Abstract
Background:
This study examines the utility of the first prefilled, rapid-acting insulin pen that can be dialed in half-unit increments. Dose accuracy and injection force were examined through a series of design-verification tests, and usability was established by human factors validation testing.
Methods:
Devices were tested for dose accuracy at 3 different doses and temperatures and under free fall, vibration, and cold storage conditioning. Injection force was measured at the maximum dose (30 units). Both experiments used the same semiautomated testing system. Usability was validated in a human factors simulated-use study that included 60 participants (patients with type 1 or type 2 diabetes [aged 10-79 years], adult caregivers, and health care providers).
Results:
The pen met the International Organization for Standardization (ISO) 11608-1:2014 requirements for dose accuracy at all settings and conditions tested. Furthermore, all individual results were within the ISO specification limits. Mean injection force across temperature settings ranged from 9.25 to 10.85 N at the highest dose. The usability validation study confirmed that use-related risks were reduced to the extent possible and that additional modifications were not likely to afford further reductions.
Conclusions:
The results from these studies demonstrated accurate dosing over the dose range (0.5-30 units) at different temperatures and conditions with an injection force that should accommodate the intended users. Use safety and usability in patients with diabetes, caregivers, and health care professionals were validated. The added convenience of this new half-unit, prefilled pen may ease the burden of diabetes management for patients who require smaller incremental dosing.
The global burden of type 1 diabetes (T1D) continues to increase (around 3% per year). 1 This disturbing and poorly understood trend is particularly evident in children where estimates show worldwide prevalence of T1D surpassed half a million for the first time in 2015 with 86,000 new cases per year (age range, 0-14 years). 1 In most Western countries, T1D accounts for over 90% of diabetes in children and adolescents. 2
Multiple daily insulin injections in children and adolescents can be challenging for both patients and caregivers, which can affect outcomes. 3 Recent data from the T1D Exchange (2013) indicated that less than one-third of pediatric patients met the HbA1c goal of <7.5% (ages one to <6 years, 27%; 6 to <13 years, 27%, and 13 to <20 years, 23%), 4 which is now the harmonized goal across all pediatric age groups.5-7 Modifications to design aspects of injection devices that diminish these challenges can help to ease the burden of treatment. The predominant injection options are vial and syringe, insulin pens, and insulin pumps,3,8-10 utilization of which varies by geographic region. 11 Both pumps and pens have distinct advantages compared with vial and syringe,8,9 but not all pediatric patients are candidates for pumps.12-14 Greater accuracy15-17 and preference for pen devices versus vial and syringe with respect to ease of use, convenience, and discreetness17-19 have been shown with better efficacy and safety outcomes,20-22 better adherence,20,22,23 and less pain.21,24
Pediatric patients with T1D often require small doses, dictated in part by low body weight and high insulin sensitivity.9,25,26 Certain adults may also have lower dosing requirements, including elderly patients and T1D patients with a high level of physical activity or high insulin sensitivity.27-29 Pen devices capable of delivering accurate, half-unit increments of insulin are available as an option for these patients, 30 including JuniorSTAR® (Sanofi S.A., Gentilly, France; JrS),31,32 NovoPen Echo® (Novo Nordisk, A/S, Bagsværd, Denmark; NPE),33-36 and HumaPen® Luxura™ HD (Eli Lilly and Company, Indianapolis, IN, USA; HPLHD).37,38 All are reusable pens with a dose range of 1 to 30 units (JrS) or 0.5 to 30 units (NPE and HPLHD).32,36,37
Prefilled (disposable) pens are another type of insulin pen. As these devices do not require replacement of cartridges, they may be easier to use, 39 and studies comparing ease of use and patient preferences for prefilled versus reusable pens have demonstrated greater ease of use and preference for prefilled pens.11,40,41 Moreover, usability in patients with strength, dexterity, or motor control deficiencies, such as children or elderly patients, may be enhanced as a result of the simplified operational aspect of prefilled pens. Children may particularly benefit from the standpoint of both physical dexterity capabilities and the convenience of a simplified injection routine under various social and recreational settings (schools, day care facilities, camps, exercise/sporting events, etc5-7,42). Recently, the first prefilled pen device designed for half-unit dosing, the Humalog® Junior KwikPen® (Eli Lilly and Company, Indianapolis, IN, USA; JrKP; Figure 1), has been approved by the US Food and Drug Administration. This device contains 3 mL of prandial insulin lispro (100 units/mL), has a dose range of 0.5 to 30 units, and dials in 0.5-unit increments.
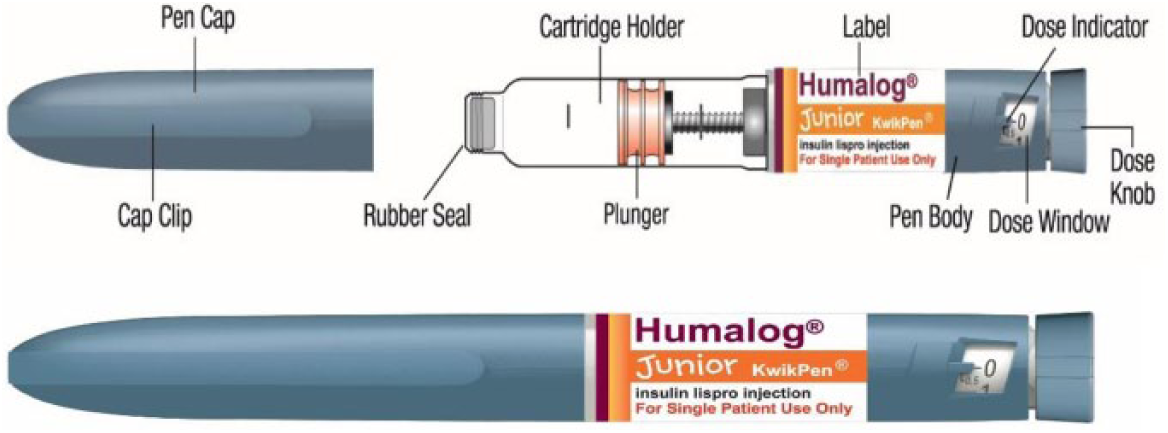
Humalog Junior KwikPen and components.
In this manuscript, we report results from 2 studies evaluating JrKP: a laboratory study that examined its functional performance and a usability study designed to validate safe and effective use in the intended populations. Specifically, dose accuracy and injection force were evaluated under different temperature settings and conditioning scenarios intended to simulate actual use circumstances. In addition, a human factors validation study assessed the use safety and usability of the device in representative patients and caregivers.
Research Design and Methods
Dose Accuracy
Dose accuracy testing was performed according to the International Organization for Standardization (ISO) standard for needle-based injection systems for medical use (ISO 11608-1:2014) 43 using a semiautomated test system. Devices were tested under a standard, cool, or warm atmosphere (23°C, 5°C, or 40°C, respectively). Automated collection of all measurements was performed by the test system. Pens were preconditioned then visually and functionally inspected. The investigator attached 31-gauge, 5-mm needles (Becton Dickenson and Company, Franklin Lakes, NJ, USA), and the devices were primed according to the instructions for use (IFU) and test system requirements prior to testing. Sixty pens were used for each temperature variation. Three different doses of insulin lispro (0.005, 0.15, and 0.3 mL [0.5, 15, and 30 units, respectively]) were dispensed from each pen from cartridges that were full, 2/3 full, or 1/3 full (20 each) according to a prespecified sequence (180 doses) to confirm accurate dosing over the dose range from full or partially filled pens. The investigator manually dialed each test dose; then a linear actuator ejected the dose with a 5-second hold time (per the IFU) directly into a catch-cup/pan assembly placed on a precision balance. The expelled dose was gravimetrically measured in grams, and the weights were converted to volume (milliliters) using the density (ρ, grams/milliliter) of the test fluid at environmental conditions. Confirmation of dose accuracy following various conditioning scenarios, including free fall, vibration, and cold storage, was also performed.
Injection Force
Injection force testing was performed using the above-mentioned test system. Sixty pens were evaluated at each of the 3 temperature settings with preconditioning and device set up executed as above. A 0.3-mL (30-unit) dose was manually dialed then ejected by the test system at the button speed of 10.2 inches/minute (flow rate, 4.6 units/second). The maximum force over the dosing interval was recorded.
Human Factors Validation Study
The study was conducted in accordance with the Declaration of Helsinki, and all participants provided informed consent. The primary objective was to validate usability and use safety of the JrKP user interface for the performance of critical tasks by the intended users. The unique user-interface elements of JrKP (half-unit dose scale and differentiation features [color, label, tradename, etc]) were the focus of an iterative series of 6 formative human factors evaluations. Safe and effective use of JrKP, including the device, carton, and IFU, was then evaluated in a human factors validation study assessing the performance of critical tasks in a typical user environment by representative users. The sequence of studies was executed according to consensus standards, 44 regulatory guidance, 45 and industry best practices. The full validation study involved 79 participants, including insulin-experienced patients (10-17 and >18 years of age) with either T1D or T2D and adult caregivers as well as health care providers and 4 color-blind individuals. Because the level of user training in actual use environments varies, both trained and untrained participants were included, and all patients/caregivers were assessed for health literacy46,47 (adult patients/caregivers also assessed for visual acuity).48,49 The critical dosing tasks were evaluated in a simulated-use investigation. For this scenario, 60 participants were included (omitting the 4 colorblind [nonpatient] individuals and 15 retail pharmacists who were specifically included for differentiation or differentiation/knowledge assessments, respectively). Participants were initially introduced to the test protocol then provided with the test materials (simulated pharmacy label, pen needle, and carton containing JrKP devices and patient IFU) and asked to inject a 7.5-unit dose into an injection pad using a production-equivalent device under a simulated-use environment (room-temperature area with standard lighting typically found in a home, school, or office and minimal distractions).
The tasks being evaluated were dialing the dose, inserting the needle into the injection pad, and injecting the dose. A moderator was present to record use-related problems but did not provide any guidance or other assistance with the injection task. Results were recorded as successes, close calls, or failures (Table 1).
Simulated-Use Tasks for the Human Factors Validation Study and Assessment Criteria. a
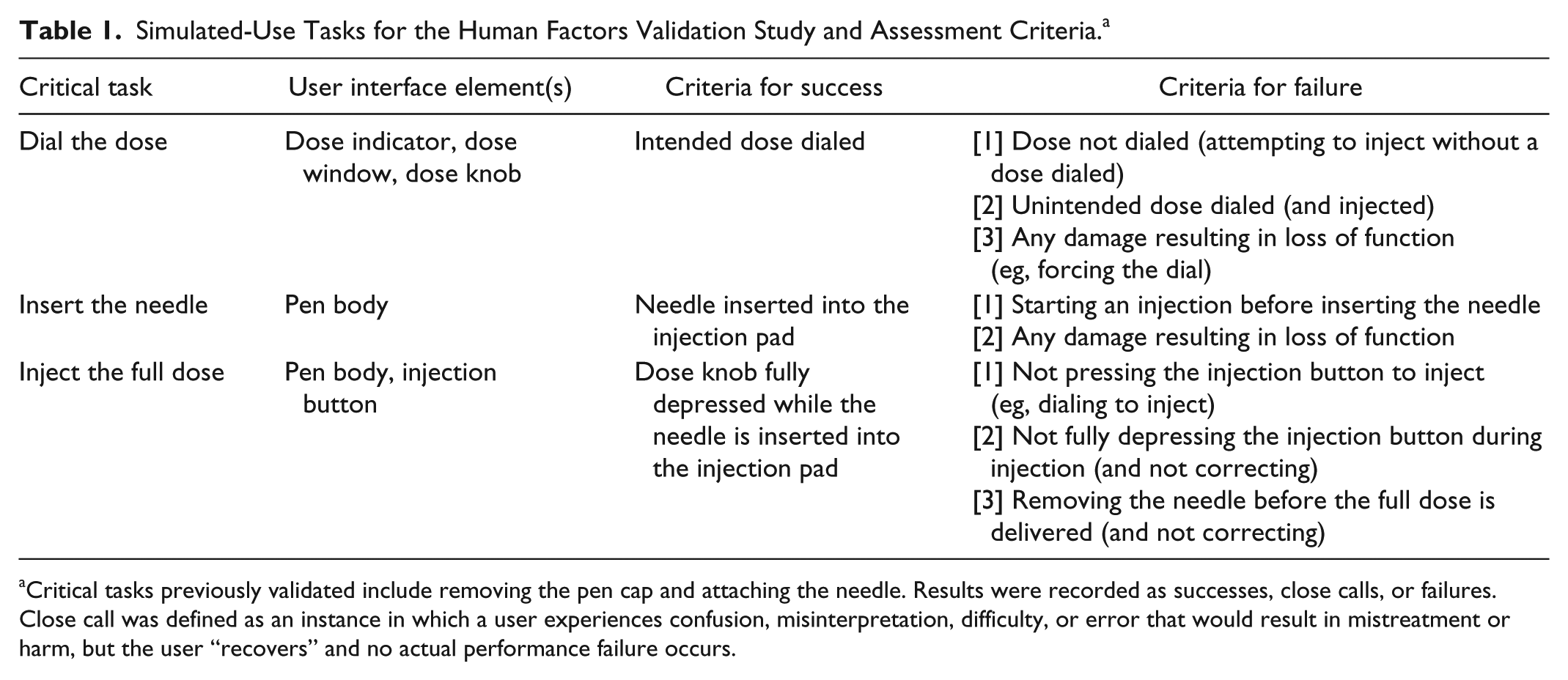
Critical tasks previously validated include removing the pen cap and attaching the needle. Results were recorded as successes, close calls, or failures. Close call was defined as an instance in which a user experiences confusion, misinterpretation, difficulty, or error that would result in mistreatment or harm, but the user “recovers” and no actual performance failure occurs.
Statistical Methods
Analyses were performed using SAS JMP 11.1.1 (SAS Institute, Cary, NC, USA). For dose accuracy testing, mean volume (standard deviation [SD]) was calculated for each data set. Two-sided target k values (tolerance limit factors [ktar]) were based on the confidence level (95%), probability content (p, 97.5%), and number of measurements. 43 Specification limits were determined from the dose plus or minus the dialing resolution (±DR, for doses ≤10 units) or a fraction of the dose (±5%, for doses >10 units). The 2-sided statistical tolerance interval was calculated from the mean expelled volume plus or minus the product of the SD and ktar. To meet the ISO standard for dose accuracy, the calculated tolerance interval, which is an estimate of the range within which 97.5% (p) of all doses are predicted to reside with 95% confidence, must fall within the predefined specification limits. For injection force analyses, means (SDs) of the maximum force values were determined for each test group.
Analyses of human factors results were conducted according to prespecified criteria (Table 1) using data from all participants completing the study. Statistical justification of sample sizes in usability testing from current standards supports inclusion of at least 15 participants for each distinct user group for a human factors validation study. 50 Qualitative analyses of all observed use problems were conducted and were based on root cause analyses, known use problems, and use errors previously identified from a risk assessment method (application failure modes and effects analyses [AFMEA]). Because statistical methodologies are inappropriate for analyses of human factors use-safety data, no statistical analyses were performed.
Results
Dose Accuracy
Dose accuracy results (mean [SD]) are shown in Table 2. Plots of individual results (Figures 2 and 3) confirm that the findings met the ISO requirements, as calculated tolerance intervals were well within ISO specification limits, validating accurate dosing across the dose range (0.5-30 units). Furthermore, the plots show that all doses fell within the limits for each test group/testing scenario.
Dose Accuracy Testing Results for Humalog Junior KwikPen.
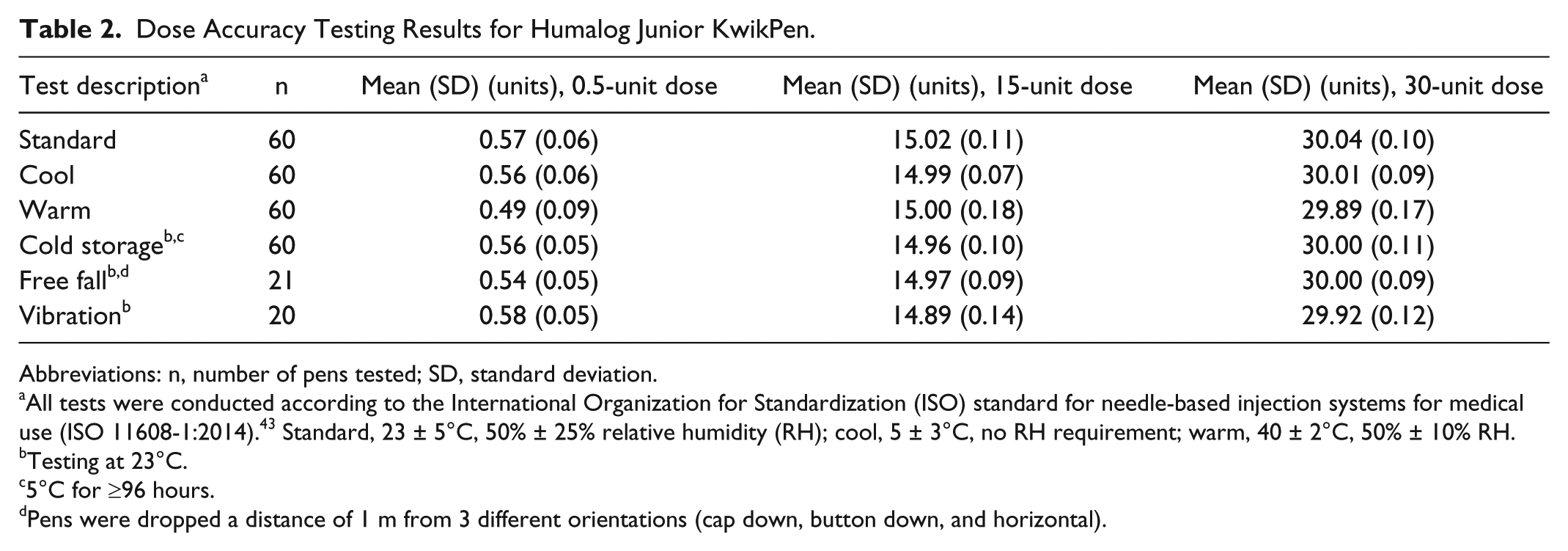
Abbreviations: n, number of pens tested; SD, standard deviation.
All tests were conducted according to the International Organization for Standardization (ISO) standard for needle-based injection systems for medical use (ISO 11608-1:2014). 43 Standard, 23 ± 5°C, 50% ± 25% relative humidity (RH); cool, 5 ± 3°C, no RH requirement; warm, 40 ± 2°C, 50% ± 10% RH.
Testing at 23°C.
5°C for ≥96 hours.
Pens were dropped a distance of 1 m from 3 different orientations (cap down, button down, and horizontal).
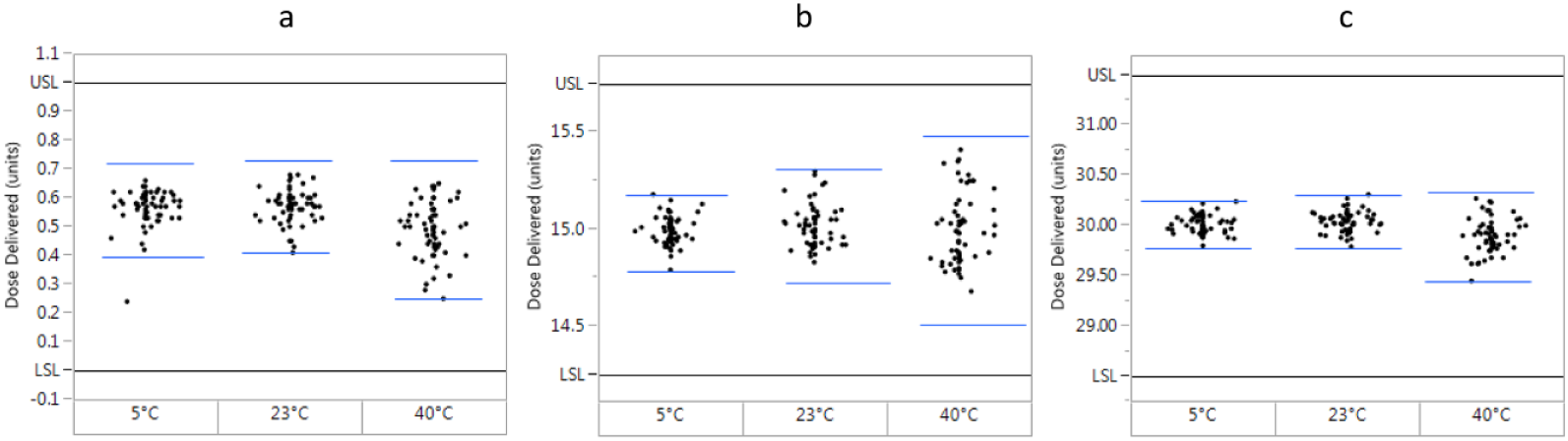
Distribution plots of actual dispensed doses at three different temperatures (n = 60). (a) 0.5-unit doses. (b) 15-unit doses. (c) 30-unit doses. Black lines represent International Organization for Standardization (ISO) upper and lower specification limits (USL and LSL, respectively, ISO 11608-1:2014), which were determined from the dose plus or minus the dialing resolution (doses ≤10 units) or a fraction of the dose (±5%, doses >10 units). Blue lines represent the tolerance intervals, which were calculated from the mean expelled volume plus or minus the product of the SD and ktar (tolerance limit factor, 2.67). To meet the ISO standard for dose accuracy, the tolerance interval must fall within the ISO specification limits.
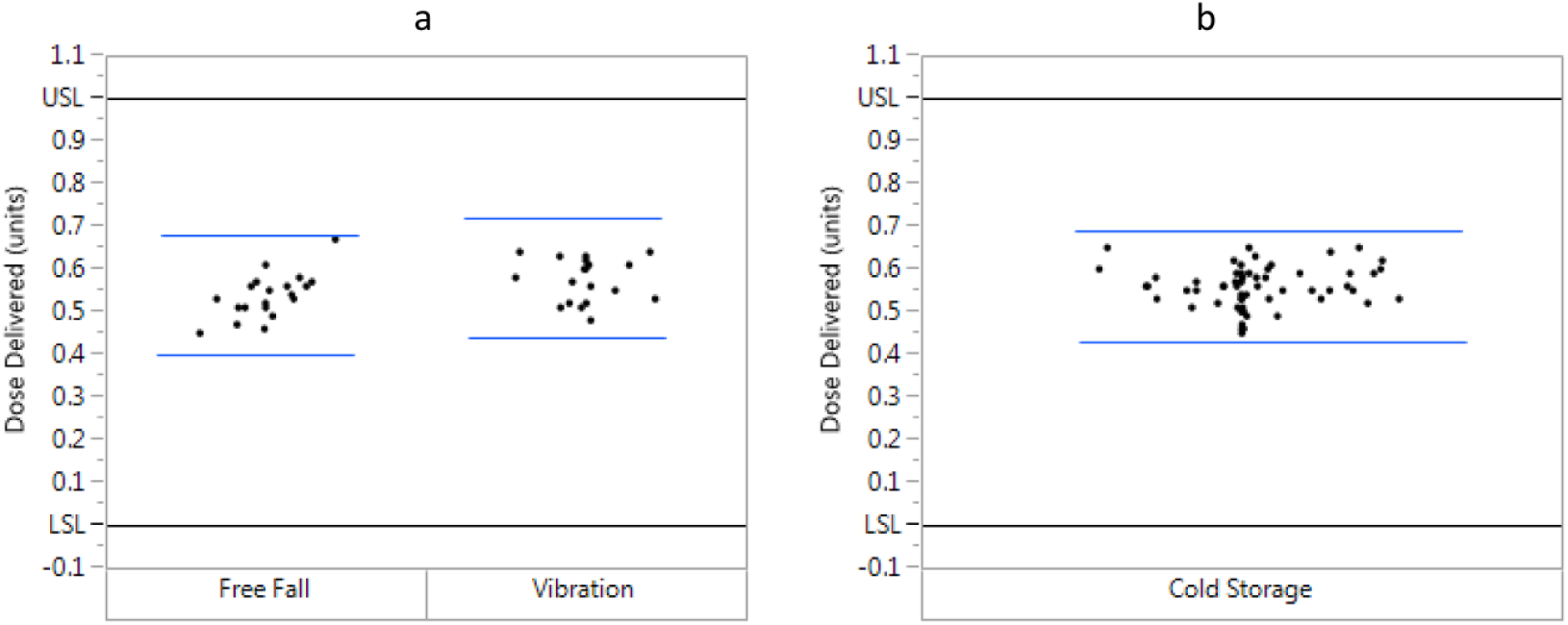
Distribution plots of actual dispensed doses (0.5-unit dose, 23°C) following conditioning. (a) Free fall (n = 21) and vibration (n = 20). (b) Cold storage (n = 60). Black lines represent International Organization for Standardization (ISO) upper and lower specification limits (USL and LSL, respectively, ISO 11608-1:2014), which were determined from the dose plus or minus the dialing resolution. Blue lines represent the tolerance intervals, which were calculated from the mean expelled volume plus or minus the product of the SD and ktar (tolerance limit factor, 2.67 [n = 60], 2.73 [n = 21], 2.76 [n = 20]). To meet the ISO standard for dose accuracy, the tolerance interval must fall within the ISO specification limits.
Injection Force
Mean injection force results for the maximum dose (30 units) at different temperatures are shown in Table 3. Under standard conditions, the pen operated with an injection force of 9.3 N.
Maximum Injection Force for the Humalog Junior KwikPen. a

Abbreviations; IF, injection force; n, number of pens tested; N, Newtons; SD, standard deviation.
Button speed, 10.2 inches/minute (insulin flow rate, 4.6 units/sec). The maximum IF over the dosing interval was recorded for each test instance. Results represent mean (SD) of these recorded values.
Standard, 23 ± 5°C, 50% ± 25% relative humidity (RH); cool, 5 ± 3°C, no RH requirement; warm, 40 ± 2°C, 50% ± 10% RH.
Human Factors Validation Study—Simulated-Use Scenario
All 60 participants completed the study, including 15 pediatric patients, 17 adult patients, 13 adult caregivers, and 15 nurses. Demographics of the study population are shown in Table 4. All participants completed the 3 critical tasks successfully (dialing the dose, inserting the needle into the injection pad, and injecting the dose) with the exception of one trained pediatric patient and one trained caregiver, both of whom dialed and injected 7.0 units instead of 7.5 units.
Population Demographics for Human Factors Validation—Simulated-Use Testing. a
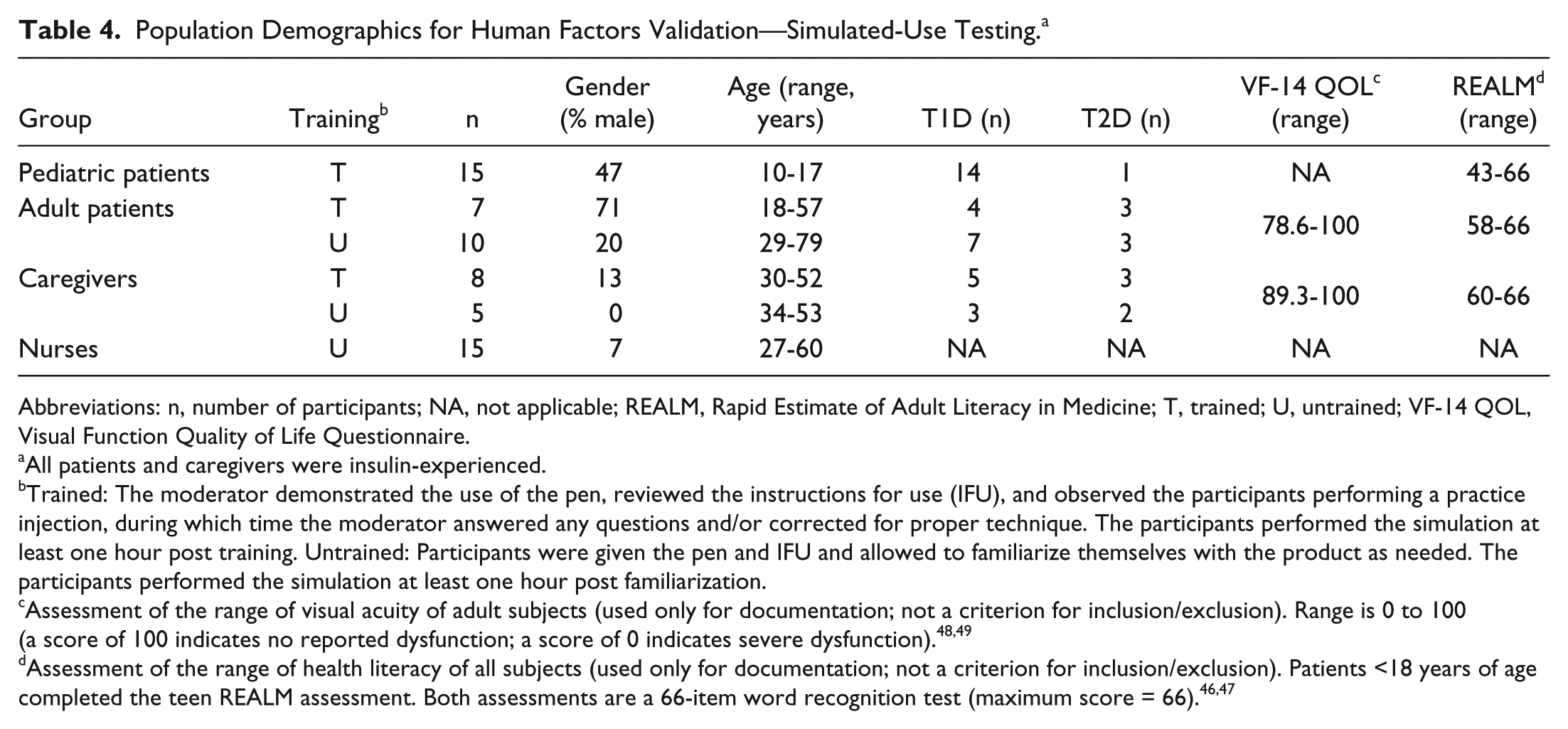
Abbreviations: n, number of participants; NA, not applicable; REALM, Rapid Estimate of Adult Literacy in Medicine; T, trained; U, untrained; VF-14 QOL, Visual Function Quality of Life Questionnaire.
All patients and caregivers were insulin-experienced.
Trained: The moderator demonstrated the use of the pen, reviewed the instructions for use (IFU), and observed the participants performing a practice injection, during which time the moderator answered any questions and/or corrected for proper technique. The participants performed the simulation at least one hour post training. Untrained: Participants were given the pen and IFU and allowed to familiarize themselves with the product as needed. The participants performed the simulation at least one hour post familiarization.
Discussion
The JrKP, containing 3 mL of prandial insulin lispro (100 units/mL), is the first prefilled pen device designed to deliver insulin in half-unit increments. ISO specification limits are more stringent for pens that dial in half-unit versus 1-unit increments at doses below 20 units (Figure 4). 43 As shown in the current study, JrKP met these requirements at all doses and conditions tested, and no individual results were outside the acceptable range. This indicates accurate dosing across the dose range (0.5-30 units) under different conditions and temperature variations intended to simulate real-world use, such as transporting or dropping a pen.
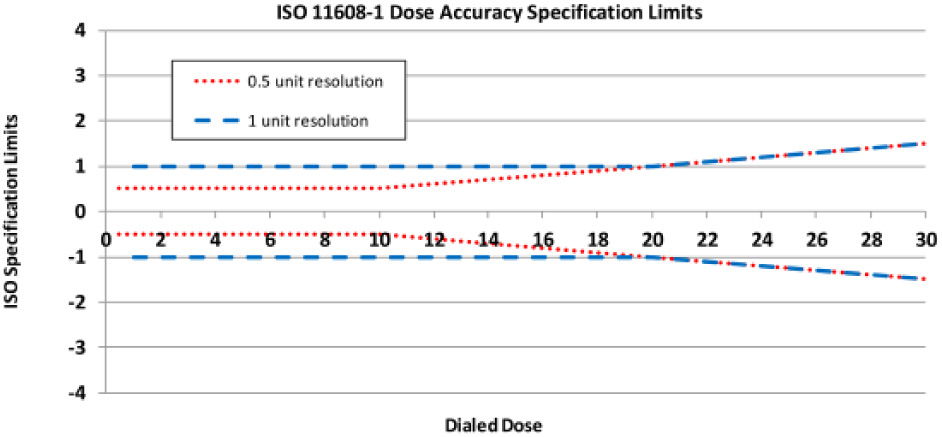
Dose accuracy specification limits for pens with half-unit and 1-unit dialing resolution (DR). ISO dose accuracy specification limits are set to the greater of ±DR or ±5% of the dose. For smaller doses, the specification limit is ±DR. Once the dose exceeds the point where the DR equals 5% of the dose (transition point), the latter is used. Thus, at doses below 20 units (0.2 mL), the limit for pens dialing in 1-unit increments is ±1 unit (±0.01 mL); for pens with half-unit DR, the limit is equal to either the DR (±0.5 units [0.005 mL] for doses below 10 units [0.1 mL]) or ±5% of the dose (10- to 20-unit doses). Beyond 20 units, specification limits are equal to ±5% of the dose for both pens.
Accurate dosing with half-unit resolution could be particularly important for children and adolescents with T1D, 3 who may require small insulin doses based on low body weight, pubertal status, 25 and/or high insulin sensitivity. 26 Infants and toddlers in particular may require precise dosing in less than 1-unit increments. 25 Other factors can also affect insulin doses, such as transient decreases in insulin requirements (following insulin initiation [honeymoon period]) and inadequate recognition of and neurological vulnerability to hypoglycemia.5,6,25,26
It is important to consider functional aspects of the half-unit device in light of the capabilities of pediatric patients. One practical aspect of an insulin pen that may impact its usability is injection force, which is a measure of the effort required by the user to depress the dose knob during an injection. The influence of injection force on usability was demonstrated in a study in patients with T2D comparing injection force and user preferences for FlexPen® versus Next Generation FlexPen (NGFP). For this comparison, a 30% lower injection force for NGFP contributed to simpler, more comfortable use. 51 No ISO standards exist for injection force; however, it should not exceed a level that can be comfortably achieved by the intended users. Injection force determined in the current study was found to be suitable relative to physical strength capabilities for pediatric patients and caregivers ascertained in a study by Peebles et al. In the Peebles study, finger-push strength measurements using the thumb or index finger were reported for various age groups spanning a broad range of users (2 to 90 years old, male and female). 52 The lowest measured strength capability for the age groups studied was associated with a mean (SD) force of 26.9 (18.8) N for a downward push with the thumb and 21.8 (7) N using the index finger (range was 23.5-53.8 and 14.7-35.0 N, respectively). In the current study, the mean injection force for JrKP under standard conditions (9.3 N [1.1]) is below the lowest mean strength measurement for all age groups, as reported in the Peebles study, and thus should accommodate the target population.
Along with injection force, the hold time (ie, period of time the needle needs to remain in the tissue after injection [dwell time]) may also impact usability and comfort.53,54 The hold time used in this study was 5 seconds (as directed in the IFU), which is similar to or lower than that of the reusable half-unit devices (HPLHD, 5 seconds; NPE, 6 seconds; and JrS, 10 seconds). 30
The JrKP has features similar to those of other KwikPen (KP) devices but with a single-dose range of 0.5 to 30 units dialed in half-unit increments. The KP platform includes several insulin pen devices, the first of which (Humalog KwikPen [Eli Lilly and Company, Indianapolis, IN, USA; HKP]) became commercially available in 2008. These devices have been used broadly in various regions and racial/ethnic populations and age groups. Several studies have validated the dose accuracy of HKP and/or assessed its functional aspects or usability.18,55-61 For example, significantly greater accuracy and user preference for HKP versus vial and syringe have been documented for several attributes. 18 Dialing and dosing JrKP uses the same steps as those for all KP products, though the dialing resolution is in half-unit increments. All KP devices use a single-step dose setting (displayed in the dose window) with tactile clicks and a dial-back feature to correct a misdialed dose. Other notable features include visibility of the cartridge contents throughout the deliverable volume, dosing restrictions that preclude dialing a dose that exceeds the amount of insulin remaining in the cartridge, and an ergonomic design to facilitate control and stability during use.
Usability of other KP-based devices has been validated in a number of human factors engineering studies (15 studies; total number of participants, 1085 [106 children/adolescents]). Human factors engineering focuses on interactions between a device and the user and is a well-established approach toward the design of safe and effective user-device interfaces. 62 For medical devices, the objective of this iterative process is to identify and minimize use-related risks/hazards and confirm these have been reduced to the lowest level that can be reasonably achieved. Accordingly, known use-related problems for insulin-pen injectors were identified and summarized along with the previously validated mitigations for each. In tandem, risk assessments of observed use errors were performed, including evaluation of the consequences and associated risk factors (AFMEA). No previously unidentified use errors were observed. Thus, in light of previous successful human factors validations of KP design features, only the potential use-related risks associated with characteristics unique to JrKP were evaluated, such as the dosing scale. In the simulated dosing validation, all critical tasks were successfully completed by all participants with 2 exceptions: one pediatric patient and one adult caregiver. Both participants were trained and insulin-use experienced, and both errors involved under-dosing by 0.5 units. Root-cause analyses determined that these were due to a simple action slip (ie, unintended action), 63 and both participants were able to dial an additional dose(s) successfully. An analysis of the residual use-related risks determined that these were consistent with those of approved insulin “dial-and-dose” pen devices, including KP devices, which have demonstrated a pattern of safe and effective use in the market. Findings from the human factors validation and residual risks analysis showed that the design of JrKP reduced use-related risks to the extent possible, given the limitations of conventional dial-and-dose technology, and that additional modifications were not likely to further reduce risk.
The usability study had limitations. Tests were simulations rather than actual clinical use studies, though the methods conform to consensus standards, regulatory guidance, and industry best practices. Also, inclusion criteria specified a minimum age of 10 years. While this is a reasonable threshold, the age at which children begin to perform self-injection is more associated with developmental maturity than a specific chronological age. 3
Conclusions
Recently approved JrKP offers a new delivery option for rapid-acting insulin therapy in pediatric and adult patients who require half-unit dose increments of insulin. This pen, which is the first prefilled insulin pen designed for half-unit dosing, meets ISO standards for dose accuracy across the full dose range (0.5-30 units) under various conditions. Other functional outcomes, such as injection force, suggest that the pen should accommodate the capabilities of the intended users, and use safety and usability for patients, caregivers, and health care providers was supported by a human factors simulation. Half-unit dose accuracy is especially important for pediatric patients, and the added convenience of a prefilled pen may simplify diabetes management with the goal of promoting both better adherence and developmentally appropriate self-care skills in this challenging age group.
Footnotes
Acknowledgements
The authors thank Kenneth Fowler and Daneen Angwin (both Eli Lilly and Company) for insight and suggestions on testing methods and analyses and Amy Kornokovich (Eli Lilly and Company) for statistical input.
Abbreviations
AFMEA, application failure modes and effects analyses; DR, dialing resolution; HKP, Humalog KwikPen; HPLHD, HumaPen Luxura HD; IDF, International Diabetes Federation; IFU, instructions for use; ISO, International Organization for Standardization; JrKP, Humalog Junior KwikPen; JrS, JuniorSTAR; KP, KwikPen; k tar , tolerance limit factor; NPE, NovoPen Echo; SD, standard deviation; T1D, type 1 diabetes; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CMK, JRK, TMM, and SMC are full-time employees of Eli Lilly and Company. JRK and SMC are stockholders in Eli Lilly and Company. TMM is a former employee of Boehringer-Ingelheim Pharmaceuticals.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Eli Lilly and Company, Indianapolis, Indiana. The funding organization participated in study design, data collection and analysis, and manuscript preparation and approval for submission.
