Abstract

Keywords
In a recent issue of your journal, Pettus and Edelman 1 outlined their recommendations for using real-time continuous glucose monitoring (rtCGM) data in type 1 diabetes. They emphasized a collaborative clinician-user relationship is vital to ensure rtCGM is effective and safe in the management of diabetes. 1 We report results of our study to investigate the issues facing clinicians managing people with diabetes in terms of self-monitoring blood glucose (SMBG) with particular emphasis on providing advice on the use of ambulatory glucose profile (AGP) applied to flash glucose monitoring (FGM) in real-life clinical practice.
We employed a two-pronged approach—first an online survey to determine attitudes to and knowledge of SMBG systems in general and AGP in particular. Second, face-to-face meetings were held with experts in the field to determine opinions and obtain suggestions on optimizing FGM plus AGP in clinical practice. Overall 122 clinicians completed both parts of the study with good agreement between replies to the online questionnaires and those from the expert meetings. There was general agreement that HbA1C cannot be the only therapeutic target in insulin-dependent patients with diabetes—as reiterated by the American Diabetes Association 2016 statement. 2 One-third of participants considered SMBG to be effective, with inadequate compliance, unreliability of patient-recorded data (in particular diaries), and missed identification of hypoglycemic episodes cited as the main drawbacks. There is a need for “user-friendly” SMBG systems that are easy to use and capture and store large amounts of data (including hypoglycemic episodes) for extended periods of time, coupled with a simple and intuitive data analysis system. Around 50% of diabetologists who took part in our online survey and 80% of those participating in the expert meetings were familiar with the AGP plus FGM the efficacy of which has been confirmed in type 1 and type 2 diabetes patients. 3
The main clinical advantages cited were the rapid and painless measurement of glucose levels, accurate assessment of glycemic variability, and the identification of nocturnal hypoglycemic episodes. Overall, feedback on the FGM plus AGP system was generally positive, in particular its ease of use and ability to capture information on glycemic variability and hypoglycemic episodes. Inadequate compliance, hypoglycemia, and lack of standardized glucose data analysis emerged as the main limitations of current glucose-monitoring technologies.
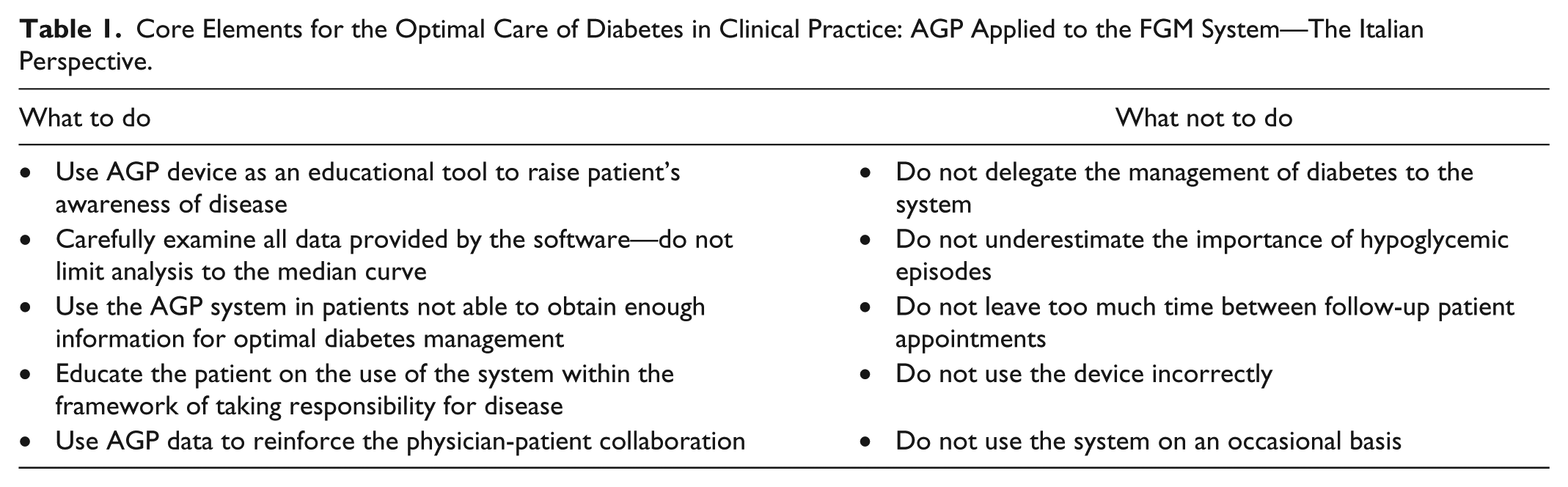
The availability of modern glucose-monitoring technologies has greatly improved the management of diabetes, but it is still not optimal. Our group developed a set of 10 do’s and don’ts in the utilization of FGM plus AGP emphasizing the importance of patient education and the professional development of the treating physicians (see Table 1). The new methodology can be used to reinforce the patient-doctor collaboration and ultimately lead to improved glucose control, particularly in those patients who do not have adequate information to optimally manage their disease. The European consensus recommendations on the use of AGP report in clinical practice suggest a week 4 review of the patient after the first AGP-based assessment and subsequent follow-up visits with AGP profile analysis every 3 to 6 months. The introduction of innovative glucose monitoring techniques and data analysis represent a major opportunity for both clinicians and patients. Irrespective of the technology employed, we believe that the patient must never be left alone with his or her disease and the physician’s intervention and supervision are an essential component of the effective management of diabetes. 4
Core Elements for the Optimal Care of Diabetes in Clinical Practice: AGP Applied to the FGM System—The Italian Perspective.
Footnotes
Abbreviations
AGP, ambulatory glucose profile; FGM, flash glucose monitoring; rtCGM, real-time continuous glucose monitoring; SMBG, self-monitoring blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AM has received lectures fees from Roche Diagnostics and A. Menarini Diagnostics and has provided advisory services to Eli Lilly, Abbott, Takeda, and Roche Diagnostics. KE has received lectures fees from Eli-Lilly, Novo Nordisk, Sanofi, Merck, and Abbott.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
