Abstract
Background:
Insulin glargine 300 U/mL (Gla-300) contains the same active ingredient as glargine 100 U/mL (Gla-100), and provides the same number of units in one-third of the volume. The SoloSTAR® injector pen has been modified to ensure accurate administration of this reduced volume and to improve user experience.
Methods:
Insulin- and pen-naïve adults with type 2 diabetes (T2DM) inadequately controlled with oral antihyperglycemic drugs, who had glycated hemoglobin (HbA1c) levels of 7.0-11.0 % (53-97 mmol/mol) were studied. They received once-daily Gla-300 in this 4-week, multicenter, open-label, single-arm study (NCT02227212). Ease of use/ease of learning (the primary endpoint), glycemic control, safety, and reliability of the disposable (prefilled) Gla-300 injector pen (secondary endpoints) were evaluated.
Results:
At week 4, 95.0% of 40 participating subjects assessed the pen as excellent/good and none as poor/very poor; 97.5% would recommend it to others. Total Diabetes Treatment Satisfaction Questionnaire scores were stable throughout the study. Mean (SD) fasting plasma glucose levels decreased from 166.1 (35.0) mg/dL at baseline to 124.2 (41.1) mg/dL at week 4. No product technical complaints (PTCs) or adverse events (AEs) related to PTCs were reported. The number of subjects experiencing hypoglycemic events of any kind and the incidence of AEs were low. No serious AEs were reported.
Conclusions:
The Gla-300 injector pen is easy to use and easy to learn to use, with demonstrable reliability and high degrees of acceptance and treatment satisfaction. Once-daily Gla-300 basal insulin treatment was well tolerated and effective in pen- and insulin-naïve adult T2DM subjects.
Long-acting basal insulin analogs, including insulin glargine 100 U/mL (Gla-100, Lantus®, Sanofi, Paris, France), have been mainstays of diabetes treatment since their introduction more than 10 years ago. The efficacy and long-term safety of Gla-100 is well documented.1-3 Once-daily insulin glargine 300 U/mL (Gla-300; Toujeo®, Sanofi, Paris, France) has been approved for use in the United States, 4 Europe, 5 Canada, 6 Japan, and Australia. 7 Gla-300 has been shown to have more stable, prolonged pharmacokinetic (PK) and pharmacodynamic (PD) profiles compared with Gla-100, 8 resulting in comparable glycemic control with less hypoglycemia versus Gla-100 in people with type 2 (T2DM)9-12 and type 1 diabetes.13,14 Gla-300 contains the same active ingredient as Gla-100 15 and provides the same number of units but in one-third of the volume.
Gla-100 is usually administered using the disposable (prefilled) SoloSTAR® insulin pen (Sanofi, Paris, France). Because of the difference in volume between Gla-100 and Gla-300, this injector pen required modification to allow accurate Gla-300 administration. In addition, certain features have been enhanced to improve the experience of users.16,17 The Gla-300 SoloSTAR injector pen (Sanofi, Paris, France) provides accurate delivery of Gla-300, with low injection force. 16 An initial survey suggested that both pen users and health care professionals who train people with diabetes in how to use pen devices find the Gla-300 injector pen easy to use. 17
This phase 3b study further investigated the ease of use/ease of learning of the Gla-300 injector pen, as well as treatment satisfaction, glycemic control, reliability, and safety of the device in insulin- and pen-naïve people with T2DM.
Materials and Methods
Participating Subjects
Participating subjects were ≥18 years of age with T2DM that was inadequately controlled with oral antihyperglycemic drugs (OADs), were considered suitable for treatment with basal insulin by the investigator/treating physician, and were insulin- and pen-naïve. Exclusion criteria included: HbA1c <7.0 % or >11 % (<53 or >97 mmol/mol); a history of T2DM for less than 1 year before screening; OAD treatment for less than 6 months; initiation of new glucose-lowering drugs and/or weight loss drug in the last 3 months; use of insulin for more than 8 consecutive days and more than 14 days in total during the previous year; previous treatment with glucagon-like peptide-1 (GLP-1) receptor agonists; only use of sulfonylureas, glinides, and/or other antihyperglycemic agents not approved in combination with insulin; and unstable proliferative diabetic or other rapidly progressive retinopathy or macular edema likely to require treatment during the study.
Study Design
This 4-week, multicenter, open-label, single-arm, phase 3b study (NCT02227212) was conducted in 7 centers in Germany between August 22 and November 20, 2014. The protocol was approved by appropriate ethics committees and the study was conducted according to Good Clinical Practice and the Declaration of Helsinki. All subjects provided written informed consent.
During the screening period, subjects were trained in self-monitoring of blood glucose (using a BGStar® meter; Sanofi, Paris, France). On day 1, subjects received face-to-face training on the appropriate use of the Gla-300 injector pen prior to first dosing. Only subjects who completed the training and demonstrated a successful complete injection (needle placement, dose setting and injection into a pad), unaided by the investigator or site staff, were eligible for the study.
Gla-300 was injected once daily in the evening, which was defined as the period from immediately prior to the evening meal until bedtime. The injection time was fixed at the baseline visit and was maintained for the duration of the study within a ±1-hour window. Subjects were individually up-titrated from a starting dose of 0.2 U/kg/day on a weekly basis (more frequently if needed, but not more often than every 3 days), seeking a fasting self-monitored plasma glucose (SMPG) target of 80-100 mg/dL (4.4-5.6 mmol/L). The titration algorithm is shown in Supplementary Table 1. Participating subjects were to continue to receive OADs at their prior dosage throughout the study (except sulfonylureas, glinides, and other antihyperglycemic agents not approved in combination with insulin, which were prohibited throughout the course of the study), unless the investigator decided that safety concerns necessitated a dose change or discontinuation of the OAD.
Baseline Characteristics (Safety Population).
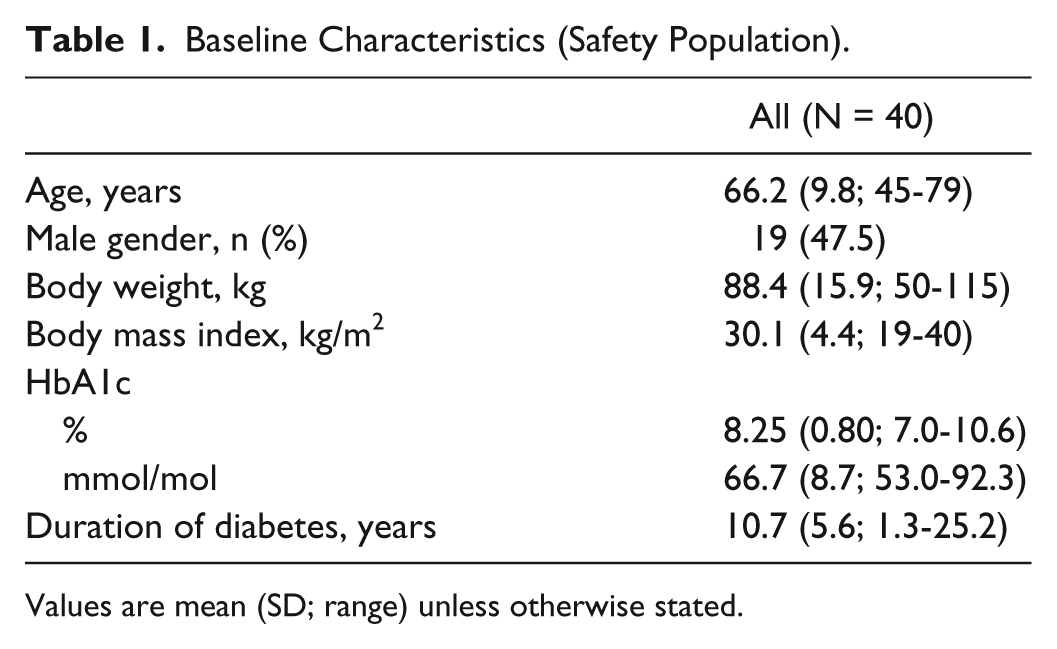
Values are mean (SD; range) unless otherwise stated.
Assessment visits occurred at screening, baseline, week 1, week 4, and 2 weeks after the final administration of treatment (posttreatment follow-up period). Mandatory telephone contacts were made at week −1, day 2, week 2, week 3, and 2 days after the final administration of treatment.
Outcomes
The primary endpoint was ease of use/ease of learning of the Gla-300 injector pen, as evaluated by the ease of use/ease of learning questionnaire, 18 completed by subjects at baseline, week 1 and week 4. The questionnaire comprised 12 items, which were assessed using a 5-point scale with scores ranging from 1 (excellent) to 5 (very poor), with the exception of item 12 (“Would you recommend the Gla-300 pen injector?”), which required a “yes” or “no” response.
Secondary endpoints included (1) treatment satisfaction (assessed by the original Diabetes Treatment Satisfaction Questionnaire, known as the status version [DTSQs], total scores ranging from 0 [very dissatisfied] to 36 [very satisfied]); 19 (2) glycemic control (assessed by change in laboratory-measured fasting plasma glucose [FPG]) and preinjection plasma glucose (assessed by obtaining SMPG 30 minutes prior to injection) from baseline to week 4; (3) change in insulin dose from baseline (starting dose) to week 4; and (4) reliability (assessed by the number of product technical complaints [PTCs] or adverse events [AEs] due to PTCs).
Although not a predefined efficacy endpoint, HbA1c levels were measured at baseline and week 4 to assess metabolic control.
Safety endpoints included the percentage of subjects experiencing at least 1 hypoglycemic event (categorized according to the American Diabetes Association definitions), 20 and the incidence of other AEs, including injection site reactions.
Data Analysis and Statistics
Descriptive statistics were performed for the primary and secondary endpoints using the safety population, which was defined as all subjects exposed to at least 1 dose of study insulin. The study was not powered to statistically assess changes in endpoints from baseline to week 4, therefore no systematic statistical testing was performed for any endpoint. The sample size for the study was based on empirical considerations and no formal sample size calculation was performed.
Results
Participating Subjects
Of 43 subjects enrolled, 3 found it difficult to attend the clinics and withdrew prior to treatment. All subjects successfully completed training on use of the Gla-300 injector pen. Therefore, 40 subjects were treated with Gla-300 and formed the safety population. All 40 subjects in the safety population completed the 4-week study period. Subjects had a mean age of 66.2 years and mean duration of diabetes of 10.7 years (Table 1).
Ease of Use/Ease of Learning
When assessing the individual functions of the Gla-300 injector pen relating to the set up and administration of Gla-300 injections (ease of use), after 4 weeks of treatment all items from the ease of use/ease of learning questionnaire were rated as “excellent” or “good” by at least 85.0% (range: 85.0-97.5%) of subjects (Table 2). The functions most consistently rated by participating subjects as “excellent” or “good” were “Ease of selecting the dose” (38/40; 95.0%), “Ease of correcting a misdialed dose” (39/40; 97.5%), and “Force/effort needed to inject insulin” (38/40; 95.0%).
Number (%) of Excellent/Good Responses on the Ease of Use/Ease of Learning Questionnaire Throughout the 4-Week Study (Safety Population).
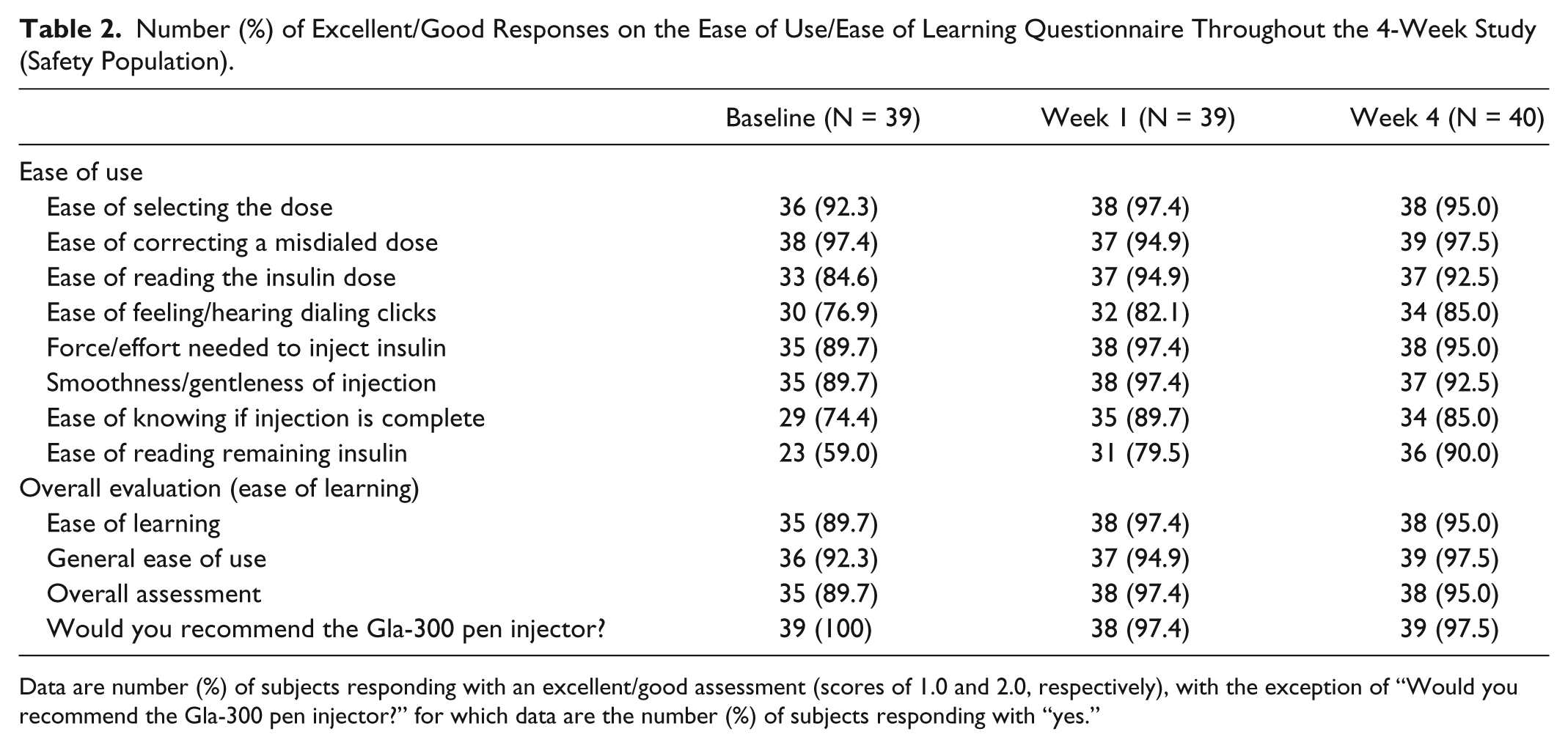
Data are number (%) of subjects responding with an excellent/good assessment (scores of 1.0 and 2.0, respectively), with the exception of “Would you recommend the Gla-300 pen injector?” for which data are the number (%) of subjects responding with “yes.”
On the “Overall assessment” score, the Gla-300 injector pen was rated very highly by subjects, with 95.0% (38/40) assessing the pen as “excellent” or “good” after 4 weeks, compared with 89.7% (35/39) at baseline (Table 2); the mean (standard deviation [SD]) overall assessment score was 1.7 (0.7) at baseline and 1.5 (0.6) at week 4. No subjects assessed the pen as “poor” or “very poor” overall at any visit. Consistent with these findings, 95.0% (38/40) and 97.5% (39/40) of subjects rated the “Ease of learning” to use the pen and the “General ease of use” of the pen, respectively, as “excellent” or “good” at week 4. Furthermore, the mean (SD) scores for these 2 items generally improved over the study period, decreasing from 1.6 (0.7) and 1.6 (0.6), respectively, at baseline to 1.4 (0.6) and 1.5 (0.6) at week 4. In addition, after 4 weeks of treatment the majority of subjects (39/40; 97.5%) reported that they would recommend the pen to others (Table 2).
Treatment Satisfaction
The total DTSQs score was relatively high at baseline (28.2), prior to the first Gla-300 dose. As the total DTSQs score can range from 0 (very dissatisfied) to 36 (very satisfied), this score indicates a good level of satisfaction with previous therapy. The total DTSQs score remained stable throughout the study, with a score at week 4 of 29.5 (5.6) (Table 3), suggesting that no change in participants’ perception of treatment burden was associated with Gla-300 therapy in combination with the Gla-300 injector pen in these previously insulin-naïve subjects. Moreover, from baseline to week 4 there were positive changes in scores for items relating to satisfaction (item 1: 0.26), recommendation (item 7: 0.26) and continuation of Gla-300 treatment (item 8: 0.28), although these were small relative to the range of possible scores (0 to 36).
Diabetes Treatment Satisfaction Questionnaire (Status Version) Scores by Visit (Safety Population).
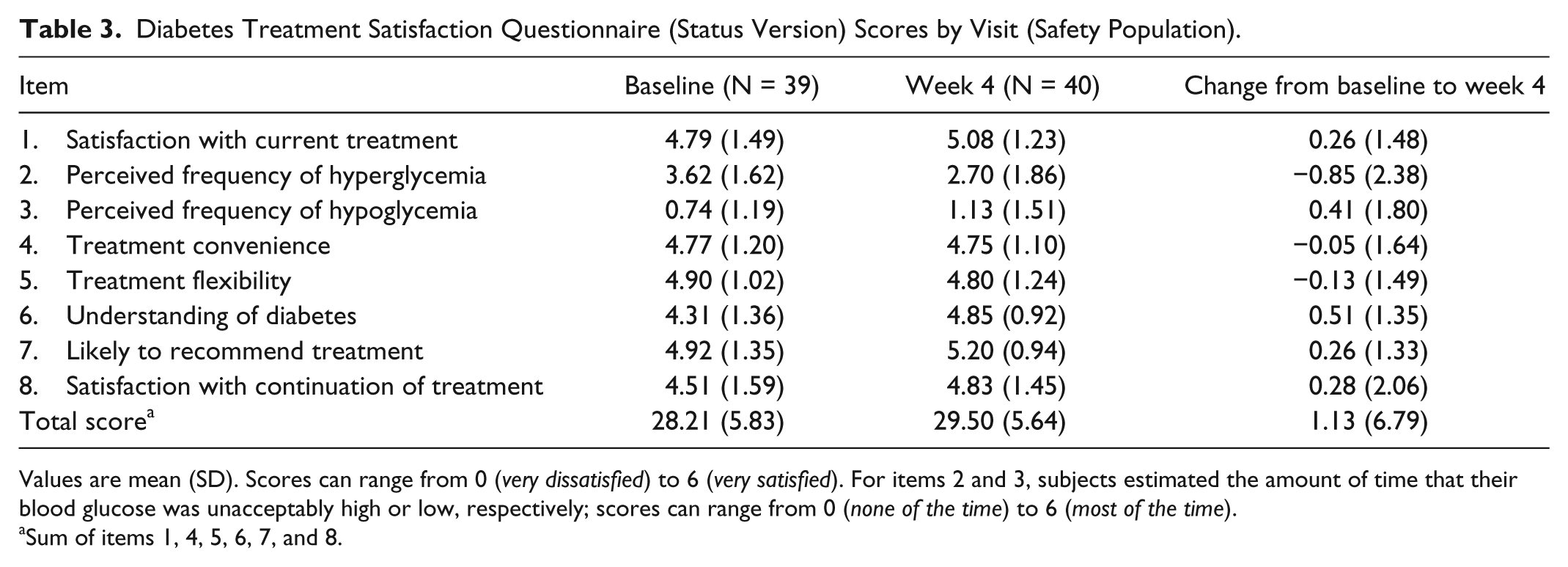
Values are mean (SD). Scores can range from 0 (very dissatisfied) to 6 (very satisfied). For items 2 and 3, subjects estimated the amount of time that their blood glucose was unacceptably high or low, respectively; scores can range from 0 (none of the time) to 6 (most of the time).
Sum of items 1, 4, 5, 6, 7, and 8.
Glycemic Control and Insulin Dose
Mean (SD) FPG levels decreased from 166.1 (35.0) mg/dL or 9.2 (1.9) mmol/L at baseline to 124.2 (41.1) mg/dL or 6.9 (2.3) mmol/L at week 4 (Table 4). Similarly, mean preinjection plasma glucose decreased from 206.0 (65.3) mg/dL or 11.4 (3.6) mmol/L at baseline to 178.3 (53.0) mg/dL or 9.9 (2.9) mmol/L over the same period. A comparable pattern was seen for HbA1c levels (Table 4).
Glycemic Control and Insulin Dose by Visit (Safety Population).
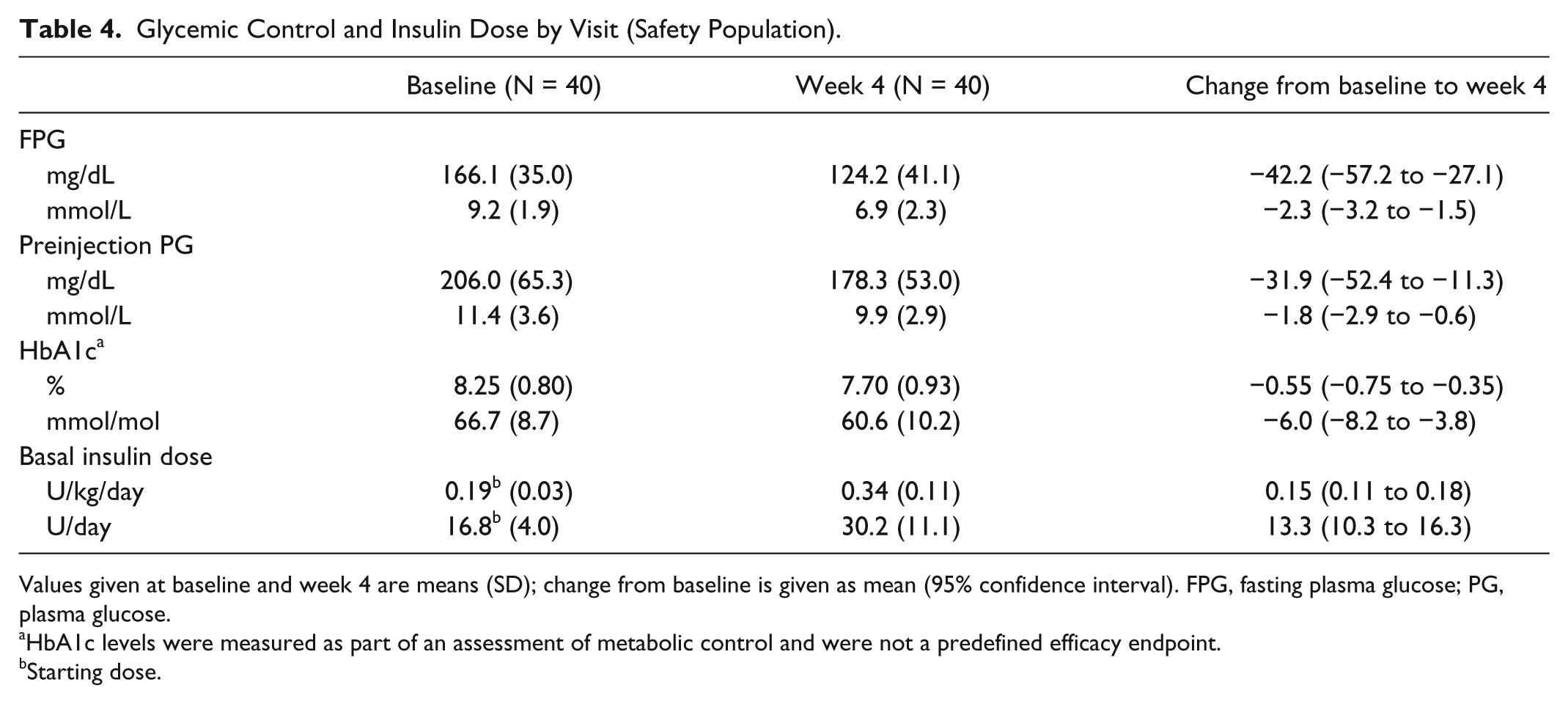
Values given at baseline and week 4 are means (SD); change from baseline is given as mean (95% confidence interval). FPG, fasting plasma glucose; PG, plasma glucose.
HbA1c levels were measured as part of an assessment of metabolic control and were not a predefined efficacy endpoint.
Starting dose.
The mean daily basal insulin dose increased from 0.19 (0.03) U/kg/day (16.8 [4.0] U/day) at baseline to 0.34 (0.11) U/kg/day (30.2 [11.1] U/day) at week 4 (Table 4), suggesting good compliance with the titration algorithm.
Reliability—Product Technical Complaints
Participating subjects did not complain about the Gla-300 injector pen. No PTCs or AEs related to PTCs were reported.
Hypoglycemia
Over the 4-week period, 7 (17.5%) subjects experienced hypoglycemia (Table 5), corresponding to 5.2 events per participant-year. Most hypoglycemic events were classified as asymptomatic (without symptoms but accompanied by a plasma glucose measurement ≤70 mg/dL [≤3.9 mmol/L]) or documented symptomatic (with symptoms and plasma glucose ≤70 mg/dL), and none was severe (requiring assistance from a third party) (Table 5).
Hypoglycemia Over the 4-Week Study Period (Safety Population).
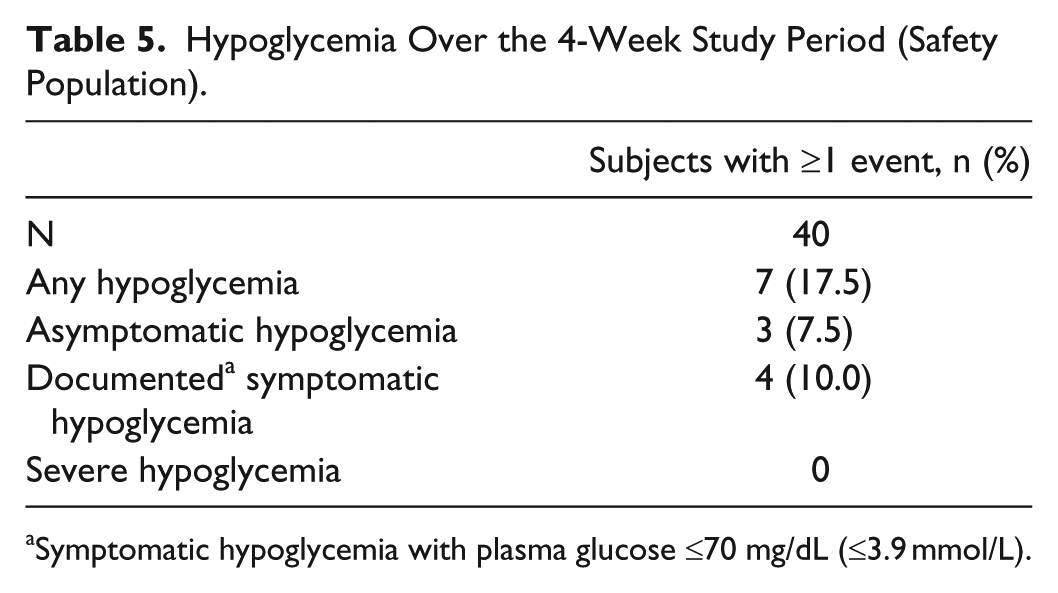
Symptomatic hypoglycemia with plasma glucose ≤70 mg/dL (≤3.9 mmol/L).
Adverse Events
A total of 11 (27.5%) subjects experienced at least 1 treatment-emergent adverse event (TEAE) over the 4-week study period, of whom 2 (5.0%) reported injection site reactions. No further local reactions or hypersensitivity reactions were observed. The most commonly reported TEAE was nasopharyngitis (4 subjects, 10.0%). None of the reported TEAEs was serious or led to either discontinuation or death.
Discussion
This open-label, 4-week study evaluated the Gla-300 injector pen among subjects with T2DM inadequately controlled with OADs only, who were insulin- and pen-naïve (and therefore also injection-naïve). The Gla-300 injector pen was rated highly on every item of the ease of use/ease of learning questionnaire, indicating a high level of satisfaction with the individual functions of the injector and overall usability. These findings are consistent with those of a recent, interview-based survey in which people with diabetes (users) and health care professionals (trainers), who were experienced in prescribing insulin pens and training pen users, compared the Gla-300 injector pen with 3 other commercialized insulin pens; 17 users and trainers ranked the Gla-300 injector pen in first place significantly more often than the other pens for “Easiest to use overall” and “Easiest to inject yourself.” In addition, the stability of the DTSQs scores throughout the current study suggests that Gla-300 basal insulin treatment administered using the Gla-300 injector pen was not associated with a change in participants’ perception of treatment burden in this population of previously insulin-naïve and injection-naïve people.
Despite recent advances in disease management, a number of well-documented barriers to insulin therapy persist among many people with diabetes, including the perception of high demands of insulin therapy such as number and timing of injections, difficulties in dose adjustment, and the anticipated pain of injections, which can lead to poor treatment adherence21,22 and therefore poor glycemic control. 23 The high ratings for overall ease of use/ease of learning reported for the Gla-300 injector pen, as well as the high scores for the individual items relating to accurate dose selection and comfortable injection, are therefore encouraging, because an insulin device that is easy to use accurately may lead to improved adherence to insulin treatment and improved glycemic control.
The phase 3 EDITION program demonstrated that treatment with Gla-300 versus Gla-100 resulted in equivalent improvements in glycemic control with less hypoglycemia9-14—however, the Gla-300 injector pen was not used in any of these studies, which were conducted before the pen had been fully developed. In addition to this, other factors such as study design (including the primary endpoint) and duration prevent meaningful comparisons between the glycemic control and hypoglycemia results reported here and those from the EDITION trials, including the EDITION 3 trial, which involved people with T2DM who were previously insulin-naïve (a study population similar to that of this study). Improvements in glycemic control seen in the current study are likely to be related to the increased insulin dose. In turn, these dose increases are suggestive of good compliance to the titration algorithm.
It has previously been shown that adherence and pen use have beneficial effects on a patient’s clinical outcome 24 and that even the use of a pen device itself can result in improved adherence to a new insulin regimen, resulting in improved glycemic control. 25 Thus, an injection device that is easy to use/easy to learn can be considered a precondition for good treatment persistence and adherence among people with diabetes, and this presumed benefit may have positive clinical implications with regard to the initiation of basal insulin treatment with Gla-300.
There are several limitations of the present study, including the potential biases introduced by the open-label nature of the design, the small study population and short study duration. Given the potential for recall bias due to the open-label protocol, the study was designed to minimize the potential for such bias by enrolling people who were both insulin- and pen-naïve. This study did not aim to distinguish between the effects of the Gla-300 injector pen and the Gla-300 insulin on the outcomes investigated. In addition, although the primary endpoint (ease of use/ease of learning) is obviously related to the insulin pen itself, certain other measured outcomes (such as glycemic control and hypoglycemia) are likely to be more related to the treatment administered than to the Gla-300 injector pen. It is important to note that any benefits experienced by subjects in this study cannot be definitively attributed either solely to the pen or to the insulin being administered. However, the treatment of diabetes requires an integrated approach and this study helps to highlight the potential comprehensive benefit of Gla-300 (with its more favorable PK and PD profiles vs Gla-100) with the improved Gla-300 injector pen. Future studies are warranted to study the clinical utility of the Gla-300 injector pen over longer periods of time and in larger, real-world populations.
Conclusions
In conclusion, over 4 weeks of once-daily dosing with Gla-300, pen-naïve and insulin-naïve adult subjects with T2DM considered the Gla-300 injector pen to be easy to use/easy to learn, and the pen was reliable. Gla-300 basal insulin treatment was both well tolerated and effective.
Footnotes
Acknowledgements
The authors thank those who participated in the study, trial staff, and investigators. Sanofi was the sponsor, and coordinated the study, monitored clinical sites, and collected and managed the data.
Abbreviations
ADA, American Diabetes Association; AE, adverse event; DTSQs, Diabetes Treatment Satisfaction Questionnaire (status version); FPG, fasting plasma glucose; Gla-100, insulin glargine 100 U/mL; Gla-300, insulin glargine 300 U/mL; GLP-1, glucagon-like peptide-1; OADs, oral antihyperglycemic drugs; PD, pharmacodynamic; PG, plasma glucose; PK, pharmacokinetic; PTC, product technical complaint; SD, standard deviation; SMPG, self-monitored plasma glucose; TEAE, treatment-emergent adverse event; T2DM, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HP has no conflicts of interest to declare. LB is a consultant and participates in speaker bureaus for Abbott, AstraZeneca, Bayer, BD, MontMed, Boehringer Ingelheim, Eli Lilly Canada, Janssen, Lifescan, Merck, Novo Nordisk, Sanofi. CB-W, JW, RD, and IN are employees of Sanofi. DK is a consultant for Ascensia, Lifecare, and Voluntis. He has received research support from Boehringer Ingelheim, BMS, Eli Lilly, Intarcia, Janssen, and Novo Nordisk, and owns stocks/shares in Tempramed.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Sanofi. Editorial assistance was provided by Simon Rees of Fishawack Communications, funded by Sanofi. This study is registered on ![]() (NCT02227212).
(NCT02227212).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
