Abstract
Background:
Glide force, average glide force, and glide force variability of the insulin lispro 200 units/mL pen (Eli Lilly and Company, Indianapolis, IN, USA) were compared to the Humalog® KwikPen® 100 units/mL pen (hereafter, KwikPen; Eli Lilly and Company, Indianapolis, IN, USA).
Methods:
Data were collected on 2 doses, 2 injection speeds, and 2 needle types.
Results:
Insulin lispro 200 units/mL pen showed significantly lower maximum glide force, average glide force, and glide force variability than the KwikPen across all combinations of dose size, dose speed, and needle type.
Conclusions:
The lower glide force observed with the insulin lispro 200 units/mL pen offers another treatment option for patients with type 1 or type 2 diabetes who require greater than 20 units of mealtime insulin daily.
Keywords
Increasing insulin doses in patients with diabetes, 1 based on rising obesity-related diseases 2 and insulin resistance, 3 have influenced development of concentrated insulins where the unit dose is delivered in a smaller amount of liquid. Insulin lispro 200 units/mL, presently under development, delivers an insulin dose in half the liquid volume of 100 units/mL Humalog® and if approved, will be available only in a prefilled pen containing 600 units, which dials from 1 to 60 units in single-unit increments.
Patients with diabetes often have concomitant hand impairments4-8 that can make injecting insulin challenging. As diabetes progresses, the likelihood of restricted hand joint mobility and reduced dexterity increases.7,9,10 Larger insulin doses and impaired hand joint mobility and dexterity are 2 of many factors influencing innovation in insulin pens.11,12
Glide force, the measure of effort required to depress the pen’s injection button, is influenced by the injection mechanism design, 13 the inner needle diameter,14,15 temperature, 16 and injection speed (hydraulic flow through the needle). 17 A pen with lower glide force requiring reduced patient effort may be helpful when injecting a large dose of insulin.
The insulin lispro 200 units/mL pen has been redesigned where the internal injection screw travel is half that of the Humalog KwikPen® 100 units/mL (hereafter KwikPen; Eli Lilly and Company, Indianapolis, IN, USA) pen, resulting in half the volume and volumetric flow rate for the same insulin flow rate and equivalent dose based on the concentration difference. This gives the insulin lispro 200 units/mL pen twice the mechanical advantage of the KwikPen, which should reduce glide force.
We compared the glide force characteristics of the new insulin lispro 200 units/mL pen to that of the currently marketed KwikPen.
Methods
Glide force was calculated as maximum and average values. Glide force variability was defined as the difference between the maximum and minimum values of glide force in the plateau region of the force graph of a delivered dose. The maximum and average glide force and glide force variability were measured by a Zwick Linear Test System (Zwick TestXpert II Version 3.4; Ulm, Germany) with a calibrated load cell for injection force characteristics (glide force and glide force variability).
Glide force characteristics were assessed by comparing 100 insulin lispro 200 units/mL pens with 100 KwikPens at injection speeds of 9 or 6 units/s with 2 different pen needles at standard temperature (23±5°C) in accordance with International Standards Organization (ISO)11608-1:2012. 18 Each pen delivered three 30-unit doses and three 60-unit doses for a total of 600 doses from each pen type. The selected injection speeds are similar to those used previously.11,13,17,19,20 Both pens were fitted with Becton Dickinson (BD) 31G/5 mm Pen Needles (Franklin Lakes, NJ). Two variations of the pen needle were used: BD Ultra-Fine 31G/5 mm Pen Needles with PentaPoint Comfort (31G, 5 mm; marketed under various brand names worldwide) and BD 31G/5 mm Pen Needles with PentaPoint Comfort & EasyFlow Technology (31G extra thin wall [XTW], 5 mm; not currently marketed in this needle gauge and length). BD pen needles with thin wall (TW) technology are marketed worldwide. XTW EasyFlow Technology is currently available on a limited set of BD pen needles but is anticipated to replace TW technology on a product-by-product and country-by-country basis. Both pen needles used in this study have the same outer diameter (ie, gauge), but 1 has a thinner inner wall and larger internal diameter, associated with significantly lower injection force characteristics. 15 Based on the anticipated replacement, both pen needle types were used in this study.
During testing, a new needle was attached to each pen. The pen was primed according to the manufacturer’s instructions, then placed into the Zwick Linear Test System with a calibrated load cell. The dose was then manually dialed and ejected by the test system in accordance with written protocols. Glide force data were collected by a calibrated load cell for the injection stroke duration, and force curves were generated.
Measurements were analyzed using Microsoft Excel (Redmond, WA) and JMP statistical software (Cary, NC). Normal quantile plots were used to evaluate data for normality. Not all data sets met the conditions for normality; therefore, all data sets were treated as nonnormal and were compared using the nonparametric, 2-sample, normal approximation median test. Summarized data are reported as medians. Statistical significance was defined as P < .05.
Results
The median glide force values for the insulin lispro 200 units/mL pen were significantly lower than those for KwikPen across all combinations of dose size, dose speed, and needle size (P < .05; Table 1). All representative force curves in the plateau region for the insulin lispro 200 units/mL pen were lower and less variable than the KwikPen across all combinations of dose size and speed (Figure 1, Supplemental Figures 1-3). The percentage decrease in the median maximum glide force from the KwikPen to the insulin lispro 200 units/mL pen was similar at both dose sizes for the TW needle, ranging from 25% to 53% (6 and 9 units/s at 30 units, respectively) and 25% to 52% (6 and 9 units/s at 60 units, respectively; data not shown). The percentage decrease in the median maximum glide force for the XTW needle ranged from 4% to 30% (6 and 9 units/s at 30 units, respectively) and 3% to 30% (6 and 9 units/s at 60 units, respectively).
Comparison of Insulin Lispro 200 Units/mL Pen Device and Humalog KwikPen 100 Units/mL Pen Glide Force Characteristics.
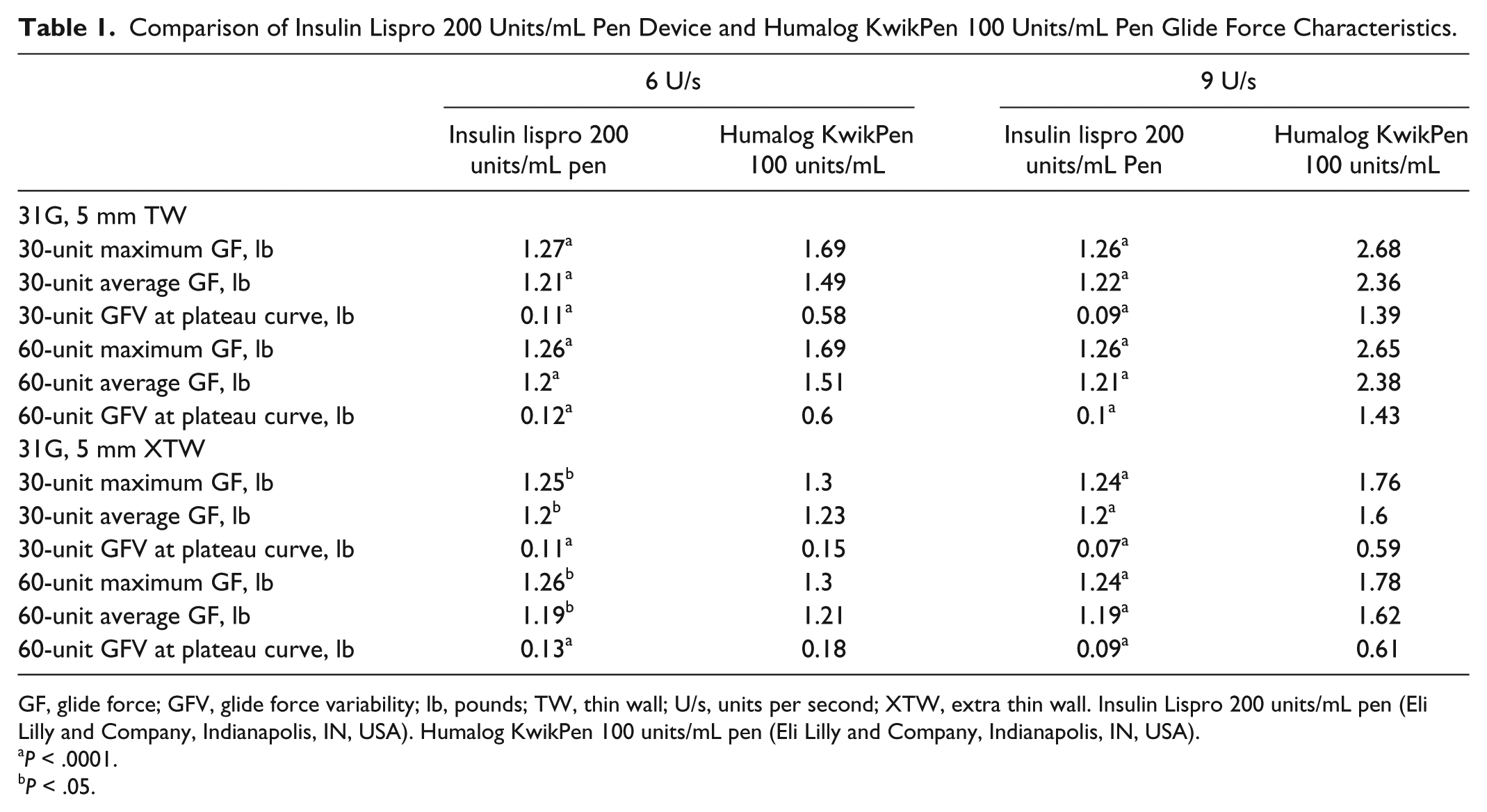
GF, glide force; GFV, glide force variability; lb, pounds; TW, thin wall; U/s, units per second; XTW, extra thin wall. Insulin Lispro 200 units/mL pen (Eli Lilly and Company, Indianapolis, IN, USA). Humalog KwikPen 100 units/mL pen (Eli Lilly and Company, Indianapolis, IN, USA).
P < .0001.
P < .05.
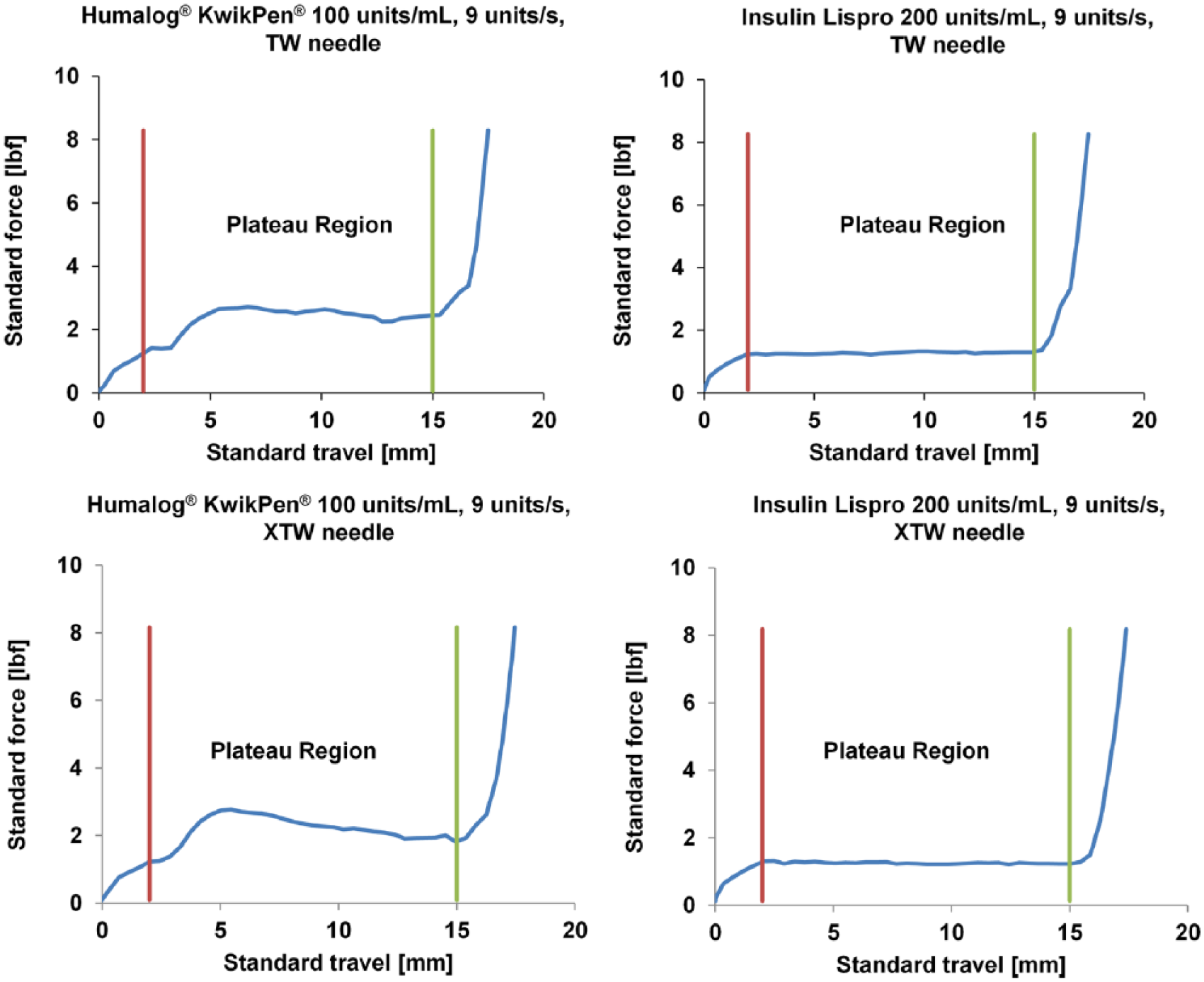
Representative glide force plateaus for the 30-unit dose size at 9 units/s (thin wall and extra thin wall needles). lbf, pound force; TW, thin wall; XTW, extra thin wall. A standard travel of 2.0 mm to 15.0 mm was used for the 30-unit dose size glide force plateau.
Discussion
The lower volumetric flow rate of insulin delivered with the insulin lispro 200 units/mL pen contributed to the observed lower glide force relative to the KwikPen.
We also observed a trend toward lower glide force based on increased internal needle diameter. The glide force of the KwikPen was numerically lower for the XTW needle than the TW needle, and a similar, but less pronounced trend was observed for the insulin lispro 200 units/mL pen. As this was not a study objective, no statistical comparisons were made between needles. Needle gauge, as a function of the increased inner diameter, enables additional reductions in glide force for the same volumetric flow rate. 15 While these forces are substantially below the maximal thumb push force published for various age groups (ranging from 6 to 45 pounds), 21 the significant reductions may be perceivable and desirable in a device used multiple times per day.
During data analysis, we found only 74 of 75 data points were collected for the insulin lispro 200 units/mL pen 60-unit dose size at 9 units/s with the TW needle. However, sufficient data existed to compare both designs.
The insulin lispro 200 units/mL pen offers another treatment option for patients with diabetes who require >20 units/day of mealtime insulin. When used as directed, taking >20 units of mealtime insulin per day ensures that the pen will be used within the in-use dating of 28 days, preventing insulin wastage or use of expired insulin. In addition, use of <20 units per day is associated with smaller doses where glide force reduction is likely to be far less perceptible to patients. As of 2012, over 60% of the US population on rapid acting insulin injects ≥20 rapid acting units a day. 22
Conclusions
The insulin lispro 200 units/mL pen exhibited significantly lower glide force and less glide force variability than the KwikPen. At 30 units, the insulin lispro 200 units/mL pen reduced maximum glide force by 53% with the TW needle and 30% with the XTW needle, respectively, at 9 units/s compared to the KwikPen. The 9 unit/s injection speed, considered a representative injection speed for patients, is consistent with those used in previous studies.11,13,17,19,20 The glide force characteristics of the insulin lispro 200 units/mL pen may be helpful for patients who require >20 units/day of mealtime insulin, with a particular benefit for those patients with hand impairments.
Footnotes
Acknowledgements
The authors thank Daneen T. Angwin (Eli Lilly and Company) for reviewing the protocols, providing technical approval, and programming the test system for data collection and Eileen Girten (inVentiv Health Clinical) for writing assistance.
Abbreviations
BD, Becton Dickinson; ISO, International Standards Organization; TW, thin wall; XTW, extra thin wall.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TMR and DAI are employees and shareholders of Eli Lilly and Company. AHL is a former employee of Eli Lilly and Company.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Eli Lilly and Company.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
