Abstract
We present the heart of a 38-year-old man with paraplegia due to a remote traumatic spinal cord injury. In the 20 years following his injury, he experienced chronic decubitus ulcers, osteomyelitis, neurogenic bladder, malnutrition, and urinary tract infections. He was admitted to the hospital with septic shock secondary to multiple decubitus ulcers and osteomyelitis and expired after a two-month hospitalization. At autopsy, there was marked replacement of left ventricle and interventricular septal myocardium by gritty, firm, yellow-white tissue. Microscopic examination demonstrated a remote infarct with marked dystrophic calcification and unexpected amyloid deposition. This example demonstrates the extraordinary extent to which dystrophic calcifications can replace myocardium and highlights multiple potential etiologies of myocardial calcifications. Of note, this is the first report documenting myocardial calcification as a complication of remote, non-iatrogenic trauma. The role of the amyloidosis in the development of calcification is unclear, but a contributory effect cannot be excluded.
Method and Results
The references cited in this report were obtained through a literature search of articles and book chapters pertaining to myocardial calcification, cardiac amyloidosis and calcifications, and the presentations of these entities in the contexts of either forensic pathology, cardiac pathology, cardiology, or critical care medicine. The patient information and history utilized in the writing of this report was obtained through statements from family members familiar with the decedent’s past medical history, review of available medical records in the electronic medical record, including laboratory values, imaging, and clinical notes from his most recent hospitalization, and examination grossly and histologically at autopsy. All images of both gross pathology and histologic sections were taken at the medical examiner office during the case workup.
Introduction
This is the case of a 38-year-old man with an unusual complication of remote spinal cord injury—massive myocardial calcification secondary to healed myocardial infarcts, likely sustained at the time of his injury due to blood loss, as well as focal deposition of amyloid within the heart. This case report serves as an opportunity to review the mechanisms and etiologies of myocardial calcification and is unique because this finding has not been described secondary to trauma. We also review the potential etiologies of the amyloid deposition, and whether it is related to the calcification. We believe this manuscript will be helpful to practicing forensic pathologists both in documenting the potential for myocardial calcification to complicate remote traumatic injury, and in reviewing the basic biologic processes behind calcium and amyloid deposition.
Case Report
The decedent was a 38-year-old man with paraplegia due to a traumatic spinal cord injury sustained from multiple gunshot wounds 20 years previously. Since his injury, he had experienced frequent readmissions for chronic sacral decubitus ulcers, osteomyelitis, neurogenic bladder with chronic urinary tract infections, anemia, and severe protein-calorie malnutrition. He was admitted again with evolving septic shock related to decubitus ulcers and osteomyelitis. Despite extensive interventions, including an exploratory laparotomy and surgical debridement of the ulcers, the patient’s condition continued to deteriorate, and he died two months after his initial admission.
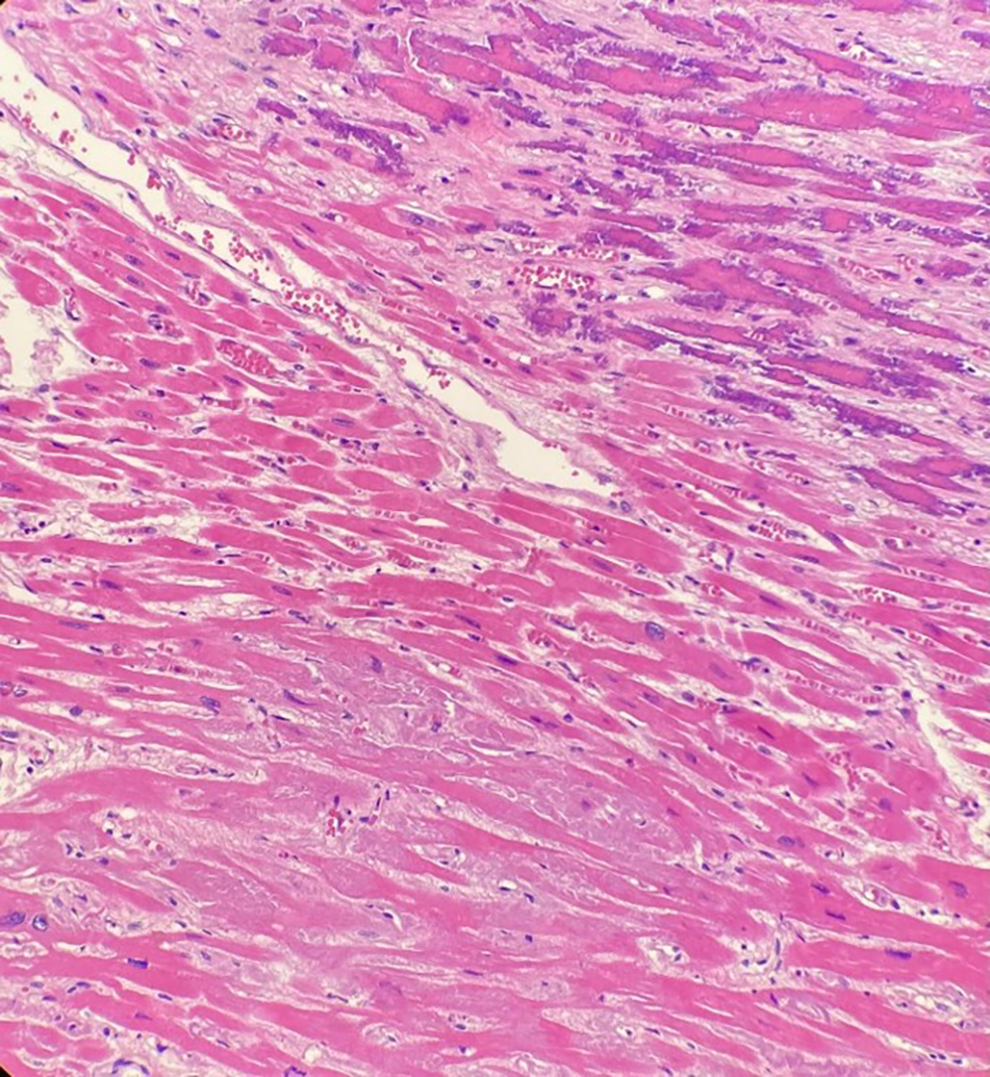
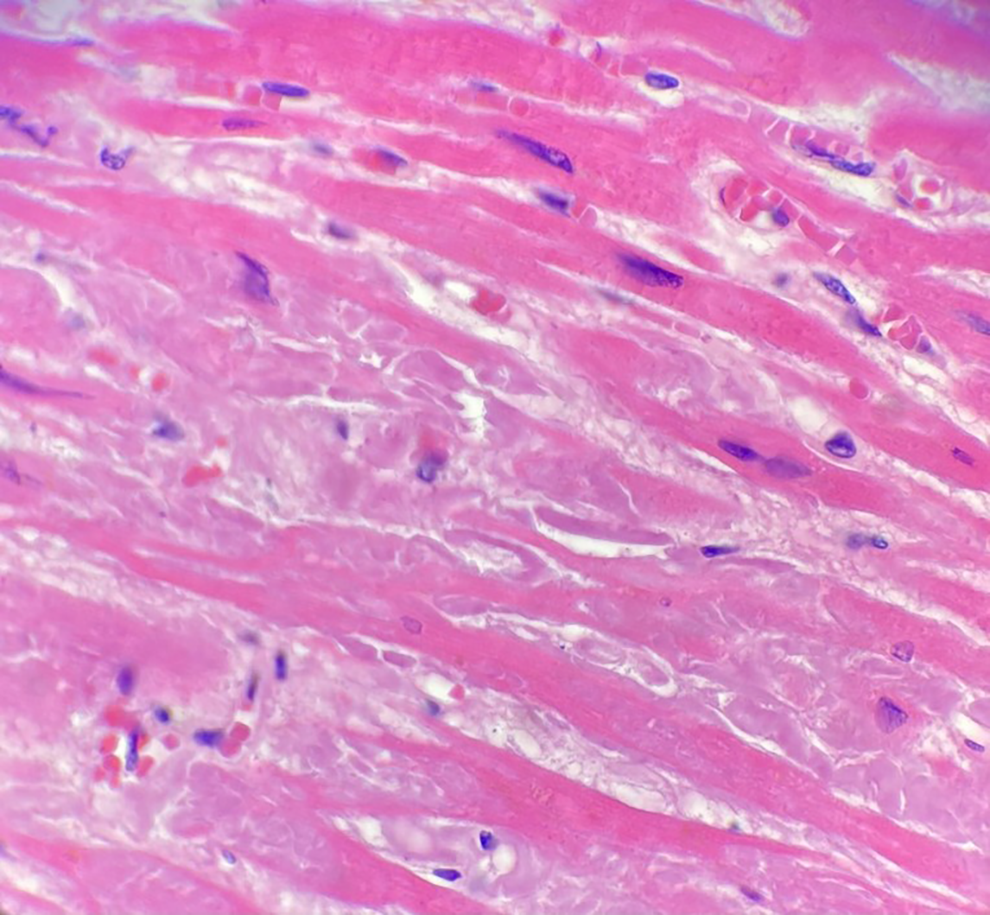
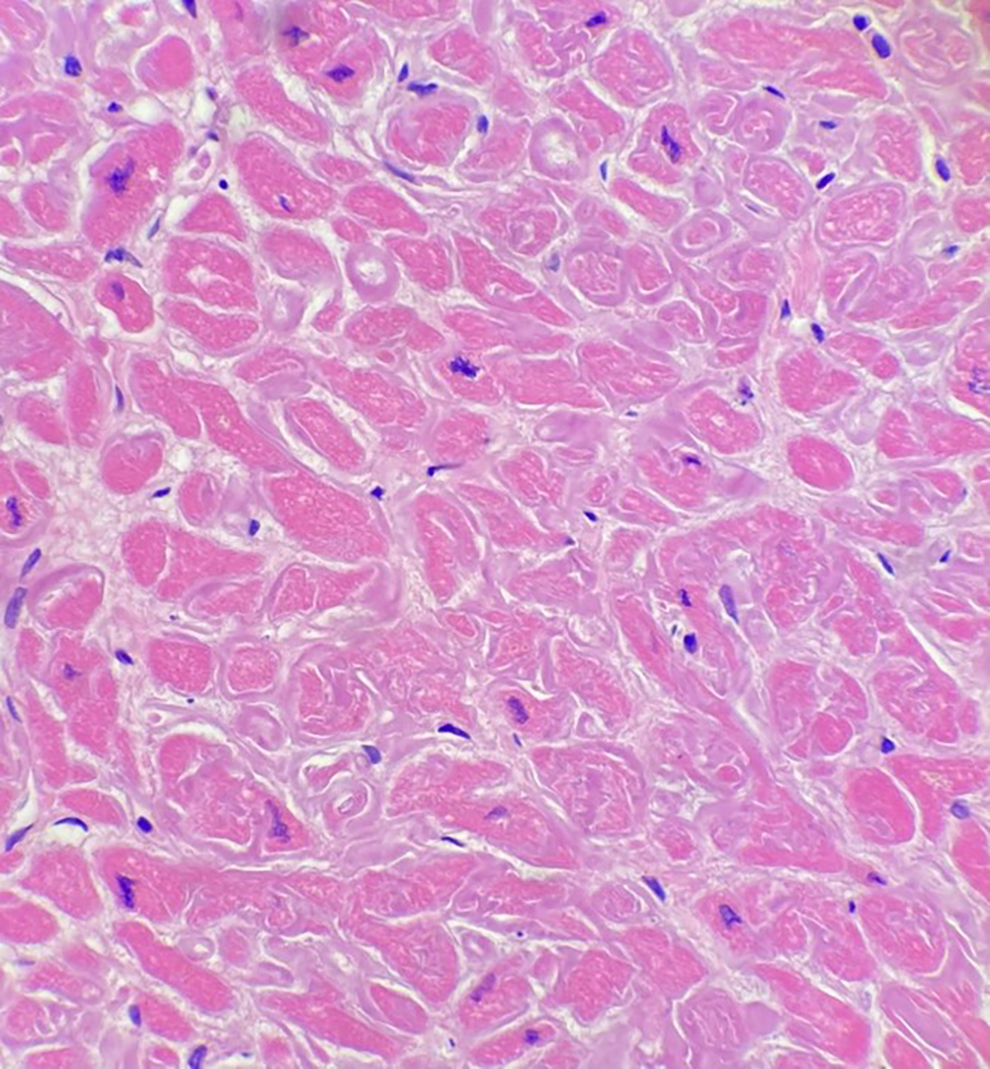
Because of the history of trauma, his body was brought to the Cuyahoga County Medical Examiner’s Office for autopsy. At autopsy, the heart felt abnormally firm. The heart weighed 430 grams and showed dilation of all four chambers (mild atrial dilation and marked biventricular dilation). The epicardium exhibited patches of white thickening and fibrinous, hemorrhagic exudate. The coronary arteries were free of atherosclerosis, luminal narrowing, or thrombosis, and the pulmonary arteries were free of thrombus or embolus. Sections of the ventricles along the short axis revealed multifocal replacement of the interventricular septum and left ventricle by gritty, yellow-white tissue in a subendocardial and near-transmural distribution (Figure 1). None of the affected areas were thinned relative to the uninvolved myocardium, which was normal in color and texture. The left ventricle measured 1.2 cm in thickness, the interventricular septum 1.3 cm in thickness, and the right ventricle 0.3 cm in thickness. The valves were also unremarkable. Microscopic examination of the grossly calcified regions revealed well-healed, collagenous scar tissue with subendocardial sparing, consistent with a remote infarct. Within the same regions were numerous calcified, mummified myocytes (see Figures 2, 3). Numerous small foci of granulation tissue and replacement fibrosis were also present, some showing interstitial calcification. Multifocal deposition of amorphous eosinophilic material, consistent with amyloid, was identified deposited in an interstitial, lace-like pattern (Figures 3, 4, 5). The cause of death was determined to be complications of paraplegia resulting from his traumatic gunshot wound injuries and the manner of death was homicide.
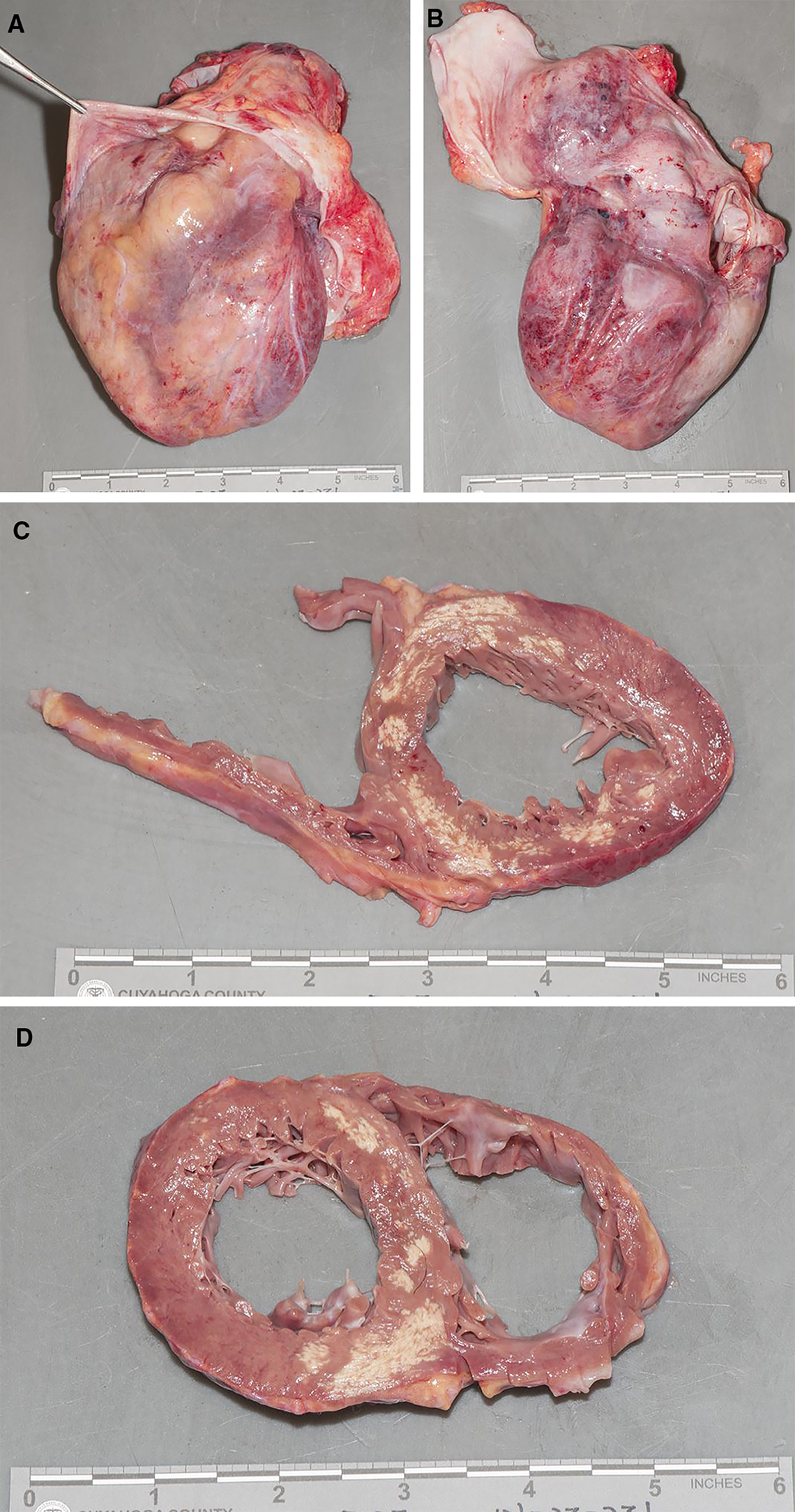
Gross images of the decedent’s heart. In A and B, one can see the epicardial surface and inner lining of the parietal pericardium are involved by scattered small patches of fibrinous exudate and white fibrous tissue. C and D are cross sections of the ventricles, which shows marked biventricular dilation as well as multiple foci of yellow-white discoloration in the interventricular septum and left ventricle. These areas were palpably firm and gritty. Note that the foci are not restricted to an area supplied by a single coronary artery.

(H&E, A 100× and B 200×) Micrographs demonstrating a remote infarct at right, with replacement fibrosis and dystrophic calcification of mummified myocytes. Relatively normal myocardium is on the left side of the image.
(H&E, 100×) Micrograph of left ventricle myocardium demonstrating dystrophic calcification of mummified myocytes and replacement fibrosis in the upper right corner. In the lower left corner, there is interstitial deposition of amorphous eosinophilic material with no associated calcification.
(H&E, 600×) Higher magnification of the interstitial amorphous eosinophilic material seen in Figure 3 which confirms it is morphologically consistent with amyloid.
(H&E, 400×) Microscopic images of ventricular myocardium showed an interstitial, lace-like pattern of amorphous eosinophilic material consistent with amyloid. This pattern of deposition is consistent with AL amyloid, although post-mortem testing was not available to confirm the amyloid subtype.
Discussion
Forensic pathologists commonly identify calcifications in the heart, typically affecting the coronary arteries, valve leaflets, and mitral valve annulus. However, calcium deposition in the myocardium itself is much less common (1 -3). While calcium deposition can reportedly occur idiopathically in the myocardium, it is usually attributable to either metastatic or dystrophic calcification (1, 3, 4). Metastatic calcification is secondary to impairment of calcium and phosphorus metabolism, which is most frequently seen in patients with chronic renal failure or hyperparathyroidism (1 -3). Dystrophic calcification, on the other hand, refers to the deposition of calcium salts in damaged or necrotic tissues in the absence of deranged calcium metabolism (1 -5). In the context of dystrophic calcification, the precipitating tissue damage can occur secondary to various clinical conditions including sepsis, myocardial infarction, radiation therapy, anatomic cardiac abnormalities, especially anomalies of the coronary arteries, and other factors leading to myocardial ischemia (1 -10).
There are several etiologies that likely contributed to the decedent’s myocardial calcification. Of note, a computed tomography (CT) scan during his initial admission for trauma showed no myocardial calcifications. Medical records from his trauma admission documented severe blood loss and hypotension which raises the possibility of myocardial infarction resulting from global hypoperfusion. Histologically, the areas of remote infarct coincide with the areas of dystrophic calcification. Because the decedent’s coronary arteries were entirely patent at autopsy, and the infarcts are not limited to a single coronary artery distribution, a global hypoperfusion event is likely the underlying culprit from the time of his original injuries. While the literature does lay bare that ischemic injury is the only substrate required to precipitate the phenomenon of extensive myocardial calcification, he was hospitalized and clinically septic secondary to various infections for nearly two months prior to his demise. A number of studies have documented myocardial calcification as a rare complication of severe sepsis, and in this context extensive myocardial calcification can appear in as little as 4 to 6 weeks (5 -8). As this complication is much rarer than sepsis, itself, some studies have attempted to identify the circumstances under which extensive myocardial calcification is more likely to present. Kapandji et al. published a case series in the Journal of Critical Care Medicine that identified prolonged hemodynamic failure, profound acidosis, high vasopressor doses, and renal failure as possible risk factors for new-onset diffuse myocardial calcification (9). As the decedent experienced nearly all of these risk factors during his two-month hospital course, it is reasonable to suspect that some degree of myocardial calcification was due to the prolonged episode of sepsis. The foci of granulation tissue appreciated histologically also suggest recent or subacute injury that would coincide with this time frame. However, a CT scan obtained early in the decedent’s hospital admission revealed “dense circumferential calcification” of the left ventricle, “suggesting previous infarction,” indicating the pathologic process preceded his final hospitalization. Recent studies have profiled a number of patients who have survived for years with extensive myocardial calcification, oftentimes being closely followed or managed with medication regimens similar to other heart failure patients (8, 9, 11). While it is possible that the decedent’s final two-month hospitalization worsened the degree of myocardial calcification, his precipitating injury was likely at an earlier period. He had many previous admissions for osteomyelitis and urinary tract infections, and it is possible any of these episodes initiated and/or exacerbated the calcification. Without regular, periodic cardiac imaging between his initial injuries that led to his paraplegia and his subsequent hospitalizations, it is difficult to state definitively which factors were most influential in precipitating the degree of myocardial calcification seen in this case.
While the presence of amyloid could not be confirmed by Congo Red or thioflavin staining due to resource limitations, the histologic features were characteristic. Cardiac amyloid deposition raises the possibility of additional pathologies at work in this case, ranging from plasma cell dyscrasias to familial amyloid conditions to chronic inflammatory conditions.
Most commonly, cardiac amyloidosis is of the light chain variety (AL). Patterns of amyloid deposition can be a distinct and useful adjunct in determining the variety of amyloidosis present. A perimyocytic pattern of amyloid deposition in the heart (as seen in the presented case) is more suggestive of AL amyloidosis than other diseases (12). AL amyloidosis is caused by the production and deposition of light chain protein fibers produced by monoclonal plasma cells in tissues of the body (12 -14). Microscopic examination of the kidneys revealed numerous sclerotic glomeruli, with nodular and diffuse expansion of the glomerular mesangium by acellular eosinophilic material. This pattern is also morphologically consistent with amyloid, and preferential involvement of the renal and cardiovascular systems is also supportive of AL amyloidosis (14). Another common form of amyloidosis involving the heart are those derived from transthyretin (TTR). TTR-amyloidosis can be either age-related (“senile” or “wild-type”) or related to inherited mutations in the TTR gene (“mutant” or TTR-associated familial amyloidosis). Histologically, TTR-derived amyloid tends to form nodular interstitial deposits of amyloid, as opposed to the perimyocytic pattern described above, which were not observed in this case. The age of the decedent also effectively excludes “senile” TTR amyloidosis, and he would be uncommonly young even for familial amyloidosis (12). The decedent’s years of chronic infections following the injury, including chronic osteomyelitis and urinary tract infections, also raise the possibility of AA amyloidosis. AA amyloidosis is a recognized complication of numerous autoimmune and inflammatory conditions, including chronic osteomyelitis (12, 15, 16) and results from misfolding and deposition of an acute-phase protein known as serum amyloid A protein (SAA) (12, 13, 15, 16). SAA concentration in the plasma increases in the presence of an inflammatory condition and, through an as-of-yet poorly understood mechanism, these increased SAA concentrations can lead to a pathway causing deposition of insoluble amyloid and the diagnosis of AA amyloidosis (12, 15, 16). Unfortunately, the diagnosis of cardiac amyloidosis was not suspected during the decedent’s final hospitalization. As such there was no serum or urine protein electrophoresis, bone marrow biopsy, or targeted testing of the TTR gene. Therefore, while AL amyloidosis is favored in this case, other etiologies cannot be definitively ruled out.
There is growing evidence in the literature that the presence of amyloid can be strongly associated with dystrophic calcifications (12, 14, 17 -19). Wei et al. reported dystrophic calcification and amyloidosis in old subcutaneous injection sites, suggesting a potential association between these two processes, while Charles et al. presented a case of bilateral intratarsal dystrophic calcification and ossification in localized immunoglobulin light chain amyloid, both drawing a possible connection between dystrophic calcification and amyloidosis (18, 19). In cardiac pathology specifically, the literature draws a relationship between valvular calcifications and amyloidosis (12, 20, 21). The exact nature of these amyloid deposits is still unknown, as immunohistochemical typing studies have not yet been able to establish a connection with any amyloid proteins known to involve the heart. Nevertheless, these specific valvular amyloid deposits are strongly correlated with degenerative changes and calcifications, as well as post-inflammatory fibrosis (12). More recent studies have shown microcalcifications in association with ATTR cardiac amyloidosis (both within and outside of the amyloid deposits) and to a lesser extent in AL cardiac amyloidosis (22). In this decedent’s heart, the vast majority of calcification corresponded spatially with remote infarcts rather than amyloid deposits, which lead us to conclude the calcification in this heart is less related to amyloid and more driven by the presence of scar tissue.
Conclusion
In conclusion, we present a unique heart from a 38-year-old man which showed extensive dystrophic calcification of remote infarcts in addition to amyloid deposition in the myocardium. The patient’s medical history and the histopathological findings suggest a multifactorial etiology for the extensive myocardial calcification, including previous myocardial infarction, prolonged sepsis, and chronic inflammatory conditions. Importantly, though, all these conditions can be traced to his initial, traumatic spinal cord injury; no other reports of post-traumatic myocardial calcification can be found on literature review.
The association between dystrophic calcifications and amyloidosis is an intriguing aspect of this case that has not been previously explored in the setting of myocardial calcification at forensic autopsy. While the exact mechanism linking these two processes remains unclear, previous studies have reported similar associations between calcification and amyloid in other contexts. The amyloid may have some local metabolic effect in promoting calcium deposition, but this is only speculation and cannot be proven at this point.
While extensive dystrophic myocardial calcification remains an exceedingly uncommon finding, there are increasing reports of this pathology in both living and deceased patients. It is in the context of the latter population that this entity is of particular importance to the forensic pathologist. When autopsy reveals a grossly firm and calcified heart, it is important to develop a differential of the possible etiologies—as exemplified by this decedent, the list of differential diagnoses should include remote trauma.
Footnotes
Ethical Approval
N/A.
Statement of Human and Animal Rights
N/A.
Statement of Informed Consent
N/A.
Disclosures & Declaration of Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Financial Disclosure
The authors received no financial support for the research, authorship, and/or publication of this article.
