Abstract
Objective:
The present retrospective study examines the influence of disease management programs on nonfatal stroke in type 2 diabetes mellitus (T2DM) patients in Germany.
Methods:
The evaluation is based on retrospective patient data from the Disease Analyzer (IMS Health). The analysis included 169 414 T2DM patients aged 40 years and older with an initial prescription of antihyperglycemic therapy between January 2004 and December 2014. A total of 86 713 patients participated in a disease management program (DMP) for T2DM and 82 701 patients received standard care. The main outcome measure of this study was nonfatal stroke. Kaplan-Meier curves of DMP and SC patients were compared using log rank test. The Cox proportional hazards model was used to provide an adjusted estimate of the DMP effect.
Results:
It is apparent from the baseline characteristics that the general health of patients receiving standard care was poorer than that of patients participating in a DMP. The baseline HbA1c value was 7.6% in the DMP group and 7.8% in the SC group. Furthermore, the SC group had a higher proportion of preexisting conditions, such as coronary heart disease (CHD), peripheral arterial occlusive disease (pAOD), and renal insufficiency. The proportion of patients who received insulin in first year therapy was higher in the SC group. Time to event analysis showed that DMP was associated with a delayed occurrence of stroke, because stroke occurred an average of 350 days later in DMP patients than in patients receiving SC (DMP: 1.216 days, RV: 866 days). The Cox model with covariable adjustment confirmed the significant association of DMPs with nonfatal stroke in patients with type 2 diabetes mellitus (HR 0.71; 95% CI: 0.69-0.74).
Conclusion:
The present study indicates that DMPs are positively associated with stroke. The possible reasons for this must be verified in further studies.
Diabetes is one of the fastest-growing diseases worldwide. In 2015, 8.8 % (95% CI, 7.2-11.4) of the world population aged between 20 and 79 were affected by the disease. About 90% of them had type 2 diabetes mellitus (T2DM).
According to estimates, this figure will increase to 10.4% by 2040 (95% CI, 8.5-13.5). Diabetes is a severe chronic disease that can lead to serious complications. 1
Disease management programs (DMPs) in Germany were introduced in 2002. These are structured treatment programs designed to ensure treatment in accordance with evidence-based guidelines. The primary aim of German DMPs is to avoid sequelae or significantly delay their occurrence. DMPs are designed to improve both self-organization of T2DM patients and the care for chronically ill people. For improved self-organization, patients participate in trainings to learn how to better manage their disease. Patients enrolled in a DMP have the advantages of better coordination between their medical partners, diagnosis and treatment in accordance with evidence based methods, continuous checks such as HbA1c measurements, and better documentation, all of which improve care. To guarantee these high standards, German DMPs must be certified regarding content and specific criteria. To justify the high expenses for DMPs, health insurance funds want to save expenses by reducing secondary diseases, misdiagnoses, hospital admissions, and emergency cases. 2
Stroke is one of the most common macrovascular complications among people with diabetes. 3 The risk of a stroke among T2DM patients is two to four times higher than among healthy people. A transient ischemic attack (TIA) is a possible harbinger of a stroke. Such an attack often precedes a stroke and usually causes symptoms similar to those of a stroke, although these disappear spontaneously within 24 hours. 4 Relatively few studies have been published on the effect of DMPs on the morbidity and mortality of diabetes patients.5-7 A recent study showed that T2D patients in DMPs achieve optimal blood glucose control significantly more often than patients receiving standard care. 8 However, there is still a lack of data on the relationship between DMPs and the risk of cardiovascular complications.
The following analysis examines the influence of DMPs on nonfatal stroke in T2DM patients in Germany. It compares patients participating in a DMP with patients receiving standard care.
Methods
Data source
This study used retrospective data from the German Disease Analyzer (IMS Health) database. The Disease Analyzer, in turn, is based on a panel consisting of approximately 2300 participating physicians’ offices within Germany. Patient data are transferred directly from the respective practice via a computer interface. Since these data include highly sensitive medical patient information, they are pseudonymized before being transmitted and again prior to their use. This ensures that they cannot be traced back to the individual patients.
The Disease Analyzer includes demographic data on patients, diagnoses (ICD-10), prescriptions (Anatomical Therapeutic Chemical Classification System; ATC), laboratory values, and specialist physician groups (specializations). Physicians’ practices are selected from the panel using stratified random sampling. The main stratification criteria are specialist physician and region. The Statistical Office of IMS Health is responsible for monitoring the panel and continually makes adjustments. Furthermore, the database was validated in terms of its representativeness. Becher et al calculated various proportional statistics from the panel and compared them with statistics that represent the population. For example, prescription structure, members of the respective health insurance funds, and age distribution of patients from the panel are very similar to various official statistics. 9 In the past, Disease Analyzer data have been used for numerous scientific studies, including studies on diabetes10-14 and the study of DMPs.8,15,16
Study population
After preparation of the data, the analysis included 169,414 T2DM patients (ICD 10: E11) with an initial prescription of antihyperglycemic therapy (ATC: A10C, A10D, A10H, A10J, A10K, A10L, A10M, A10N, A10P) between January 2004 and December 2014. A total of 86 713 (51.2%) patients participated in a DMP for T2DM and 82 701 (48.8%) patients received standard care. Patients were included in the analysis if they were at least 40 years old and had more than 183 days of therapy information. Patients who have had a stroke but less than 365 days of preobservation time, like prescriptions, laboratory parameters, or other physician consultations, were excluded. This step was necessary to ensure incidence regarding stroke. Incidence is important because the probability of a further stroke is much higher if a patient has already had a stroke in the past. To avoid uncertainty about this, these patients were excluded from this analysis.
A patient was censored at the time of the previous prescription if the time to the next prescription was longer than 365 days. If the difference between the first and second prescription was more than 365 days, the time variable was null. Those patients were excluded due to the censoring algorithm. In most cases, there were no gaps longer than 365 days; therefore, censoring was performed on the last observed prescription. Figure 1 illustrates the process for selecting study cohorts.
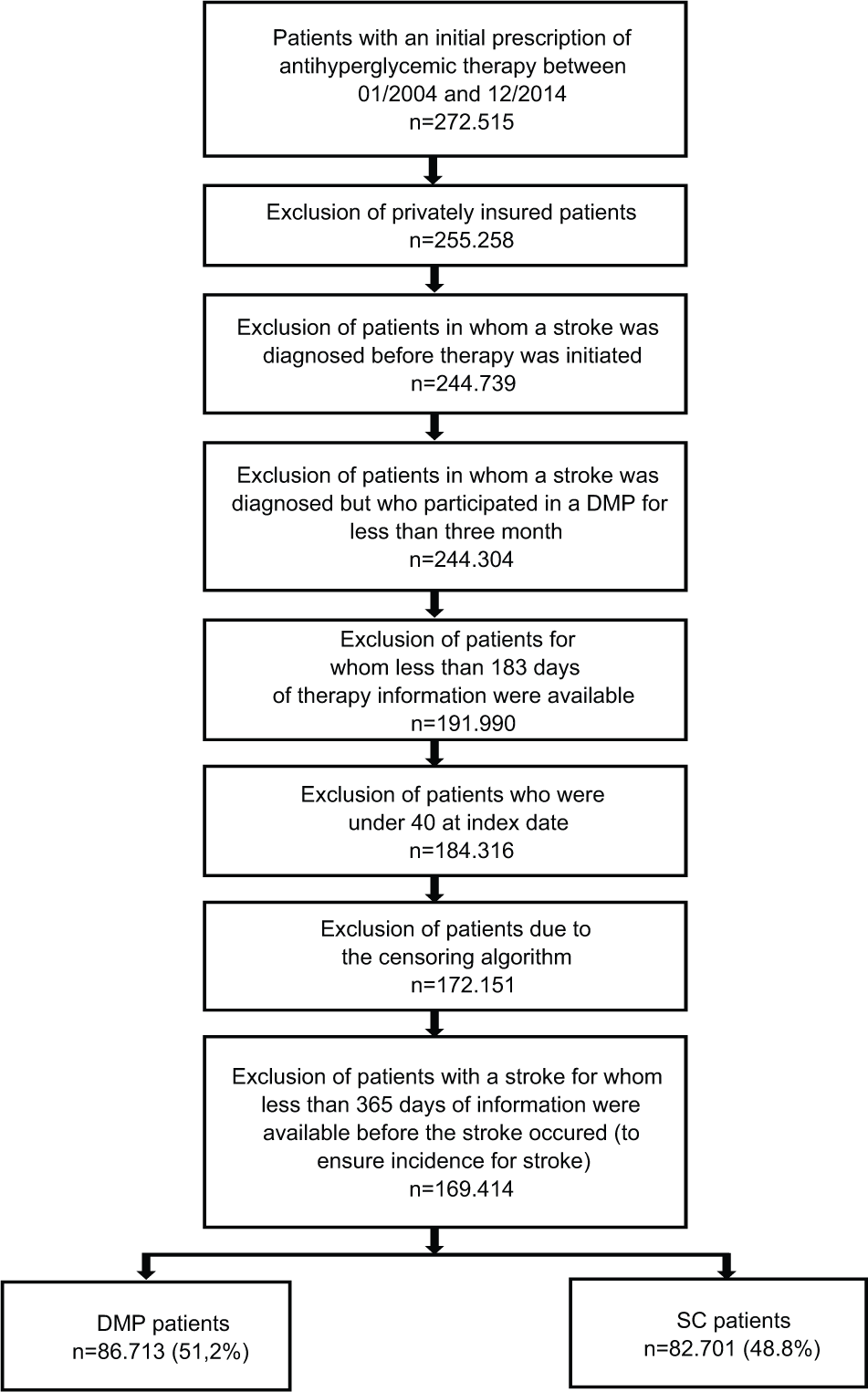
Selection of study patients.
Outcome
The primary endpoint of the analysis is the initial confirmed diagnosis of nonfatal stroke including TIA (ICD 10: I63, I64, G45), recorded using the “Event” variable. At the same time, this variable can also be interpreted as a censoring variable. Thus, all patients who did not suffer a stroke are considered censored, since nothing is known about their subsequent progress. Fatal stroke was not part of this analysis because data came from physicians’ offices, not from hospitals. Therefore, information about fatal stroke events was not available.
The time of censoring was determined by means of an algorithm as the time of the previous prescription, if the time to the next prescription was longer than 365 days. Furthermore, the variable “time” measures the period until the stroke occurred and/or the observation period. The maximum observation period was limited to ten years to ensure stable estimates, even in the case of long observation periods.
In addition to the outcome, the following covariates were included:
- Participation in DMP T2DM
- Demographic data: gender, age, region of medical practice, diabetologist (diabetological practice)
- Medical parameters: HbA1c baseline measurement, BMI, systolic blood pressure, diastolic blood pressure, index date. The index date is the prescription first recorded, which in most cases corresponds to the start of therapy. Where the start of therapy is mentioned later in this analysis, this refers to the index date.
- Participation in DMP coronary heart disease (CHD)
- Physician’s office with at least one DMP T2DM enrollment
- Form of therapy in the first year after the index date (metformin, insulin, sulfonylureas, DPP 4 inhibitors, SGLT 2 inhibitors, glinides, glitazones, alpha-glucosidase)
- Diseases prior to the index date: myocardial infarction, coronary heart disease, peripheral arterial occlusive disease, dialysis treatment
Statistical Methods
Standardized bias (SB) was used to compare baseline characteristics. Probability of initial diagnosis stroke was represented using Kaplan-Meier curves. Subsequently, the Cox Proportional Hazards Model was used to provide an adjusted estimate of the DMP effect. The analysis was carried out using the R 3.2.2. statistical software.
Results
Baseline Characteristics
SB was used to compare baseline characteristics. SB can be considered the standardized difference between the covariate distributions. SB is calculated by the absolute difference of means (or proportions) between DMP and SC, divided by the standard deviation. The major advantage of SB compared to tests of significance is that SB does not depend on sample size. Furthermore, tests of significance are usually not constructed to show the equality of two groups.
The demographic and clinical characteristics of the patients in the DMP and SC groups are shown in Table 1. The mean age was 64.9 (SD: 11.0) among DMP patients and 66.1 (SD: 11.8) among SC patients (SB = 0.101). The proportion of women was nearly identical in the DMP and SC groups (48.9% vs 49.0%, SB = 0.002). BMI differed by 0.1 (SB = 0.015) between the two groups. A total of 89.3% of patients in the DMP group and 87.4% of patients in the SC group were overweight (BMI > 25). Of these, 56.9% and 56.6%, respectively, were obese (BMI > 30).The variables DMP CHD (0.346), diabetologist (0.269), insulin (0.211), and metformin (0.205) exhibited the largest standardized bias.
Baseline Characteristics of Study Patients.
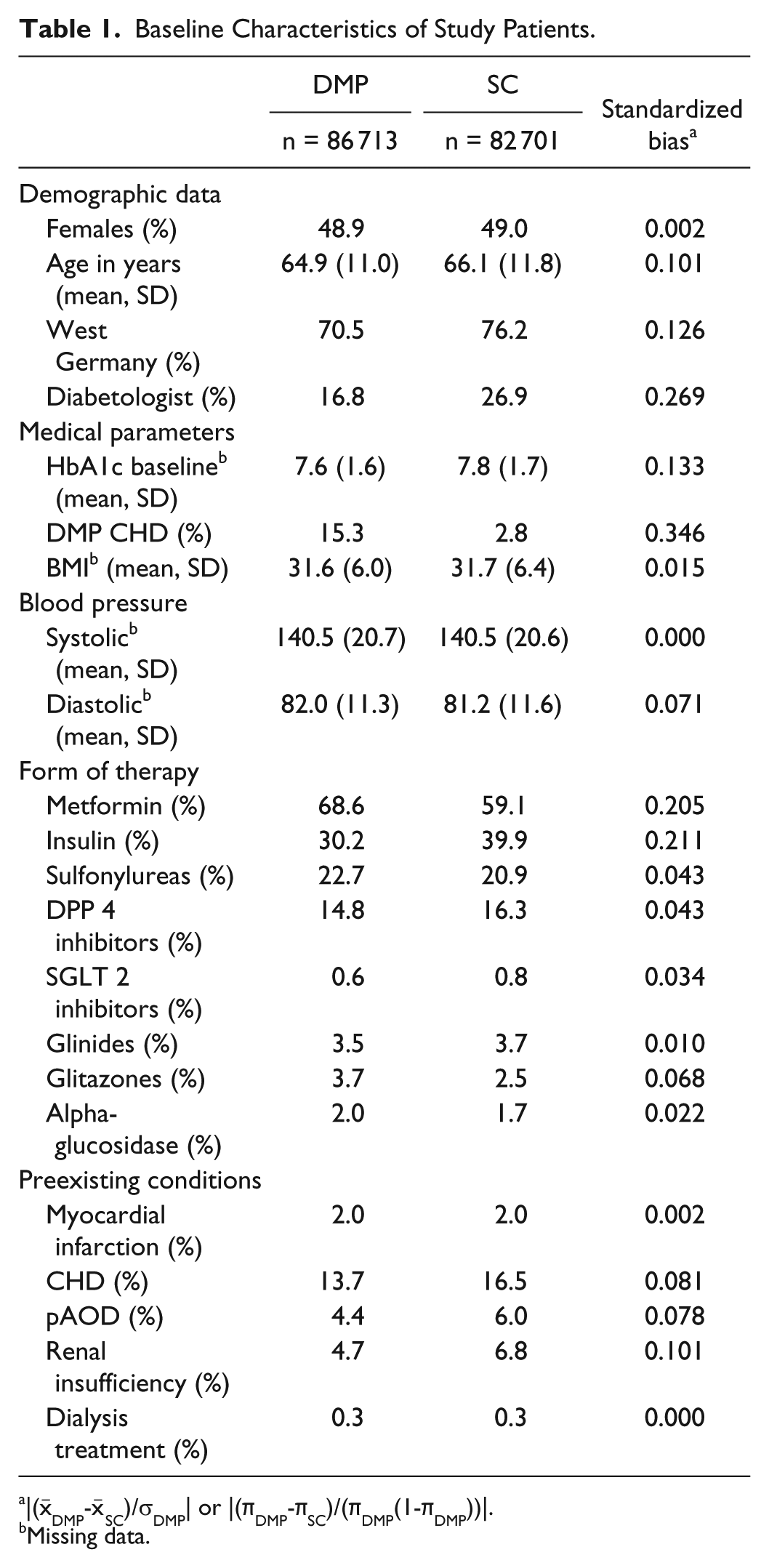
|(
Missing data.
HbA1c baseline, BMI, and blood pressure were missing many values (66% HbA1c baseline, 90% BMI, 85% blood pressure). Due to the high proportion of missing data, imputation was not appropriate. However, a complete case analysis indicated no selection effects.
Impact of the DMP on the Risk of Stroke
Figure 2 shows, that stroke occurred an average of 350 days later in DMP patients than in patients receiving SC (DMP: 1216 days, SC: 866 days). Kaplan-Meier curves showed that the probability for stroke among SC patients was higher than among DMP patients (log-rank test, P < .001) (Figure 3). It is important to note that the Kaplan-Meier curves of the DMP and SC groups do not reflect the proportion of strokes in the data set since only a few patients had an observation time of 10 years. Therefore, Kaplan-Meier curves at the end of 10 years indicate the probability for stroke, which is not equal to proportions.
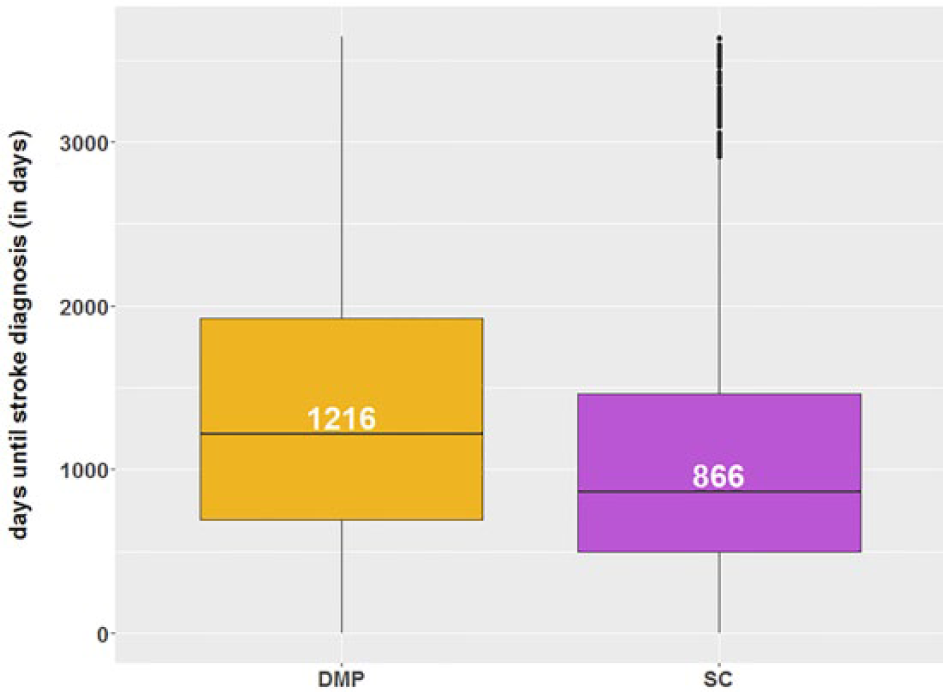
Average time to stroke in DMP and SC patients.
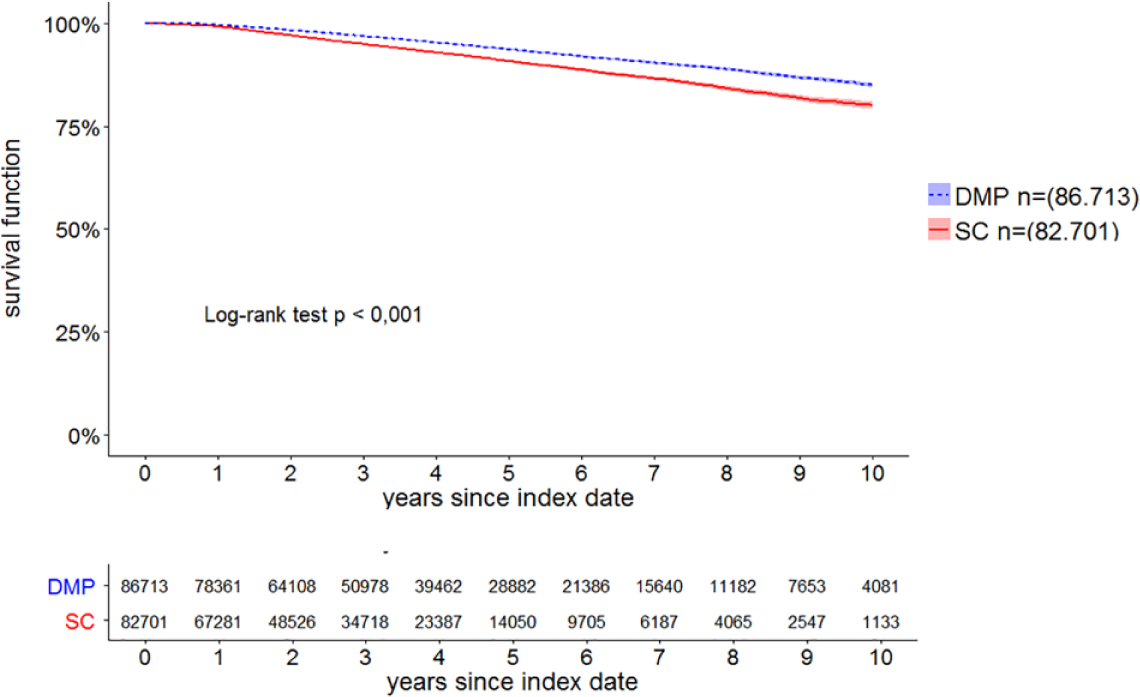
Kaplan-Meier curves for time to stroke in type 2 diabetes patients in DMP and SC.
The adjusted estimation of the DMP effect was calculated using the Cox proportional hazards model. Adjustments included demographic variables, participation in DMP-CHD, form of therapy, and preexisting conditions. It became apparent that there is a significant association between DMP and stroke (HR: 0.71, 95% CI: 0.69-0.74).
Discussion
Before the introduction of DMPs in Germany, the effectiveness of DMPs was not assessed by means of randomized controlled trials (RCTs). While specific study protocols existed for RCTs, the German national Association of Statutory Health Insurance Funds did not support them. 17 In the United States by contrast, DMPs were tested for their effectiveness by means of RCTs. However, the results of US studies may not be transferable to Germany due to the substantial differences between DMPs in the United States and in Germany. 18
Within Germany, increased attempts have been made in recent years to establish the effectiveness of DMPs by means of retrospective analyses. A systematic review of relevant publications compiled by Fuchs et al provides important insights in this regard. After extensive research in the basis of 17 publications, they presented an overview of the studies currently available in Germany. The high relevance of the work results from the fact that it only included publications that used a control group design to measure the DMP effect. Significant effects on mortality were found in favor of the DMP group. The results in terms of morbidity and quality of life differed greatly. The study concluded that it was not possible to make any general statements about the effectiveness of DMPs due to the heterogeneous outcomes of the various studies. In particular, only a few studies consider specific parameters. 19
The present study was aimed at contributing further insights, especially regarding hard endpoints. The main result of this study is the finding that there is a significant association between DMP and stroke (HR: 0.71; 95% CI: 0.69-0.74). Furthermore, it is apparent from the baseline characteristics (Table 1) that the general health of patients receiving standard care was poorer than that of patients participating in a DMP. This is indicated by the higher proportions of individual preexisting conditions in the SC group and the different proportions of forms of therapy in the first year in the DMP and SC groups. Looking at previously published studies, one finds that Drabik et al also found that DMPs had a positive effect on the risk of a stroke. Whereas 1.14% of patients receiving SC suffered a stroke, only 0.91% of patients participating in a DMP suffered the same rate. However, the occurrence of stroke was not the primary outcome measure, which is why only a descriptive analysis was carried out in this regard due to the multiple test problem. The significantly lower cumulative incidence compared to the present study is due to the shorter observation period, which was limited to a maximum of four years. 5
One possible reason why patients in the DMP group have a lower stroke risk could be the higher rate of adherence among DMP patients. It became apparent, for example, that patients in the DMP group received a greater number of prescriptions than patients in the SC group (DMP 6.62 versus SC 6.20). Furthermore, patients in the DMP group had more HbA1c measurements on average (3.70, SD 1.04) than patients in the SC group (3.07, SD 1.37). Linder et al also found that patients participating in a DMP had increased drug consumption rates. 6 However, further analyses are required to verify this aspect because higher prescription and HbA1c measurement can only be indicators for higher adherence. Studies on DMPs in other therapeutic areas found that adherence among DMP patients is better. In a study of breast cancer patients, for example, the treatment discontinuation rate was significantly lower in patients participating in a DMP than in patients receiving standard care (32.7% DMP versus 39.6% SC, P < .001). 15
The present study has several limitations. One such limitation arises from the fact that some socioeconomic (eg, education and income, etc) and demographic information was not available. It should be clear that the regression analysis can only take existing covariates into account and that unobserved variables may continue to cause selection effects. On the other hand, although a study by Graf et al found that the level of education of DMP patients is only slightly higher than that of patients receiving standard care, this does not necessarily apply to the present analysis. The study by Graf et al only included members of the BARMER health insurance fund. In addition, the multivariate factor analysis included other covariates than those used in the present study. 20 Another possible limitation is that the variables BMI, blood pressure, and baseline HbA1c value were not included in the regression analysis due to the high proportion of missing values.
A further difficulty in identifying the time of censoring, especially in retrospective studies, is determining when a patient is considered as no longer observable. For example, in the present study, information on the patient may be missing due to a change in physician. We tried to mitigate this problem by means of a censoring algorithm developed specifically for this study. In view of the regular therapy history one would expect to be recorded for chronically ill people, a patient was censored at the time of the previous prescription if the time to the next prescription was longer than 365 days.
In summary, this study indicates that DMPs are positively associated with stroke. The possible reasons for this must be verified in further studies. Especially in Germany, there are still too few studies that measure the DMP effect on the basis of hard endpoints. Moreover, the few studies that have been published in this area differ greatly in terms of their qualitative data, although recent studies frequently use an appropriate control group design. Although existing studies have provided some insight, further studies are needed to better understand the impact of DMPs.
Footnotes
Abbreviations
ATC, Anatomical Therapeutic Chemical Classification System; CHD, coronary heart disease; DMP, disease management program; DPP4i, dipeptidyl peptidase-4 inhibitors; GLP-1, glucagon-like peptid 1; HbA1c, hemoglobin A1c; ICD, International Classification of Diseases; pAOD, peripheral arterial occlusive disease; SB, standardized bias; SC, standard care; SD, standard deviation; SGLT-2, sodium dependent glucose transporter; T2DM, type 2 diabetes mellitus.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
