Abstract
Background:
Optimal usage of continuous glucose monitoring (CGM) requires adequate training of the users. Providing patients with a CGM system without such a training usually doesn’t lead to the intended improvement in metabolic control.
Methods:
In Germany we developed a structured training program (“SPECTRUM”) to ensure a high quality standard for the use of CGM systems.
Results:
This program is suitably for patients of all age groups and is applicable to all CGM systems and all forms of insulin therapy. A curriculum was also developed so that training centers with less experience with CGM could become capable of offering comprehensive CGM training.
Conclusions:
We believe that usage of such a program can be an important step forward in achieving more widespread acceptance and use of CGM systems. Translations in other languages and evaluation with a controlled clinical trial are planned.
Keywords
Continuous glucose monitoring (CGM) represents a significant advance for the treatment of patients with diabetes mellitus. 1 Usage of CGM enables an increased understanding of the effects that various influences of daily life have on changes in glucose levels, for example, diet, insulin, and physical exercise.
Providing patients with a CGM system without special training and professional coaching does not usually improve their metabolic status.2,3 Patients need a wide range of information and skills in order to use the full potential of a CGM system and also to avoid potential negative aspects, for example, overcorrection. At present such training services are mostly given in individual and often unstructured consultations by a physician or (more often) by a certified diabetes educator. These are not ideal conditions for a structured training session. Structured training sessions are not only necessary at the start of CGM usage, but also through the long-term use of CGM if the patients are unable to cope with the system or if the therapy targets are not achieved.
The Diabetes Technology Working Group (AGDT) and the Pediatric Diabetology Working Group (AGPD) of the German Diabetes Association (DDG) have recognized the need for a comprehensive and independent CGM training program. A team of about 20 diabetes educators, diabetologists and psychologists from both groups jointly developed a program that
Ensures a high-quality standard for the use of CGM systems
Is suitable and specifically adapted to the needs of patients of different age groups
Is applicable to all CGM systems and all forms of insulin therapy
Is manufacturer-independent (however, in the first stage of the development, representatives of the manufacturers were consulted with regard to technical aspects)
The program is named “SPECTRUM” (

SPECTRUM training file with printed curricula and slides from the memory stick.

Examples of slides from the 3 versions of SPECTRUM for adults, for parents with young children, and for adolescents with type 1 diabetes. (a) SPECTRUM was developed as a manufacturer independent educational program by 2 working groups of the German Diabetes Association. This example shows the opening slide of module 1 for adults. (b) This is an example taken from the pediatric version of SPECTRUM for young kids and their parents. Possible advantages and challenges of CGM use are discussed prior to the decision on using CGM. (c) Patients are involved actively into the educational program. In this example taken from the adult version of SPECTRUM the patients should note their personal expectations connected with CGM use on a special card. This card will be stored until the end of SPECTRUM by the diabetes team and then discussed again. (d) As strict product neutrality is crucial for reimbursement of a patient educational program in Germany, we developed a neutral CGM display. This example is taken from the adolescents’ version of SPECTRUM and patients are asked to discuss the correct reaction to a low alarm. (e) This example is taken from the adult version of SPECTRUM and shows how patients are trained for interpreting their CGM display by discussing small “real” cases. Patients are asked in a very structured way to describe what they see on the display, which causes may underlie, and what the patient should do now in this situation. The next slides show the same case a few hours later (not shown). (f) The patients are suggested a structured way of interpreting CGM data (retrospective analysis) in 4 steps: detect recurrent hypoglycemia and assess glucose levels during nighttime and before and after meals. This algorithm is practiced with own CGM data during the training sessions.
Structure of SPECTRUM
To make the best use of CGM, the patients need to have a good understanding of a variety of physiological and technical aspects. 2 The most important aims of the training and treatment program are
Support in improving the quality of life and somatic parameters through the use of CGM
Motivation and empowerment through the use of this new diagnostic option
Help with cognitive and emotional management to prevent from “data overload”
Development of problem-solving abilities, for example, the prevention of hypoglycemic events and high postprandial glycemic excursions, or the safe correction of hypoglycemia and increased glucose values in everyday life while avoiding of overtreatment in those situations
Strengthening the individual ability to make decisions based on the interpretation of the current or saved CGM data and the adjustment of the alarm settings in everyday life
Optimization of the calibration procedure of CGM systems
The adult version of SPECTRUM consists of an introductory module, followed by 6 training modules each about 90 minutes long (Table 1). The pediatric versions (one for the adolescents and one for the parents with mainly younger children) consist of an introductory module and 5 training modules (Table 2). No content was left out for the pediatric versions. The different number of training sessions is due to the different setting with smaller group size in the pediatric versions and more outpatient visits at follow-up.
Basic Structure of the Adult Version of SPECTRUM.

Basic Structure of the Pediatric Versions of SPECTRUM.

The number of patients that should attend the training modules for adults is intended to be 2 to 6 patients. For parents of very young children the group size should be small, or individual training sessions should be offered.
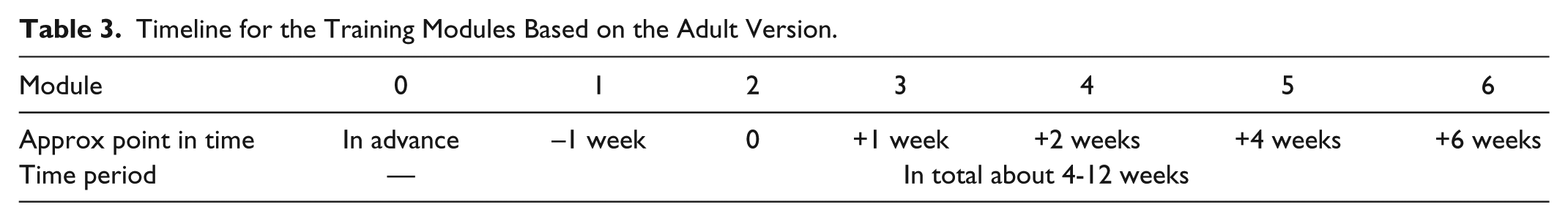
For patients that are not hospitalized a time frame of about 8 weeks should be planned for participation in the whole training program (a time range between 4 and 12 weeks can be considered, see Table 3). For children and teenagers, variations in the time frame are optional, in particular when used in the context of inpatient care or day clinics.
Timeline for the Training Modules Based on the Adult Version.
Performance of SPECTRUM
Patients who are interested in using a CGM system should first take part in the introductory module (Module 0). This module can be employed in different settings, for example, as part of an introductory meeting, or as part of a training session for patients to be trained in intensified insulin therapy (IIT) or insulin pump therapy. This module is freely available on the internet (on the homepages of the AGDT and AGPD). Reimbursement of this introductory session will not be possible in Germany, therefore this session is defined as “module 0.”
After participation of this initial module, those patients who are seriously interested in CGM should speak to their diabetes team for further approach. When a patient or the parents of a child decide that they want to use a CGM system and the system is available to them, then the actual training program can start (Table 1: adult version, Table 2: pediatric versions of SPECTRUM)
In Module 1, the most important basic CGM principles are covered (eg, CGM display, difference between glucose levels in interstitial fluid and blood, calibration)
In Module 2 the patients insert for the first time a glucose sensor, program the first alarm settings, and learn the “start rules” for the first days with CGM
In Module 3 the interpretation of the data shown on the CGM display is discussed and the alarm settings are adapted to the first experiences
In Modules 4 and 5 CGM data are analyzed together with the trainer and practical experiences exchanged from the first few weeks of CGM usage are discussed
In Module 6 patients receive support for long-term CGM use and for special situations
To address typical pediatric challenges, several psychological elements were included in the pediatric modules with worksheets on realistic expectations and targets for adolescents and parents and specific pediatric practical aspects, like CGM in nursery and school, child-appropriate explanation, CGM in challenging social situations, alerts at night and day, skin protection, and pain reduction at sensor insertion.
Materials Used by SPECTRUM
This training and treatment program consists of specific sets of slides for adults (introductory session + 6 training modules) and for parents with younger children and adolescents (introductory session + 5 training modules for each version). The slides are digitally available on a memory stick and in 2 different measurement units (mg/dl or mmol/l). In addition, a SPECTRUM folder is available that contains the detailed curricula in printed form for each of the 3 versions. The memory stick also contains a range of worksheets and checklists for the patients that can be printed for each participant in their required measurement unit.
SPECTRUM was published in German in January 2016 by Kirchheim Verlag, Mainz. Translations into other languages are planned.
Summary
SPECTRUM, a training and treatment program for CGM, has been developed for patients with diabetes to make the best use of CGM in daily life. To our knowledge SPECTRUM is the first comprehensive and product neutral CGM training and treatment program for patients of all age groups.
Footnotes
Abbreviations
AGDT, Diabetes Technology Working Group; AGPD, Pediatric Diabetology Working Group; CDE, certified diabetes educator; CGM, continuous glucose monitoring; DDG, German Diabetes Association; IIT, intensified insulin therapy.
Members of the SPECTRUM Group (Adult Version)
Dr med Bernhard Gehr, Diabetes- und Stoffwechselzentrum, Fachklinik Bad Heilbrunn (Overall coordinator, coordinator for SPECTRUM for Adults, coordinator for Module 1)
Dr med Eberhard Biermann, Munich
Sabine Carstensen, CDE, Diabetologische Schwerpunktpraxis, Bremen (until 2/2014)
Dr med Guido Freckmann, Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm
Prof Dr rer nat Lutz Heinemann, Düsseldorf
Elke Kerth, CDE, Diabetologische Schwerpunktpraxis, Ludwigshafen (organization and coordination of the photo shootings)
Dr med Ralf Kolassa, Diabetologische Schwerpunktpraxis, Bergheim
Prof Dr phil Dipl-Psych Bernhard Kulzer, Forschungsinstitut Diabetes-Akademie Bad Mergentheim (Coordinator for Module 0)
Dr med Andreas Liebl, Diabetes- und Stoffwechselzentrum, Fachklinik Bad Heilbrunn
Rosalie Lohr, CDE, Diabetologie Campus Innenstadt, Ludwig-Maximilians-Universität Munich
Dipl-Psych Berthold Maier, Forschungsinstitut Diabetes-Akademie Bad Mergentheim (editorial preparation of the slides and the curricula; organization and coordination of the photo shootings)
Kerstin Remus, CDE, Kinder- und Jugendkrankenhaus auf der Bult, Hannover
Claudia Sahm, CDE, Diabetologische Schwerpunktpraxis, Herrsching (Coordinator for Modules 4, 5 and 6)
Sandra Schlüter, Diabetologische Schwerpunktpraxis, Northeim
Marcella Schulz-Braun, CDE, Saarlandklink Kreuznacher Diakonie, Fliedner Krankenhaus Neunkirchen
Dr med Thorsten Siegmund, Klinik für Endokrinologie, Diabetologie und Angiologie, Städtisches Klinikum München Bogenhausen (Coordinator for Module 3)
Ulrike Thurm, CDE, Diabetologische Schwerpunktpraxis, Berlin (Coordinator for Module 2)
Marita Wernsing, CDE, Christliches Krankenhaus Quakenbrück
Members of the SPECTRUM Group (Pediatric Version)
Dr med Martin Holder, Olgahospital, Klinikum Stuttgart (Coordinator for the pediatric versions of SPECTRUM; organization and coordination of the pediatric photo shootings)
Dr med Dorothee Deiss, Endokrinologikum Berlin
Dr med Bettina Heidtmann, Katholisches Kinderkrankenhaus Wilhelmstift, Hamburg
Prof Dr rer nat Karin Lange, Medizinische Psychologie, Medizinische Hochschule Hannover (Editor of the pediatric curricula)
Dr med Carmen Ludwig-Seibold, Klinik für Kinder und Jugendliche, Oberschwabenklinik, Ravensburg
Kerstin Remus, CDE, Kinder- und Jugendkrankenhaus auf der Bult, Hannover
Dr med Simone von Sengbusch, Klinik für Kinder- und Jugendmedizin, Universitätsklinikum Schleswig-Holstein, Campus Lübeck
Tanja Wadien, CDE, Olgahospital, Klinikum Stuttgart (organization and coordination of the pediatric photo shootings)
Dr med Ralph Ziegler, Diabetologische Schwerpunktpraxis für Kinder und Jugendliche, Münster
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BG reported advisory activities for Roche Diagnostics and received speakers’ honoraria and reimbursement of travel costs from Dexcom, Nintamed, Roche Diagnostics and Medtronic. MH received speaking fees and reimbursement of travel costs from Medtronic. BK received speakers’ honoraria or consulting fees from Abbott, Bayer, Berlin-Chemie, LifeScan, Novo Nordisk, Roche Diagnostics, Sanofi and Lilly. KL received speakers’ honoraria or consulting fees from Bayer, BDI, Lilly Deutschland, Medtronic, Menarini Berlin-Chemie, Merck Serono, MSD Sharp & Dohme, NovoNordisk, Roche Diagnostics and Sanofi-Aventis. AL reported advisory activities and received speaking fees and reimbursement of travel costs from Dexcom, Nintamed, Roche Diagnostics and Medtronic. CS received speaker’s honoraria from Roche and Medtronic. SvS reported advisory activities for Abbott, Medtronic and NovoNordisk and received speaking fees and reimbursement of travel costs from Abbott, Lilly, Medtronic, NovoNordisk and Pfizer. SS received speakers’ honoraria from Abbott, Astra Zeneca, Bayer, Berlin-Chemie, Lilly, Nintamed, Novo Nordisk, Roche Diagnostics, Sanofi-Aventis and advisory activities for Abbott. TS received speakers’ honoraria from Abbott, Ascensia, Astra Zeneca/Bristol-Myers Squibb, Bayer Vital, Berlin Chemie, Boehringer Ingelheim, Eli Lilly, Medtronic, MSD, Novartis, Novo Nordisk and Sanofi, reported advisory activities for Abbott, Bayer Vital, Boehringer Ingelheim, Eli Lilly, Janssen, Medtronic, MSD, Novo Nordisk and Sanofi, and reported research funding from Astra Zeneca/Bristol-Myers Squibb, Becton Dickinson, Eli Lilly, MSD, Novo Nordisk and Sanofi. UT reported advisory activities for Lilly and Animas and received speaking fees and reimbursement of travel costs from Bayer Healthcare, Berlin Chemie, DiabetikExpress, Novo Nordisk, and Medtronic. RZ received speaker’s honoraria or consulting fees from Abbott, Animas, Novo Nordisk and Roche Diagnostics. GF received speakers’ honoraria or consulting fees from Abbott, Bayer, Berlin-Chemie, Becton-Dickinson, Dexcom, LifeScan, Menarini Diagnostics, Novo Nordisk, Roche Diagnostics, Sanofi, and Ypsomed. LH is a member of a number of national and international Advisory Boards for companies developing novel diagnostic and therapeutic options.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support by 2 working groups of the German Diabetes Association (DDG), AGDT and AGPD.
