Abstract
Evidence for the value of self-monitoring of blood glucose (SMBG) with regard to the achievement of treatment targets, detection of postprandial glucose excursions, and minimization of glycemic variability is increasing. In large clinical trials, SMBG is a key component for the optimization of diabetes treatment in insulin-treated diabetes. It also plays an essential role in outcome studies. However, details of SMBG use in both the methods and results sections of clinical articles are frequently scarce. Also, a discussion of the SMBG data and its impact on insulin therapy is valuable. In the recently completed Outcome Reduction with an Initial Glargine INtervention (ORIGIN) trial, SMBG was described in detail and insulin titration was largely driven by SMBG. Both aspects largely contributed to the fact that near-normal control was achieved over a long-term period.
The Role of Self-Monitoring of Blood Glucose
Self-monitoring of blood glucose (SMBG) in the management of diabetes plays a key role in many large-scale outcome studies, acting as an important contributor to results. SMBG has many proven benefits, such as aiding the achievement of hemoglobin A1c (HbA1c) targets,1,2 minimizing glucose variability, 3 and helping to predict severe hypoglycemia. 4 In an epidemiological cohort study, SMBG has also been reported to be associated with decreased diabetes-related morbidity and all-cause mortality in type 2 diabetes. 5 SMBG can also heighten patients’ awareness of the disease and the impact of lifestyle on blood glucose levels.3,6 A recent review discusses key publications on SMBG in type 2 diabetes (T2D) and puts them into perspective with regard to results from the 2012 Cochrane analysis where applicable. 7
Given the numerous benefits of SMBG, it is unsurprising that many international and regional guidelines recommend its routine use for successful diabetes management and therapy.8-10 Guidelines recently published by the American Diabetes Association, for example, identified SMBG as a key technique for health care professionals and patients alike to assess the effectiveness of a particular management plan on glycemic control. 10
In this commentary, we briefly explore how SMBG was used in a number of recent landmark clinical trials (Table 1), with a particular focus on studies involving cardiovascular disease (CVD), the leading cause of morbidity and mortality in patients with diabetes. 11 The majority of the trials chosen by the authors were large-scale, international studies. However, other trials were also selected to broaden the picture of the true clinical impact and benefit of SMBG use in patients with type 1 diabetes (T1D) or type 2 diabetes (T2D).
Key Findings From Outcome Studies With SMBG Use.
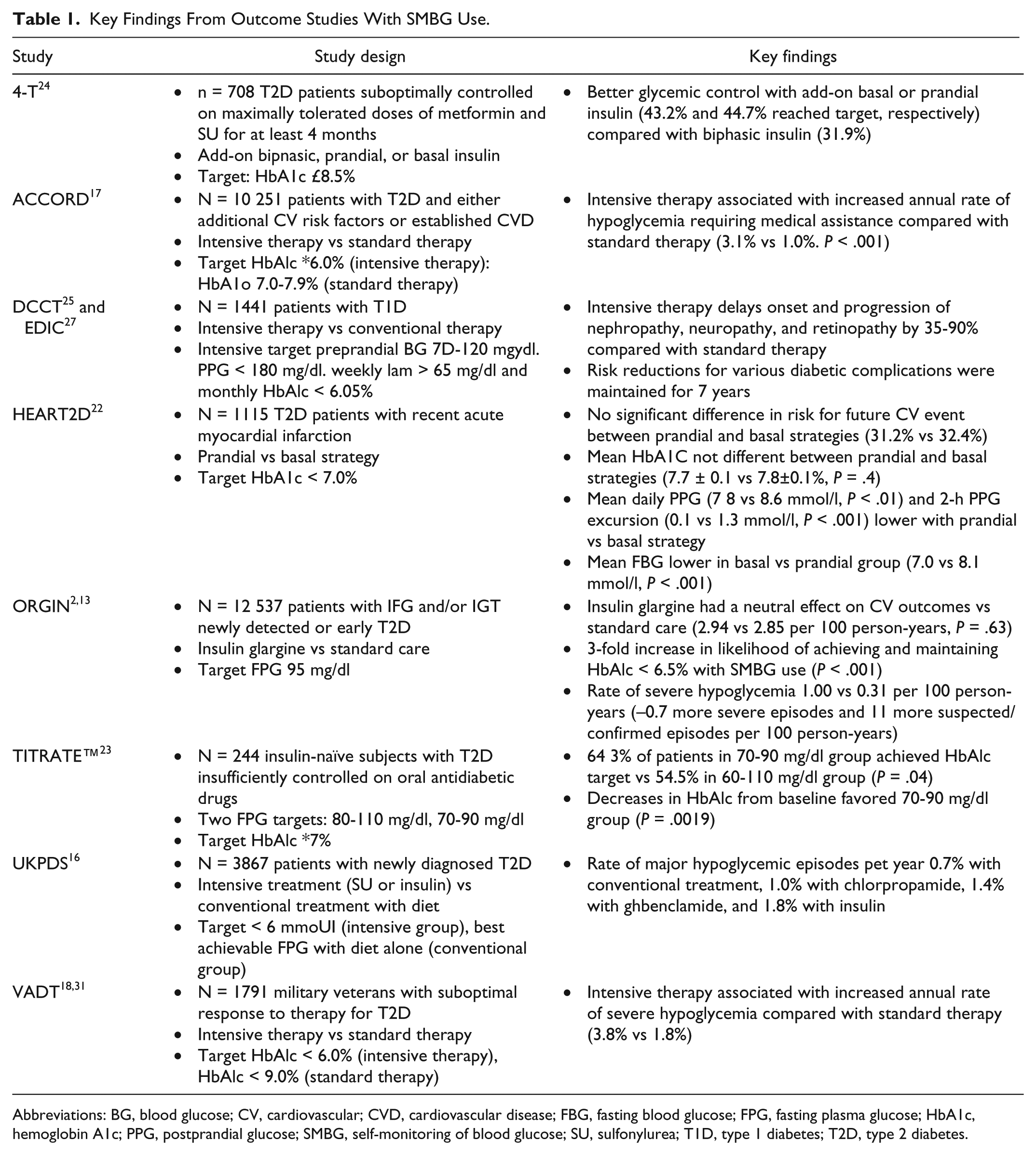
Abbreviations: BG, blood glucose; CV, cardiovascular; CVD, cardiovascular disease; FBG, fasting blood glucose; FPG, fasting plasma glucose; HbA1c, hemoglobin A1c; PPG, postprandial glucose; SMBG, self-monitoring of blood glucose; SU, sulfonylurea; T1D, type 1 diabetes; T2D, type 2 diabetes.
Self-Monitoring of Blood Glucose—An Essential Contributor to Clinical Trials
Reducing Hemoglobin A1c Levels, Minimizing Glucose Variability, and Detecting Severe Hypoglycemia in the Outcome Reduction With an Initial Glargine INtervention Trial
In the recently completed Outcome Reduction with an Initial Glargine INtervention (ORIGIN) trial conducted in dysglycemic patients with cardiovascular (CV) risk factors, all eligible participants were taught to measure their own blood glucose levels at trial onset prior to randomization. SMBG served as a key pillar of diabetes management for patients receiving insulin glargine. Participants in this treatment arm were instructed to take daily fasting plasma glucose (FPG) readings until they reached self-measured FPG levels between 72 mg/dl and 95 mg/dl, and then at least twice per week. Insulin glargine was uptitrated weekly as necessary through the use of an algorithm. Patients with T2D, present at baseline or developed during the trial, receiving standard care were provided with self-glucose monitoring equipment and support. 12
Although data are not available on how many, or which, patients self-titrated, as opposed to being titrated by their physicians, what is deducible from the results is the fact that titration of basal insulin took a relatively long time. After 1 year, 50% of patients in the insulin glargine group achieved the target FPG level (≤94 mg/dl), 13 which might be advantageous from the point of view of avoiding the high morbidity and mortality levels seen in other CV outcome studies. Importantly, the target fasting blood glucose (FBG) level was maintained throughout the entire trial period in the insulin glargine group, 13 the lowest mean FBG observed in any major diabetes outcome trial. This is the best single piece of evidence supporting the value of SMBG.
Insulin glargine had a neutral effect on CV outcomes, with incidence rates of 2.94 and 2.85 per 100 person-years observed in the insulin glargine and standard care groups, respectively. There was no significant increase in the risk of death from CV causes, nonfatal myocardial infarction or nonfatal stroke in patients with preexisting CV disease versus those with no CV history. 13 Individuals using SMBG to adjust their daily dose of basal insulin were reported to be approximately 3-fold more likely to achieve and maintain near-normal glycemic levels, defined as HbA1c <6.5%, compared with individuals receiving standard care (P < .001). 2
Since severe hypoglycemia often follows a specific blood glucose fluctuation pattern identifiable from SMBG, partial prediction of imminent severe hypoglycemia is possible and can help trigger self-regulation of significant hypoglycemia. 5 Expert groups therefore recommend the use of SMBG in patients with T2D who use oral glucose lowering agents and/or combined treatments to help detect hypoglycemia.14,15 Although the incidence of a first episode of severe hypoglycemia in ORIGIN was significantly higher with insulin glargine versus standard care (1.00 vs 0.31 per 100 person-years, P < .001), the absolute increase in risk was low (approximately 0.7 more severe episodes and 11 more suspected or confirmed episodes per 100 person-years) compared with other insulin studies. 13
In the United Kingdom Prospective Diabetes Study (UKPDS), for example, the rate of major hypoglycemic episodes per year was 1.8% versus 0.7% with insulin and conventional treatment, respectively, an absolute risk difference of 1.1%. 16 Other examples of studies where patients receiving intensive therapy reported higher rates of severe hypoglycemia compared with those receiving standard care include the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study 17 and the Veterans Affairs Diabetes Trial (VADT). 18 In ACCORD, the annualized rate of hypoglycemia requiring medical assistance was 3.1% versus 1.0% (P < .001)—an absolute risk difference of 2.1%. 17 In VADT, the absolute risk difference in the annual rate of severe hypoglycemia was 2.0% (3.8% vs 1.8%, respectively). 18
These findings emphasize the long-term beneficial role SMBG plays in keeping HbA1c <6.5% over 6 years, with a low risk of hypoglycemia and a low mean amplitude of glycemic excursions (MAGE). A further substudy of ORIGIN confirmed that insulin glargine does not significantly affect glycemic variability (standard deviation of mean initial glucose concentration and MAGE), as measured by the continuous glucose monitoring system. 19 Minimizing glycemic variability helps to reduce adverse events, such as the impairment of endothelial function and cognitive performance.20,21 The findings from the ORIGIN trial provide further evidence for SMBG use in diabetes management.
Guiding Treatment in the Hyperglycemia and Its Effect After Acute Myocardial Infarction on Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus Trial
Another clinical trial that made use of SMBG was the large-scale Hyperglycemia and Its Effect After Acute Myocardial Infarction on Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus (HEART2D) trial, which involved 1115 T2D patients with recent acute myocardial infarction. The aim of this trial was to compare the effect of prandial versus basal strategies on CV outcomes. Participants were assigned to 1 of 2 treatment groups (prandial or basal strategy), targeting an HbA1c level <7.0%. In addition, individuals in the prandial treatment group (mealtime insulin lispro 3 times daily) had a self-monitored postprandial glucose (PPG) target of <135 mg/dl, while those in the basal group (neutral protamine hagedorn twice daily or insulin glargine once daily) had an FBG target of <121 mg/dl. Treatment doses were adjusted accordingly when HbA1c levels were >8.0% on 2 consecutive visits and patients were to be followed for up to 7 years. 22
The trial was stopped after approximately 3 years for lack of efficacy when no difference in risk for future CV event rates was observed between the 2 groups. The lower FBG levels obtained in the basal group compared with the prandial group (7.0 vs 8.1 mmol/l; P < .001) may have contributed to the fact that despite significant differences in PPG and glucose excursions between the 2 groups, no differences in CV outcomes were observed. In addition, the difference in PPG levels did not reach the goal of 2.5 mmol/l assumed for the power calculations to achieve the primary objective. 22 Undoubtedly, SMBG was a key element in the study, helping to keep HbA1c levels at comparable levels between the 2 groups.
Supporting Patient Empowerment Through Disease Self-Management—The Treating to Target in Type 2 Diabetes Study and the TITRATE Study
The use of SMBG in clinical trials allows patients to become more involved in their own treatment and this sense of empowerment has been found to be essential to motivating patients to achieve treatment targets. 23
Such patient-directed titration was prominently used in the Treating to Target in Type 2 Diabetes (4-T) study, which examined the efficacy and safety of add-on biphasic, prandial or basal insulin to oral therapy in patients with T2D suboptimally controlled on maximally tolerated doses of metformin and sulfonylurea (SU). Patients had 6 scheduled visits during the first year of the 3-year study, followed by quarterly visits thereafter, and were asked to measure their capillary glucose levels 3 times prior to each visit. Between visits, regular SMBG measurements were taken to monitor glucose levels and hypoglycemia, with investigators and patients encouraged to vary and amend doses between visits according to the recommendations of an online trial-management system, if deemed clinically appropriate. The study concluded that add-on basal or prandial insulin provided better glycemic control compared with biphasic insulin, with 43.2% and 44.7% of patients in the respective groups obtaining an HbA1c level of ≤6.5% compared with 31.9% of those on biphasic insulin. 24 It highlights how SMBG enables patients to clearly visualize the impact of various insulin treatment approaches on HbA1c levels and provides useful information that can be used to inform decisions about treatment optimization.
Another trial where patients played an important role in directing their treatment by SMBG was the Treat to Target with Once-Daily Insulin Therapy: Reduce A1C by Titrating Effectively (TITRATE) study. TITRATE was the first prospective randomized study to examine the effect of different FPG targets on glycemic control using patient-directed titration of once-daily insulin detemir. The aim of the study was to compare the efficacy and safety of 2 FPG titration targets (80-110 mg/dl and 70-90 mg/dl) in insulin-naïve subjects with T2D insufficiently controlled on oral antidiabetics. A patient-directed, treat-to-target algorithm was used based on which subjects self-titrated their insulin detemir dose every 3 days according to the mean FPG level of daily readings taken in the previous 3 days. Patients received further support in the form of a patient card (to assist them with calculations and dosing), a patient training booklet, education and counseling sessions, office visits, and frequent phone contact. 23
Results at the end of the 20-week study suggested that the lower FPG target of 70-90 mg/dl showed superior efficacy compared with the higher target of 80-110 mg/dl, resulting in a decrease in mean HbA1c from baseline of 1.2% and 0.9%, respectively (P = .0019). Overall, both treatment targets led to significant reductions in HbA1c levels and the majority of patients achieved an HbA1c level <7%. This trial provides key evidence on the utility of SMBG-guided insulin titration in T2D to achieve more aggressive goals. The authors concluded that empowering patients to adjust their basal insulin dose using a simple, patient-directed titration algorithm with the help of SMBG can lead to measurable improvements in their condition. 23
Diabetes Control and Complications Trial and the Epidemiology of Diabetes Interventions and Complications—Establishing the Standard of Care for Patients With T1D
Although the results from the Diabetes Control and Complications Trial (DCCT) were published in 1993, this landmark trial broadened our knowledge and helped establish SMBG as the standard of care for patients with T1D.25,26 It demonstrated the impact of near-normal glycemic control on reducing the development and progression of microvascular and neurological complications in patients with T1D. Participants receiving intensive therapy used SMBG, and additional factors such as dietary intake and expected exercise, to adjust their insulin dosage. Readings were taken ≥4 times daily to meet target preprandial glucose levels of 70-120 mg/dl, PPG levels of <180 mg/dl, weekly 3
SMBG was 1 of the key technologies that made the facilitation of DCCT possible, allowing participants to be followed for a mean of 6.5 years. 25 Results from the trial provided evidence that intensive insulin therapy effectively delays the onset and progression of nephropathy, neuropathy, and retinopathy by 35-90% compared to conventional insulin therapy. The authors concluded that intensive intervention was most effective when begun early, before complications were detectable.25,27
Similarly, use of SMBG was also employed in the follow-up trial to the DCCT—the Epidemiology of Diabetes Interventions and Complications (EDIC) study. The aim of the new study was to determine the long-term impact of maintaining HbA1c levels as close to normal as feasible on the risk of developing microvascular and macrovascular outcomes. Over 36% of patients in both treatment groups of EDIC monitored their own blood glucose levels at least 4 times daily. 28 Findings from EDIC support a strategy of early insulin use with titration by SMBG, reporting that risk reductions for various diabetic complications were maintained throughout the 7 years of follow-up. 27
Summary
Guidelines from expert bodies such as the International Diabetes Federation, the European Society of Cardiology, and the European Association for the Study of Diabetes recognize SMBG as an indispensable prerequisite for the successful management and therapy of diabetes. They highlight that the effective use of SMBG fosters self-management and empowerment, and help form the rational basis for a partnership between patients and their health care team to help improve outcomes.3,9 Specific schemes for SMBG exist, including those recently recommended by a group of European experts. 29
As discussed, SMBG can play an essential role in outcome studies, providing a means by which participants can monitor their glucose levels and manage their treatment accordingly. For example, patients can use SMBG to assess the relative contributions of postprandial and basal hyperglycemia, and tailor their insulin dosage appropriately to avoid the onset of hypoglycemic episodes.14,15
The usefulness of SMBG in outcome trials is plausible but cannot be proven in insulin-treated patients by a treatment arm without SMBG. In addition, there is strong evidence to suggest that SMBG, when used effectively, can help patients achieve good glycemic control and reduce the risk of diabetes-related complications.12,25,27,30,31 This effect has been most recently evidenced in the ORIGIN trial, where the use of SMBG to adjust basal insulin dose resulted in a significant increase in the ability of patients to maintain near-normal glycemic levels versus standard care. FPG levels of <108 mg/dl were achieved and maintained for at least 5 years in over 75% of patients receiving insulin glargine, and median HbA1c remained ≤6.3% throughout the study. 13 While the use of SMBG is frequently described in the methods section of clinical articles, a recognition of its impact on results is often lacking in discussions of findings and is hence recommended for inclusion. The discussion of the SMBG data and its impact on insulin therapy is valuable and highly warranted.
Footnotes
Abbreviations
4-T, Treating to Target in Type 2 Diabetes; ACCORD, Action to Control Cardiovascular Risk in Diabetes; BG, blood glucose; CV, cardiovascular; CVD, cardiovascular disease; DCCT, Diabetes Control and Complications Trial; EDIC, Epidemiology of Diabetes Interventions and Complications; FBG, fasting blood glucose; FPG, fasting plasma glucose; HbA1c, hemoglobin A1c; HEART2D, Hyperglycemia and Its Effect After Acute Myocardial Infarction on Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus; MAGE, mean amplitude of glycemic excursions; ORIGIN, Outcome Reduction with an Initial Glargine INtervention; PPG, postprandial glucose; SMBG, self-monitoring of blood glucose; SU, sulfonylurea; T1D, type 1 diabetes; T2D, type 2 diabetes; TITRATE, Treat to Target with Once-Daily Insulin Therapy: Reduce A1C by Titrating Effectively; UKPDS, United Kingdom Prospective Diabetes Study; VADT, Veterans Affairs Diabetes Trial.
Authors’ Note
The contents of this article and opinions expressed within are those of the authors, and it was the decision of the authors to submit the manuscript for publication. The authors were responsible for the conception of the article and contributed to the writing, including critical review and editing of each draft, and approval of the submitted version.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Markolf Hanefeld has received speaker honoraria from Takeda, GSK, Roche, BAYER, Lilly, and Sanofi-Aventis, and advisory board honoraria from Sanofi-Aventis, Takeda, BMS, and GSK.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Editorial support was provided by Sandy Ong and Lisa Swanson (Havas Life Medicom) and funded by Sanofi.
