Abstract
Background:
Fluorescent glucose-sensitive nanosensors have previously been used
Methods:
Glucose-sensitive nanosensors were encapsulated in two different commercially available gelling agents: gel 1 and gel 2. Multiple formulations of each gel were assessed
Results:
Five gel formulations had encapsulation efficiencies of the nanosensors greater than 90%. Additionally, they retained up to 20% and 40% of the nanosensors over 24 h for gel 1 and gel 2, respectively.
Conclusions:
Encapsulating glucose nanosensors in two injectable gels prolonged nanosensor lifetime
Introduction
The worldwide prevalence of diabetes has spurred interest in continuous glucose monitoring systems as an alternative to the finger-prick method. Glucose monitors such as DexCom™ STS™ Continuous Glucose Monitoring System and Medtronic's Guardian® REAL-Time Continuous Glucose Monitoring System are commercially available and approved by the Food and Drug Administration to track trends in glucose levels.1,2 However, further research into novel approaches for glucose monitoring is still of interest in order to prolong sensor lifetimes, improve accuracy, and minimize invasiveness of measurements. Several reviews, such as those written by Wang, 3 Pickup and coauthors, 4 Steiner and coauthors, 5 and Cash and Clark, 6 cover the scope of these developments that include the extension of nanotechnology to glucose sensing. For example, glucose microsensors and nanosensors provide the benefits of rapid response times and ease of implantation due to their large surface-area-to-volume ratio and small size. 6 Chaudhary and coauthors 7 are developing a “smart tattoo” composed of dissolved-core alginate microspheres to be implanted into the skin and monitor glucose levels in interstitial fluid. These sensors use fluorescence resonance energy transfer and a competitive binding mechanism with a noncatalytic mutant of glucose oxidase 8 to repeatedly monitor reversible changes in glucose at physiological levels. In other work, nanosensors using boronic acids as a nonbiological recognition element for glucose have also been investigated because of boronic acids' affinity for 1,2 diols. 5 A common sensing mechanism involving boronic acids is the competitive binding between a fluorescent reporter and glucose for the boronic acid binding site. The fluorescence of the reporter is different in the bound and unbound states, yielding a change in measured signal as the reporter is displaced from the boronic acid. Wang and coauthors 9 developed glucose sensing vesicles using this mechanism with phenylboronic acid, glucose, and the fluorescent reporter alizarin red S (ARS). 9 Alizarin red S was electrostatically coupled to cationic quaternary ammonium salts that self-assembled into vesicles in solution. As glucose was added to the system, ARS was displaced by glucose, which resulted in a decrease in intensity, because the unbound ARS is significantly less fluorescent. Our group utilizes a similar competitive binding scheme as the basis for functional nanosensors, but the sensing components are embedded in a lipophilic, highly plasticized polymeric particle into which glucose is extracted.10,11 The hydrophobic particle design has several advantages that include isolation of the sensing components from biological fluid to prevent biofouling 12 and tunability of the system to adjust the dynamic range. 13
Our glucose-sensitive nanosensors successfully tracked changes in glucose levels when injected subcutaneously along the backs of mice.
10
However, the monitoring time was limited to 1 h because of the loss of signal intensity at the injection site. Studies conducted by Gopee and coauthors
14
with intradermally injected quantum dots found that quantum dots migrated from the site of injection, with 60% of quantum dots remaining at the injection site after 24 h. Our nanosensors were implanted similarly in the skin, and migration was assumed to be the main cause of signal loss over time. To mitigate sensor migration in our system, sensor geometry was altered into microworms that limited sensor diffusion and prolonged their lifetime in the skin more than the nanosensors alone.
15
However, the yield of microworms was not sufficient for
Materials
Matrigel™ basement membrane matrix (gel 1; growth factor reduced, phenol-red-free, lactate-dehydrogenase-elevating-virus-free) and 31 G insulin syringes were purchased from BD Biosciences (Franklin Lakes, NJ). Extracel-X® hydrogel kit (gel 2) was purchased from Glycosan Biosystems Inc. (Alameda, CA). Bis(2-ethylhexyl) sebacate (DOS), tridodecylmethylammonium chloride (TDMAC), alizarin (dye content 97%), tetrahydrofuran (THF; anhydrous, ≥99.9%, inhibitor-free), and chloroform (Chromaslov®, ≥99.8%) were purchased from Sigma-Aldrich (St. Louis, MO).
Octylboronic acid was purchased from Synthonix Inc. (Wake Forest, NC). Gibco® phosphate-buffered saline (PBS) (1x, pH = 7.4) was acquired from Life Technologies (Grand Island, NY) and poly(vinyl chloride) carboxylated (PVC-COOH) was purchased from Scientific Polymer Products Inc. (Ontario, NY). Amicon Ultra centrifugal filters (molecular weight cutoff 100 kDa) were obtained from Millipore (Billerica, MA). 1,2-distearoyl-
Methods
Optode Cocktail
Macrosensors and nanosensors were fabricated from an optode cocktail that contained all sensing components: 30 mg of PVC-COOH, 60 μl of DOS, 3 mg of octylboronic acid, 4 mg TDMAC, and 1 mg of alizarin, all dissolved in 500 μl of THF. The selection, optimization, and characterization of these components for glucose sensing are described elsewhere.10,11
Nanosensor Fabrication
Fabrication of glucose nanosensors have been described previously. 10 Briefly, the optode cocktail was dried for at least 4 h on a glass plate. It was then removed from the plate and placed into a scintillation vial along with 5 ml of PBS, 5 mg of PEG 550, and 500 μl of total chloroform. The mixture was sonicated using a Branson digital sonifier (Danbury, CT) for 3 min at 40% amplitude. The nanosensors were concentrated using Amicon Ultra centrifugal filters prior to encapsulation in the gelling agents.
Gel Preparation
Two commercially available gels were selected for encapsulating the nanosensors. Each gel has applications in cell encapsulation and tumor growth models.17,18 Gels were prepared according to the manufacturer's instructions and diluted with nanosensors and PBS according to the ratios in
Nanosensor Encapsulation Efficiency
Prepared gel (100 μl) with sensors or the no-gel control was injected into a 96-well optical bottom well plate. Fluorescence measurements were acquired on a SpectraMax Gemini EM plate reader (Molecular Devices, Sunnyvale, CA) at 460 and 570 nm for excitation and emission wavelengths, respectively. After the initial reading, the gels were washed with 100 μl of PBS, the PBS was removed, and then a second measurement was acquired. The encapsulation efficiency was calculated as the fluorescence intensity of the after wash measurement divided by the initial measurement for each sample. This ratio was then expressed as a percentage.
Glucose Permeability through Gels
Glucose macrosensors were used to test glucose permeability because they can be adhered to a surface, eliminating the possibility of unencapsulated nanosensors contributing to sensor response. The macrosensors are formed by pipetting the optode cocktail (2 μl) onto glass discs adhered to the bottom of a 96-well optical bottom well plate and then dried. A total of 200 μl of PBS was added to each well, and the sensors were hydrated in PBS for at least 4 h until their fluorescence intensity stabilized. All fluorescence measurements were acquired at 460 and 570 nm for excitation and emission wavelengths, respectively, using a SpectraMax Gemini EM plate reader. After hydration, 10 0 μl of gels were injected over the macrosensors and adjusted until they covered the entire bottom of the well plate. Gels were prepared according to the ratios in
Gel Retention of Nanosensors
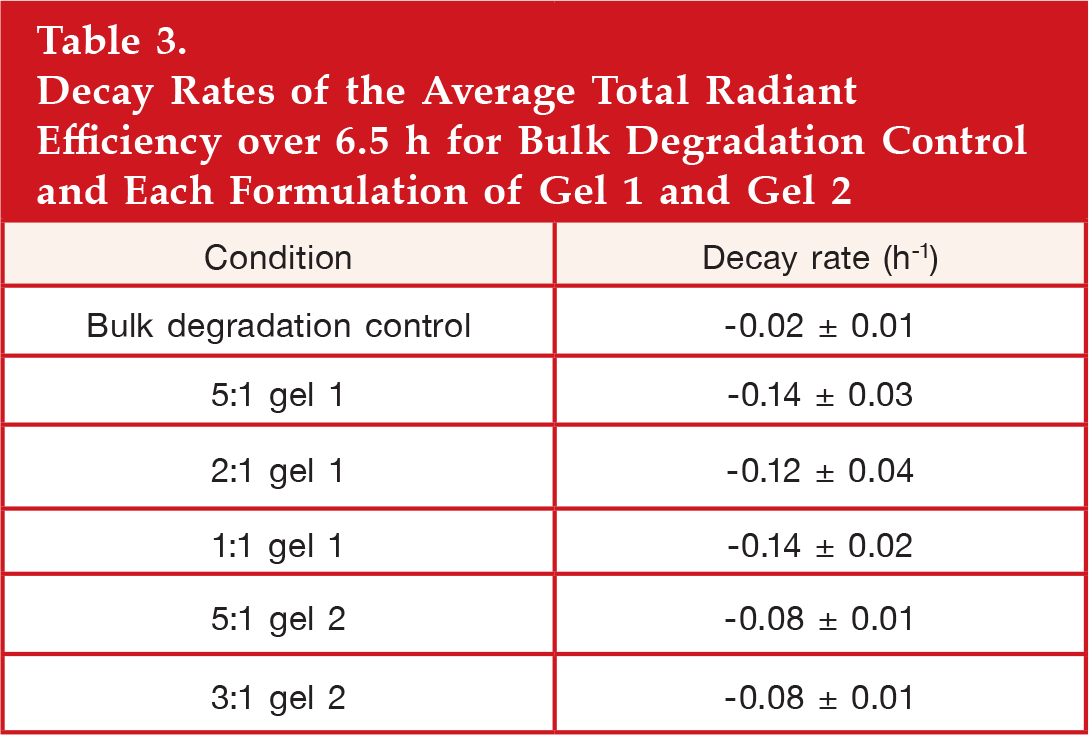
Glucose nanosensors were encapsulated in gels as described in the gel preparation section. A total of 40 μl of gel was injected onto a glass bottom petri dish that was then filled with 4 ml of PBS. The gels were placed in an incubator at 37 °C for 10 min and then imaged using an IVIS Lumina II small animal imager (Caliper Life Sciences, Hopkinton, MA). Images were acquired with 465 nm excitation and 580 nm emission filters. They were recorded approximately every half hour for the first 2 h and then at 6.5 h and approximately 24 h. Between measurements, the gels were stored in an incubator at 37 °C. The bulk degradation of the nanosensors (sensor degradation, component leaching, photobleaching) over this time was also monitored to separate these effects from the loss of intensity caused by diffusion. For the bulk degradation controls, 40 μl of glucose nanosensors in PBS was placed in microcentrifuge tubes and imaged at the same time points as mentioned earlier. In between measurements, the nanosensors were stored in an incubator at 37 °C. For analysis, the total radiant efficiency from a region of interest encompassing either the gel or the nanosensors for the bulk degradation control was selected, normalized to the time 0 measurement, and plotted over time to determine the rate of nanosensor diffusion out of the gel.
In Vivo Testing
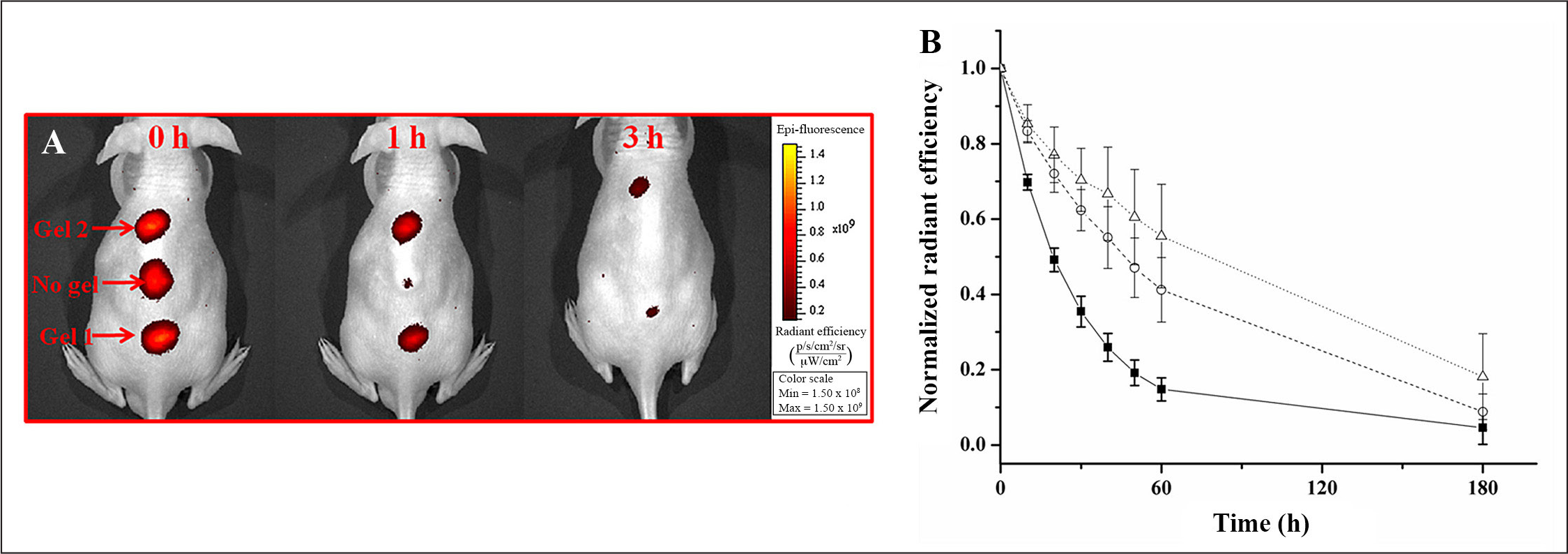
All animal procedures were approved by Northeastern University's Institutional Animal Care and Use Committee. Glucose nanosensors were encapsulated in gels according to the protocol in the gel preparation subsection or diluted with PBS for the no-gel control. SKH1-E mice were anesthetized, and 40 μl of each gel with nanosensors and a no-gel control were injected intradermally along their backs. Intradermal injections were achieved by pinching the skin and then injecting nanosensors with or without gel while the syringe was inserted parallel to the skin. Characteristic of intradermal injections, a bleb or small skin welt was visible after injection. 20 Mice were imaged with an IVIS Lumina II small animal imager with excitation and emission filters at 465 and 580 nm, respectively. Images were acquired for the first 60 min at 10 min intervals, and an additional measurement was taken 3 h post-injection. Similar to the gel retention experiments, the total radiant efficiency from a region of interest surrounding each injection spot was selected for analysis. An additional region of interest with skin only was also selected to determine the background skin fluorescence. The background fluorescence from the skin was subtracted from the region of interest, and the data were normalized to time point 0.
Results
In Vitro Characterization
Both gelling systems were assessed for three critical characteristics: encapsulation efficiency, glucose permeability into the gel, and sensor retention over time. Gel formulations were down-selected based on these
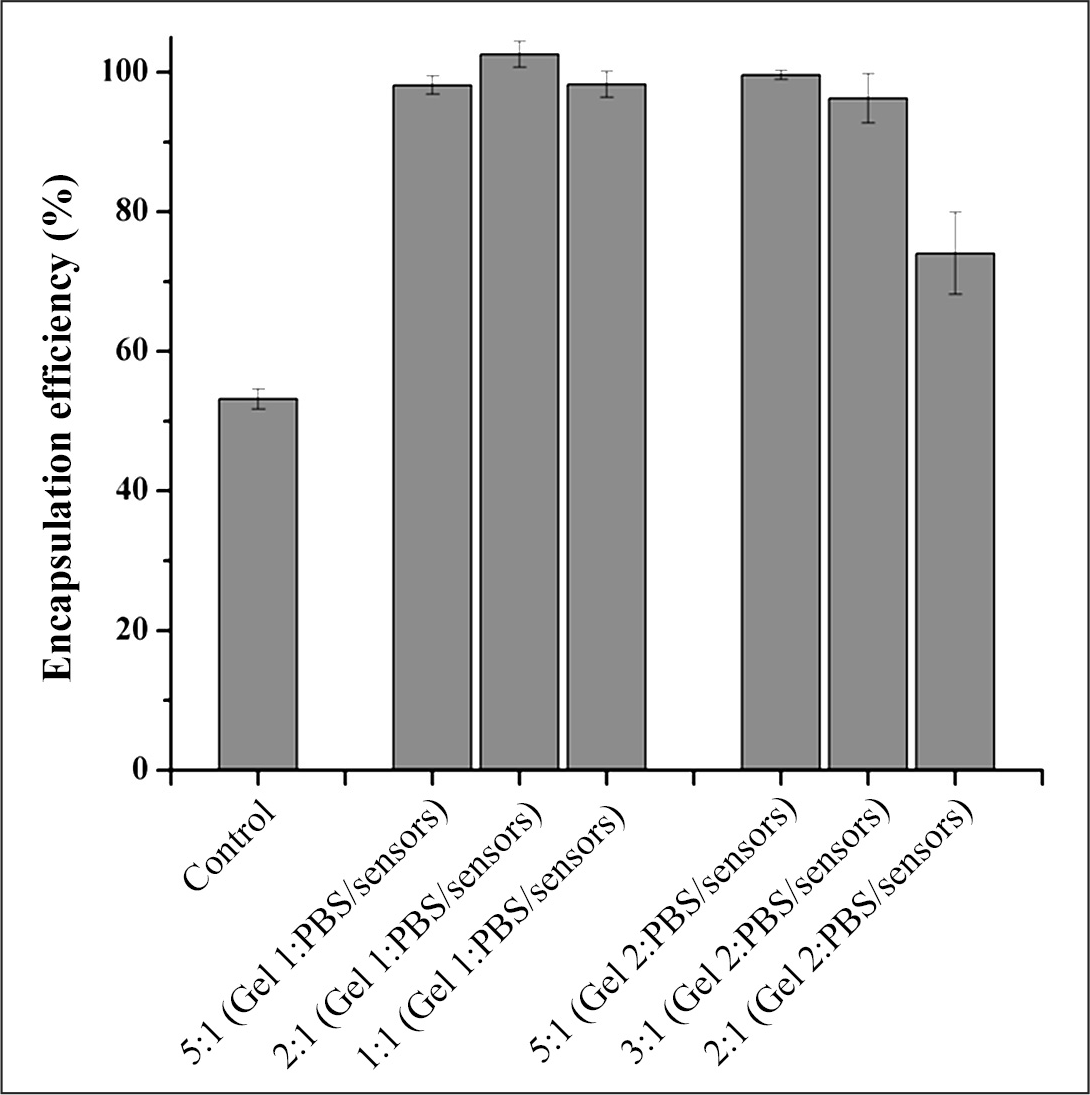
Encapsulation efficiency of gels. Glucose nanosensors were mixed with gelling agents with variable stiffness and allowed to gel. Shown here is the percentage change in fluorescence intensity after the gels were washed with PBS.
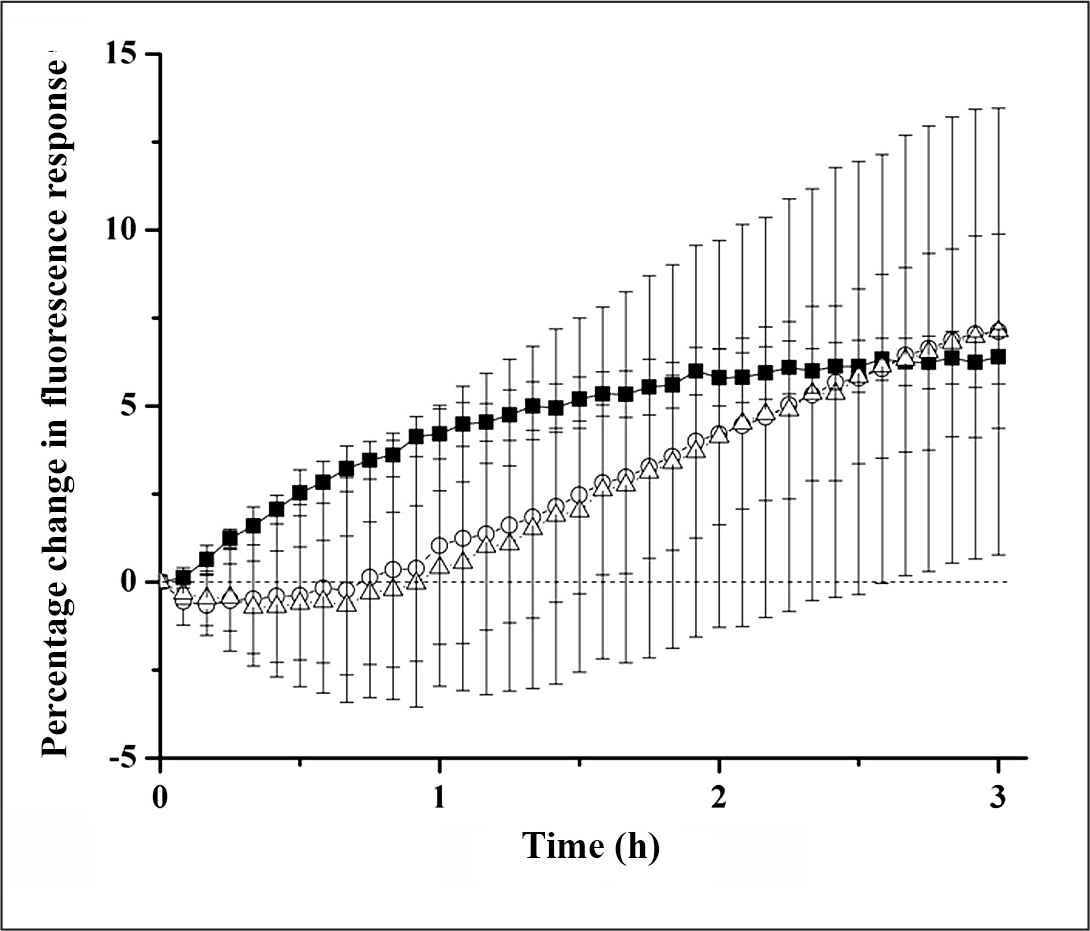
Gel encapsulation of the nanosensors may affect their ability to monitor real-time changes in glucose concentration because of limited-to-no glucose permeability into the gel and potential delays in response. Therefore, glucose permeability into the gels was tested. To determine glucose permeability, glucose macrosensors were used rather than nanosensors to eliminate unencapsulated nanosensors from contributing to the fluorescence response to glucose. Additionally, glucose macrosensors respond over the course of 1 h
10
and thus added to the overall monitoring time required. The percentage change in signal of the glucose macrosensors as a function of time are shown in Glucose permeability through gel 1. Glucose macrosensors were layered with different gel 1 formulations of variable stiffness and then exposed to 0 and 9000 g/dl of glucose for the control and experimental groups, respectively. The percentage difference between the controls and experimental groups are plotted against time for no gel (▪), 5:1 (gel 1: PBS; ◯), 2:1 (gel 1: PBS; Δ), and 1:1 (gel 1: PBS; ▽). Glucose permeability through gel 2. Glucose macrosensors were layered with different gel 2 formulations of variable stiffness and then exposed to 0 and 9000 g/dl of glucose for the control and experimental groups, respectively. The percentage difference between the controls and experimental groups are plotted against time for no gel (▪), 5:1 (gel 2: PBS; ◯), and 3:1 (gel 2: PBS; Δ). Gel 1 retention of glucose nanosensors. Fluorescent images of gel 1 with glucose nanosensors over time for 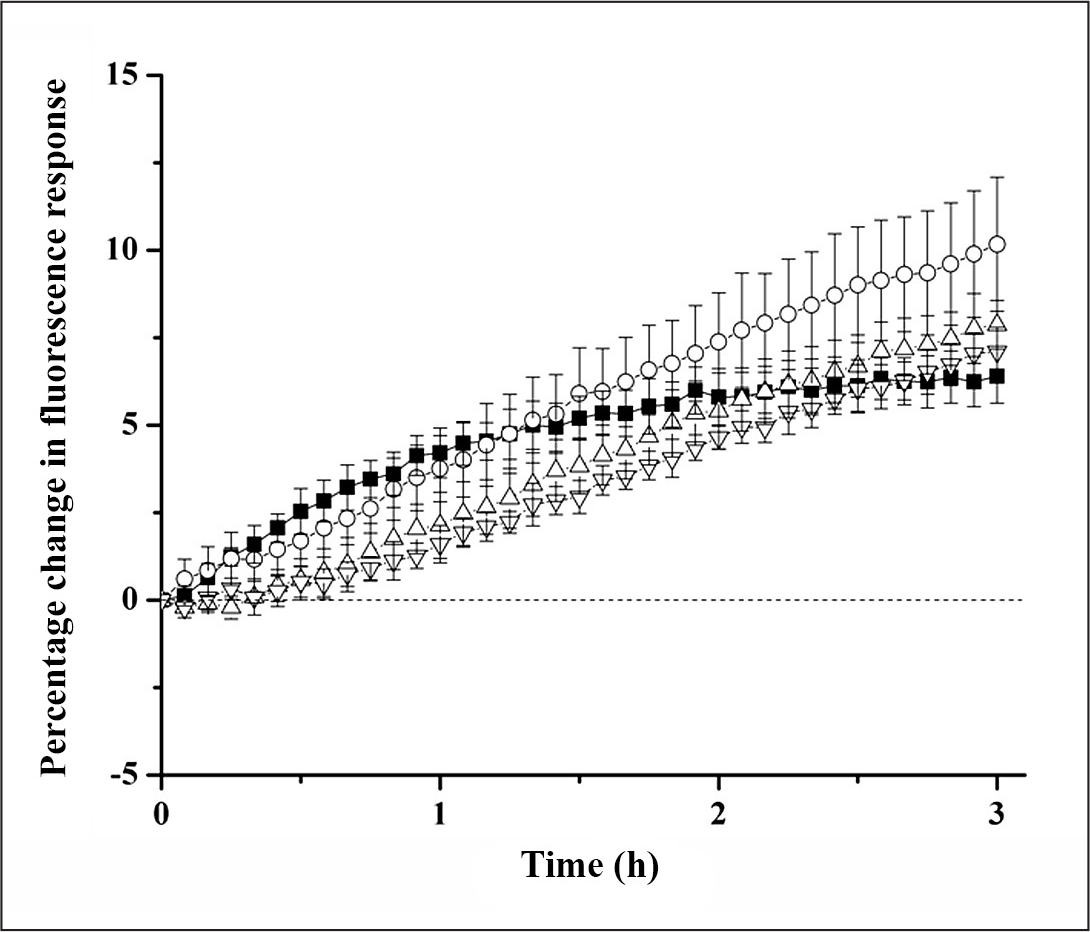
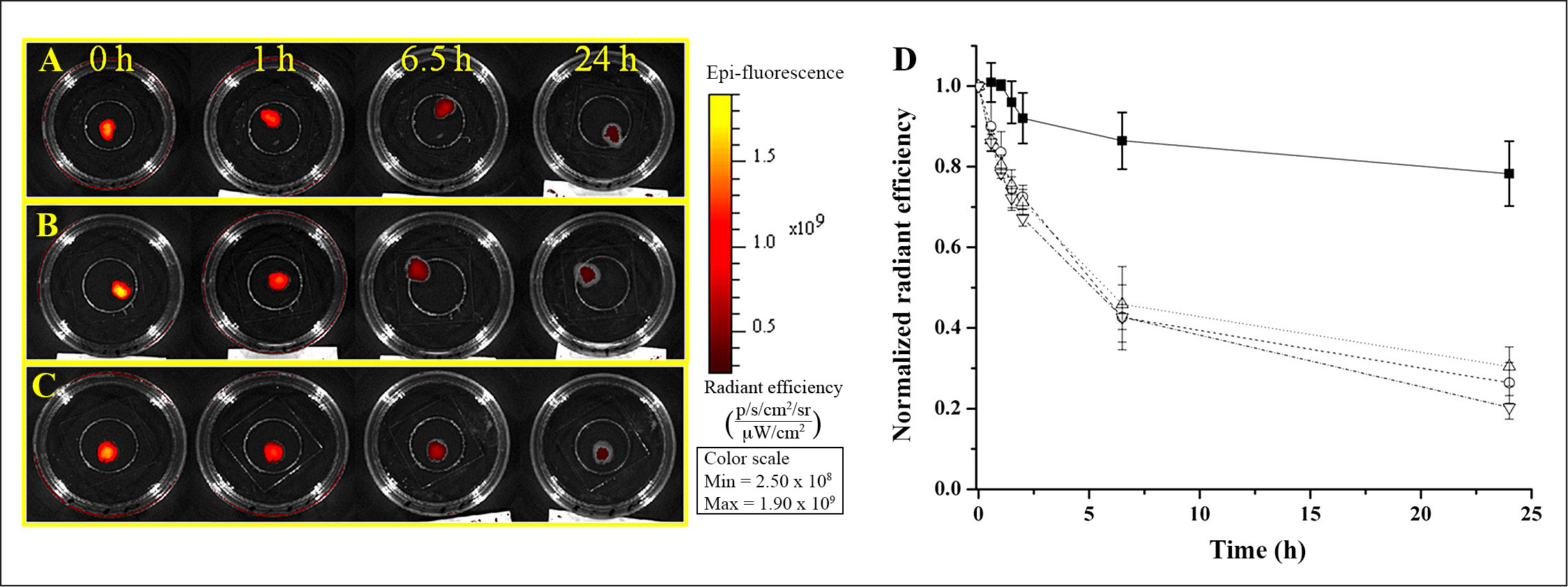
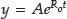
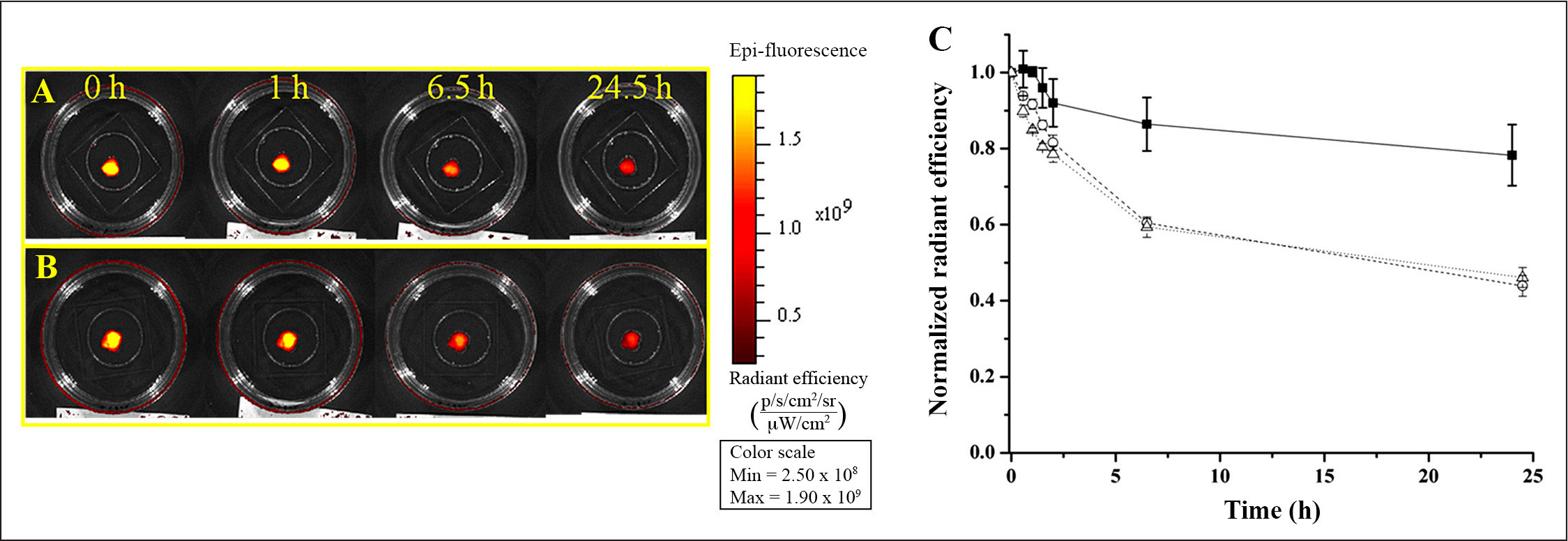
Gel 2 retention of glucose nanosensors. Fluorescent images of gel 2 with glucose nanosensors over time for
Decay Rates of the Average Total Radiant Efficiency over 6.5 h for Bulk Degradation Control and Each Formulation of Gel 1 and Gel 2
In Vivo Demonstration
Discussion
Gel 1 and gel 2 were selected for these studies because they can be easily modified for stiffness through dilution,18,19 and they are amenable to nanosensor incorporation during the gelling process. Three parameters were used to assess and downselect the optimal formulations for
The particles being suspended had the potential to inhibit gelation, as was noted with other matrices (data not shown). At several different formulations for gel 1 and gel 2, incorporating a sufficiently high concentration of nanosensors for
Lastly, the diffusion of the nanosensors out of the gels was investigated to determine efficacy of sensor retention over time. The average size of the glucose nanosensors is 74 nm, 10 and gel pore sizes less than this would be desirable for greater retention. Reported values of pore size for gel 1 range from 26 nm to 2 μm for 1:1 dilution,19,25 with pore size varying with gel 1 concentration. 19 Drastic changes in pore size of gel 1 was not evident from our results since all gel 1 formulations performed similarly, and we predict that the average pore size for these matrices was greater than the size of the nanosensors. On the other hand, gel 2 has been shown to retain proteins as small as 70 kDa. 18 The average pore size for gel 2 may be smaller than gel 1 and thus explains the greater retention of nanosensors over time.
Previous
Conclusions
Two commercially available injectable gels sufficiently encapsulated glucose nanosensors while maintaining glucose permeability. Both gels prolonged sensor lifetime
Footnotes
Abbreviations:
This work was funded by the IGERT Nanomedicine Science and Technology program at Northeastern University from National Cancer Institute and National Science Foundation grant DGE-0504331 and National Institutes of Health Grant 5RO1GM084366.
Acknowledgments:
We thank Kevin J. Cash and Ankita Shah for their technical guidance in preparing this article.