Abstract
Objective:
The purpose of this study was to determine whether an approach of multisensor technology with integrated data analysis in an armband system (SenseWear® Pro Armband, SWA) can provide estimates of plasma glucose concentration in diabetes.
Research Design and Methods:
In all, 41 subjects with diabetes participated. On day 1 subjects underwent an oral glucose tolerance test (OGTT) and on day 2 a 60-minute treadmill test (TT). SWA plasma glucose estimates were compared against reference peripheral venous glucose concentrations. A continuous glucose monitoring device (CGM) was also placed on each subject to serve as a reference for clinical comparison. Pearson coefficient, Clarke error grid (CEG), and mean absolute relative difference (MARD) analyses were used to compare the performance of plasma glucose estimation.
Results:
There were significant correlations between plasma glucose concentrations estimated by the SWA and the reference plasma glucose concentration during the OGTT (
Conclusions:
Plasma glucose concentration estimation by the SWA’s noninvasive multisensor approach appears to be feasible and its performance in estimating glucose approaches that of a CGM. The success of this pilot study suggests that multisensor technology holds promising potential for the development of a wearable, noninvasive, painless glucose monitor.
Keywords
Adequate glycemic control in people with diabetes decreases the risk of developing both microvascular and macrovascular complications.1,2 To achieve adequate glycemic control, frequent self-monitoring of blood glucose (SMBG) is often necessary. The American Diabetes Association recommends that individuals with diabetes monitor their blood glucose as frequently as necessary to safely manage their condition, often as many as 4 to 6 times daily. 3 The current standard method for SMBG biochemically measures capillary glucose with a portable meter and requires lancing the skin to obtain blood. Although this method is often numerically and clinically accurate, it has shortcomings. Lancing the skin is often associated with pain, which discourages long-term compliance. Also, glucose meter technology relies on episodic testing and thus requires a motivated individual to remember to test several times a day. There are continuous glucose monitoring devices (CGM) available for continuous estimation of plasma glucose concentration, but these require insertion of a cannula into the skin and thus are still invasive. Clearly, there is a need for alternative device technologies, which ideally should be noninvasive, painless, and easy to use while providing reasonable estimations of plasma glucose concentrations to make informed decisions on diabetes self-management.4,5 Even when glucose estimates are not perfectly accurate, close estimates can be of clinical value in determining glucose trends that help inform self-management.
The BodyMedia SenseWear® Pro Armband (SWA; BodyMedia, Inc, Pittsburgh, PA) is a multisensor noninvasive device worn around the upper arm and employs multiple sensors to measure skin and ambient temperature, galvanic skin response, heat flux, and body motion. Mathematical algorithms integrate signals from those sensors as surrogate measures to predict physical activity levels and determine caloric expenditure in free-living individuals. In pilot studies, it was observed that there were correlations between plasma glucose concentration and integrated signals from skin temperature, heart rate, heart rate variability, and galvanic skin response. 6 These correlations led us to hypothesize that integrated multisensor technology signals could be employed to indirectly estimate plasma glucose concentrations. Plasma glucose has subtle influences on physical tissue properties such as conductivity, heat capacity, density, and impedance in several tissue compartments (eg, interstitium, blood, cells), making such a hypothesis plausible.7-12 Previously, the SWA utilized a physiologically inspired set of dynamic differential equations in a Wiener model to estimate glucose concentrations in a free-living environment. 13 Further mathematical refinement suggested that a better glucose-estimating model could be developed into the software without dynamic differential equations, applying instead context-dependent static regressions based on its multisensor data. Combining this refined model in the software with the multisensor technology in the SWA could create new opportunities to develop a wearable, noninvasive, and painless plasma glucose estimating system.
In this pilot study, we tested whether the multisensor approach with this refined model could provide estimates of circulating plasma glucose concentrations that would be at least as good as that of a CGM system. We examined its performance in 2 separate conditions: an oral glucose tolerance test (OGTT) to elicit rapid glucose excursions and a treadmill test (TT).
Methods
Human Subjects
The study was approved by both the Human Subjects Research Review Board of the US Army Medical Research and Material Command and the University of Pittsburgh’s Institutional Review Board. All participants gave informed consent. Exclusion criteria were debilitating arthritis, cardio-respiratory disease, skin edema or infection, pregnancy, implanted devices, hemoglobin < 9.0 g/L, recent diabetic ketoacidosis, or fasting glucose level > 300 mg/dL. Participants were 18 to 65 years old and had type 1 diabetes (T1DM; n = 18) or type 2 diabetes (T2DM; n = 23). The majority were women (32/41, 78%). Mean age was 42.1 ± 13.8 years. The average duration of diabetes was 22.7 ± 11.3 years in those with T1DM, and 7.1 ± 10.0 years in those with T2DM. Mean BMI was, respectively, 26.9 ± 7.6 kg/m2 and 36.2 ± 10.0 kg/m2 in T1DM and T2DM.
OGTT
Participants were studied after an 8-hour overnight fast. Eighty minutes prior to testing, participants were fitted with the SWA around the upper left arm and connected to an ECG electrode placed on the ipsilateral arm and shoulder to obtain precise measurements of heart rate and variability (Figure 1A). For SWA data downloads, CMS prerelease software version 1.0 was used. The first plasma glucose of the session was used for calibration of the devices. At the same time, a CGM was connected to the abdominal skin and calibrated according to manufacturer’s instructions (iPro, Medtronic, Northridge, CA). This was done 60 minutes prior to data recording to ensure proper calibration. For reference glucose concentrations, an IV catheter was placed into a warmed peripheral hand vein to obtain arterialized blood, from which plasma glucose was measured by an YSI 2300 STAT glucose analyzer (YSI, Yellow Springs, OH). Blood was sampled at −20, −10, −1, 15, 30, 60, 90, 120, 150, 180 minutes. At time 0, subjects with T2DM drank 75 g of glucose (Glucola), whereas 25 g was used for those with T1DM to prevent critically high hyperglycemia.
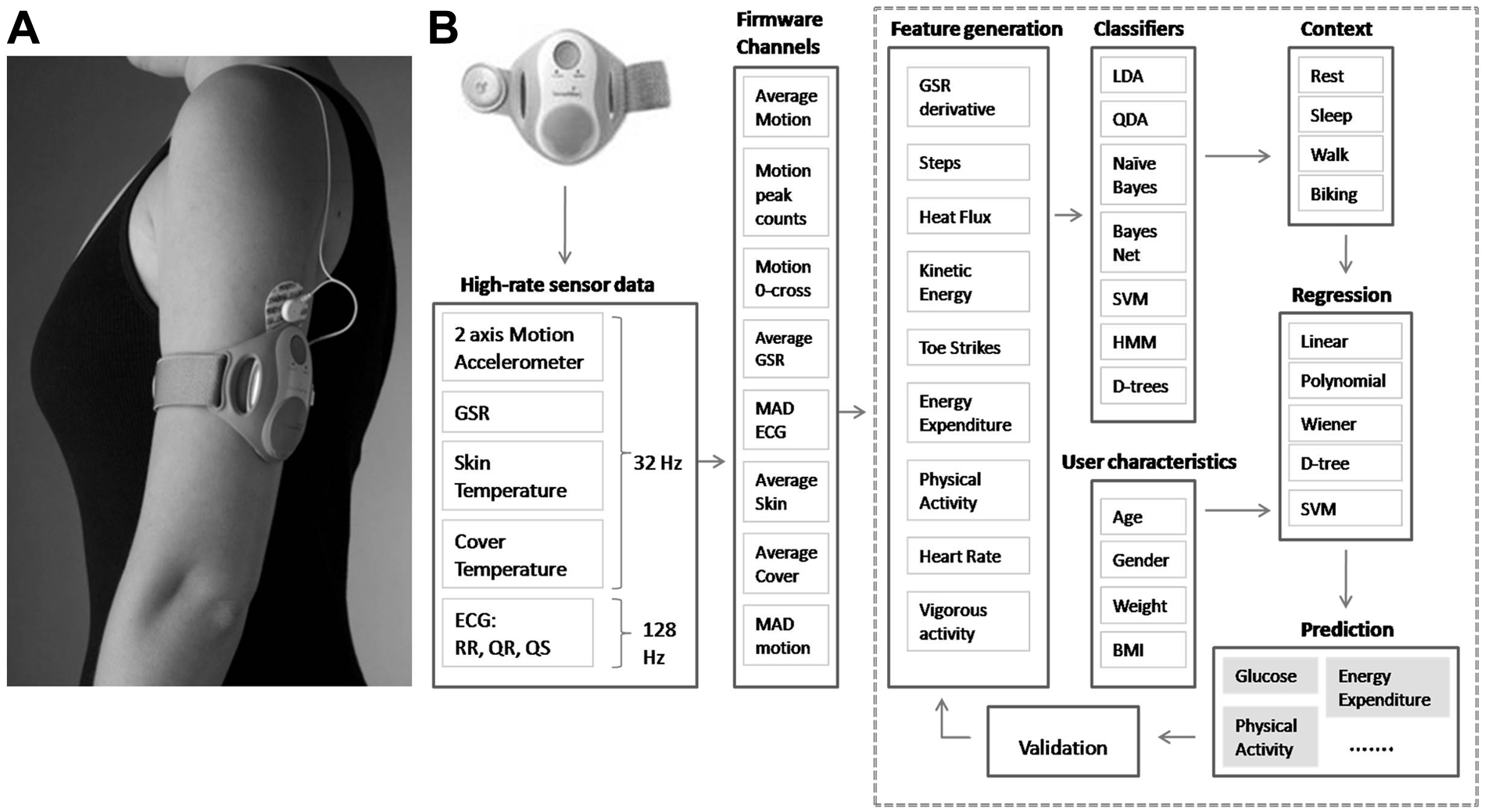
A: A representative image of the Senswear® Pro Armband worn on the upper arm during the study. B: An example of the SWA’s sensor set and model development procedure. The items listed after each subheading serve as examples of information used and are not an exhaustive list, except for the high-rate sensor data. The ECG sensors operate at 128 Hz and all other sensors at 32 Hz. The SWA’s firmware processor transforms the raw data collected from the channels and stores useful information at the rate of 1 sample per minute. The feature generation phase extracts other derived information from the data recorded by the firmware channels. The modeling procedure then identifies the activity context using the suite of classifier algorithms. Then the regression models are developed per each context from the suite of regression algorithms to provide the predicted output. All models go through rigorous validation procedures that include a by-subject cross-validation. This model development procedure is iterative and predicted outputs can be part of input features for new predictions in the next iteration. For example, energy expenditure is a predicted output using this procedure and then is used as an input for glucose estimation in the next iteration. Abbreviations: MAD: Mean Absolute Difference, LDA: Linear Discriminant Analysis, QDA: Quadratic Discriminant Analysis, SVM: Support Vector Machines, HMM: Hidden Markov Models, D-Trees: Decision Trees.
TT
Subjects returned on a separate day for the TT. For safety reasons, subjects were nonfasting. Placement and calibration of the SWA and CGM followed the same protocol used in the OGTT and IV access was established to obtain multiple reference glucose concentrations. At time 0, subjects were asked to walk on a treadmill at an initial pace of 1 mph, gradually increasing to 2.5 mph over the first 5 minutes and remained at this speed until completion (T = 60 min). During the TT, plasma glucose from the warmed peripheral IV line was measured every 5 minutes. Subjects were instructed to maintain natural arm motion during the test. To prevent hypoglycemia, participants on insulin were asked to reduce their doses before the tests; however, a few instances of hypoglycemia were still observed. In the few instances of hypoglycemia, the treadmill was stopped and oral carbohydrate in the form of a snack was given to raise plasma glucose concentrations. Plasma was still sampled at the prespecified time points and none of the hypoglycemic numbers were excluded from analysis.
Armband System
The SWA is worn around the upper arm and produces a plasma glucose estimation every minute, providing sequential glucoses over time. It contains 5 sensors: (1) a 2-axis accelerometer that tracks the movement and position of the upper arm and body, (2) a heat-flux sensor that measures the heat loss along a thermally conductive path between the skin and vent on the side of the SWA, (3) thermistors that measure skin temperature and near-armband temperature, (4) a galvanic skin response sensor that measures the conductivity of the subject’s skin, and (5) ECG electrodes worn on the arm and shoulder. 14 The SWA contains a proprietary algorithm to estimate heart rate from the ECG signals captured from the arm. To estimate plasma glucose concentration, supervised machine learning based algorithms similar to those applied in the SWA’s energy expenditure applications were utilized. 15 A brief overview of the supervised machine learning based model development procedure is provided in Figure 1B. For plasma glucose estimation, a “contextual-regression” strategy was employed that uses a classification algorithm to separate activity into context first and then applies an appropriate regression equation for that context. In this study, the classification algorithm disambiguated the exercise and nonexercise contexts. For example, the classification algorithm relies on motion parameters to disambiguate context between an OGTT and a TT. The regression model uses features derived from galvanic skin response, skin temperature, heart rate, heart rate variability, and motion. The features used in the regression model were automatically selected from a prepopulated pool of features using a forward search method. 16 Type 1 and type 2 regression models were trained separately, providing 4 separate batches of prepopulated features, varying for each combination of type of diabetes (type 1 and type 2) and session type (OGTT and TM). Once the features were decided as above, the parameter fitting part of the regression model was trained and evaluated using a k-fold (or by subject) cross-validation technique due to subject-specific traits present and the time-dependent nature of the data.
Once the features were decided as above, the parameter fitting part of the regression model was trained and evaluated using a k-fold (or by subject) cross-validation technique due to subject-specific traits present and the time-dependent nature of the data. In k-fold cross-validation, the data are partitioned into similarly sized k-folds and the technique “omits” data from the k-th fold and trains the algorithm on the rest of the data. The trained model is then used to test the data on the k-th fold. This procedure is repeated until each fold is “omitted” exactly once. This is a well-known procedure to estimate the performance of a machine-learning based method, as it applies an algorithm prospectively for each subject based on the other subjects and does not do a retrospective analysis where a given subject’s data are used to create that very algorithm that is used for them. The test results of the technique are a good estimate of the performance on unseen data. 17
Analyses
Correlation coefficients were calculated by Pearson analysis. To quantify numerical accuracy, we calculated the mean absolute relative difference (MARD).
18
To address clinical accuracy, we utilized Clarke error grid (CEG) analysis.
19
Two-tailed
Results
There were no instances of SWA malfunction; comparisons were not possible in 2 individuals due to poor IV access during OGTTs. With the CGM, there were instances of technical malfunction with data loss in 6 OGTTs and 11 TTs. The data points missing during these 17 tests were not factored into the accuracy estimations for the device.
Numerical Accuracy
Table 1 shows the numerical accuracy of the SWA and CGM each as compared to the reference during rapid glycemic excursions induced by the OGTT. There was a statistically significant correlation between the values obtained from the SWA and the reference plasma glucose concentration (
Numerical Accuracy of the SWA and CGM Against Plasma Glucose (Reference).
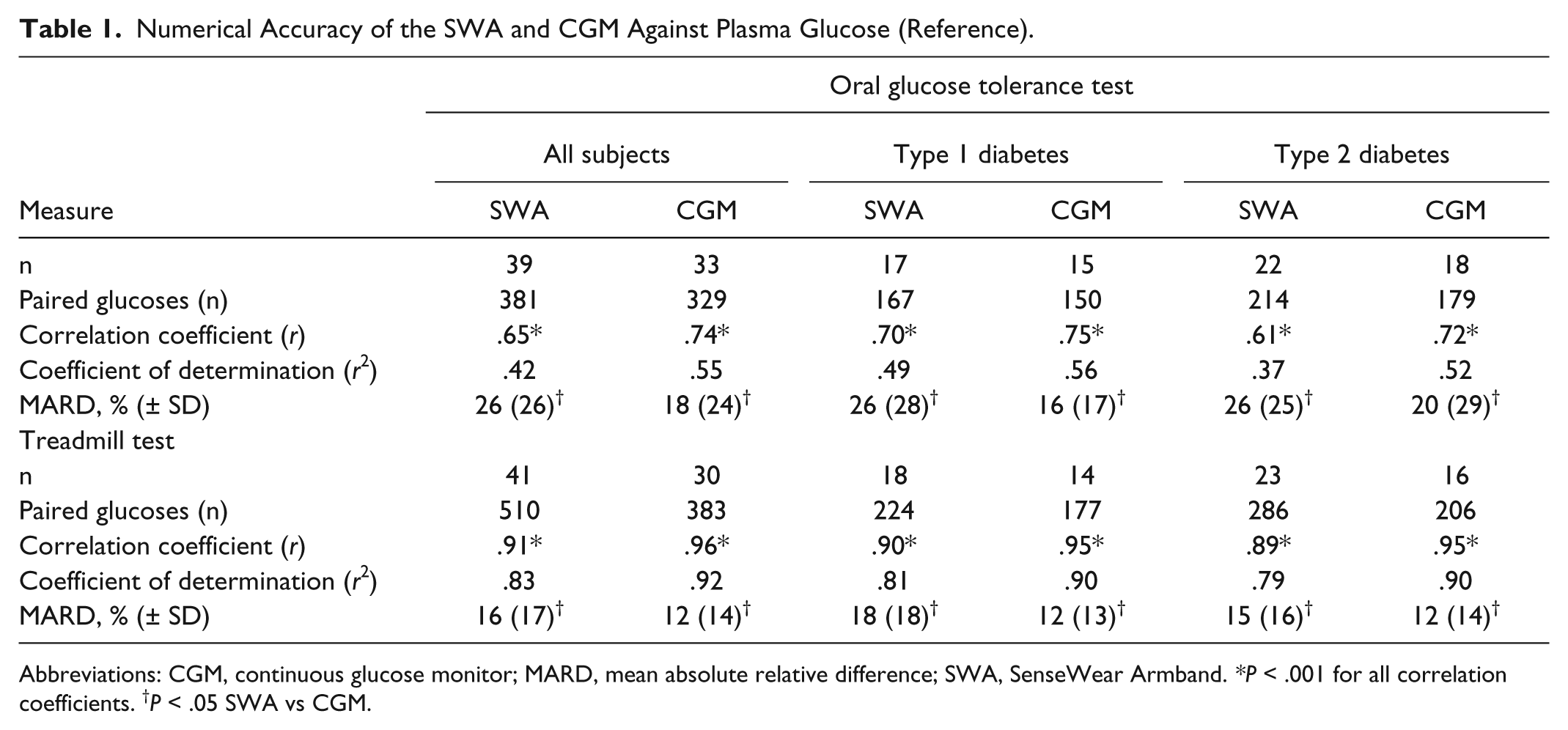
Abbreviations: CGM, continuous glucose monitor; MARD, mean absolute relative difference; SWA, SenseWear Armband.
During the TT, plasma glucose concentration estimates from both devices were highly correlated with the reference plasma glucose concentration (SWA:
Tracking of Glucose Over Time
Figure 2 shows representative individual glucose curves for each subject over time for the OGTT (Figure 2A) and TT (Figure 2B). In addition, the average of all glucose estimates made by the SWA and CGM tracked with reference plasma glucose concentration over time during the OGTTs and TTs is included as the final plot in each figure, respectively. On average, both devices tracked the upward and downward phases of the reference plasma glucose excursions. However, in certain individuals, the SWA or the CGM estimates had large deviations from the reference plasma glucose measure.
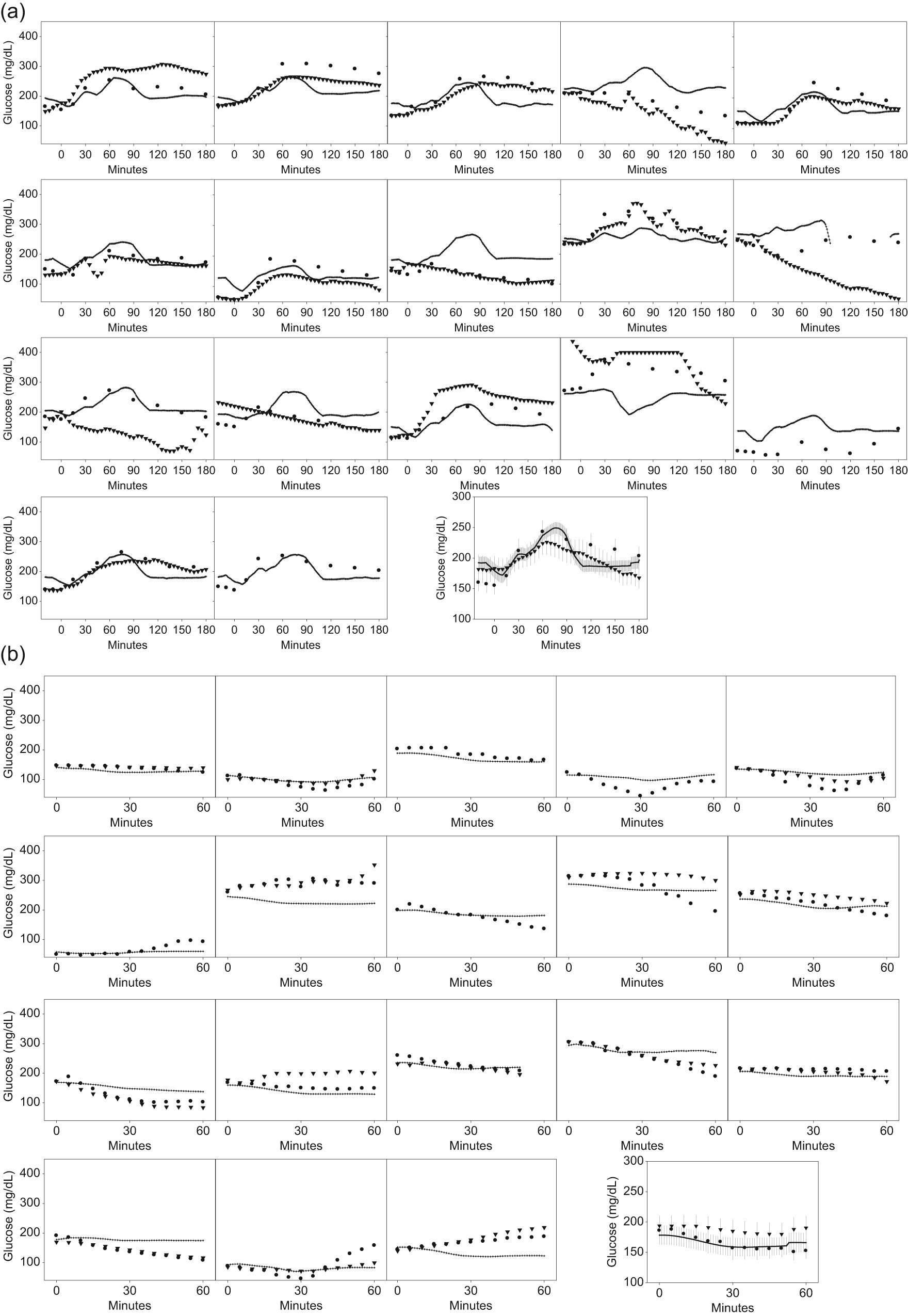
(A) Individual plots of glucose estimates during OGTTs. Reference glucose values (circles), CGM (triangles) and SWA estimates (continuous lines) are plotted for 17 OGTTs. The last figure shows the mean ± SE of all curves combined. Due to space limitations, only data for subjects with type 1 diabetes are presented. (B) Individuals plots of glucose estimates during treadmill tests. Reference glucose values (circles), CGM (triangles), and SWA estimates (continuous lines) are plotted for 18 tests. The last figure shows the mean ± SE of all curves combined. Due to space limitations, only data for subjects with type 1 diabetes are presented.
Clinical Accuracy by CEG Analysis
During the OGTT, the SWA recorded a total of 381 plasma glucose estimates, compared to 329 plasma glucose estimates recorded by the CGM. The SWA (Figure 3A) and the CGM (Figure 3B) both displayed a high percentage of plasma glucose values in the clinically acceptable zone A+B (SWA: 93%, CGM: 95%,
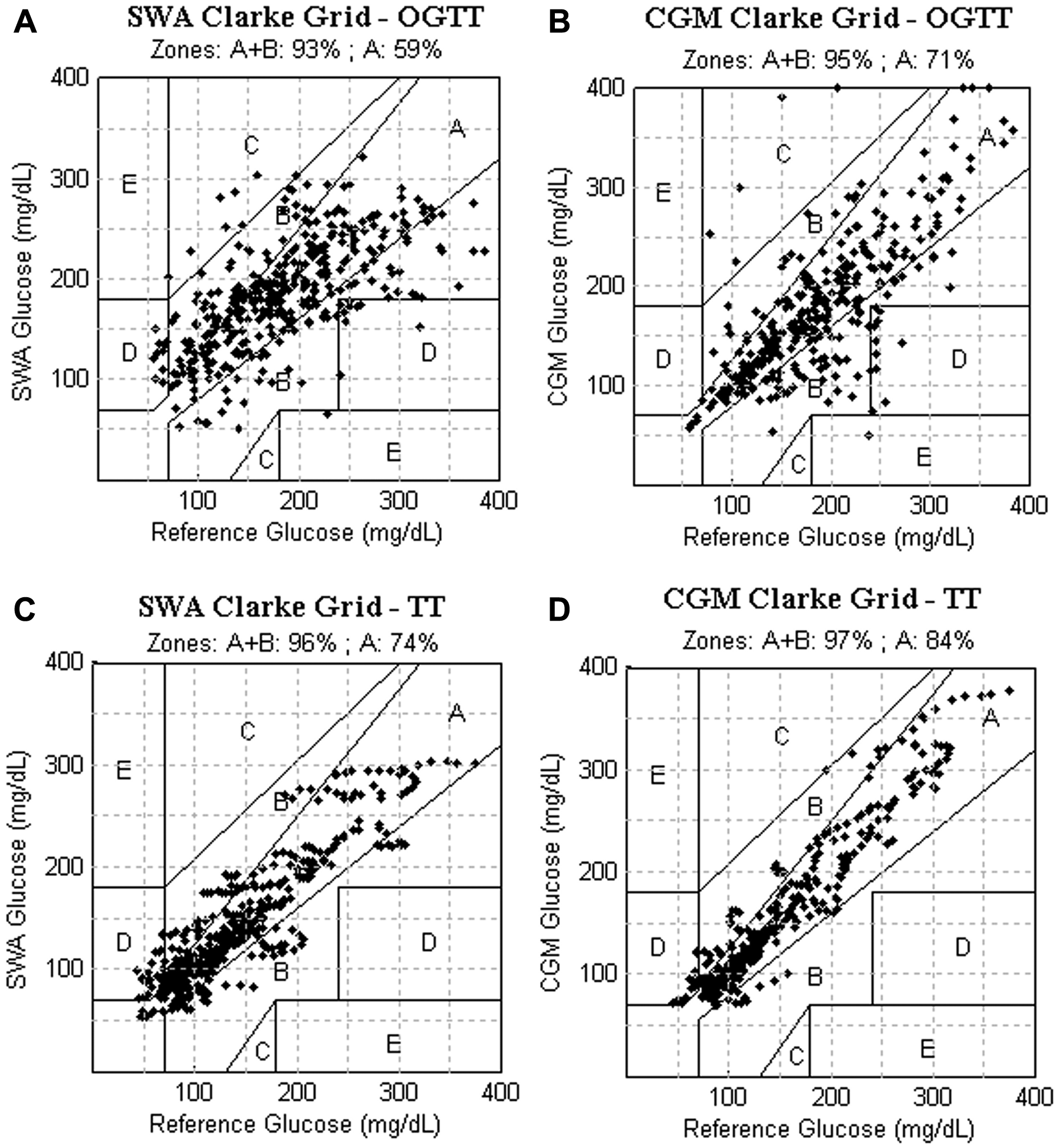
Clinical accuracy examined by Clarke error grid analysis: Glucose estimates were plotted against reference venous plasma glucose measured by an automated glucose meter (YSI 2300 STAT). Panel A: SWA vs reference during OGTT. Panel B: CGM vs. reference during OGTT. Panel C: SWA vs. reference during treadmill tests (TT). Panel D: CGM vs. reference during TT. For each grid, the percentage of clinically acceptable estimates (zones A and B) is shown at the top.
During the TTs, the SWA recorded a total of 509 plasma glucose estimates and the CGM recorded a total of 382 plasma glucose estimates. Of SWA plasma glucose estimations 96% fell in zone A+B, which was nearly indistinguishable from the 97% accuracy obtained by the CGM (
When all OGTT and TT data were combined, the percentage of points in zones A+B was again statistically indistinguishable between the SWA and the CGM (95% vs 96%, respectively,
Figure 3. Clinical accuracy examined by Clarke error grid analysis: Glucose estimates were plotted against reference venous plasma glucose measured by an automated glucose meter (YSI 2300 STAT). Panel A: SWA vs reference during OGTT. Panel B: CGM vs. reference during OGTT. Panel C: SWA vs. reference during treadmill tests (TT). Panel D: CGM vs. reference during TT. For each grid, the percentage of clinically acceptable estimates (zones A and B) is shown at the top.
Safety
No patient reported pain associated with the use of the SWA. Despite medication adjustments in preparation for the studies, readings under 70 mg/dl were observed. However, no patients were symptomatic. There were a total of 45 reference hypoglycemic readings among 10 different subjects, 6 of whom had T1DM. Of the 45 hypoglycemic readings, 11 were recorded during the OGTT. Two subjects arrived hypoglycemic and remained so for the first several measurements of the test; this was thought to be due to inadequate insulin dose adjustment while fasting. The SWA did not provide any clinically acceptable estimates of these OGTT hypoglycemic points (zone A or B), 91% of the SWA estimates were in zone D and 9% in zone E. The CGM estimated 36% of the hypoglycemic points in the clinically acceptable range (zone A), 0.3% were in zone D, whereas no estimates were recorded for the remaining 54% of the reference points. The remaining 34 reference hypoglycemic measurements were recorded during the TT. The SWA provided correct estimates in 26% of these instances (zone A) and incorrect estimates in 74% (zone D). The CGM provided correct estimates of hypoglycemia in 0% of these instances (zone A), incorrect estimates in 32% (zone D) and did not provide estimates in 68%. Overall, out of 45 hypoglycemic readings, the SWA correctly estimated hypoglycemia in 20% (9/45). Appropriate detection of hypoglycemic readings by the CGM was lower at 8.9% (4/45). Failure to detect occurred due to incorrect glucose estimation (12/45) or unrecorded signals (29/45).
Discussion
There continues to be an unmet need for accurate noninvasive plasma glucose monitoring. The current SMBG standard of care remains invasive, causes pain, and is associated with poor rates of compliance. CGM devices that measure interstitial glucose in the skin have been developed but remain invasive since they require introduction of a cannula into the skin and are complex to operate. They are also more expensive than portable glucose meters. Therefore, new solutions for SMBG are still needed. Ideally, alternative technologies should be painless and easy to operate while at the same time providing reasonably accurate estimations of plasma glucose concentration that are sufficient for clinical decision making.
Noninvasive glucose monitoring systems are still in an early phase of development, but they offer the potential to become viable adjuncts in SMBG in the future. One approach to noninvasive plasma glucose estimation is to combine multisensor technologies with integrated data analysis that algorithmically derives an estimation of plasma glucose concentration from the physiological signal outputs of those sensors. Other multisensor approaches and models have been tested and suggest that accurate plasma glucose estimation may eventually become practical.20-23 A concern about noninvasive glucose monitoring devices is demonstration of clinically acceptable accuracy since no direct measurement of plasma glucose concentration is performed. Even CGM devices, which estimate plasma glucose concentrations indirectly from interstitial glucose measurements, currently lack the precision necessary to fully replace standard enzyme-based portable glucose meters. In this study, we set out to determine the potential of a multisensor and integrated analysis approach to estimate plasma glucose concentration accurately using the SWA. It is important to note, however, that there are several methods by which technical and numerical accuracy can be measured and no single metric may fully reflect the practical value of a device. In fact, many authors have identified the need to establish accuracy standards applicable across devices.5,18 Furthermore, deciding on cutoff points of minimally acceptable accuracy is potentially fraught with subjectivity. For these reasons, to provide a reference for interpretation of the accuracy of the SWA, estimates were compared to readings obtained by a clinically approved CGM device. We found that SWA plasma glucose concentration estimates correlated with the venous glucose, and correlations were nearly identical to those of the CGM. The MARD was better for the CGM than for the SWA. However, the SWA’s MARD approached that of the CGM, suggesting that this performance gap could be plausibly closed in future development. In some subjects the SWA estimates deviated from the reference. Although this limitation will need to be addressed and resolved for all individuals, it is a limitation that was observed for both the SWA and CGM (Figures 2A and 2B).
While numerical accuracy quantifies differences in blood glucose estimates from a statistical standpoint, they do not take into account the clinical significance of those differences. CEG analysis, however, provides a method to quantify the performance, taking into account the clinical relevance of those differences. 19 In the CEGs, the percentage of plasma glucose estimates in the clinically acceptable zone A+B was similar between the SWA and the CGM. Although a greater proportion of the plasma glucose values estimated by the SWA made up zone B to achieve a high percentage of plasma glucose values in zones A+B, according to the interpretation of CEGs, this would indicate a benign error not impacting clinical decision making. Considering this was a pilot study, the similarity in clinical accuracy between the SWA and CGM suggests that the SWA’s approach to monitor plasma glucose concentration is rather promising.
Conditions of dynamic plasma glucose concentration fluctuations have been shown to negatively influence CGM systems. 24 One of the strengths of our study was the fact that we tested performance in nonstatic conditions that mimic more realistic usage scenarios. Few studies of noninvasive glucose monitors have included interventions given the concern that background noise could be introduced, potentially blunting the signals picked up by the sensors and thereby affecting the quality of the plasma glucose concentration estimates. We employed a more rigorous approach. In our study, OGTTs were chosen to predictably elicit dynamic changes in plasma glucose levels. TTs were chosen to mimic light-to-moderate physical activity with arm motion. This is not a trivial issue since other noninvasive experimental sensors have been shown to be negatively affected by motion artifact and body temperature.12,25 We found that even in those dynamic conditions the SWA estimations performed fairly well. However, the correlation coefficient and MARDs were noted to be better during TTs than the OGTTs, suggesting that the performance of both devices is less optimal during rapid plasma glucose excursions.
Comparing the SWA’s accuracy against other noninvasive or minimally invasive devices would be difficult for several reasons. For one, the metric of accuracy can be reported in a variety of ways. In addition, some accuracy estimates are based off of studies done in a small or distinct population of subjects (healthy subjects or subjects with only T1DM). Often, plasma glucose is maintained within a narrow range with euglycemic clamps, or hypoglycemic readings are excluded from analysis.12,22-27 A final issue is the fact that the reference plasma glucose chosen varies among studies: sometimes plasma glucose is used24,28-31 and at other times fingertip capillary blood glucose is measured by portable meters, devices that we know are subject to their own error.18,32-34 Strengths in our study include that subjects with both T1DM and T2DM were tested, dynamic conditions were used to elicit glucose fluctuations, plasma glucose was used as a reference, and we conducted a simultaneous comparison of the SWA’s relative performance to that of a CGM. Cognizant of the challenges in comparing accuracies across studies, the SWA’s combined MARD of 20% is in fact in line with studies that examined the numerical accuracy of present-day minimally invasive CGM devices using plasma glucose concentration as the reference. In those studies, the MARD reported was in the 15% to 20% range.24,28-31 When compared in terms of CEG-based clinical accuracy, the performance of the SWA (95% of estimates in zones A+B) is in line with that of CGM devices, in which it generally ranges from 93 to 98%.18,24,28-31,33
One limitation of the SWA’s design is that it requires 2 electrodes to measure heart rate and obtain heart rate variability measures. There are clear limitations to the practicality of this design. However, a newer version of the SWA armband where a dry electrode is attached to the strap of the armband is in an advanced stage of development and should overcome that limitation. Nevertheless, because heart rate measurements are required, we suspect that the SWA will remain incompatible with patients on beta-blockers or those with arrhythmias for a foreseeable future. In addition, the SWA’s approach should be tested in free-living conditions and in studies specifically designed to test accuracy during hypoglycemia. Our study was not designed for hypoglycemia, but the fortuitous occurrence of hypoglycemic readings in this study provided some preliminary data. There were too few hypoglycemic readings, but one could infer that the SWA’s performance during hypoglycemia was not satisfactory. However, the performance of CGM was not satisfactory either. This illustrates the ongoing challenge of clinically reliable detection of hypoglycemia by indirect means of minimally or noninvasive devices. 35
Further research is needed to meet the stringent requirements of a wearable, noninvasive, and accurate glucose monitor. This may be addressed by the addition of more sensors or the development of improved algorithms that interpret the physiologic inputs. The present learned models employed by the SWA could be overfit, and this requires additional rigorous testing to further address the potential of developing a clinically accurate noninvasive glucose monitor. With the present results of this pilot study, it is clear that the SWA is not ready for clinical use; however, it provides encouraging data on the potential of development of a multisensor, noninvasive glucose prediction system.
Conclusions
The SWA’s performance in this pilot study suggests that multisensor technology holds promising potential for the development of a wearable, noninvasive, painless glucose monitor.
Footnotes
Acknowledgements
The authors would like to express their gratitude to the nurses and laboratory staff at the University of Pittsburgh Clinical and Translational Research Center for careful execution of the experimental protocol. The authors would also like to acknowledge Rachel Jackson, John Stivoric, and Kara Hawkins for assisting in the implementation of the study.
Abbreviations
ADA, American Diabetes Association; CEG, Clarke error grid; CGM, continuous glucose monitor; D-Trees, decision trees; ECG, electrocardiogram; HMM, hidden Markov models; MAD, mean absolute difference; MARD, mean absolute relative difference; OGTT, oral glucose tolerance test; SMBG, self-monitoring of blood glucose; SWA, SenseWear armband; TT, treadmill test; YSI, Yellow Springs Instruments.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NV and DA were consultants for BodyMedia during the study. FGST received research grant support from BodyMedia in connection with the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This was an investigator-initiated study supported by an unrestricted research grant from BodyMedia, Inc through the US Army Medical Research and Material Command. The study took place at the University of Pittsburgh Clinical and Translational Research Center (NIH grant UL1 RR024153).
