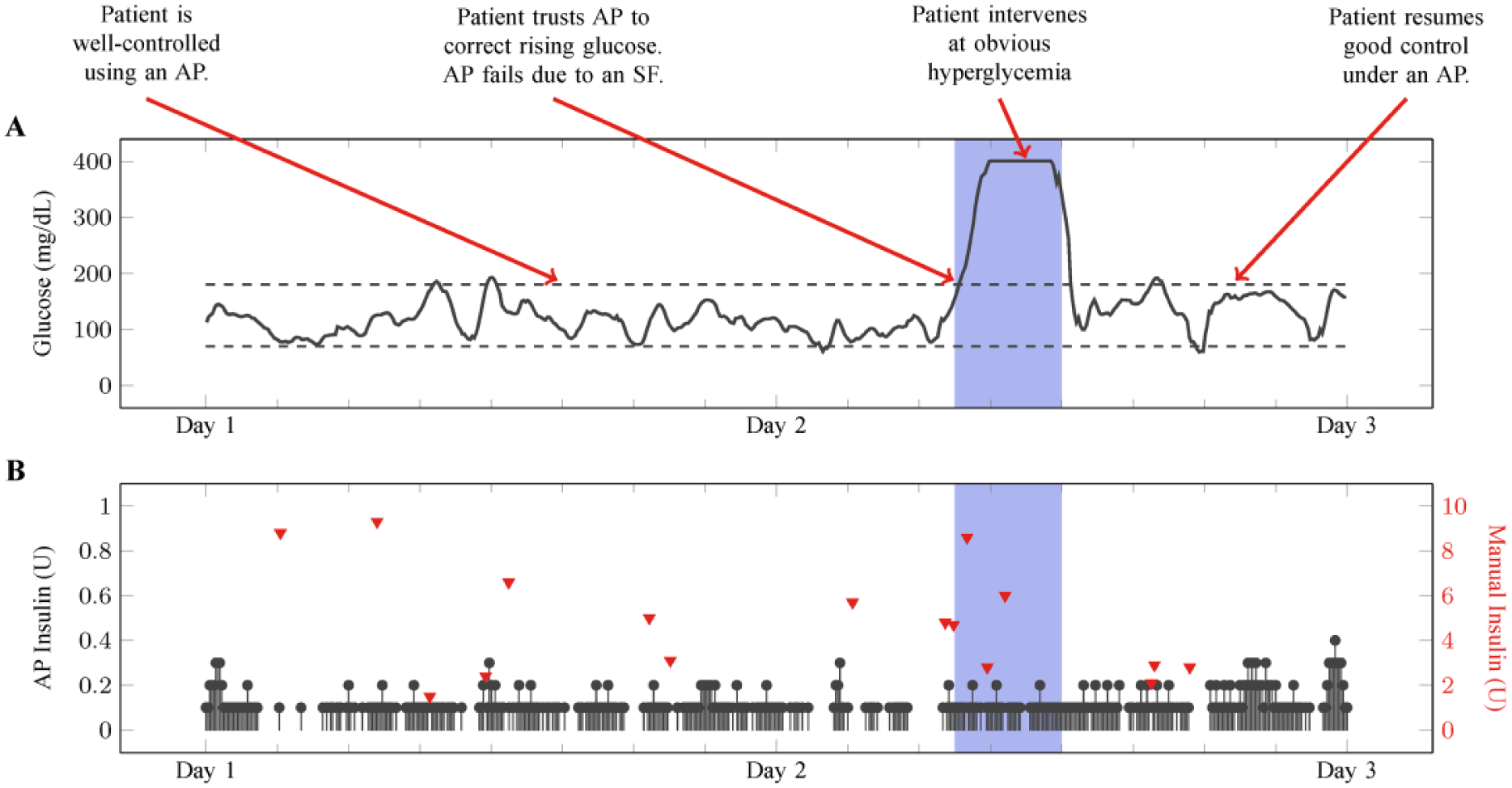
Actual SF in an artificial pancreas. The shaded time period represents the 4.5-hour time frame after the infusion set failed and before the patient intervenes. The patient must intervene by replacing the infusion set to resume insulin delivery.
SFs are the most common and most problematic issue with infusion pump therapy and will continue to occur with artificial pancreas systems. The term “infusion site failure” (SF) is used here to describe any malfunction associated with the entire process of insulin delivery, from leaving the reservoir to ultimately entering the bloodstream and affecting glucose levels. SFs may be caused by catheter kinking, insulin precipitation, insulin leakage, site inflammation, set detachment, or otherwise unknown reasons and lead to hyperglycemia and ketonemia if left uncorrected.15,16 Although SFs are often classified into different failure modes for diagnostic purposes, they result in the same action from the patient, that is, inserting a new infusion set in a different location. Inability to recognize SFs can lead to diabetic ketoacidosis (DKA), a high-cost acute complication that may be fatal.17,18 Although the incidence of DKA is not increased by CSII pump use over multiple daily injections,17,18 problems with the pump (eg, SFs) are the largest contributor to development of DKA19,20 and incidence of DKA is associated with higher rates of CSII pump discontinuation. 21 Early SF detection promises to decrease both exposure to hyperglycemia and incidence of DKA.
Previous studies in the literature have retrospectively evaluated SF detection in both open-loop and closed-loop scenarios, primarily in simulation. The interested reader is referred to Howsmon et al 22 and the references therein for an in-depth discussion of SF detection approaches. However, there has been no study to date that investigates real-time SF detection, where patients and clinical personnel can immediately give feedback on alarm performance (ie, true/false positives) and missed SF events. In an effort to address this gap in knowledge, an artificial pancreas using zone model predictive control (Zone-MPC)23-27 was outfitted with an SF detection algorithm. 22 Patients were instructed to wear infusion sets for up to seven days, significantly beyond the recommended use-time of two to three days,16,28 in an effort to provoke a higher incidence of SFs. This report marks the first study that evaluates the performance of real-time SF detection in advanced diabetes technology.
Methods
Study Design
Twenty subjects were enrolled at two clinical centers (Barbara Davis Center and Stanford University) in a six-week, outpatient randomized crossover controlled clinical trial consisting of two blocks (first patient enrolled: May 2016, last patient completed: July 2016). Randomization occurred in a 1:1 ratio at each clinical site. Group A started with Zone-MPC outfitted with the SF detection algorithm and Group B started with sensor-augmented pump (SAP) therapy. There was one week of washout in between each evaluation period. Each block consisted of one week of sensor run-in followed by two weeks with prolonged infusion set and sensor wear. Both groups were remotely monitored for sensor glucose values <60 mg/dL for more than 30 minutes or a sensor glucose >300 mg/dL for more than 60 minutes. The Zone-MPC + SF detection group was further remotely monitored for SFs.
In each block, patients wore an infusion set for up to seven days in an effort to increase the number of SFs. The Roche Accu-Chek Spirit Combo was used in the Zone-MPC + SF detection arm and each patient’s personal pump was used during the SAP arm; the Dexcom G4 PLATINUM CGM System with Share was used in both study arms. A new infusion set was inserted at the beginning of each week in each block. Both steel and Teflon infusion sets were used and infusion sets used in the Zone-MPC + SF detection were similar, but not identical to the patient’s usual choice of infusion set. SF was defined in a similar manner to previous studies:15,29 (1) a meter glucose value >300 mg/dL and ketones >0.6mmol/L; or (2) a failure of a correction dose to lower glucose value by at least 50 mg/dL/h, for a meter glucose >250 mg/dL obtained ≥2 hours postprandially, or (3) a pump occlusion alarm. Inflammation and insulin leaking around the insertion site were also noted upon detection of SFs. SFs were detected by the SF detection algorithm in the Zone-MPC + SF detection arm and by patients/remote monitoring in the SAP arm. In the SAP arm, clinician review of glucose-insulin records via remote monitoring prompted correction doses for testing criterion (2); in the Zone-MPC + SF detection arm, SF detection alarms prompted correction doses for testing criterion (2). Each SF event was confirmed or refuted (and thus classified as true positive or false positive events) following immediate communication with the patient and patients were instructed to change infusion sets and infusion set locations for confirmed SFs. Glycemic outcomes, sensor functioning, and Zone-MPC AP performance are described in Forlenza et al. 30
The study (www.clinicaltrials.gov,NCT02773875) was approved by the US Food and Drug Administration and the institutional review boards at Stanford University and the University of Colorado Denver. Briefly, subjects 18-55 years old with a clinical diagnosis of type 1 diabetes requiring daily insulin therapy for at least 12 months, a total daily dose (TDD) of insulin greater than 0.3 U/kg/day, and at least three months of experience with using an insulin pump were enrolled in the study. In addition, each subject was required to have living with them an adult companion that was willing to attend to the subject if safety concerns arose. Finally, female participants were required to use an adequate method of contraception and were not pregnant. Subjects were excluded from consideration if they had DKA or severe hypoglycemia in the past six months, required intermediate or long-acting insulin as part of their daily insulin needs, used other antidiabetic medications within the past eight weeks, used an oral/inhaled glucocorticoid, had a skin condition affecting sensor placement, or had other medical conditions that, in the opinion of the investigators, would interfere with study completion.
SF Detection
The SF detection algorithm has been described previously.
22
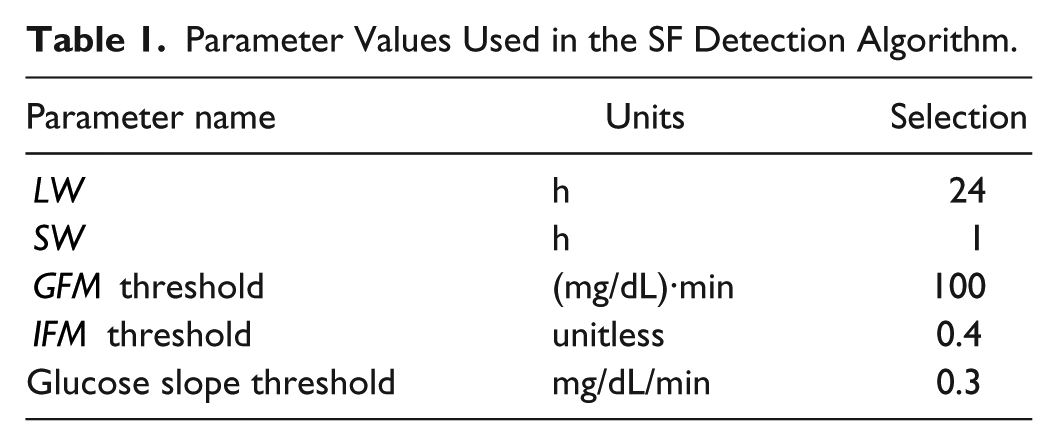
Briefly, adaptive, patient-specific metrics for hyperglycemia exposure (glucose fault metric,
Parameter Values Used in the SF Detection Algorithm.
Zone-MPC and System Architecture
This study used a Zone-MPC artificial pancreas23-27 on the DiAs platform. 31 This algorithm employs a predefined glucose range as the explicit target for the controller. The DiAs platform collects data on an Android smartphone and frequently transmits data to a secure server. The SF detection algorithm then used data from the server to update predictions and transmit any SF detection alerts to the clinical team. The clinical team notified patients of these potential SFs and patients confirmed or refuted SF events, provided descriptions of their insertion site, and confirmed all infusion set changes.
Retrospective Analysis of SF Detection Under Open-Loop
The open-loop portion of the trial was analyzed retrospectively as done previously. 22 Briefly, the time and characteristic of each set removal was documented as described in the Study Design. To classify alarms produced by the algorithm, an estimated failure start time is determined from the most previous trough in CGM readings prior to a documented SF. Then, the SF detection algorithm runs on the retrospective data and records the time instants at which alarms would have been issued. Since SFs are rare events and the overwhelming majority of time points should be classified as true negatives (TNs), all metrics are reported on the basis of alarm events, rather than individual time points. If an alarm is issued in between the proposed SF start time and the documented infusion set removal time, the SF is detected as a true positive (TP); however, if no alarm is issued in this time period, a false negative (FN) is recorded. If an alarm would have been issued at any other time, a false positive (FP) is recorded. Since specificity cannot be determined on a per event basis, the FP rate is reported as the average number of FPs per day.
Statistical Analysis
Data are presented either as mean ± SD or median (IQR). The main objective of this study was to assess the sensitivity and FP rate of the SF detection algorithm in real time and under closed-loop conditions as done previously. 22 As in the retrospective analysis, alarms generated by the algorithm were classified as TP or FP events while SFs that were not detected by the algorithm were classified as FN events. The major difference between the real-time and retrospective analysis is that SF start times do not need to be calculated as the infusion set status is determined by the patient and clinical staff for each alarm. A secondary outcome was a comparison between infusion set survival in SAP and Zone-MPC. The Kaplan-Meier estimates of the survival curves are plotted, and the effect of the intervention strategy is assessed with the log-rank test. The survival analysis was conducted using the “survival” library in R. 32 The total numbers of infusion sets censored after seven days in SAP and Zone-MPC were compared with a chi-square test for independence. Finally, the relationships between the hyperglycemia end point (minutes >250 mg/dL in previous four hours) and (normalized) carbohydrate intakes were analyzed via F-tests for regression.
Results
Nineteen patients (10 at Stanford and 9 at Barbara Davis) successfully completed the six-week study, leading to 38 patient-weeks of prolonged infusion set wear under both SAP and Zone-MPC + SF detection scenarios for a total of 76 patient-weeks. One participant was excluded midstudy due to failure to comply with the study protocol. Summary demographics are provided in Table 2.
Patient Demographics.
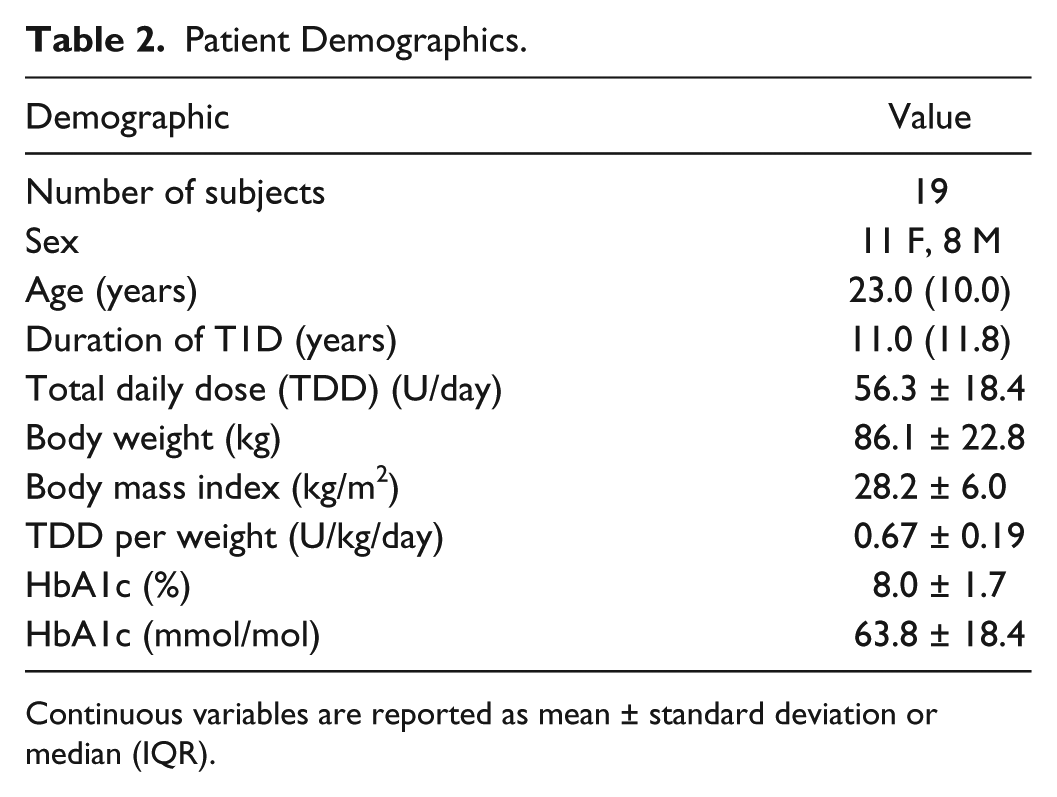
Continuous variables are reported as mean ± standard deviation or median (IQR).
Retrospective Analysis of SF Detection Under SAP
The SAP portion of the trial was analyzed retrospectively. One SF was removed from analysis due to a CGM dropout that started three hours before and continued until two hours after the documented SF. Of the 15 remaining failures, the SF detection algorithm correctly detected 11 SF events (sensitivity of 73.3%) while only issuing 0.27 FPs/day (Table 3). Furthermore, when the algorithm correctly detected an SF, if an alert had been responded to in real time, there would have been 33% less time spent in hyperglycemia in the four hours prior to SF detection, compared to when the patient detected the SF.
Algorithm Performance and Comparison With Previous Retrospective Analysis.
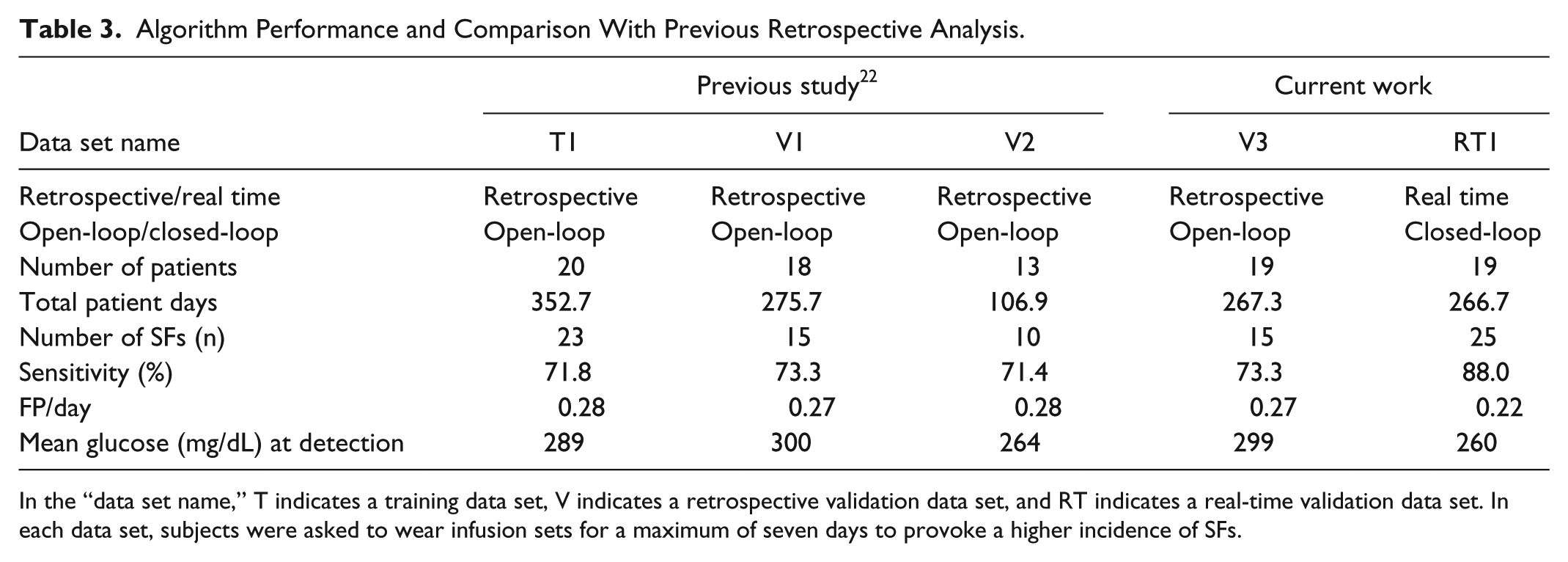
In the “data set name,” T indicates a training data set, V indicates a retrospective validation data set, and RT indicates a real-time validation data set. In each data set, subjects were asked to wear infusion sets for a maximum of seven days to provoke a higher incidence of SFs.
Performance of Real-Time SF Detection Under Zone-MPC
The Zone-MPC portion of the trial was performed in real time. Under Zone-MPC, the SF detection algorithm achieved a sensitivity of 88.0% (n = 25) and issued only 0.22 FPs per day, on average (Table 3). This translates to one FP alarm every 4.55 days. Mean glucose at detection was 260 mg/dL. There were three FNs during the closed-loop portion of this study.
Characteristics of SFs
Statistical analysis of observed SFs reveals no association between either infusion set duration or the total number of SFs with the intervention strategy. In 76 weeks of prolonged infusion set wear, there were 25 and 16 patient-confirmed SFs under Zone-MPC + SF detection and SAP conditions, respectively (Table 4, Figure 2). The most common cause of set failures was unexplained hyperglycemia that failed to respond to a correction dose. There was only 1 occlusion alarm generated by the pump over all subjects and in both arms. Analysis of the Kaplan-Meier estimates of infusion set survival (Figure 3) produces a P value of 0.58, indicating that no association between infusion set survival and intervention strategy was observed. Furthermore, the median first-weekly infusion set duration coincided with the censoring time of seven days (Figure 2). The observed infusion set duration is comparable to infusion set durations observed in previous studies.15,29 No association was found between the intervention strategy and the number of infusion sets that lasted the full seven days (P = 0.64).
Classification of SFs Into Different Failure Modes.
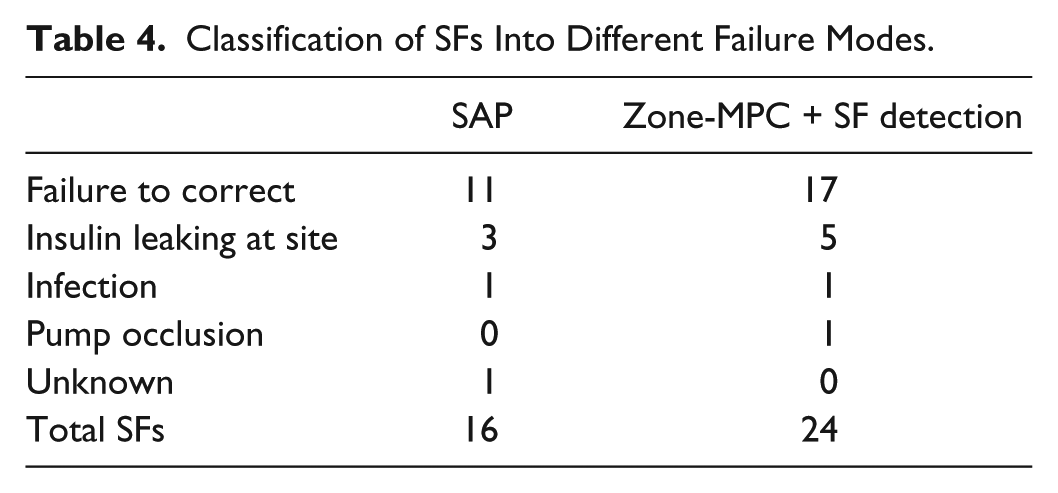
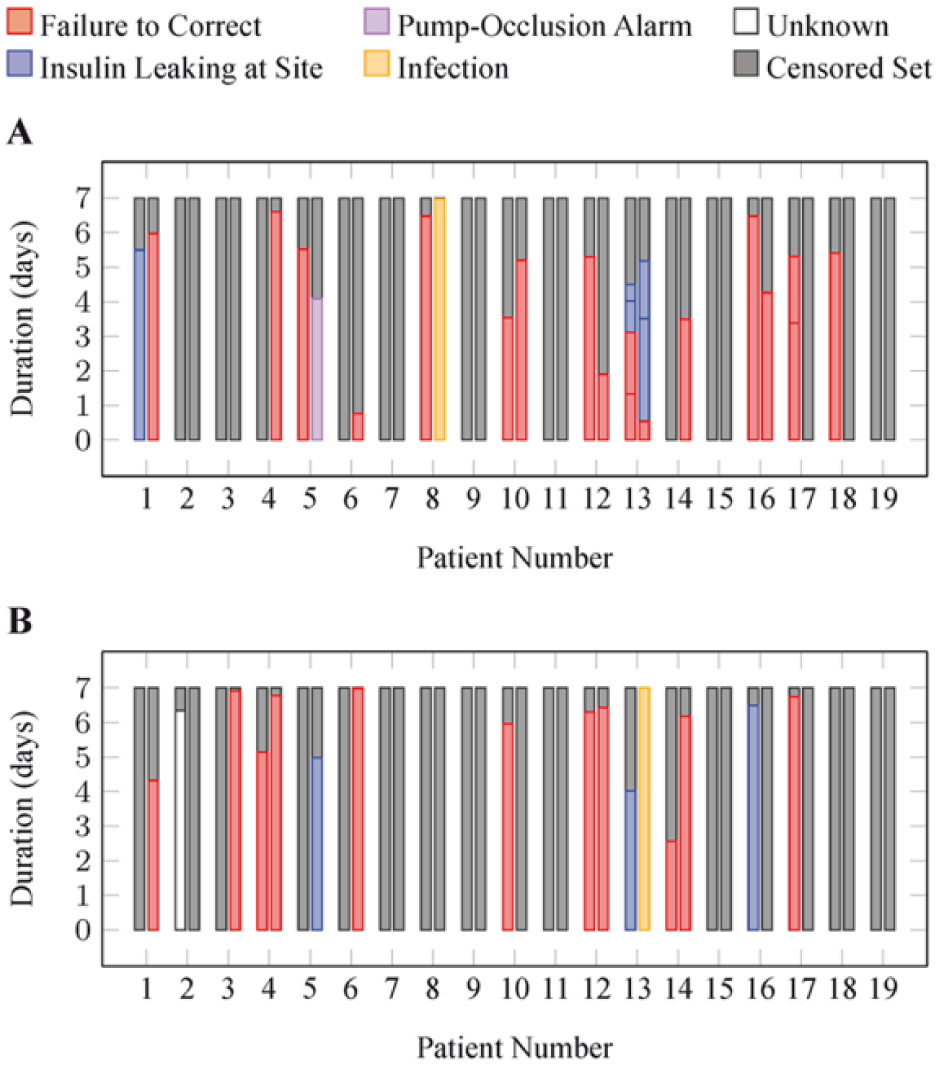
Duration and characteristics of each infusion set insertion under (A) Zone-MPC + SF detection and (B) SAP. The two bars for each patient number represent the two 7-day observation periods under each intervention. Colors designate the status of the infusion set insertion upon removal. Censored sets are those that were removed at the end of each 7-day period even though they had not failed.
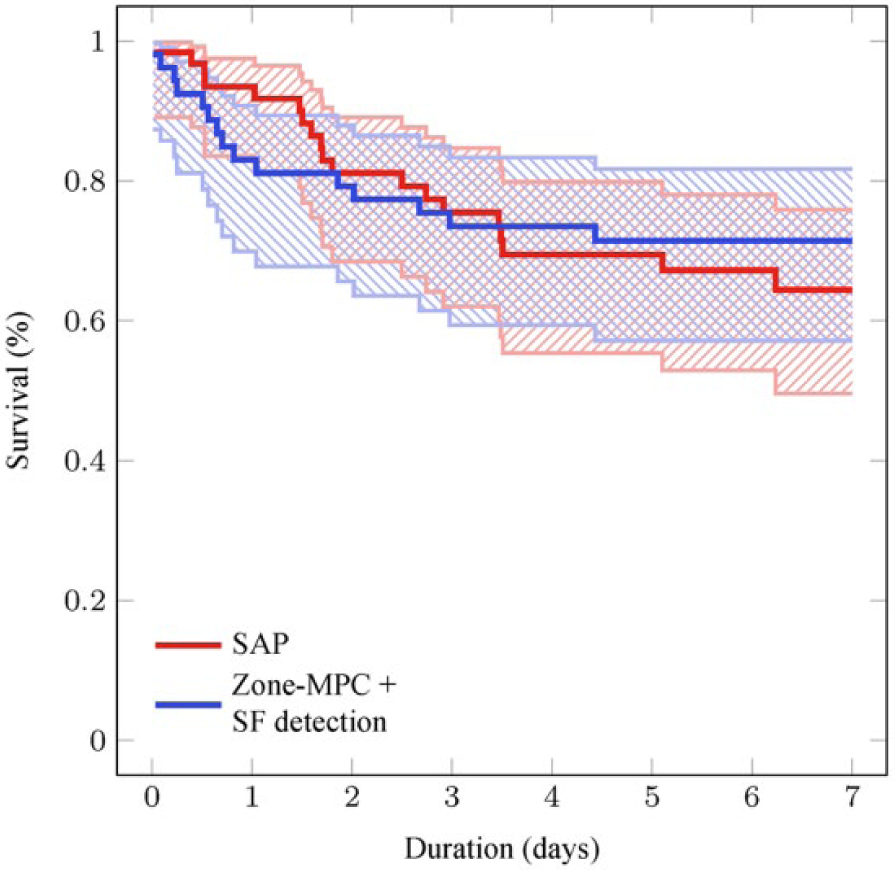
Kaplan-Meier estimates for infusion set survival in a 7-day period. Shaded regions indicate the 95% confidence intervals for the curves (P = 0.58).
Impact of SF Detection on SF-Induced Hyperglycemia
Although this pilot study was conducted to evaluate the performance of the SF detection algorithm under closed-loop conditions, limited results are presented regarding the impact of SF detection on SF-associated hyperglycemia. To evaluate the effect of the SF detection algorithm on SF-induced hyperglycemia, the duration of hyperglycemia (>250 mg/dL) in the four hours preceding an SF was determined for conditions of SAP, SAP + retrospective SF detection, and Zone-MPC + real-time SF detection (Figure 4). Retrospective analysis of the SAP arm indicates a median reduction in hyperglycemia duration from 92 to 63 minutes (P = 0.085) if the SF detection algorithm had been active. Furthermore, real-time SF detection in the Zone-MPC arm further reduced this hyperglycemia duration from 63 to 30 minutes (P = 0.060). It was observed that subject 13 may have skewed the distribution due to the total number of failures recorded for this subject. Removing subject 13 from the analysis produces a reduction in median hyperglycemia duration from 99 to 67 minutes (P = 0.086) when the SF detection is applied retrospectively to the SAP portion and from 67 to 23 minutes (P = 0.087) when SF detection is applied in SAP versus Zone-MPC, alleviating concerns that this subject skews the statistical significance of the results. Furthermore, no association was observed between this duration of hyperglycemia preceding an SF and the amount of carbohydrates consumed in the six hours preceding an SF detection, even when the amount of carbohydrates was normalized by either the patient’s body weight or their average daily carbohydrate intake (all P > 0.6). Additional studies are needed to fully characterize the effects of real-time SF detection and various intervention strategies on SF-associated hyperglycemia.
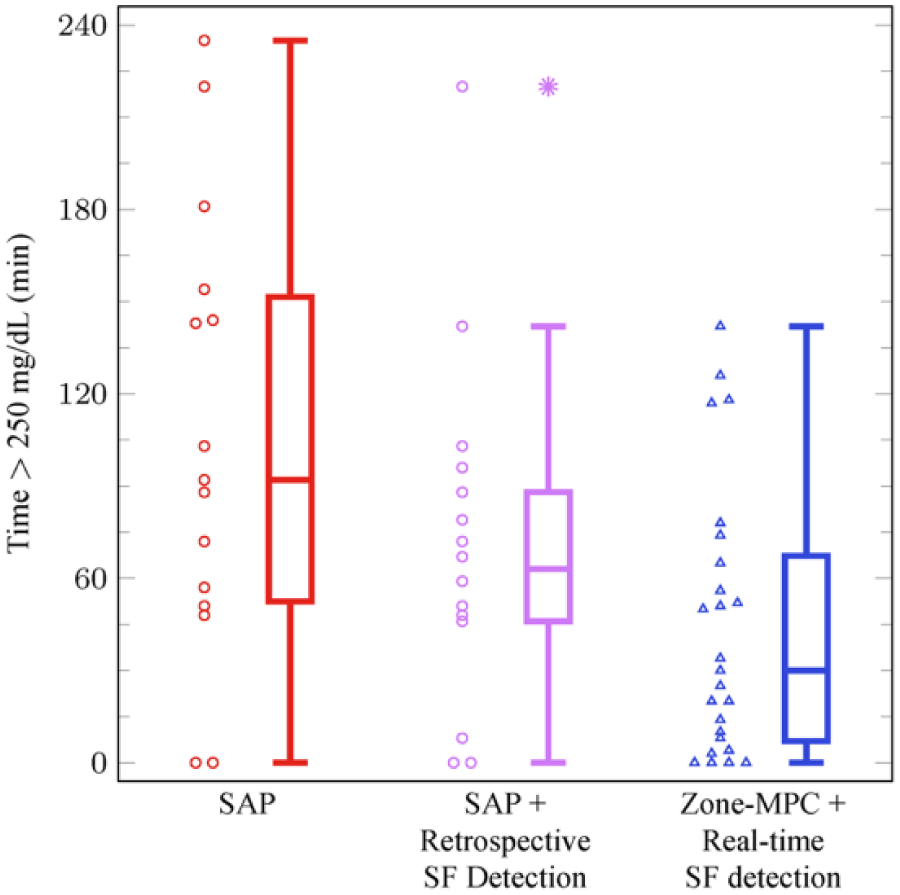
Distribution of the hyperglycemia outcome: the number of minutes the patient experiences hyperglycemia (>250 mg/dL) in the 4 hours preceding an SF (SAP vs SAP + Retrospective Detection: P = 0.085; SAP + Retrospective SF Detection vs Zone-MPC + Real-Time SF Detection P = 0.060). Asterisks mark outliers in the data.
Discussion
The SF detection algorithm performed well in real-time, closed-loop conditions. Retrospective analysis of the SAP arm indicates a sensitivity of 73.3% with 0.27 FPs/day, agreeing closely with previous results 22 (Table 3), indicating that the validation strategy used to guard against overfitting the algorithm parameters served its intended purpose. Furthermore, SFs were detected at a higher sensitivity of 88.0% and while only issuing 0.22 FPs/day under real-time, closed-loop conditions (Table 3). Since the SF detection algorithm requires the glucose to be elevated relative to each patient’s specific baseline, FPs may alert patients to other scenarios of inadequate control, such as unannounced meals. The SF detection algorithm could be made more specific to SFs instead of missed meals if meal information was provided explicitly to the algorithm. However, since it does not rely on user-provided meal announcement, the SF detection algorithm may be used in conjunction with a fully closed-loop artificial pancreas that also does not require meal announcement. Overall, these results suggest that the SF detection algorithm is applicable to both open-loop and closed-loop scenarios, with arguably better performance in closed-loop. Additional studies investigating SF-associated hyperglycemia outcomes in closed-loop with and without the SF detection algorithm are needed to explicitly measure the impact of the SF detection algorithm on SF-associated hyperglycemia in closed-loop.
Although three SFs were not detected by the SF detection algorithm in real time (Figure 5, discussed below), retrospective analysis on the Zone-MPC arm suggests that one FN was due to a diligent patient response and the other two FNs were attributed to prolonged SF dynamics resulting from gradual increases in insulin feedback directed by the artificial pancreas. The similarity in the results from the real-time analysis presented herein and the retrospective analysis conducted previously 22 helps to mitigate concerns surrounding the retrospective determination of the start of SFs. Moreover, real-time analysis can better assess the performance of these algorithms than simulated scenarios with assumed SF dynamics.
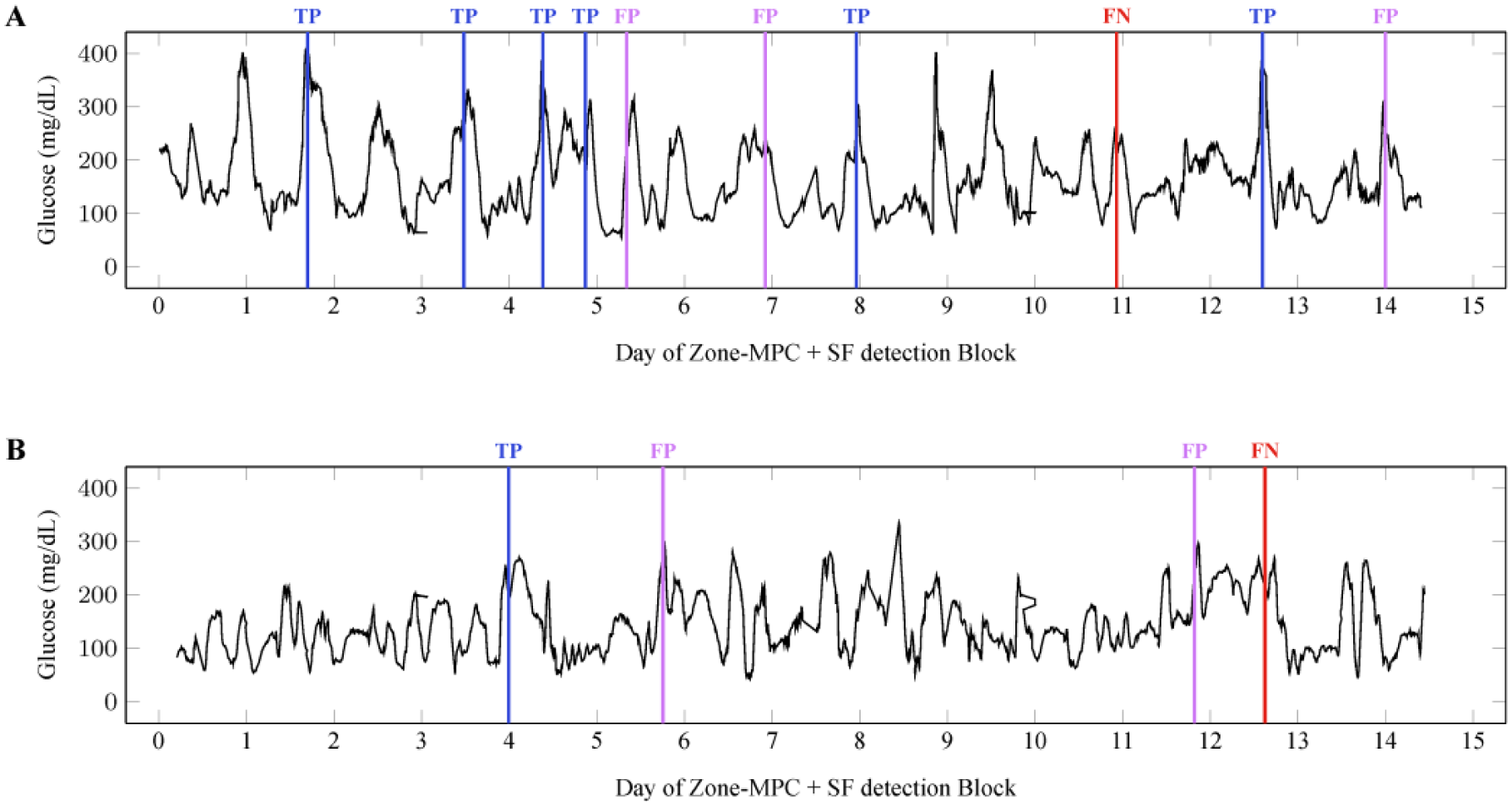
Case study of false negatives. (A) One patient experienced a much higher incidence of SFs, potentially leading this patient to be more diligent. Only one out of seven SFs was missed for this patient. (B) An example of one patient where the SF dynamics were much slower than those observed in the training set (and those detected as TP).
Of the three FNs, one undetected SF was detected very early by a patient who noticed insulin was leaking at the insertion site (Figure 5A). This SF occurred on the eleventh day of the closed-loop portion of the trial and the patient had already experienced five SFs in this study arm, potentially increasing this patient’s attention to their infusion set. It should be noted that each fault metric in the SF detection algorithm was either increasing or had already exceeded its threshold, indicating that the SF detection algorithm may have detected this event, albeit at a later time than the patient. The other two undetected SFs presented in a similar way (Figure 5B), thus only one will be discussed in detail. The patient experienced a slight trend upward in their glucose values during the previous day, with normal glucose excursion due to manual bolusing and meals. An FP was issued after the glucose level reached 300 mg/dL, but the set was not deemed to have failed at this point because a correction bolus had its intended effect. However, the glucose remained elevated overnight and into the next day despite frequent boluses. The infusion set was finally deemed unresponsive to correction boluses and was replaced. The SF detection algorithm issued an FP, indicating that it had detected the unusual trends in glucose and insulin; however, its failure to continue issuing alarms resulted in the FN. The failure to continuously issue an alarm is attributed to the increased insulin delivery due to the feedback control in the artificial pancreas, enabling the fault to present with slower dynamics than in the open-loop cases on which the algorithm was trained. Furthermore, the expected dynamics of the TP and the slower, unexpected dynamics of the FN experienced by the same patient (Figure 5B) suggest that the time scale of the failure cannot be explained by differences in patient demographics.
Fault detection strategies can augment a safety layer for artificial pancreas applications. Current alarm systems for advanced diabetes technology focus on alerting patients of current or impending hyper- and hypoglycemia. However, if a patient receives an alert for hyperglycemia, they do not know if the hyperglycemia is due to insufficient insulin administration (eg, due to an underbolused meal) or due to an SF. These root causes of hyperglycemia require very different interventions of either giving an additional insulin bolus or replacing the infusion set, respectively. Informed alarms dictating the appropriate patient-directed intervention potentially increase safety in advanced diabetes technology by improving the interactions between patients and technology. 29
Conclusion
The short duration of infusion set life continues to be a hindrance for artificial pancreas applications. It is currently recommended that insulin infusion sets are changed every two to three days,16,28 but patient surveys indicate that there is considerable variation in the actual frequency with which patients exchange infusion sets, with mean change intervals ranging from 2.7 to 4.8 days with a range from 1 to 30 days. 33 Problems with infusion sets continue to be a major contributor to diabetes-related adverse events34-36 and a reason for patients discontinuing insulin pump therapy.21,37-39 More reliable infusion sets and SF detection algorithms may potentially reduce the diabetes technology discontinuation rate by reducing patient burnout with changing infusion sets and relieving concerns over complications related to SFs. Furthermore, alarm systems that alert patients to specific manual interventions, such as providing a correction dose, changing an infusion set, or taking a specific amount of carbohydrates for treating hypoglycemia, can potentially increase market penetration of artificial pancreas systems by enhancing human/device interactions and ameliorating concerns over troubleshooting these devices.
Footnotes
Acknowledgements
The authors would like to first thank the patients who participated in the clinical trial as well as their families and support teams without whose contributions this work would not have been possible. The authors would also like to thank the University of Virginia Center for Diabetes Technology for their support with use of the DiAs AP platform. The authors would also like to acknowledge the work by the diabetes technology teams at Stanford and the Barbara Davis Center who contributed many overnight and weekend hours to this project. Study data were collected and managed using Research Electronic Data Capture (REDCap) electronic data capture tools hosted at Stanford University. 40
Abbreviations
AP, artificial pancreas; CGM, continuous glucose monitor; CSII, continuous subcutaneous insulin infusion; DKA, diabetic ketoacidosis; FN, false negative; FP, false positive; SAP, sensor-augmented pump; SF, infusion site failure; TDD, total daily dose; TN, true negative; TP, true positive; Zone-MPC, zone model predictive control.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BAB has received research support from Medtronic, Dexcom, Insulet, Roche, Tandem, and Bigfoot Biomedical and is on advisory boards for Sanofi, Novo-Nordisk, and Becton Dickenson, and was a consultant for Dexcom. GPF is a consult for Abbott Diabetes Care and receives grant funding from Medtronic, Dexcom, Animas, Tandem, Bigfoot and Insulet. TTL has received research funding from Medtronic and Tandem and is currently employed by Insulet. DMM is on the advisory board for Insulet, is a consultant for Abbott Diabetes Care and receives research funding from Medtronic, Roche, and Dexcom. RG receives royalty payments on intellectual property related to the MPC algorithm employed in this study. FJD and ED have patents on the underlying MPC algorithms used in the study, and are currently receiving royalty payments on these patents. BWB has served as a consultant for Becton, Dickinson and Company. All other authors report no COI as it pertains to this work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by a grant from the Juvenile Diabetes Research Foundation (grant number 17-2013-471). The Clinical Translational Research Unit at Stanford was supported by NIH grant UL1 TR001085. The development of the Zone-MPC system was supported by grants from the National Institutes of Health (grant DP3DK094331 and DP3DK104057). Research device support was provided by Roche AG (Basel, Switzerland) and Dexcom, Inc (San Diego, CA, USA).
