Abstract
Objective:
The objective was to assess the reliability of a novel automated closed-loop glucose control system developed within the AP@home consortium in adults with type 1 diabetes.
Research Design & Methods:
Eight adults with type 1 diabetes on insulin pump therapy (3 men; ages 40.5 ± 14.3 years; HbA1c 8.2 ± 0.8%) participated in an open-label, single-center, single-arm, 12-hour overnight study performed at the clinical research facility. A standardized evening meal (80 g CHO) accompanied by prandial insulin boluses were given at 19:00 followed by an optional snack of 15 g at 22:00 without insulin bolus. Automated closed-loop glucose control was started at 19:00 and continued until 07:00 the next day. Basal insulin delivery (Accu-Chek Spirit, Roche) was automatically adjusted by Cambridge model predictive control algorithm, running on a purpose-built embedded device, based on real-time continuous glucose monitor readings (Dexcom G4 Platinum).
Results:
Closed-loop system was operational as intended over 99% of the time. Overnight plasma glucose levels (22:00 to 07:00) were within the target range (3.9 to 8.0 mmol/l) for 75.4% (37.5, 92.9) of the time without any time spent in hypoglycemia (<3.9 mmol/l). Mean overnight glucose was 7.8 ± 1.3 mmol/l. For the entire 12-hour closed-loop period (19:00 until 07:00) plasma glucose levels were within the target range (3.9 to 10.0 mmol/l) for 84.4% (63.3, 100) of time. There were no adverse events noted during the trial.
Conclusions:
We observed a high degree of reliability of the automated closed-loop system. The time spent in target glucose level overnight was comparable to results of previously published studies. Further developments to miniaturize the system for home studies are warranted.
Despite significant improvements in the care of type 1 diabetes, achieving tight glycemic control while avoiding hypoglycemia1,2 remains a challenge for many patients and novel therapeutic approaches are required. Closed-loop insulin delivery is an emerging treatment option for patients with type 1 diabetes. 3 Compared to preprogrammed basal insulin rates in insulin pumps, closed-loop insulin delivery systems modulate insulin delivery in glucose-responsive fashion based on subcutaneous sensor glucose levels utilizing advanced computer algorithms. 3
Several studies have evaluated the safety and efficacy of closed-loop under laboratory conditions and shown promising results. These include evaluations using a randomized design by our group in youths,4,5 adults, 6 and pregnant women 7 and by others using the model predictive control algorithm,8,9, the proportional-integral-derivative approach,10,11 and the fuzzy logic controller.12,13 Insulin and glucagon coadministration have also been applied in randomized studies.14-16 Several of above studies4-7 were conducted in the manual operational mode where sensor glucose values were entered into a laptop computer running the algorithm followed by manual adjustment of the insulin pump. More recently automated closed-loop control systems utilizing an ultra-portable laptop computer 17 or mobile phone platform 18 have also been described. In February 2010, the European Union granted funding for the AP@home project (Artificial Pancreas at Home), a consortium of European academic medical centers, biotechnology companies, and industrial partners, to carry out closed-loop glucose control research. 19 The ultimate goal of the AP@home project is to develop a closed-loop system for use outside clinical research centers. The primary objective of the current study was to evaluate the reliability of the first automated closed-loop glucose control system developed fully within the AP@home consortium, laying the foundations for future system developments. Secondary objectives included evaluation of overnight glucose control and accuracy of Dexcom G4 sensor.
In contrast to previous systems where control algorithm resides within a commercially available laptop or mobile phone, the current system incorporates a dedicated artificial pancreas platform (APP) powered by a main processor with an additional supplementary safety processor capable of periodic integrity checks on the main processor.
Methods
Patients and Study Design
The study protocol, participant information sheet, and consent form were approved by Cambridge Central Research Ethics Committee. The study was an investigator-initiated, prospective, single-center, single-arm, open-label feasibility study performed at the NIHR/Wellcome Trust Clinical Research Facility at Addenbrooke’s Hospital, Cambridge, UK.
Study participants were recruited from March 2013 to May 2013. Key inclusion criteria were adults (≥18 years) with a diagnosis of type 1 diabetes over at least 1 year and treated with insulin pump therapy. Key exclusion criteria were concurrent illness or medications likely to interfere with interpretation of study results, poor glycemic control (HbA1c >10%), recurrent severe hypoglycemia, significant hypoglycemia unawareness, and clinically significant nephropathy, neuropathy, or retinopathy. All participants provided written informed consent prior to study related activities.
Automated Closed-loop System
The automated closed-loop system (Figure 1) comprised (1) CE marked Dexcom G4 Platinum continuous glucose monitoring (CGM) system (Dexcom Inc, San Diego, CA), (2) a purpose-built embedded APP unit (Triteq Ltd, Berkshire, UK) running a model predictive control (MPC) algorithm, (3) CE marked Accu-Chek Spirit Combo insulin pump (Roche, Burgdorf, Switzerland), and (4) a laptop PC providing the graphical user interface and USB connections for the APP unit and the Dexcom G4 CGM. The algorithm used in the current study is broadly similar to the Cambridge closed-loop algorithm used in previous studies.4,6
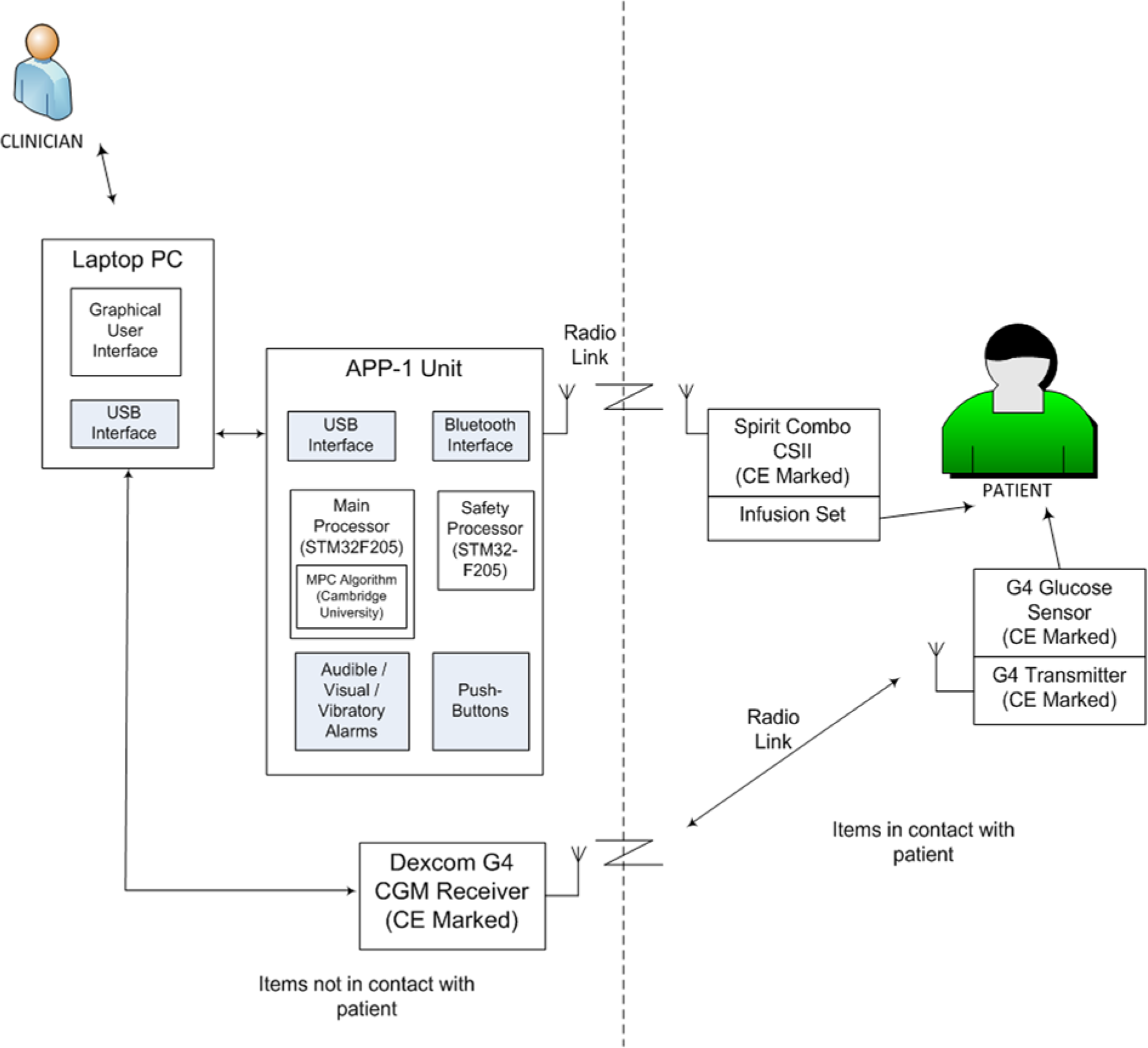
Design of the closed-loop system. On the right side of the figure separated by the dashed line are the components attached to the patient (Spirit Combo insulin pump and Dexcom G4 transmitter). artificial pancreas platform (APP) unit contains a dedicated main processor running the algorithm and a safety processor. APP unit and the Dexcom receiver are attached to a laptop computer that has the user interface.
The sensor was inserted in either the anterior abdominal wall or upper arm. The APP unit contained a main STM32 processor (STMicroelectronics, Geneva, Switzerland), capable of running advanced control algorithms together with a safety processor that carried out a periodic integrity check on the main processor. In addition the APP unit was able to raise audible, visual, and vibratory alarms in the case of a malfunction.
Every 15 minutes, the algorithm calculated a new insulin infusion rate, which was wirelessly sent to the study insulin pump. The calculations utilized a compartment model of glucose kinetics 20 describing the effect of rapid-acting insulin and the carbohydrate content of meals on glucose levels. Information about the preprogrammed basal insulin delivery rates, carbohydrate intake, and meal time insulin bolus information was provided to the algorithm via the user interface. In addition, participant’s weight and total daily insulin dose were entered at setup. The treat-to-target control algorithm aimed to achieve glucose levels between 5.8 and 7.3 mmol/l. Algorithm version 0.3.24 was used (University of Cambridge).
Study Procedures
Continuous glucose monitoring was commenced 48 to 72 hours prior to each study visit. Participants were instructed to calibrate the sensor according to manufacturer instructions using quality-controlled Bayer Contour Next (Bayer plc, Berks, UK) glucometer. On arrival at the clinical research facility (approximately 17:30), participants were connected to the study insulin pump infusing insulin analogue Aspart (Novo Nordisk, Bagsvaerd, Denmark) according to usual basal rates. An intravenous cannula was inserted to allow for frequent venous sampling starting at 18:40. In all patients the sensor was calibrated using finger-stick glucose value before the start of closed-loop operation. Venous blood samples were collected at 15-minute intervals for the measurement of plasma glucose and insulin levels for the first 3 hours between 19:00 and 22:00 hours, followed by every 30 minutes thereafter until 07:00 hours following morning.
Participants consumed a self-selected evening meal containing 80 g of carbohydrates at 19:00. Ten minutes before the evening meal, an insulin bolus was delivered, calculated using subject’s standard insulin pump bolus calculator settings and premeal finger-stick glucose levels. The insulin bolus was given with the meal if finger-stick glucose ≤ 4.0 mM. In addition subjects were also offered an optional 15 g snack at 22:00 without any bolus insulin or informing the algorithm. The purpose of the snack was to offer participants a choice. It was not given to prevent hypoglycemia. All 8 participants decided to have the bedtime snack. Closed-loop glucose control was commenced at 19:00 and continued for the next 12 hours terminating at 07:00 the following morning.
Assays
We used a YSI2300 STAT Plus Analyzer (YSI, Lynchford House, Farnborough, UK) for determination of plasma glucose and an immunochemiluminometric assay (Invitron, Monmouth, UK; intraassay CV 4.7%; interassay CV 7.2 to 8.1%) for determination of plasma insulin.
Statistical Analysis
Investigators agreed on the outcome measures and the analysis plan in advance. The primary reliability outcome was the percentage of time the system functioned as intended in active closed-loop mode. Secondary reliability outcomes included failure rates of various system components such as CGM device, the artificial pancreas unit, the user interface and communication failures. Other secondary outcomes were time spent with glucose levels between 3.9 and 8.0 mmol/L and 3.9 to 10 mmol/L as measured by reference (YSI) glucose, time spent above and below the target glucose range, mean and standard deviation of plasma glucose, total number of insulin units infused, plasma insulin concentration, absolute and relative difference between matched pairs of subcutaneous glucose sensor and reference plasma glucose. The safety evaluation comprised of number of episodes of hypoglycemia (plasma glucose ≤ 3.3 mmol/L), serious hypoglycemia (plasma glucose < 2.8 mmol/L), number of episodes of significant ketosis (plasma ketones >3 mmol/l), and nature and severity of any other adverse events. Reliability and safety outcome were calculated for the entire study period from 19:00 to 07:00 hours. Glucose based outcomes were calculated for the entire study period as well as overnight between 22:00 and 07:00 period.
Calculations were carried out using SPSS version 19 (IBM Software, Hampshire, UK). Outcomes were calculated by GStat software version 1.3 (University of Cambridge, UK). Values are given as mean (SD) or median (interquartile range [IQR]).
Results
Study Participants
All 8 recruited subjects (3 males) completed the study; age 40.5 ± 14.3 years, body mass index (BMI) 24.5 ± 1.9 kg/m2, diabetes duration 28.2 ± 14.1 years, HbA1c 8.2 ± 0.8% (66 ± 9 mmol/mol), and prestudy total daily insulin dose 0.61 ± 0.17 U/kg.
System reliability
Except in 1 subject the system performed as intended 100% of time. In the remaining subject closed-loop was interrupted for 20 minutes due to accidental detachment of the power cable of the laptop and battery drainage. Overall time spent in closed loop was 99.6% (including the interruption due to battery failure). No failures were noted with any of the system components.
Glucose control and insulin administration
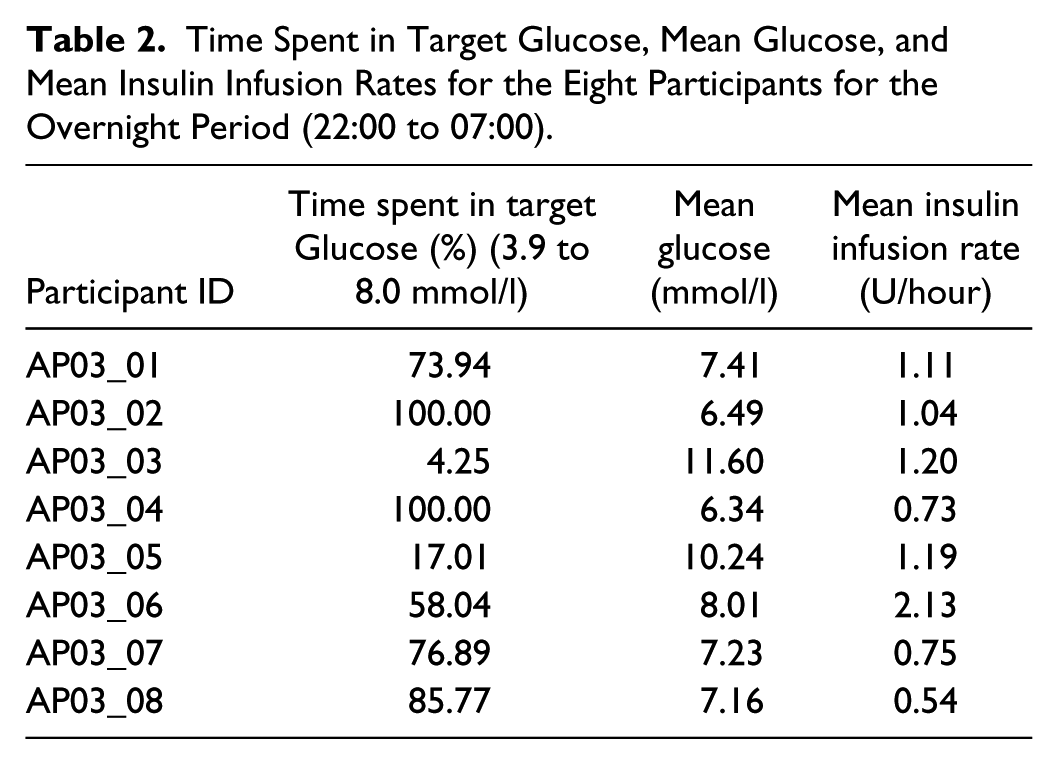
The study efficacy outcomes for the overnight (22:00 to 07:00) as well as for the full duration (19:00 to 07:00) are shown in Table 1. For the overnight period, the percentage of time with plasma glucose in the target range between 3.9 and 8.0 mmol/l was 75.4% (37.5, 92.9) with a mean glucose of 7.3 (6.8, 9.1) mmol/l. The percentage of time with plasma glucose in the wider target range from 3.9 to 10.0 mmol/l for the entire study period was 84.4% (63.3, 100) with a mean glucose of 7.6 (6.7, 9.1) mmol/l. The mean insulin infusion rate overnight was 0.96 ± 0.33 U. Profiles of plasma glucose, insulin infusion rates, and plasma insulin are shown in Figure 2. Individual participant data for the overnight period are shown in Table 2.
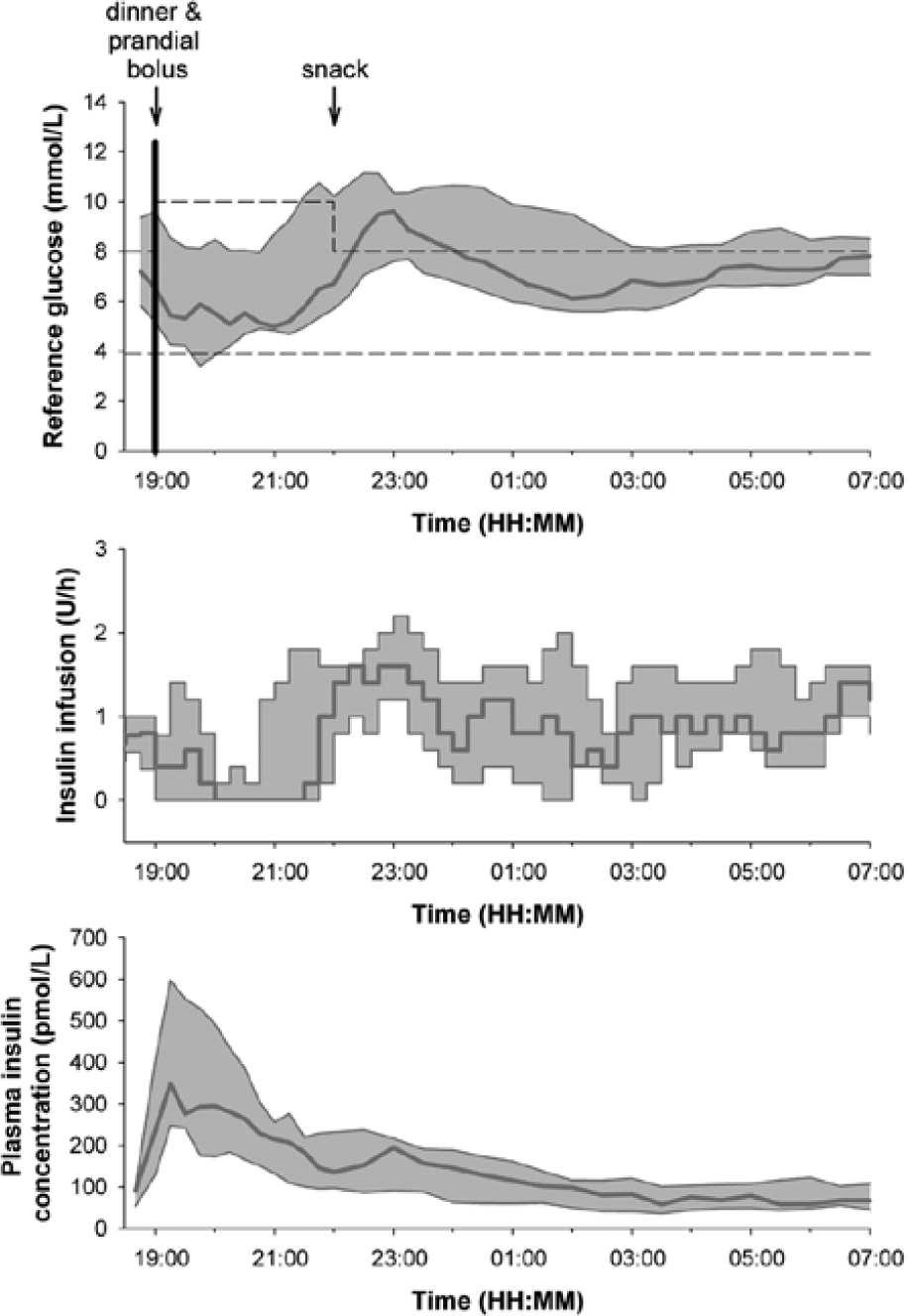
Plasma glucose, insulin infusion, and plasma insulin levels. Data shown are median (IQR).
Study Outcomes and Insulin Infusion Information Based on Reference Glucose.
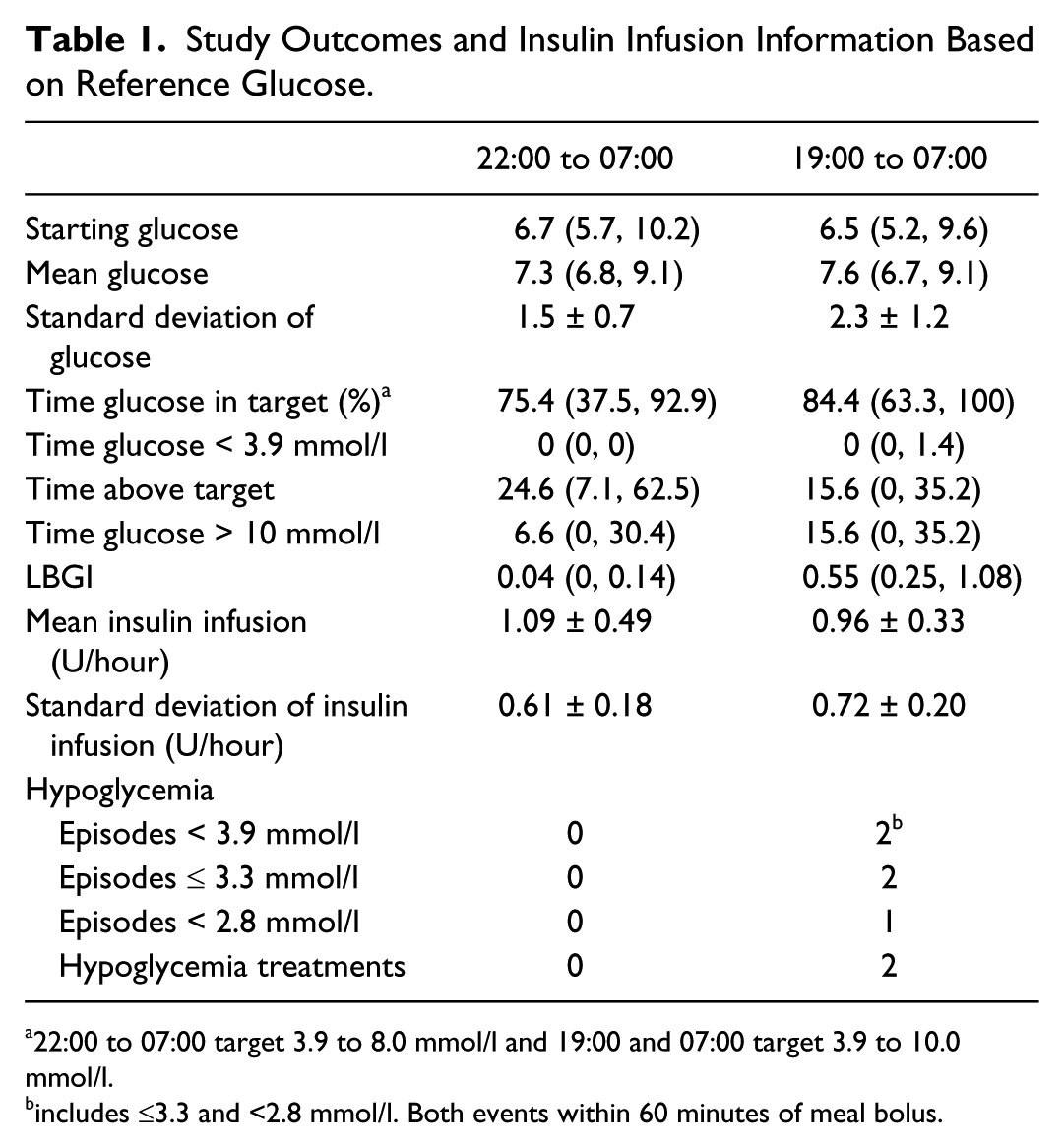
22:00 to 07:00 target 3.9 to 8.0 mmol/l and 19:00 and 07:00 target 3.9 to 10.0 mmol/l.
includes ≤3.3 and <2.8 mmol/l. Both events within 60 minutes of meal bolus.
Time Spent in Target Glucose, Mean Glucose, and Mean Insulin Infusion Rates for the Eight Participants for the Overnight Period (22:00 to 07:00).
Safety Aspects
There were 2 hypoglycemic events, both of which occurred within 60 minutes of the evening meal bolus and could not be prevented despite complete cessation of insulin delivery by the algorithm. There were no other adverse events.
Sensor Performance
Sensor performance was good with the median absolute deviation of 0.6 (0.3-1.1) mmol/l and the median absolute relative deviation (MARD) of 8.3% (4.0-14.5). Detailed clinical and numerical sensor accuracy metrics are shown in Table 3. Of the values, 87% were in Clarke error grid zone A (Figure 3), which indicates a high degree of clinical accuracy (<20% deviation from reference glucose).
Numerical and Clinical Accuracy of Dexcom G4 Sensor Over the 12-Hour Study Period (19:00 to 07:00 Following Day).
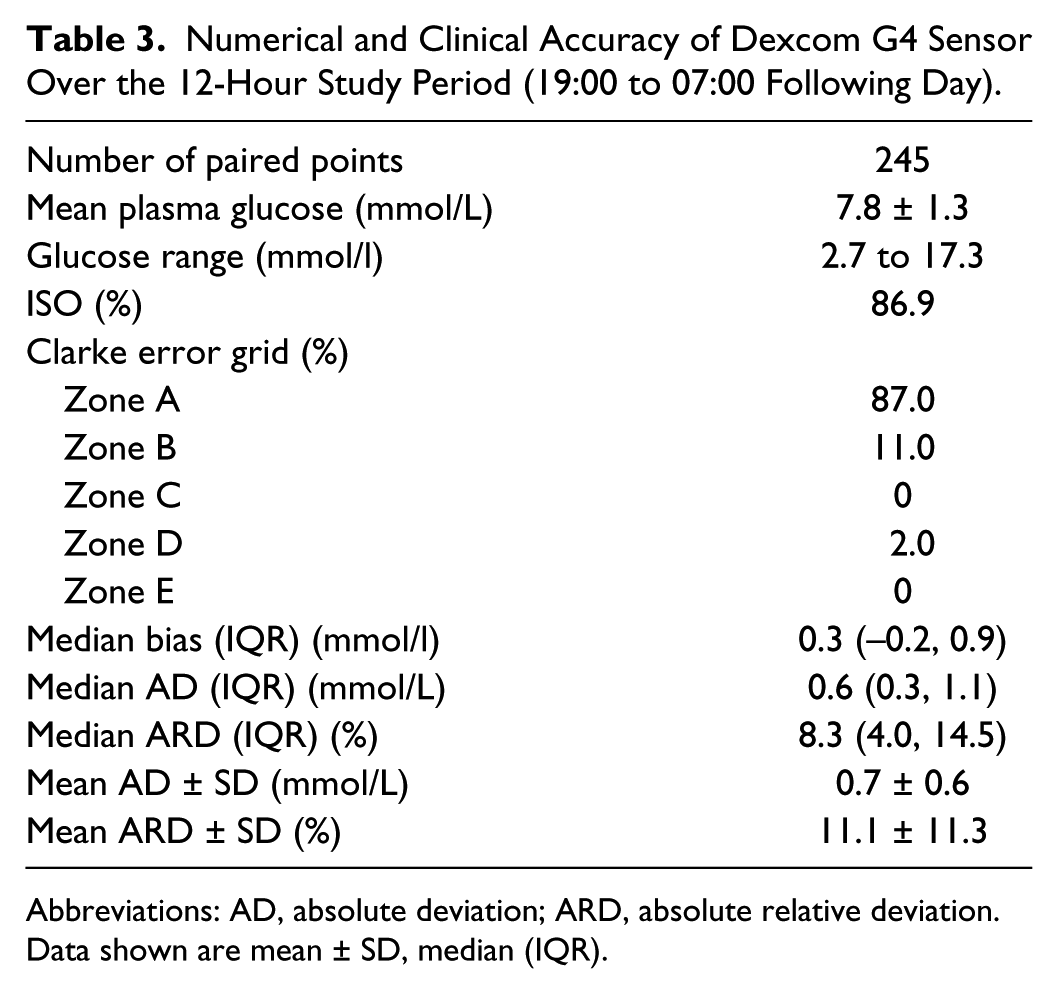
Abbreviations: AD, absolute deviation; ARD, absolute relative deviation. Data shown are mean ± SD, median (IQR).
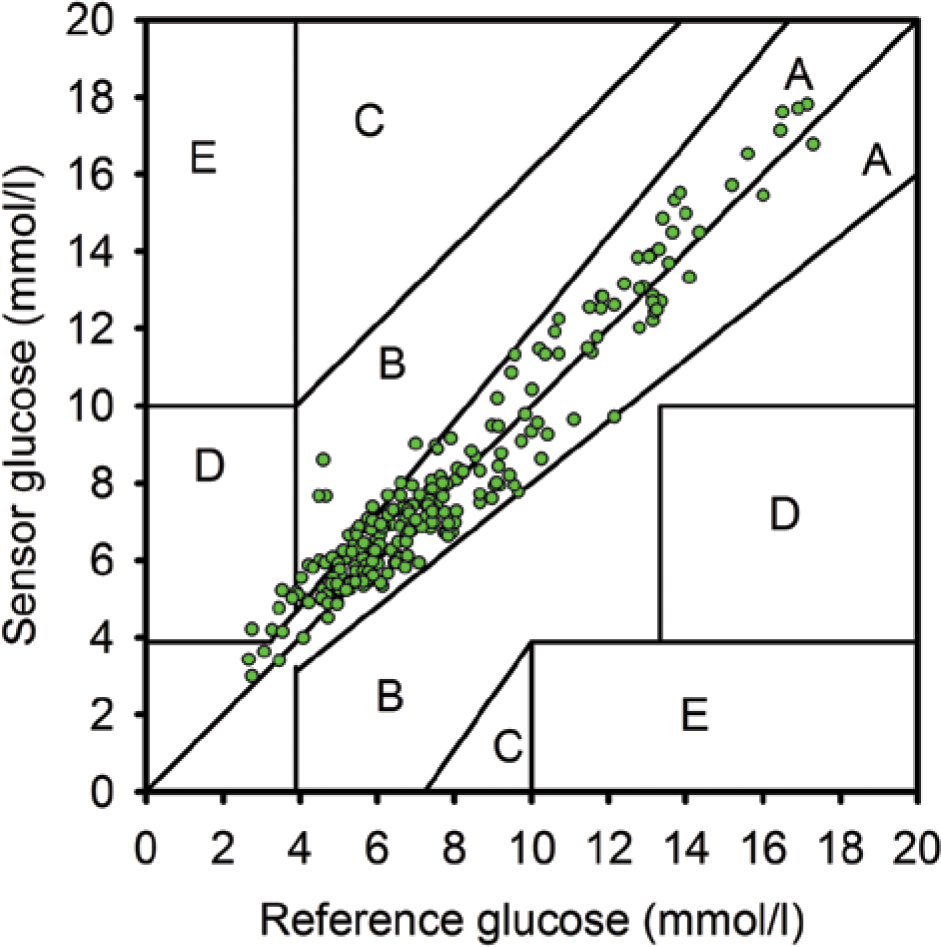
The Clarke error grid of sensor and reference glucose levels.
Discussion and Conclusions
Results of our study show that the first automated closed-loop system developed fully within the AP@home consortium is reliable. The time spent in target glucose level overnight was comparable to previous studies using Cambridge algorithm.4-6 In particular we observed a reliable system performance and no communication errors. We used a purpose built high performance APP capable of supporting a number of industry standard connections, allowing flexibility for connecting to different sensors and output devices. The platform contained an embedded code library, encapsulating the control algorithm, allowing its safe execution.
The timing of the 2 hypoglycemic events, 1 after 30 minutes and a second after 60 minutes of the evening meal bolus, despite complete cessation of basal insulin delivery by the algorithm suggests overaggressive insulin to carbohydrate ratio or the need for a dual phase bolus for the evening meal in these 2 participants. Paradoxically these 2 patients showed the lowest time spent in overnight target range between 3.9 and 8.0 mmol/l (5% and 25%) as both participants demonstrated rebound hyperglycemia after the correction of hypoglycemia.
Reliability and accuracy of the CGM device is critical for the safe and effective function of closed-loop systems. We used a single sensor that was inserted 48 to 72 hours before the experiments and calibrated according to manufacturer’s instructions with an additional calibration before the start of the experiment. All calibrations performed using finger-stick glucose. We observed a high degree of accuracy of the Dexcom G4 Platinum sensor which was noticeably better than our previously reported accuracy with Dexcom Seven Plus sensor (median ARD 8.3% vs 12.6%, G4 vs Seven Plus, Clarke error grid zone A 87% vs 71%). 21 We did not notice any large sensor overreading with the current sensor. We could only find 1 previous publication assessing the accuracy of the Dexcom G4 Platinum sensor showing an aggregate mean ARD of 13% 22 and 1 conference abstract with a mean ARD of 10.8%. 23 The Dexcom sensor was used in anterior abdominal wall, which is an approved location, and this study was not designed to assess sensor performance based on location.
Limitations of our study include the single-center, nonrandomized study design and small number of subjects. Since the efficacy of Cambridge MPC algorithm during overnight closed-loop insulin delivery has been previously demonstrated in randomized studies,4,5 we deliberately choose a nonrandomized design in keeping with our primary objective of assessing system reliability.
In conclusion, we observed a high degree of reliability of the automated embedded closed-loop system. Further system developments to miniaturize the system for home studies are warranted.
Footnotes
Acknowledgements
We thank study participants and staff at NIHR/Wellcome Trust Clinical Research Facility, Cambridge University Hospitals NHS Foundation Trust, Addenbrooke’s Hospital, Cambridge, UK.
We thank Dexcom Inc and Roche for support; these companies provided no funding and played no other role in clinical studies or data analysis.
Abbreviations
AD, absolute deviation; APP, artificial pancreas platform; ARD, absolute relative deviation; BMI, body mass index; CE, Conformité Européenne; CGM, continuous glucose monitoring; CHO, carbohydrate; CL, closed-loop; CSII, continuous subcutaneous insulin infusion; CV, coefficient of variation; IQR, interquartile range; ISO, International Organization for Standardization; LBGI, low blood glucose index; MARD, mean/median absolute relative deviation; MPC, model predictive control; SD, standard deviation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LL, HT, JMA, MN, and MEW have no conflicts of interest. KP and SL are employees of Triteq Ltd. MLE reports having received speaker honoraria from Abbott Diabetes Care and Animas and serving on advisory board for Medtronic, Roche, Cellnovo. RH reports having received speaker honoraria from Minimed Medtronic, Lifescan, Eli Lilly, and Novo Nordisk, serving on advisory panel for Animas and Minimed Medtronic, receiving license fees from BBraun, and having served as a consultant to BBraun, Profil, and Sanofi.
