Abstract
Background:
Self-monitoring of blood glucose (SMBG) is a key pillar of personal diabetes management. The objective of this observational study was to analyze diabetes self-management (DSM) and glycemic outcomes before and during system implementation in real-life settings of a blood glucose meter system with a color-coded display of glucose levels, which helps identify out-of-range levels.
Methods:
A total of 193 insulin-treated diabetes patients (11% T1DM; 55% male, age 60 ± 4 years, mean diabetes duration 14 ± 9 years, HbA1c 8.68 ± 1.2%) were enrolled into the study. Both the Diabetes Self-Management Questionnaire (DSMQ) and glycemic control were analyzed at baseline and 3 and 6 months after study initiation.
Results:
DSMQ general perception improved significantly by the end of the study period (“Sum Scale,”
Conclusion:
This real-world study demonstrates that SMBG implemented via this new blood glucose meter not only leads to an improvement in metabolic control, but also is associated with a significant improvement in diabetes management.
Self-monitoring of blood glucose (SMBG) plays an important role in diabetes management.1,2 SMBG has been shown beneficial for the achievement of glycemic targets and reducing glycemic variability, allowing even for hypoglycemia anticipation.3-6 In patients with type 2 diabetes treated with insulin, SMBG is not only associated with an improved metabolism, it also strengthens patient responsibility,7-11 increasing disease awareness thereby influencing lifestyle. 12 Furthermore, an epidemiological cohort study demonstrated a positive influence of frequent SMBG on diabetes-related morbidity and overall mortality. 13
Various national and international guidelines recommend regular SMBG for diabetes management and treatment support. The American Diabetes Association guideline recommends SMBG to both health providers and patients for glycemic control assessment. 1 Additional recommendations, such as the guidelines by the International Diabetes Federation (IDF), 14 the European Society of Cardiology (ESC), 15 and the European Association for the Study of Diabetes (EASD) 16 consider SMBG an integral part of diabetes self-management (DSM). Moreover, European consensus recommendations stress individual SMBG strategies for patients with diabetes.17-20
OneTouch® Verio® (Johnson & Johnson Medical GmbH; Geschäftsbereich Johnson & Johnson Diabetes Care Companies; Johnson & Johnson Platz 2, 41470 Neuss, Germany) is a new blood glucose meter that informs users via a color-coded display system whether the measured values are within (green), below (blue) or above (red) their target range, which would be individually tailored to the patient by their health provider. It provides easy-to-understand measurement results, increasing patient awareness, thus allowing for better self-management of their blood glucose levels, irrespective of whether exercise, food choice or insulin dose adjustment is required. The system stores measurements and therefore allows for tracking progress. Patients are informed and encouraged by the system via automatic on-screen messages. For instance, continuously within-target values, or getting back into range after being too high are reaffirmed by success messages.21-23 A summary of device features can be observed in Figure 1.
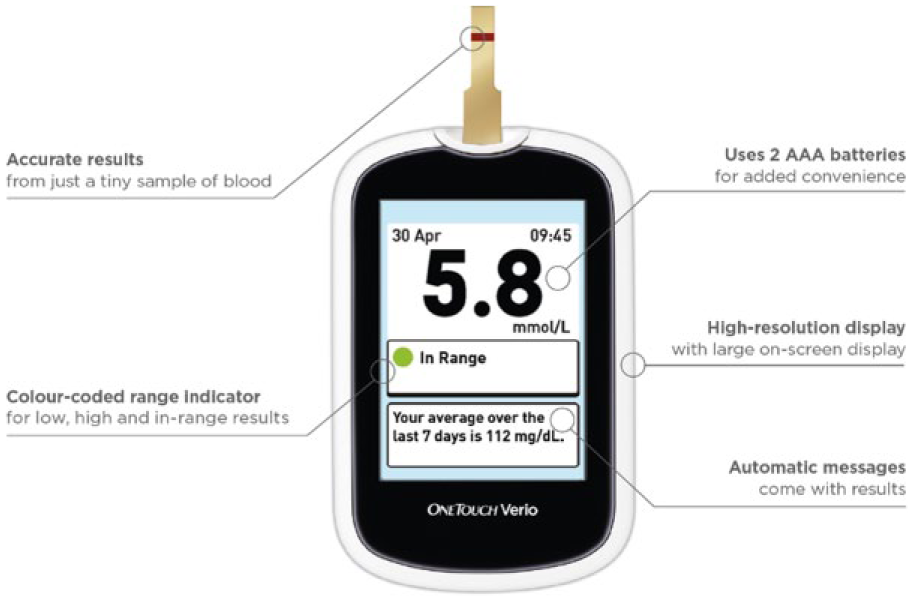
Main features of the OneTouch® Verio® blood glucose meter.
In this observational study, we intended to analyze DSM before and during system implementation in real-life settings and demonstrate its effects on glycemic control.
Methods
We performed a national multicenter (9 trial sites) prospective observational study. The trial was performed in agreement with the requirements from the Bavarian Medical Council. All patients provided written informed consent after being properly informed in writing about the objectives and particulars of the study. The duration of the observational study was 6 months; values were analyzed upon launch (baseline), at 3 months and at 6 months.
Biological parameters examined included blood glucose and HbA1c levels, which were evaluated by means of quality-controlled laboratory methods as were lipids (LDL, HDL, and triglycerides). For analysis of biological factors, the usually accepted reference values/intervals were employed. Anthropometric data included the patient’s body mass index (BMI) and waist size. Therefore, weight and height were noted at baseline and weight followed up over the course of the study. To get a general overview over the patient’s general health status, relevant medical data (ie, diabetes-related diseases) was extracted from the patient’s medical history. Moreover, systolic and diastolic blood pressures were monitored throughout the study.
Patients with type 1 diabetes and patients with type 2 diabetes were eligible if they had been treated with insulin for at least 6 months prior to enrollment. Key inclusion criteria were a glycated hemoglobin level of 7.5% or more, and a good knowledge of SMBG plus regular blood glucose metering for at least 6 months or longer before inclusion. Key exclusion criteria included known diabetes mellitus without insulin treatment, febrile diseases upon enrolment and/or gastric resection, or any sort of pancreas condition (chronic pancreatitis, pancreatic cancer, total pancreatectomy, hemochromatosis or cystic fibrosis). Endocrine disorders such as hypercortisolism, hyperthyroidism, Addison’s disease, Cushing’s disease, pheochromocytoma, acromegaly, Conn syndrome or glucagonoma, or cortisone therapy precluded patient recruitment. Under age (<18 years old), pregnant or breast-feeding patients were also excluded from participation in the study.
Patients provided their information/data during their regular care visits to the recruitment centers: At baseline, where they provided informed consent on trial participation and two further visits 3 and 6 months after study initiation. In addition, patients were instructed on meter use and their personal glucose targets established with their health care provider.
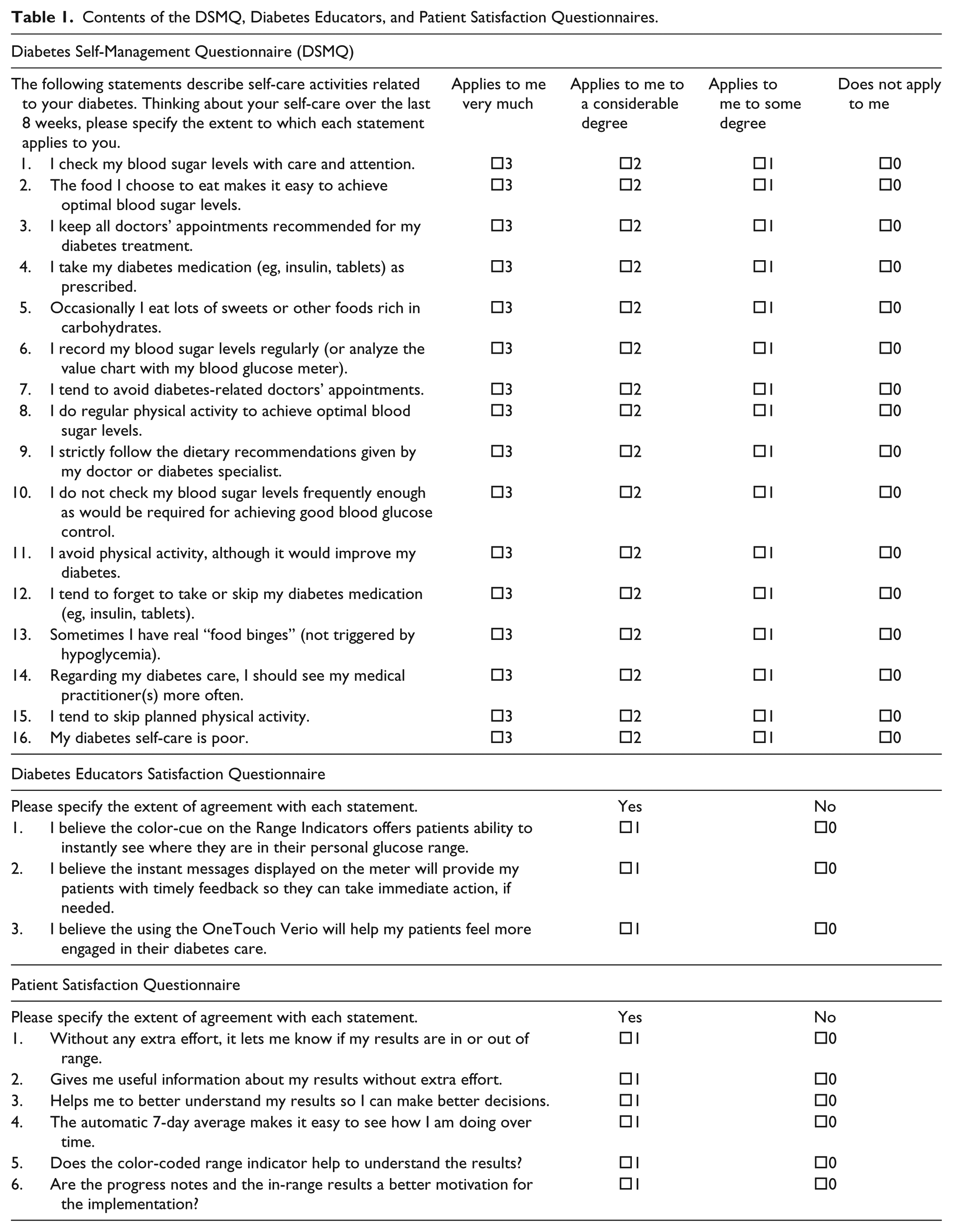
In each visit, patients were questioned about SMBG frequency and biological parameters like HbA1c and blood glucose, among others, were evaluated for further comparison. As data were extracted from routine patient visits, there was no extra requirement to analyze any biological parameter such as lipids or else. Patients were also asked to fill out a validated Diabetes Self-Management Questionnaire (DSMQ) consisting on 16 distinct questions on various points, 24 to assess the possible changes to their disease self-management brought about by the intervention, that is, by use of the OneTouch meter in their daily SMBG routines. DSMQ subscales (“Glucose Management,” questions 1, 4, 6, 10, 12; “Dietary Control,” questions 2, 5, 9, 13; “Physical Activity,” questions 8, 11, 15; “Health-Care Use,” questions 3, 7, 14) scores were calculated according to Schmitt et al. 24 A total score “Sum Score” including all DSMQ questions and representative of overall DSM was also calculated as in Schmitt et al. 24
Moreover, in the follow-up visits at 3 and 6 months after trial launch, patients and diabetes educators at the recruitment sites were asked to submit further satisfaction evaluations by filling out a short binary (yes/no) questionnaire. A description on the content of the three questionnaires can be found in Table 1.
Contents of the DSMQ, Diabetes Educators, and Patient Satisfaction Questionnaires.
Target criteria for the observational study include changes in subjective perceptions regarding diabetes management as well as changes in laboratory parameters. The primary endpoint was the DSMQ “Sum Score.” It was analyzed in 136 patients, in whom the DSMQ was obtained at all three visits. The analysis of the DSMQ subscales was part of secondary endpoints. The additional important secondary endpoint was changes to HbA1c levels.
Data were gathered by the site investigators, and transmitted for analysis to Sciarc GmbH. Statistical analysis of results was performed with the SPSS software package. For continuous variables, as well as for the analysis of DSMQ Scores, a Friedman Test corrected for multiple comparisons (post hoc Nemenyi test) was used; P values refer to differences between baseline, 3 months and 6 months. For binary variable analysis a McNemar test was used.
Results
From July 2015 until January 2016, a total of 193 patients were screened and enrolled in the study; of these patients, 150 (77.7%) attended the last follow-up visit at an investigator site.
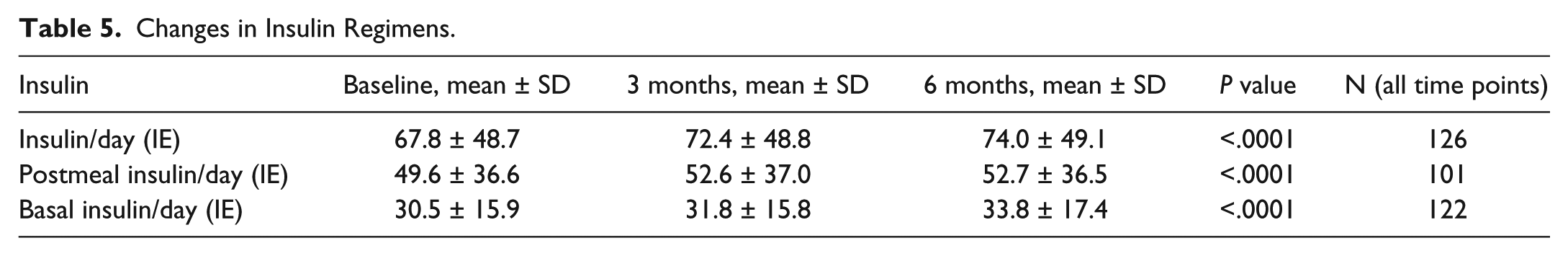
Demographic and clinical characteristics of the patients at baseline are summarized in Table 2. Average age was 60 years old and 106 (54.9%) were male. 172 (89.1%) were patients with T2DM under insulin treatment, while the rest were patients with T1DM. The overall mean diabetes duration was 13.7 years, and the mean glycated hemoglobin level was 8.68%. At baseline, the average SMBG reported frequency was over three times a day (3.34 ± 1.49). Insulin therapy at baseline consisted on average of 67.8 ± 48.7 (IE) per day, where 49.6 ± 36.6 IE corresponded to postmeal insulin and a further 30.5 ± 15.9 IE to basal insulin. Furthermore, nearly 60% of patients received metformin; other antidiabetic medications, like GLP-1 RA or DPP-4 inhibitors were also part of the standard diabetes therapy, but were used by far less patients as metformin (13.4 and 12%, respectively). In association with a study population with a majority of T2DM, patients were on average overweight as demonstrated by average BMI (29.3 ± 5.78).
Baseline Characteristics of Patients Enrolled in the Study.
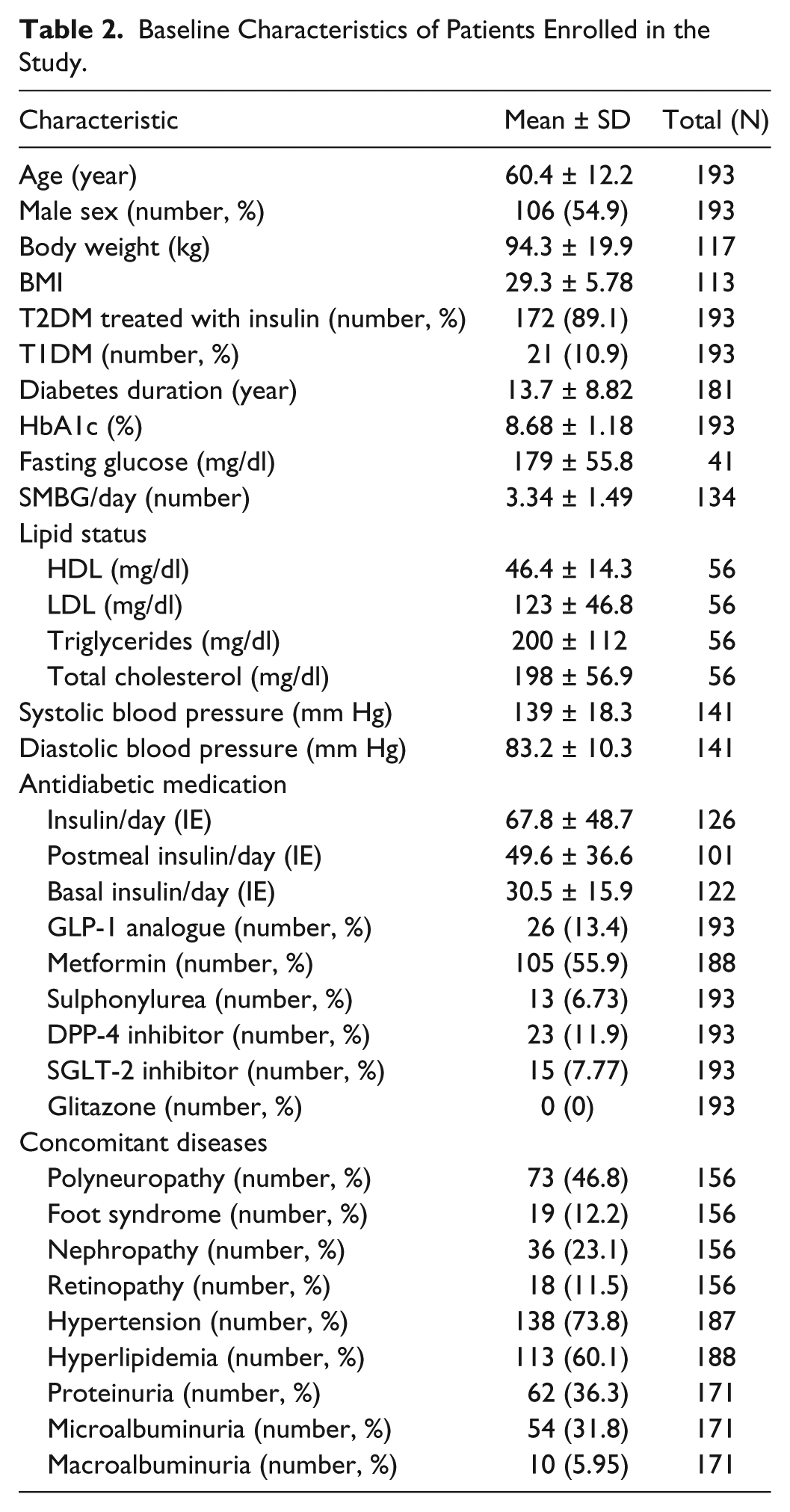
As expected from patients with a relatively long disease duration, many patients suffered from diabetes-related microvascular ailments such as polyneuropathy (46.8%), foot syndrome (12.2%), retinopathy (11.5%) or nephropathy (23.1%). In association with renal damage, 36.3% of enrolled patients suffered from proteinuria (31.8% microalbuminuria; 5.95% macroalbuminuria). The lipid status of patients at enrollment showed hyperlipidemia in 60.1% of cases, with average total cholesterol levels of 198 ± 56.9 mg/dl, and triglyceride, HDL and LDL cholesterol levels within healthy limits but nearly out of range (Table 2). Since high blood pressure is associated with diabetes, hypertension was also found in almost 74% of enrolled patients, with an average systolic and diastolic blood pressure of 139 ± 18.3 mmHg and 83.2 ± 10.3 mmHg, respectively.
In terms of primary end-point assessment, the analysis of the changes in response patterns from patients to the DSMQ over the course of the study showed a significant improvement of the “Sum Score” (
Differences in DSMQ Scores for the Different Subscales and Total Score at All Time Points.
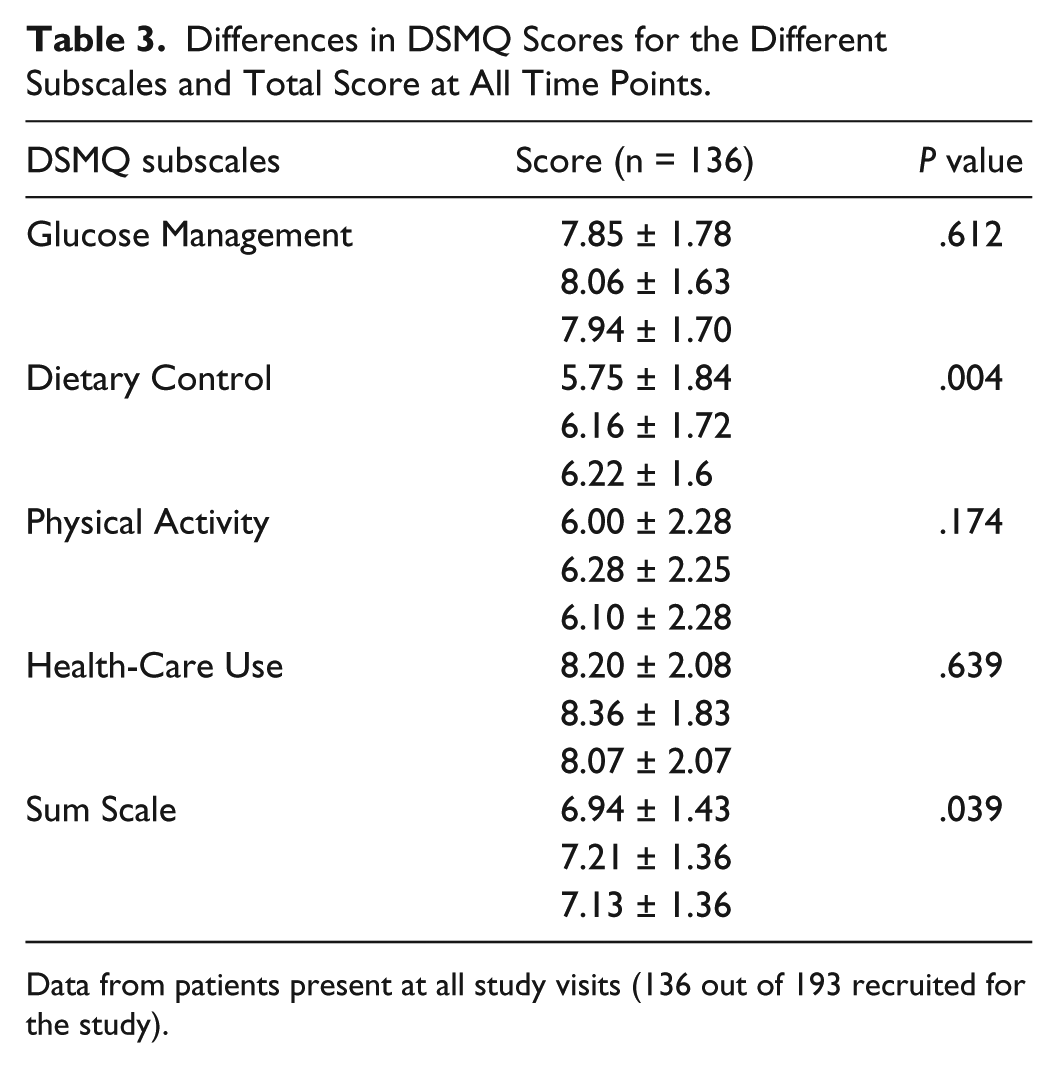
Data from patients present at all study visits (136 out of 193 recruited for the study).
Over the course of the study, there was a significant improvement of glycemic control as shown by the decrease both in HbA1c (
Secondary Biological Outcomes.
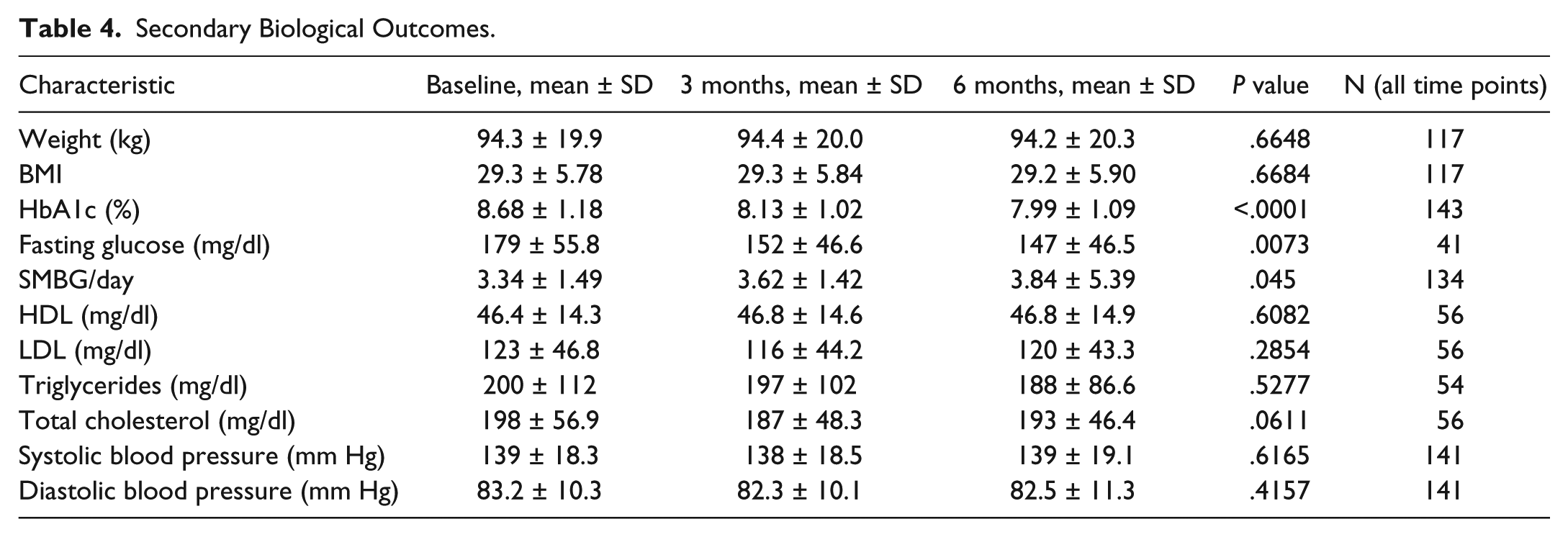
Other parameters such as BMI or body weight did not change over the course of the study, nor did the average blood pressure or the overall lipid status of study participants (Table 4), even when a tendency toward improved total cholesterol levels could be detected (198 ± 56.9 to 193 ± 46.4 mg/dl;
SMBG frequency did also significantly increase during the intervention, demonstrating an increase of one more time a day to a total of nearly 4 times a day after 6 months (
Changes in Insulin Regimens.
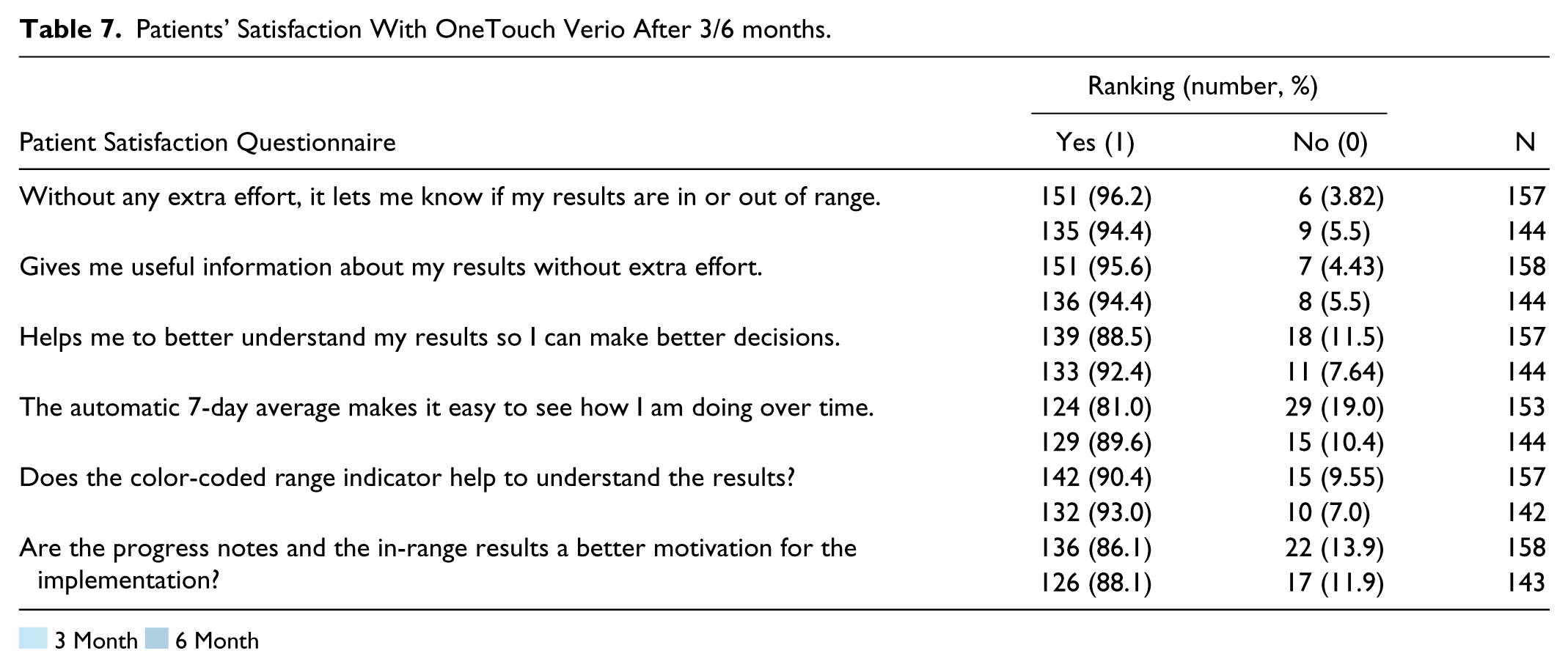
Three and six months after trial launch, both diabetes educators and the patients themselves believed, with between 72.3% to 96.2% agreement to the different satisfaction statements, that the use of the color coded SMBG meter OneTouch was helpful for DSM (Tables 6 and 7) .
Diabetes Educators’ Satisfaction With OneTouch Verio After 3/6 months.

 3 Month
3 Month  6 Month
6 Month
Patients’ Satisfaction With OneTouch Verio After 3/6 months.
 3 Month
3 Month  6 Month
6 Month
Discussion
In this observational study, we could show that OneTouch, a color-coded display SMBG meter with extended functionality, 21 has significant effects on DSM during and after system implementation in real-life settings, and were able to demonstrate its effects on glycemic control.
The analysis of self-reported disease management via DSMQ, showed a significantly improved subjective perception on the quality of their personal diabetes management (“Sum Scale,”
When asked about the influence of using the OneTouch meter in their DSM, both patients and diabetes educators agreed (72.3-96.2% assent) on its positive effects and usefulness for therapy adaptation. This self-reported improved diabetes management due to the use of the OneTouch meter over a continuous period of 6 months could have been the reason for the significant decrease of HbA1c levels (
This improved self-management is also reflected by the significant change in blood glucose measurements/day observed after 6 months (
No significant changes to body weight/BMI or other biological variables were observed over the span of the study, which could be explained due to the relative short duration of the trial for achievement of noticeable changes in certain parameters such as blood pressure or lipid status, even when a general tendency toward better total cholesterol levels was observed (
SMBG is recognized as a valuable tool for diabetes management, having been demonstrated to help patients achieve better biological outcomes,3-6,13,25-29 and to increase their disease self-awareness and improve on their management strategies, both at a clinical and lifestyle levels.3,7-11,13,30-34 The results of this 6-month observational study show that SMBG via OneTouch blood glucose meter, with its characteristics, including its color-coded display and easy-to-understand results, contributes to helping not only to improve glycemic control but also to may lead to a better disease self-management.
Despite the positive results of the study, it is important to remember that as an observational study, patients were not blinded to study intervention, and we cannot disregard the possibility of certain study-related effects that might have influenced patient compliance, for instance. On the other hand, as such there is no control group therefore we cannot establish if OneTouch is better in achieving glycemic control and improving self-diabetes management than other devices, but this study confirms that it can effectively help achieve these goals.
Conclusion
In conclusion, this medium-term, real-life study demonstrates that OneTouch® Verio® assisted SMBG leads not only to a significant improvement in glycemic control, but also to a significant improvement in DSM.
Footnotes
Acknowledgements
We thank all the patients, investigators, and trial-site staff members involved in the trial, as well as M Koetzner-Schmidt for organizational support.
Abbreviations
BMI, body mass index; DPP-4, dipeptidyl-peptidase-4; DSM, diabetes self-management; DSMQ, Diabetes Self-Management Questionnaire; GLP-1 RA, glucagon-like-peptide 1 receptor agonist; HDL, high density lipoprotein; LDL, low density lipoprotein; Q, question; SMBG, self-monitoring of blood glucose; T1/2DM, type 1/2 diabetes mellitus.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All the authors had confidential access to the final trial results and actively contributed to manuscript preparation. OS and MH were responsible for study design. A working group including the first and fourth authors wrote the first draft of the manuscript, which was revised and approved by all the authors, who made the decision to submit the manuscript for publication. The authors assume responsibility for the accuracy and completeness of the data. Editorial support was funded by the sponsor. OS has acted as member of advisory boards and/or given lectures under support from Abbott, Astra Zeneca, Bayer Healthcare, Boehringer-Ingelheim, Eli Lilly, Medtronic, Novartis, Roche Diagnostics, Sanofi; and is CEO and founder of Sciarc GmbH. GK and MH have no conflicts of interest. BG is an employee of Johnson & Johnson GmbH.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Study was supported by LifeScan, Johnson & Johnson Medical GmbH.
