Abstract
The incidence of insulinopenic diabetes mellitus is constantly increasing, and in addition, approximately a third of all hyperinsulinemic diabetic patients develop insulinopenia. Optimal glycemic control is essential to minimize the risk for diabetes-induced complications, but the majority of diabetic patients fail to achieve proper long-term glucose levels even in clinical trials, and even more so in clinical practice. Compliance with a treatment regimen is likely to be higher if the procedure is simple, painless, and discreet. Thus, insulin has been suggested for nasal, gastrointestinal, and inhalation therapy, but so far with considerable downsides in effect, side effects, or patient acceptance. The stratum corneum is the main barrier preventing convenient drug administration without the drawbacks of subcutaneous injections. Recently, devices with miniaturized needles have been developed that combine the simplicity and discretion of patch-based treatments, but with the potential of peptide and protein administration. As this review describes, initial comparisons with subcutaneous administration now suggest microneedle patches for active insulin delivery are efficient in maintaining glycemic control. Hollow microneedle technology could also prove to be efficient in systemic as well as local delivery of other macromolecular drugs, such as vaccines.
Diabetes Mellitus and Insulin Treatment
Insulin deficiency is the hallmark of diabetes mellitus (DM) type 1 and plays an important role in type 2 DM. Before the discovery of insulin, type 1 DM was a uniformly mortal disease, but even with today’s plethora of insulin delivery systems, such as insulin pumps, short and long-acting insulin analogues, and transplantation of islets of Langerhans, or whole pancreas, DM still causes to significant morbidity and mortality. The most important risk factor for complications is poor control of blood glucose.1-4 At the same time, strict glycemic control is associated with a markedly greater risk of hypoglycemia,3,5,6 and recent systematic reviews have not been able to show any improvement in all cause mortality.5,6 However, even adequate glycemic control is difficult to achieve for a large number of patients. 7 This can be traced to difficult to regulate disease in some patients, but in the majority it is tied to poor adherence to insulin-treatment and glucose monitoring.
The problem is that multiple daily injections, and blood samples, which are painful and cause trauma to the skin makes it difficult for diabetic patients to adhere to an optimal treatment regimen. It is known that the majority of diabetic patients are not confident to manage their disease themselves, 8 and that 1 out of 5 diabetic children place their injections inappropriately. 9 Every fourth diabetes patient treated with insulin describes some anxiety regarding self-injection.8,10 Furthermore, the perceived risk of hypoglycemia is another important factor that reduces adherence to strict glucose control. 11
Injections are necessary since the oral bioavailability of insulin is poor because it is poorly absorbed and quickly degraded in the gastrointestinal tract. In addition, as a protein insulin do not permeate the skin without enhancement strategies. An alternative to daily injections is the subcutaneous insulin pump which has been shown to achieve glycemic control, and reduce the risk of complications in diabetic patients. 1 On the other hand, insulin pump therapy requires bulky equipment and the insertion of the subcutaneous catheter, which may be painful.
Compliance with a treatment regimen is likely to be higher if the procedure is simple, painless, and discrete. 12 Insulin has so far been suggested for nasal, gastrointestinal, as well as inhalation therapy.10,13 Gastrointestinal and nasal administrations have been unsuccessful so far, but an insulin inhaler was recently developed and approved. 14 However, since insulin is a potent growth factor, there is concern that intra-alveolar deposition of insulin could impair pulmonary function,15,16 and adoption has been slow. Other routes are therefore to prefer, which do not compromise the comfort and future health of diabetic patients. This review aims to discuss recent research on microneedle drug administration through the skin barrier.
The Skin and Drug Delivery
The skin has 2 main functions: It is a physical barrier, and a receiver of external stimuli such as pain. The skin consists of 3 main sections; the epidermis, dermis, and hypodermis, or subcutaneous tissue (Figure 1). The outer part, epidermis, is approximately 100 µm thick and consists of a main barrier part, stratum corneum, where stacked, dead cells are continuously replaced by outward moving, new cells produced in the more basal layer. The underlying dermis is more than 1000 µm thick and contains blood vessels, sweat glands, hair follicles, and nerves. The dermis, especially the inner dermis, is very rich in nociceptors. The subcutaneous tissue, finally, is a layer of adipose tissue serving as an energy reservoir and thermal insulation. 17
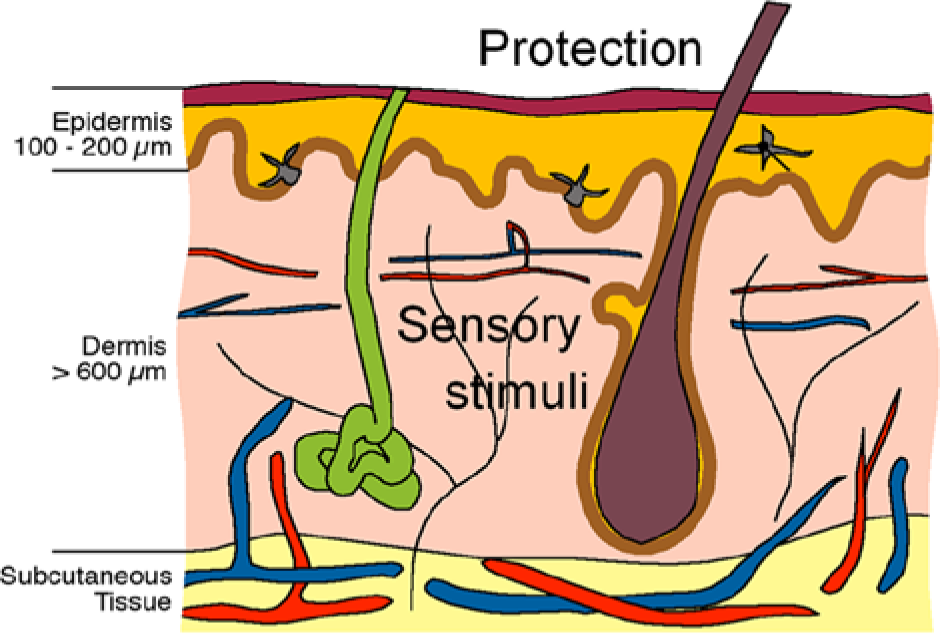
Schematic illustration depicting a cross section of the skin, with epidermis, dermis, and subcutaneous tissue. 46
Transdermal drug delivery means that a pharmaceutical compound is moved across the skin for subsequent systemic distribution. For hydrophobic drugs that easily diffuse across the skin, patch-based drug delivery provides an excellent route of administration. The drawback of this approach for insulin is that the skin’s outer stratum corneum provides an insurmountable barrier. Micrometer-scale needles represent a unique technological approach to enhance drug permeation across the stratum corneum and thereby enable delivery of drugs that would normally require injection. Microneedles are usually designed to penetrate down to the dermal layer of skin, but without reaching and stimulating the dermal nerves. The needles are usually arranged in arrays (Figure 2) and can be used in several ways to enhance transdermal drug transport. The needles can be inserted into the skin before applying the drug in question, to create micron-sized pores that are big enough to increase permeability of macromolecules and even microparticles. Drugs may also be coated onto the microneedles and administrated by inserting the needles into the skin, and there are even reports of polymer microneedles that dissolve upon application. 18 Hollow microneedles, however, are used to directly inject dissolved drug into the skin (Figure 3).

Scanning electron microscopy picture showing a complete microneedle device displayed next to 2 standard 20 G and 27 G syringes. 35
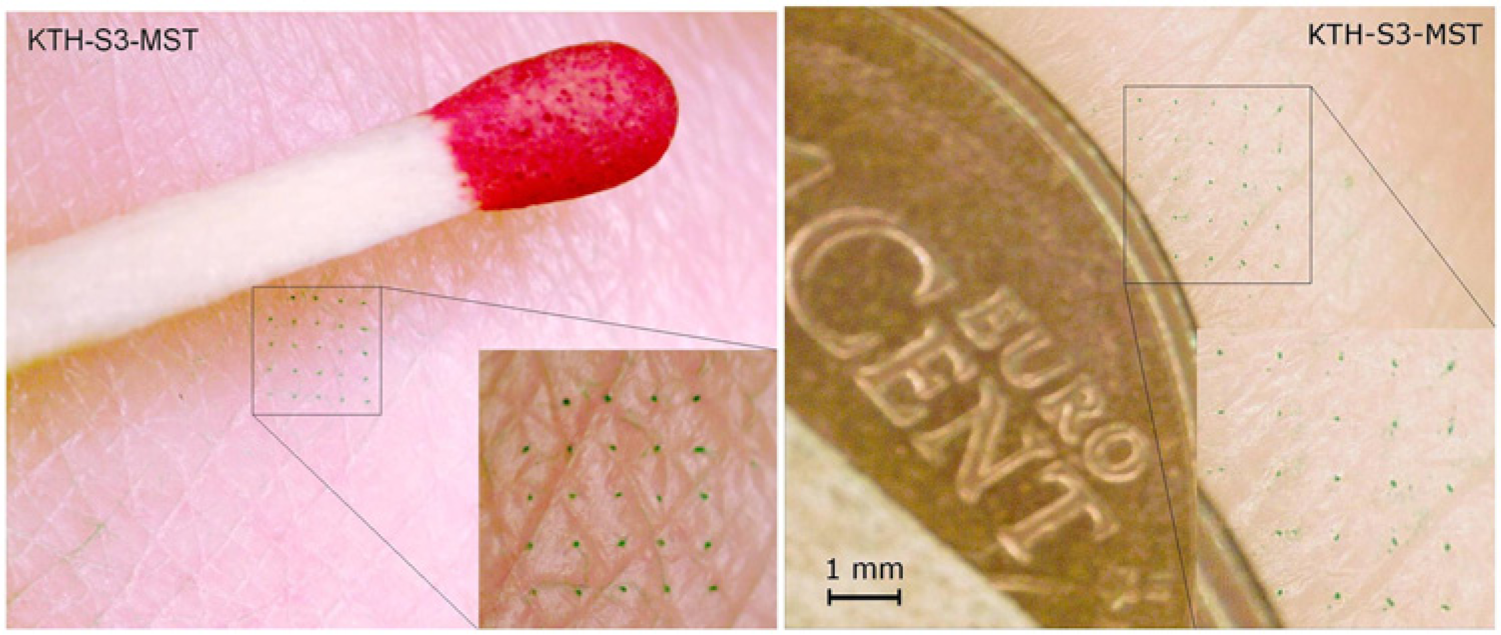
Photomicrograph showing a pattern in the skin of a human subject after dye injection through the patch-like microneedle device. 28
Microneedle Technology
In 1998, the Prausnitz Lab at Georgia Institute of Technology reported the fabrication and insertion of microneedles long enough to cross the stratum corneum, but short enough to not stimulate most nociceptors, which makes the technology minimally invasive and painless.19,20 Their report for the first time describes the use of microfabricated microneedles to enhance drug delivery across skin. 21 In 2003, the same lab reported novel microfabrication techniques for solid and hollow microneedle arrays in the micrometer scale made from silicon, metal, as well as biodegradable polymer, 20 and that same year, Griss et al published the first side-opened hollow out-of-wafer-plane silicon microneedles. 22 In 2005, hollow microneedles were proven to be are clinically effective in bypassing the stratum corneum for drug injections, 23 and later, Kim et al fabricated out-of-plane hollow silicon dioxide microneedle arrays and investigated stratum corneum and skin insertion by altering needle width and cross-section. 24 Recently, several research groups have developed devices with miniaturized needles that combines the simplicity and discretion of patch-based treatments with the potential of peptide and protein administration.24-28 The perhaps most promising drug delivery devices are achieved with a technique known as a Micro-Electro-Mechanical System (MEMS). MEMS is microfabrication technology integrating mechanical elements with sensors, actuators, and electronics on a silicon substrate, rendering possible the production of complete systems-on-a-chip that could completely control insulin treatment (Figure 4).
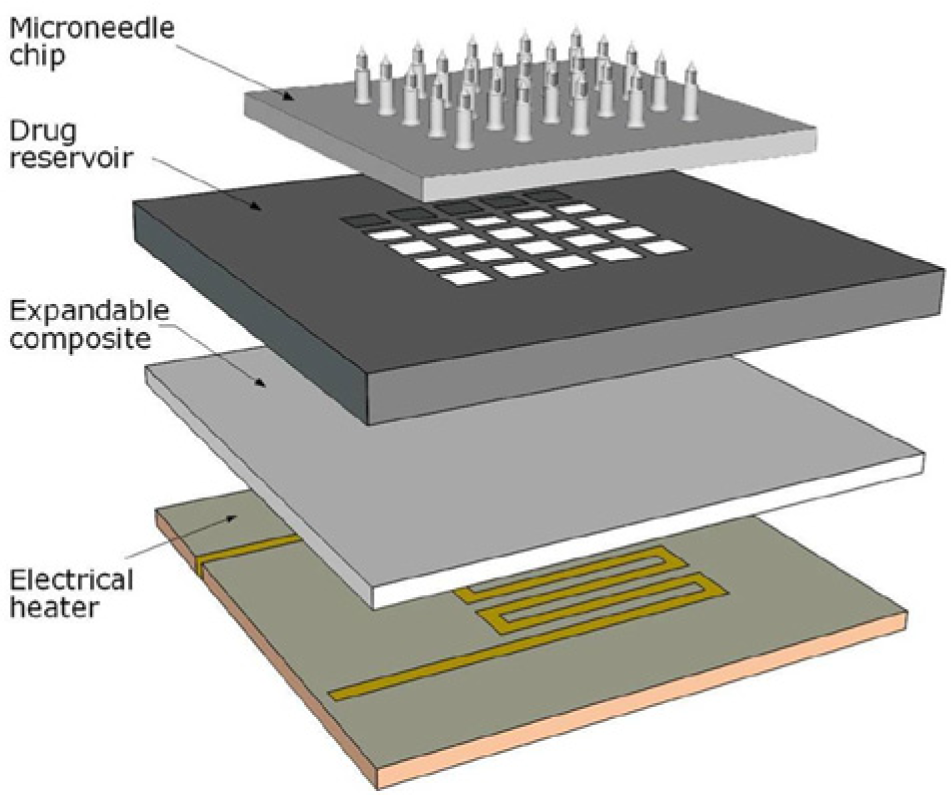
Schematic overview of a Micro-Electro-Mechanical System (MEMS) device.
The main advantage of the parenteral approach is that the drug is introduced into the body without being passed through the human body’s various defense systems. The length of the microneedles means that they reach the capillary-rich layers of the skin where insulin can be readily absorbed and distributed via the circulation. If sufficient bioavailability can be obtained using this technique it would have the advantages of subcutaneous drug delivery, but in a discreet, painless, and minimally invasive manner.
Initial studies on microneedle devices revealed that the needles can be inserted into the skin in a predictable manner. 29 The first reports of enhanced transdermal delivery of insulin in vivo regarded the use of solid microneedles for increasing insulin delivery and decreasing blood glucose levels. 30 Later, infusion in vivo and into human cadaver skin also showed promising results.31,32 In all studies, the human subjects describe microneedle insertions as painless19,20 and in a recent study, all tested microneedles were described as significantly less painful than a 26-gauge hypodermic needle. 33
In 2003, McAllister et al demonstrated microliter infusion into skin in vivo using hollow microneedles, including microinjection of insulin to reduce blood glucose levels in diabetic rats. 20 Since then, several groups have tested microneedle devices for insulin delivery and demonstrated insulin infusion as well as effective reduction of blood glucose concentrations in diabetic rats.30,34,35 Studies so far show no disadvantage using transdermal patches as compared to subcutaneous injection. Subcutaneous injections are known to cause a substantial variability in dose delivered, 11 and intradermal delivery may achieve a more controlled distribution with lower variability. Recently the use of active microneedle delivery was shown to be effective in conscious, chronically diabetic animals, 35 which is important since earlier studies were mostly in short time diabetes without diabetes induced skin changes, 36 and some studies have been performed on anaesthetized rats with minimal physiological registration. 34
Intradermal Glucose Monitoring
Perhaps even more painful than hypodermic insulin injections are the lancets used for conventional blood collection for blood glucose monitoring. 37 However, 0.2 µl is sufficient monitor blood glucose, indicating that the microneedle approach is a feasible alternative. 37 In MEMS technology, sensors can be incorporated into a system to gather information from the environment through measurements of, for example, mechanical, biological, or chemical phenomena. The electronics then process the obtained information and direct the actuators to respond by increasing or decreasing the infusion rate. Thus, it should be possible to develop devices with incorporated blood glucose sensing and self-controlled insulin rates. In 2000, Smart et al described painless blood glucose testing with a silicon microneedle device, drawing less than 0.2 microliters of blood into a microcuvette, where an assay was performed automatically. 38 In 2005, Wang et al demonstrated the use of microneedles for extraction of dermal interstitial fluid for glucose monitoring.39,40 The team reported that the glucose concentrations measured in the interstitial fluid correlated well with blood glucose concentrations measured on rat tail vein blood and on human subjects with a conventional lancet method. Were these studies to be followed up, such a sensor could be integrated with the insulin device to further improve diabetes management.
Concluding Remarks
Recent research shows that the newly developed microneedles are a plausible treatment strategy for insulin delivery to diabetes patients. Further studies should aim at investigating the microneedle devices in terms of adjusted flow rate and biocompatibility. Recent research presents the microneedles are also good candidates for controllable and patient-friendly insulin delivery. The clinical and preclinical data set the stage for microneedle arrays used as intradermal patches in clinical practice and might introduce a more efficient treatment for diabetic patients. This microneedle technology could also prove to be efficient in systemic as well as local delivery of other macromolecular medications, skin-impermeant drugs and biopharmaceuticals, such as protein and DNA vaccines, and endocrine substances.41-45
In parallel with avoiding multiple injection regimens, needle anxiety, and social difficulties associated with self-injecting, microneedle administration route requires minimal training and attention. It is not at all unlikely that compliance with insulin treatment would be higher if the treatment procedure was simple and painless, as with patch-like microneedle devices. Thus, microneedles are a feasible option to improve glycemic control, could set the stage for improved and more comfortable diabetes management and reduce the risk of long-term diabetes complications.
Footnotes
Abbreviations
DM, diabetes mellitus; MEMS, micro-electro-mechanical system.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was funded by the Swedish Society for Medical Research.
