Abstract
Background:
The closed-loop (CL) system delivers insulin in a glucose-responsive manner and optimal postprandial glycemic control is difficult to achieve with the algorithm and insulin available. We hypothesized that adjunctive therapy with liraglutide, a once-daily glucagon-like peptide-1 agonist, would be more effective in normalizing postprandial hyperglycemia versus insulin monotherapy in the CL system, in patients with type 1 diabetes.
Methods:
This was a randomized, controlled, open-label, crossover design trial comparing insulin monotherapy versus adjuvant subcutaneous liraglutide 1.2 mg and insulin, using the CL system in 15 patients. Blood glucose (BG), insulin, and glucagon concentrations were analyzed.
Results:
The liraglutide arm was associated with overall decreased mean BG levels (P = .0002). The average BG levels from 8:00
Conclusion:
This is a proof-of-concept study showing liraglutide can be a potential adjunctive therapy in addition to CL with insulin to reduce postprandial hyperglycemia in type 1 diabetes.
The closed-loop (CL) system has potential to change the lives of patients with type 1 diabetes. The system delivers insulin in a glucose responsive manner resulting in periods of euglycemia.1,2 Current open-loop insulin pump therapy requires meticulous carbohydrate counting and entering of carbohydrate and finger-stick blood glucose (BG) measurements into the system for estimation of insulin bolus amount. 3 Even in the most fastidious patients, postprandial hyperglycemia is common, as demonstrated by continuous glucose monitoring (CGM). 4 Furthermore, this occurs when using the CL system. 5
The CL algorithm for calculating insulin delivery fails to have a timely response to the glucose absorption from a meal 6 and results in postprandial hyperglycemia followed by hypoglycemia. In diabetes, there is paradoxical immediate postprandial hyperglucagonemia resulting in postprandial hyperglycemia 7 and dysregulated glucagon-like peptide- 1 (GLP-1) seems to be part of the etiology.7,8 Dupre et al demonstrated native GLP-1 improves postprandial hyperglycemia in type 1 diabetes, possibly by suppressing glucagon. 8 Our earlier study showed reduction in postprandial glucose levels following a preprandial injection of exenatide in adolescents with type 1 diabetes. 9 Subsequently, we demonstrated that exenatide with CL was effective in reducing postprandial hyperglycemia compared to CL with insulin monotherapy. 10
Liraglutide is a human GLP-1 receptor agonist with 1 amino acid substitution and C-16 palmitic acid chain attached via a glutamyl spacer. Modifications were made to slow subcutaneous tissue absorption, cause reversible albumin binding, and GLP-1 resistance to inactivation by dipeptidyl peptidase-4.11,12 Unlike exenatide, with a half-life of 2.4 hours and renal elimination, liraglutide has a half-life of 12 hours and is 99% bound to albumin with free liraglutide, degraded by endogenous peptidases. As opposed to twice daily exenatide, liraglutide is a once-daily injection and better tolerated as shown by the LEAD-6 study. 13 LEAD-6 compared the efficacy and safety between exenatide and liraglutide in type 2 diabetes. It also demonstrated that liraglutide significantly improved HbA1C when compared to exenatide. 13 Although a more recent study suggests liraglutide treatment did not improve HbA1C in type 1 diabetes, 14 several studies have shown improvement in HbA1C and postprandial hyperglycemia by CGM; when liraglutide is used adjunct to insulin. 15
We hypothesized that liraglutide could normalize postprandial hyperglycemia. This study is the first to examine the role of once-daily liraglutide plus the CL system in type 1 diabetes.
Methods
This was a randomized, controlled, open-label, crossover design trial comparing adjuvant liraglutide and insulin therapy versus insulin monotherapy using the external physiological insulin delivery (ePID) CL system. Albert Einstein College of Medicine Institutional Review Board (IRB) and Food and Drug Administration (FDA) approval was obtained. Investigations were carried out according to the Declaration of Helsinki.
Patients were recruited from Montefiore diabetes clinics and through local advertising and online postings on clinical trial sites. Twenty subjects were screened, ages 18-40, with a clinical diagnosis of type 1 diabetes for at least 1 year and on insulin pump therapy. Subjects had an HbA1C ≤ 8.5%, Hgb ≥ 12 g/l, weight > 50 kg, normal serum creatinine, and no other medical conditions (except hypothyroidism, stable). Females required a negative pregnancy test and could not be breastfeeding. Subjects were excluded if they had a history of celiac disease, gastroparesis, disordered intestinal absorption, QT prolongation or cardiovascular disease, alcohol or drug dependency (assessed by CRAFFT behavioral health questionnaire), 16 elevated calcitonin, medullary thyroid carcinoma, or multiple endocrine neoplasia type 2. Subjects could be on stable treatment of the following medications: oral contraceptives, antihypertensive, lipid-lowering agents, or antidepressants. Patients taking any medication known to affect BG, acetaminophen and vitamin C, glucocorticoid therapy, or sensitivity to serotonin receptor antagonists were also excluded.
Screening evaluations were performed 4 to 8 weeks prior to study enrollment and consisted of informed consent, medical history, physical examination (height, weight, vitals), blood draw (complete blood count, HbA1C, serum electrolytes, amylase, lipase, calcitonin, HCG pregnancy test), and a 12-lead electrocardiograph. After screening, subjects who qualified were randomized to study order, Study A (CL with insulin monotherapy [control arm]) or Study B (CL with insulin + liraglutide, single daily subcutaneous injection [treatment arm]), based on a 2-period crossover design. Pharmacy was responsible for randomization using an online program, Randomization.com. Investigators as well as participants were not masked to treatment assignment.
Study A, Day 1
Subjects were admitted to the inpatient unit at the Montefiore study location in Bronx, New York, at 2
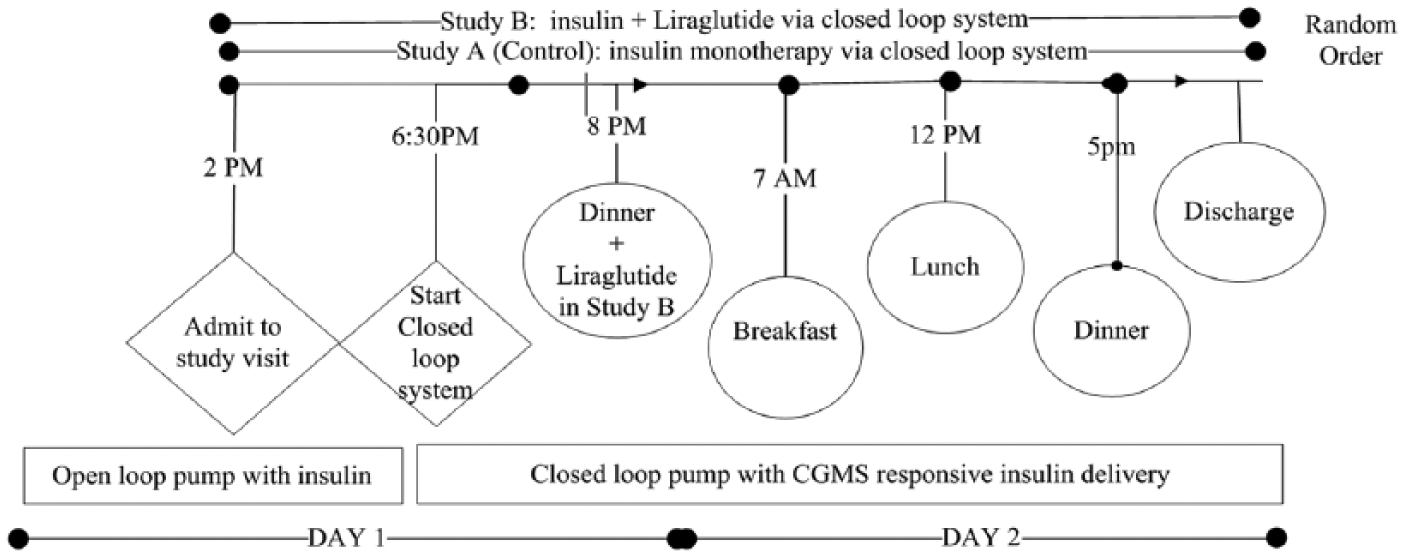
Design of the study evaluating overnight and daytime closed-loop delivery. A schematic illustration indicating the timing of the main study procedure. CGMS, continuous glucose monitoring system.
Day 2
Breakfast was served at 7
Study B
This study was identical to Study A except before dinner, on day 1 (0 minutes), subjects were given a subcutaneous injection of liraglutide (Victoza®, Novo Nordisk Inc., Plainsboro, NJ) 1.2 mg. Liraglutide dose calculations were based on previous studies by Varanasi et al. 15 Visits were at least 10 days apart (washout period). Identical to Study A, the AUC for insulin, glucagon, and glucose were calculated postprandially on day 2.
The CL system was provided by Medtronic (Northridge, CA, USA) and consisted of: Medtronic Paradigm® insulin pump (Medtronic MiniMed, Inc., Northridge, CA), Medtronic MiniLink™ real-time transmitter (MMT-7703, Medtronic MiniMed, Inc., Northridge, CA) adapted for 1-minute transmission, 2 Medtronic Enlite™ (MMT-7008, Medtronic MiniMed, Inc., Northridge, CA) sensors (study and backup), Enlite™ (Medtronic MiniMed, Inc., Northridge, CA) insertion device, Medtronic Silhouette® (MMT-399, Medtronic MiniMed, Inc., Northridge, CA), and Medtronic ePID algorithm (Medtronic MiniMed, Inc., Northridge, CA). The investigator purchased 1-time use supplies. Algorithm calculations were performed by a computer receiving glucose sensor signals every minute from a radiofrequency transmitter and delivered insulin commands to the pump by radiofrequency signaling. Signaling between the pump and the computer was occasionally interrupted for a few minutes, for example, when patient was in the bathroom or if the pump or transmitter needed to be repositioned. The ePID controller used a proportional-integral-derivative algorithm modified to include insulin feedback.17,18 Although the system uses 1 sensor, 2 sensors were placed. Control of CL was set to sensor 1, but was switched to sensor 2 at the discretion of the investigator if sensor 1’s function deteriorated (accuracy did not improve after calibrating twice). The backup sensor was used at least once during all visits. Sensors were calibrated at the start of CL and when Analox BG and sensor error exceeded 20%. The detection limit for the BG Analox system includes a calibration of 10.08 mg/dl and sensitivity of 0.108 mg/dl. 19 Analox was approved for use by the FDA. Target BG was 100.8 mg/dl.
Primarily, the effect of a single-dose subcutaneous liraglutide injection as an adjuvant therapy to insulin in the CL setting was assessed in comparison to CL with insulin monotherapy. In addition, safety and efficacy was evaluated. Furthermore, the effects of liraglutide on insulin requirements and on insulin and glucagon concentrations were assessed. Outcomes were evaluated at the completion of all visits. Protocol deviations and adverse events were reported to the IRB, FDA, and data safety monitoring board.
Assays were performed in the core laboratories of Einstein Institute for Clinical and Translational Research. 20 Glucagon was measured using a Mercodia enzyme-linked immunosorbent assay kit (cat no 10-1271-01, Winston Salem, NC), and insulin was measured using an EMD Millipore radioimmunoassay kit (HI-14K, Billerica, MA). Lower limit of quantification for insulin was 3 uU/mL and glucagon was 25 pg/ml.
The study is designed to detect a 25% difference in mean AUC of glucose for postprandial periods. Based on a power of .8, alpha of .05, correlation coefficient of r = .5, 9 and standard deviation (SD) excursion measure AUC for Analox glucose approximately 0.8 SD, sample size was estimated to be 16 subjects. With a 10% dropout rate, we needed to recruit 17 subjects.
Statistical comparisons were done using repeated measures ANOVA to determine the effect of liraglutide on plasma glucose, insulin and glucagon. We used GraphPad Prism 6.0 software (GraphPad Software Inc, San Diego, CA) to analyze the comparative and descriptive statistics. The mean AUC was calculated using the trapezoidal method for insulin, glucagon, and glucose over time as well as 2-hour postprandial levels after breakfast (660-780 minutes), lunch (960-1080 minutes), and dinner (1260-1380 minutes) and analyzed using a 2-way ANOVA followed by a 1-way ANOVA followed by post hoc Bonferroni or Tukey’s test. All data are expressed as mean ± SD. The data for variability in AUC (treatment vs control) for 2-hour postprandial periods is expressed as mean ± 95% confidence interval (CI).
Sensor accuracy was calculated as mean absolute relative deviation (MARD) of the sensor glucose level from the plasma venous glucose level for all paired points (mean ± SD). The differences (treatment vs control) were considered to be statistically significant at P < .05. Hypoglycemic events were analyzed between control and treatment arms using Fisher’s exact test.
Results
Table 1 depicts baseline characteristics of participants. A total of 15 subjects (n = 15) completed both visits. There were 2 screen fails, 1 due to elevated HbA1C and 1 who reported history of pancreatitis (the investigator chose not to enroll for safety concerns). There was 1 dropout who chose not to participate in the second visit due to scheduling conflicts and 2 screens that qualified but had scheduling conflicts. Nine subjects were randomized to the control arm first. The first patient was enrolled on February 2, 2013 and the last study visit was August 19, 2014.
Baseline Characteristics of the Participants.
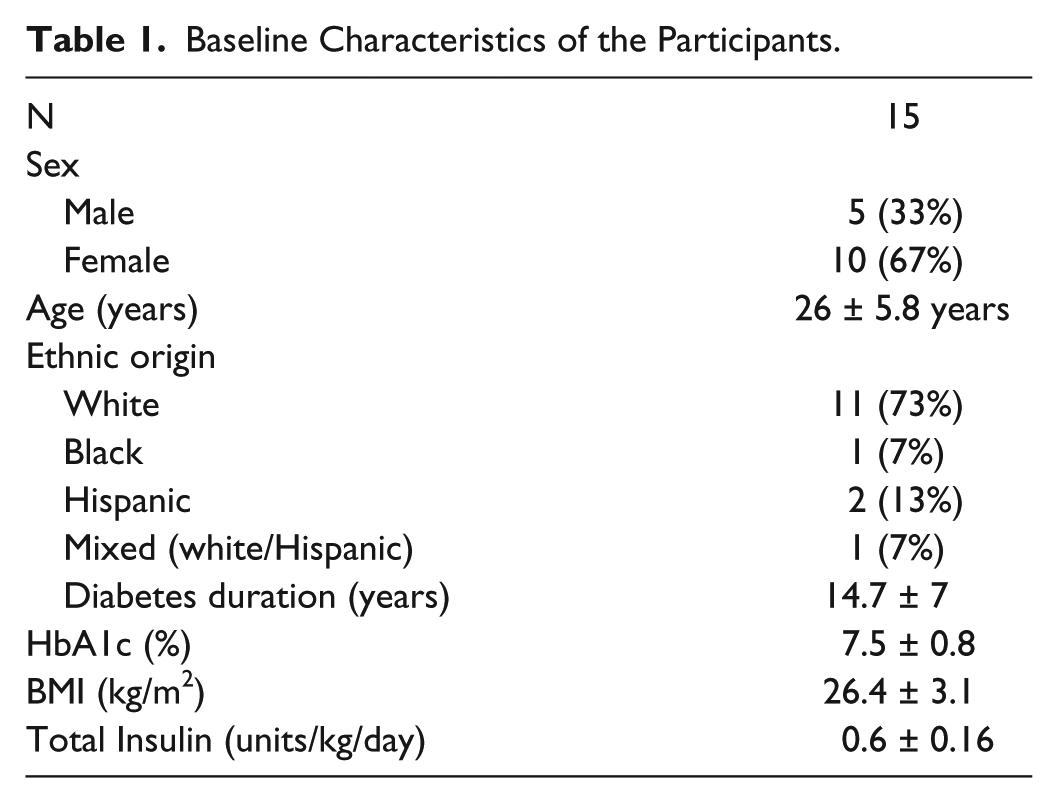
Figure 2A depicts BG concentrations during the entire study period. BG values were significantly lower in the treatment compared to the control arm (P = .0002). The average BG from 8
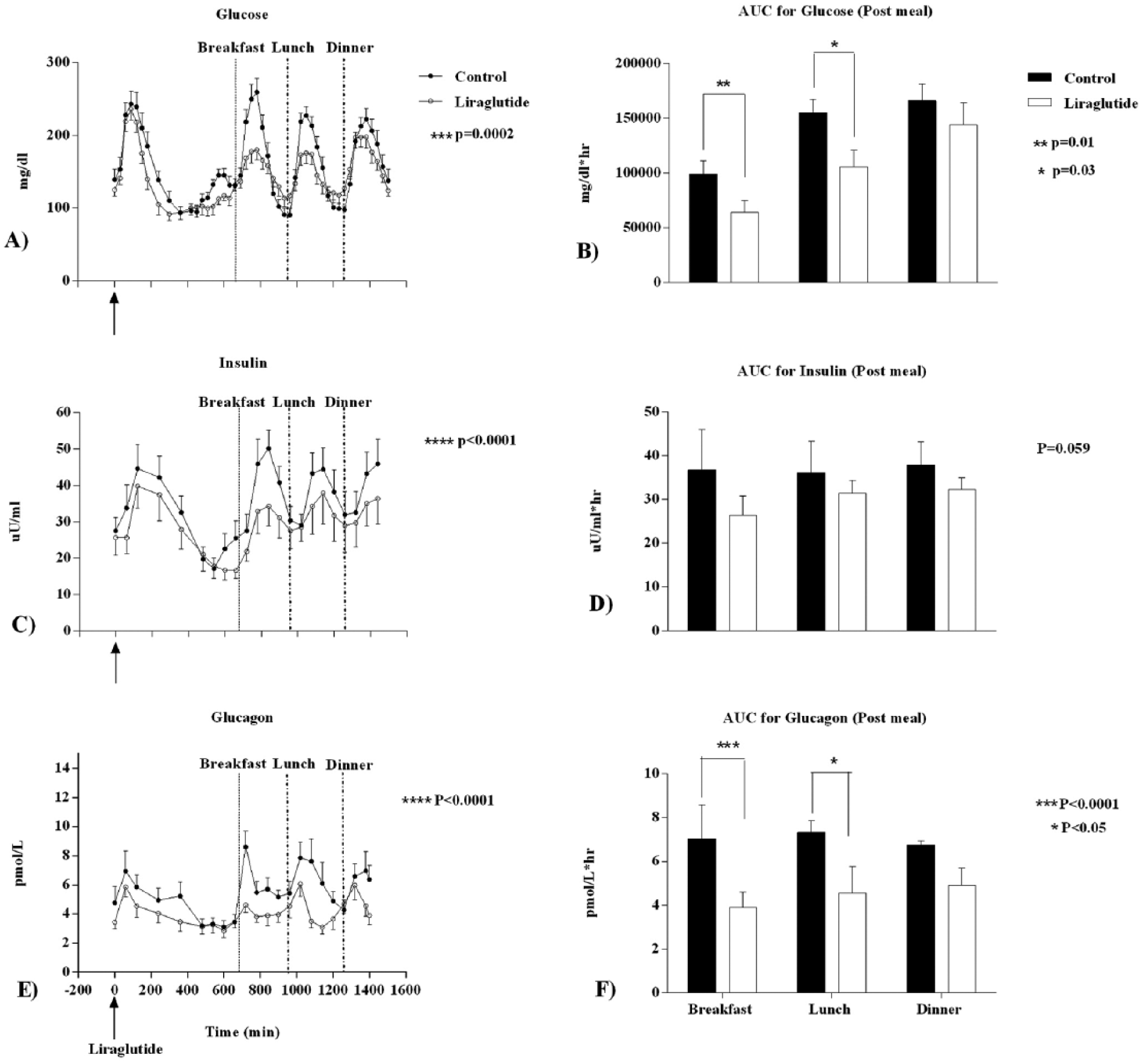
Glucose, insulin and glucagon concentrations in CL with and without ligraglutide (A, C, and E) and area under curve values following postprandial changes in glucose, insulin, and glucagon with meals (B, D, and F). Glucose, insulin, and glucagon concentrations (A, C, and E) were analyzed during the time points 0-1440 minutes and are represented as black circles for control and white circles for treatment (liraglutide). AUC for glucose, insulin, and glucagon (B, D, and F) were calculated postprandial 2 hours after each meal (660-780 minutes after breakfast, 960-180 minutes after lunch, and 1260-1380 minutes after dinner) and are represented as black bars for control and white bars for treatment (liraglutide). All data are expressed as mean ± SD. The difference between the treatment vs control arm is expressed as mean ± 95% confidence interval (CI). A P value < .05 was considered to be statistically significant.
Secondary outcomes (Figures 2C and 2E) suggest insulin and glucagon concentrations were significantly reduced overall (P < .0001) in the treatment arm. Compared to controls, insulin levels were lower in the treatment arm (209.6 ± 56.6 vs 172.4 ± 41.94 pmol/L, P < .0001) and there was a significant difference in insulin levels between the treatment and control arm −37.3 pmol/L (95% CI −49.58, −24.95). Similarly the glucagon levels were significantly lower in the treatment arm (5.5 ± 1.5 vs 4.1 ± 0.9 pmol/L, P < .0001), the difference being −1.4 pmol/L (95% CI −1.98, −0.898). The AUC (Figure 2F) for glucagon concentrations were also significantly lower for breakfast (630.8 ± 897.6 vs 1174 ± 1680 pmol/L*hr; P < .0001) and lunch (1070 ± 1622 vs 1482 ± 2196 pmol/L*hr; P < .05). The mean difference in AUC 2 hours postbreakfast is −542.8 pmol/L*hr (95% CI −2487, 1402) and 2 hours postlunch is −412 pmol/L*hr (95% CI −1842, 1018). The AUC for insulin excursions (Figure 2D) were also lower, but did not reach significance (−16 918 pmol/L*hr [95% CI −37 222, 3387]). Insulin requirements in the CL system for the study period were also lower in the treatment arm (2.5 ± 0.3 vs 2.1 ± 0.3 units/hr; P < .01). However, insulin use per hour during the postprandial period in control versus treatment arm was not different (breakfast [4.4 ± 0.8 vs 3.1 ± 0.6; P = .19], lunch [3.9 ± 0.8 vs 2.9 ± 0.4, P = .17], and dinner [4.0 ± 0.7 vs 3.1 ± 0.3, P = .19]).
There were no serious adverse events. Expected adverse events included nausea (n = 8), headache (n = 1), and emesis (n = 2). Removal of the subjects with emesis from analysis did not alter results; therefore, they were included in final analysis. Subjects proceeded, using ondansetron, as per protocol. None of the subjects in the control arm had any side effects.
Hypoglycemia occurred most frequently before meals and was treated with glucose tablets. Episodes were considered hypoglycemic if BG was below 70 mg/dl at any of the 47 required BG time-points. There were 45 episodes of hypoglycemia in the control arm and 42 in the liraglutide arm (3 episodes per visit per subject versus 2.8 per visit per subject), suggesting no significant differences in the hypoglycemic episodes between the 2 arms (P = .83, Fisher’s exact test). None of the episodes were severe (requiring another person to administer a resuscitative action) and no patient withdrew due to hypoglycemia.
Discussion
In the current study, we demonstrated that CL with insulin and subcutaneous liraglutide is effective in reducing overall glucose excursions without causing increased hypoglycemia compared to CL with insulin monotherapy, in type 1 diabetes. A CL system with insulin monotherapy is effective in reducing postabsorptive hyperglycemia, however remains ineffective in reducing postprandial hyperglycemia. Liraglutide, a once-daily subcutaneous injection, lowers insulin requirements and glucose excursions.
Failure of glucagon to rise in response to hypoglycemia and decrease postprandially is perturbed in type 1 diabetes. Recent studies suggest that postprandial hyperglucagonemia worsens glycemic control and results in severe decline in beta-cell function in children with type 1 diabetes. 21 However, there has been a failure to emphasize the role of alpha-cell dysregulation in type 1 diabetes. As a result, there are no approved medications besides pramlintide22,23 as adjunct therapy. Pramlintide, an amylin analog deficient in type 1 diabetes, must be administered subcutaneously multiple times each day to be effective.3,24,25 This results in a very complicated treatment algorithm especially if using multiple-hormonal therapy in CL. 25 Hence, our research has focused on studying newer analogs such as exenatide9,10 (twice daily) and now liraglutide (once daily), to improve postprandial glucose concentrations in type 1 diabetes, thus reducing treatment complexity. As improved treatment options emerge, efficacy in CL must be investigated.
In future studies, C-peptide measurements at baseline could be measured. In C-peptide negative patients with type 1 diabetes, delaying of gastric emptying, as well as reduced glucagon levels, can explain the effect of a GLP-1 agonist on glucose control postprandially. However, in patients with some preservation of beta-cell function (which is about 10% in patients with type 1 diabetes) the endogenous insulin secretion may also be affected by liraglutide.26,27
One limitation is the lack of participant and investigator blinding. However, blinding would have been difficult due to gastrointestinal side effects and ease of the liraglutide dosing pen. Furthermore, a larger study is needed to corroborate our findings and study long-term effectiveness on a metabolically diverse population. Results may not be generalizable because sample size was limited.
Nausea is an expected side effect of GLP-1 analogs, which improves over time.28,29 Since this was a small study done with a single dose of liraglutide, it could be possible that a lower dose and ramping up of the dose be considered. Slowly increasing dosage would ensure patient tolerability. Larger studies in the outpatient setting will also be very important to determine tolerability of medication with stepwise dose increment.
Regarding hypoglycemia, our previous study using exenatide plus CL also had no differences in hypoglycemic episodes compared to the control arm. Hypoglycemia in CL can be mitigated by fine tuning the algorithm to account for use of adjunct medications. In addition, compared to other studies that used only CGM to monitor BG, the current study used CGM as well as Analox BG for confirmation.30,31 Studies that failed to confirm CGM with BG may have had undetected and underreported hypoglycemia.
In conclusion, our study provides a novel approach to CL therapy using adjuvant liraglutide and demonstrates significant improvements in glucose concentrations as a consequence of decreasing both glucagon and insulin concentrations with less insulin use.
Footnotes
Acknowledgements
The authors acknowledge support from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) and the Clinical and Translational Science Awards (CTSA). We also thank Medtronic for the equipment and algorithm support and Montefiore Medical Center for providing additional resources.
Abbreviations
ANOVA, analysis of variance; AUC, area under the curve; BG, blood glucose; CBC, complete blood count; CGM, continuous glucose monitoring; CI, confidence interval; CL, closed loop; CTSA, Clinical and Translational Science Awards; DSMB, Data Safety Monitoring Board; ePID, external physiological insulin delivery; FDA, Food and Drug Administration; GLP-1, glucagon-like peptide-1; HbA1C, hemoglobin A1C; IRB, institutional review board; MARD, mean absolute relative deviation; NCATS, National Center for Advancing Translational Sciences; NIDDK, National Institute of Diabetes and Digestive and Kidney Diseases; NIH, National Institutes of Health; TDD, total daily insulin dose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MC is an employee of Medtronic, the supplier of the closed loop system and data analyzer of the closed loop ePID data. RAH is a shareholder in Thermalin Diabetes and consultant for Lexicon, Merck, and Mannkind. RAH is also participating in trials with Sanofi, Covance, and Lexicon.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This trial was funded by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) and supported in part by the Clinical and Translational Science Awards (CTSA) Grant 1 UL1 TR001073, 1 TL1 TR001072, 1 KL2 TR001071 from the NCATS, a component of NIH. Medtronic provided the equipment and retrieved the data from the software and provided the ePID data for analysis.
