Abstract
Background:
4-Fluoroethylphenidate (4F-EPH) is a psychoactive substance, sold primarily over the Internet as a `research chemical'. Recreational and `functional' use of this drug has been reported by online user fora. Scientifically-based data on the pharmacological, physiological, psychopharmacological, toxicological, and epidemiological characteristics of this molecule is non-existent. The aim of this paper is to remedy this situation.
Methods:
Recent literature (including ’grey‘) was searched to update what is known about 4F-EPH, especially its toxicity. This was supplemented by netnographic examinations of internet sites.
Results:
The resultant information is presented, including details of the first reported death involving 4F-EPH use in 2016. There are no international controls imposed on 4F-EPH. However, it has been made a controlled drug in several European countries, including the United Kingdom since 31 May 2017, as well as Canada.
Conclusions:
It is vital that any other cases, including non-fatal overdoses, are documented so that a firmer scientific evidence-base can be established for this molecule. This will then help inform clinical practice.
Keywords
Introduction
There have been constant changes in recent years in the creation, manufacture, diffusion, and use of novel psychoactive substances (NPSs). When or where a molecule has been designed de novo, rather than being rediscovered by an aberrant chemist, information about its pharmacology, metabolism, toxicity, and psychoactive properties is normally either very scant or nonexistent. Typically, it is only the chemical structure of that molecule which has undergone scientific investigation by its creator and producer; often this is to evade legal controls. Consequently, the potential harms and risks posed by the molecule are unknown to the manufacturer, supplier/retailer, and consumer.
The main area of growth, over the past 17 years or so, in the number of NPS identified and reported to international agencies, such the European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) and the United Nations Office on Drugs and Crime (UNODC), has been that of psychostimulants (1,2). Within this category, the largest group is synthetic cathinones. Other stimulants belong to the aminoindane, arylalkylamine, phenethylamine, piperazine derivate, and piperidine and pyrrolidine groups. The last-mentioned group includes ethylphenidate (Ritalin, Concerta) and several analogs such as 4-fluoroethylphenidate (4F-EPH; 4F-TEP).
This article’s main aim is to deepen and enhance the knowledge base concerning 4F-EPH, first identified in Europe about seven years ago, through updating current information but particularly its toxicology. Regular monitoring and surveillance of drug-related fatalities helps to identify both the nature of deaths related to NPS consumption and the epidemiological characteristics of NPS users (3). A secondary aim is to present information regarding user experiences and how this molecule compares to other stimulants.
Methods
Recent literature (including “gray”) was searched to update what is known about 4F-EPH, especially its toxicity. Google Scholar, Scopus, and PubMed were searched systematically on November 1, 2020 using the following search terms in combination: “4F-EPH,” “4-fluoroethylphenidate”; “ethyl 2-(4-fluorophenyl)-2-(2-piperidyl)acetate”; “ethyl 2-4-fluorophenyl-2-(piperidin-2-yl)acetate”; “p-fluoroethylphenidate”; “effects”; “death”;“ fatal*”; “toxic”; “poison*”; “overdose.” In addition, alerts were set up to identify any subsequent relevant research.
This was supplemented by netnographic examinations of “snapshots” taken on November 1, 2020 of Internet sites considered likely to report experiences with 4F-EPH: www.erowid.org; www.chemeurope.com, http://www.druglibrary.org/, www.bluelight.org, https://tripsit.me/, https://drugs-forum.com/, https://psychonautwiki.org/wiki/Main_Page, and https://www.reddit.com/. Relevant material, from the last five sites listed, was extracted into an ad hoc Excel spreadsheet for analyses. The thematic analysis approach was used to identify aspects of potential interest and relevance, emerging topics were then identified, categorized, and grouped for reporting (see Supplementary Material).
The first death contributed to by 4F-EPH is described here. It is based on postmortem and toxicology reports, as well as legal records relating to the death investigation, provided to the lead author by relevant stakeholders and/or in the public domain.
Findings
The following sections outline the main themes and findings that resulted from these investigations. There is some overlap of material as information from one site is also shared/reported on other sites (e.g., PsychonautWiki and Reddit) or across different threads on the same platform (e.g., Reddit). The number of reports/threads deemed to be relevant are as follows: Reddit 32, Bluelight 24, Drugs-forum 13, PsychonautWiki 2, and Tripsit 0. Full details are available in the Supplementary Material.
Chemistry
4F-EPH seems to have been created by “research chemists,” probably based in China, as a replacement for ethylphenidate (Ritalin) following its control in countries such as the United Kingdom.
The 4F-EPH (4-fluoroethylphenidate) molecule appears to have two International Union of Pure and Applied Chemistry (IUPAC) names: (1) ethyl 2-(4-fluorophenyl)-2-(2-piperidyl)acetate and (2) ethyl 2-4-fluorophenyl-2-(piperidin-2-yl)acetate. Another variation appears to be p-fluoroethylphenidate (4). The UNODC refers to it as 4-F-EPH. Other variations are 4FEPH, 4-FEPH, and 4F-TEP. The molecule does not appear to have any street names. Its CAS (base) number is 2160555-59-7.
It belongs to the piperidine class of chemicals (Figure 1) and is the ethyl derivative of 4-fluoromethylphenidate (methyl 2-(4-fluorophenyl)-2-(2-piperidyl)acetate) and the 4-fluoro derivative of ethylphenidate (ethyl 2-phenyl-2-(piperidin-2-yl)acetate). This molecule is a chiral compound. Its structure is described as similar to that of amphetamine but with the following differences: “a substitution at Rα which is incorporated into a piperidine ring ending at the terminal amine of the phenethylamine chain. It contains an ethyl acetate bound to R2 of its structure and is fluorinated at R4 of its phenyl ring” (5).
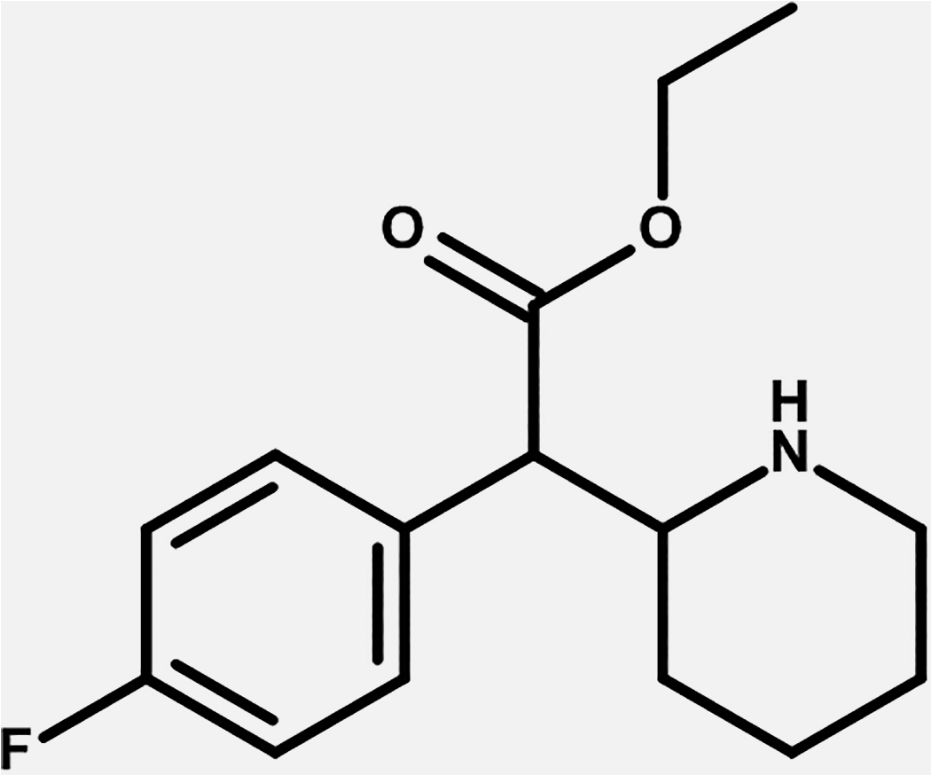
Chemical structure of 4F-EPH. 4F-EPH indicates 4-fluoroethylphenidate.
Pharmacology
Little information, including academic literature, is currently available on 4F-EPH’s pharmacology. As a result, parallels are drawn with ethylphenidate, for which the molecule was supposedly marketed as a replacement (6). Reference is made to ethylphenidate’s action as both a dopamine and norepinephrine reuptake inhibitor.
The PsychonautWiki site states that 4F-EPH may be more efficient than methylphenidate (Ritalin) as a dopamine reuptake inhibitor, thereby giving rise to euphoric and stimulant effects (7 -9). It is also speculated that, based on its similarity to other related analogs of methylphenidate, the molecule exerts “its activity as some form of double or triple monoamine reuptake inhibitor” (5).
Use of 4F-EPH
There is no epidemiological information, regular or otherwise, on the use of 4F-EPH.
According to the European Database on New Drugs (EDND), the first identifications of 4F-EPH in the European Union were made in France and the United Kingdom in March 2016 followed later that year by Denmark, Slovenia, Spain, Estonia and Sweden, and Latvia in 2017. In 2020, the first identifications of the molecule were made in the Netherlands (July) and Hungary (September) (6).
At of early November 2022, the UNODC Early Warning Advisory on New Psychoactive Substances database records 32 reports of 4F-EPH, the first ones being in 2016 from mostly European countries (i.e., Austria, Denmark, Estonia, France, Latvia, Lithuania, Sweden) and Japan (10). In 2017, there were reports from Canada, Poland, Sweden, and Switzerland. Reports were made in 2018 from Brazil, Finland, France, Poland, and Sweden. Five countries made reports in 2019: Brazil, the Netherlands, Poland, Slovenia, and Sweden. More recently, reports were made in 2020 by Finland, Germany, Spain, and the United States, see also (11); 2021 saw reports from France, the United Kingdom, and the United States.
An examination of Google Trends searches for “4F-EPH” data indicates that the first ones were conducted about November 2015 and continued until February 2017, after which there was a falloff with sporadic bouts of interest until a spike around October 2018 (Figure 2). This interest rapidly fell away until December 2019, since when more attention has been paid to the molecule. It is worth noting that some of the interest in May 2020 could have been generated by the authors undertaking Google searches!
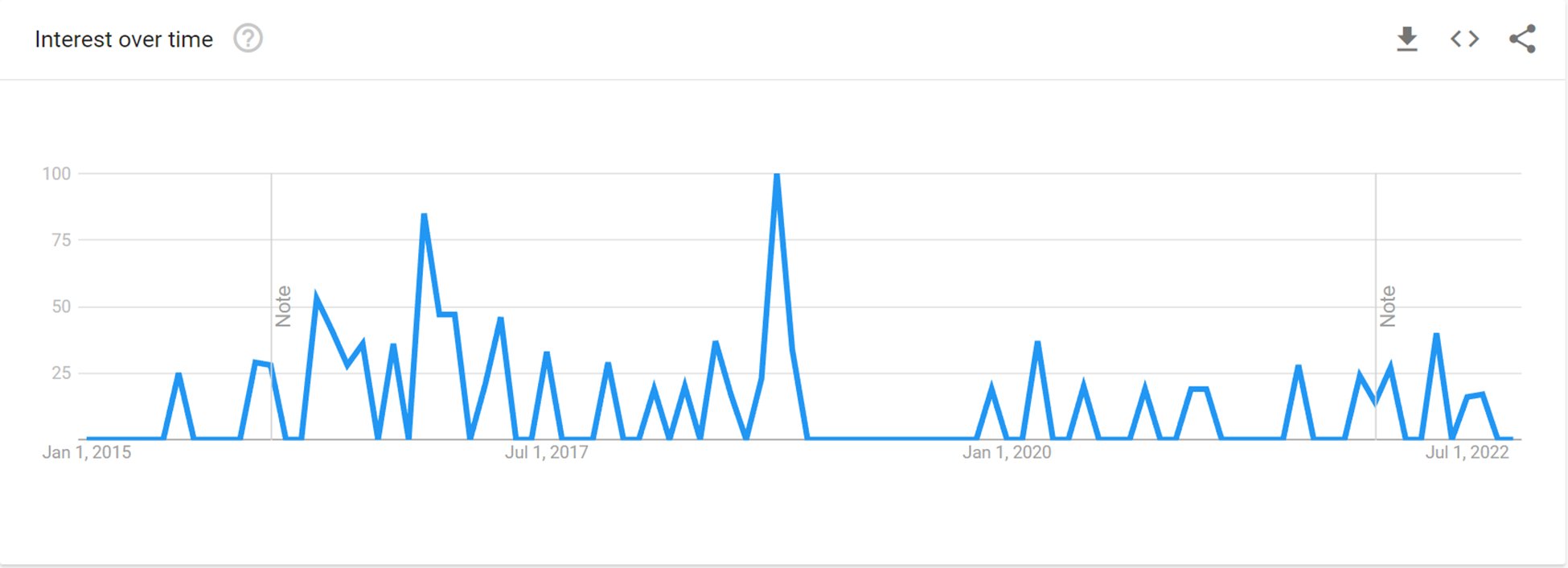
Google Trends searches for 4F-EPH over time. Note from Google Trends: Values are calculated on a scale from 0 to 100, where 100 is the location with the most popularity as a fraction of total searches in that location, a value of 50 indicates a location which is half as popular. A value of 0 indicates a location where there was not enough data for this term. Data source: Google Trends (https://www.google.com/trends).
Over the course of the period November 2015 to November 2022, most interest proportionately appears to have been in Sweden, followed by Poland, the United Kingdom, and the United States (Figure 3).
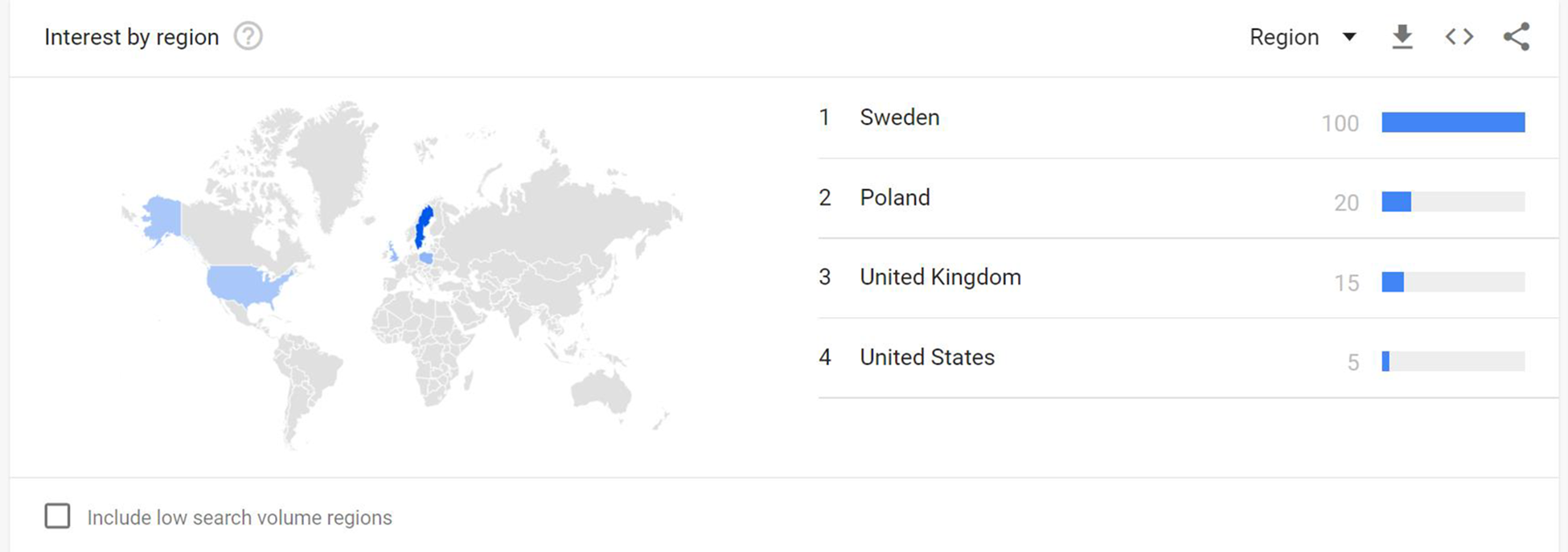
Google Trends searches for 4F-EPH by region. Note from Google Trends: Values are calculated on a scale from 0 to 100, where 100 is the location with the most popularity as a fraction of total searches in that location, a value of 50 indicates a location which is half as popular. A value of 0 indicates a location where there was not enough data for this term. Data source: Google Trends (https://www.google.com/trends).
Online resources, principally PsychonautWiki, suggest that there is little by way of history of 4F-EPH being used recreationally (5). No surveys have captured information on this molecule. User reports examined for this present study suggest that the first online discussions about 4F-EPH took place about mid-October 2015 on Bluelight.
This substance seems to have been hardly used, known about, or encountered by law enforcement and forensic science agencies or emergency rooms. This is even true in Europe where its use appears to have been concentrated. For these reasons, there is little scientific information about the drug, its pharmacology, metabolism, effects, toxicity, and epidemiology. Information about its chemistry, effects, and so on provided on vendors’ websites is heavily “borrowed” from the PsychonautWiki entry for the molecule.
Availability
The earliest officially documented appearance of 4F-EPH is January 2016 (6). However, one user experience cited in Trott states that this molecule was “released at the end of 2015” (12). This is supported by it being reported as a “new entry” for the last quarter of 2015 into the “Top Ten New Psychoactive Substances” listed by Wedinos (13). It was offered for sale online as early as July 2016 (https://www.gr8researchchemicals-eu.com/).
User reports suggest that 4F-EPH first appeared as a response to the U.K. government making ethylphenidate (4F-MPH) and several other related substances the subject of a Temporary Class Drug Order on April 15, 2015 (14). 4F-EPH filled this gap during the period leading up to the passing of the Psychoactive Substances Act 2016 a year later. The molecule was legal at a time when other “RC phenidates” were in short supply, and it was cheaper than 4F-MPH. However, as will be seen below, it was considered inferior to 4F-MPH and thus did not attract as much interest.
The most common source of supply is from the online market, where it is typically advertised as a “research chemical” or “research compound” with claimed purities of >94% to 99.7%. Statements are carefully worded to convey a message that purports these products are “only intended for research purposes in a controlled laboratory for forensic or scientific study and are NOT intended for human or animal consumption” and that “since the chemical is so new research is not available.” Other vendors offer it as a “legal alternative for Adderall, speed, amphetamine or coke.” A vendor that appears to be based in Turkey (www.highfarmbotanicals.com) states that it sells “4F-EPH for in vitro research purposes only. It is not suitable for human consumption.” Some sites making such statements then provide information on dosages, recommending harm reduction practices if purchasers choose to the product. These and other ploys have been previously reported for selling molecules that were once “legal highs” subsequent to them being controlled in the United Kingdom (15,16).
Descriptions of 4F-EPH seizures reported to the EDND all refer to white powder. However, online vendors offer both powder and crystal forms, including “crystal rock.” Users give varying descriptions: “a very fine off-white powder, with a few small clumps mixed in which break up easily. It seems to be quite dense. It is slightly less white than 4F-MPH and has an almost identical smell and taste”; “white powder with lumps”; “crystalline pure white powder with negligible smell in the UK batch”; “white shiny powder.”
There does not appear to be any reported academic research on the purity levels of 4F-EPH, or possible diluents, cutting agents, or contaminants. The Wedinos website (https://www.wedinos.org/) has only one entry (July 2019) for an intended purchase of 4F-EPH; this turned out to be pure. This was also the case for two sample test results reported on Erowid (https://www.erowid.org/). There are no user reports relating to purity.
Online vendors are based in various parts of the world. For example, one based in Shijiazhuang, China, advertises a minimum sale of 1 g and claims to be able to supply 10 kg/day, with >99% purity. A Netherlands-based supplier (https://chemsterdam.nl/product/4f-eph/) refers to its products as “high quality Dutch research chemicals,” with prices ranging from 1 g for €16.45 to 50 g for €411. Another Dutch supplier (https://www.dutchcitysales.net/product/4f-eph-crystals/), which claimed (October 2020) to be registered with the Chamber of Commerce, advertises a 20% discount, offering 1 g for €9.60 up to 100 g for €560 (October 2020 and March 2022 prices).
There appears to be no documented sales of 4F-EPH on the street, or in “smart” or “head” shops.
Legal Status
There are no international controls imposed on 4F-EPH. However, it is a controlled drug in Canada, Estonia, Finland, Germany, Lithuania, Norway, Sweden, Switzerland, Turkey, and the United Kingdom. In April 2021, 4F-EPH became controlled in Hungary (17).
The lead author (JC), as a member of the Novel Psychoactive Substances Committee of the U.K.’s Advisory Council on the Misuse of Drugs (ACMD), took part in drafting a report from the ACMD to the Home Office Minister for Vulnerability, Safeguarding and Countering Extremism sent on March 10, 2017 (18). He provided an update on behalf of the EU-MADNESS project on Scottish deaths involving methylphenidate-related molecules and specifically brought attention to 4F-EPH (paragraph 31). The Minister responded, a week later, indicating that her “officials and I are pursuing parliamentary approval for the control of these substances” as Class B drugs (19). A Home Office Circular (008/2017) was sent to police forces and other relevant agencies on June 1, 2017 drawing attention to the fact that 4F-EPH and 11 other methylphenidate-related molecules were to become Class B drugs from May 31, 2017 (20), following the passing of relevant secondary legislation (statutory instruments) under the Misuse of Drugs Act 1971 (21 -23). It is illegal to possess, produce, or supply 4F-EPH.
Experience Reports
Most of the information published on the effects of 4F-EPH is derived from first-hand personal accounts presented in discussion fora. However, Wedinos has one entry, relating to London in July 2019, containing only 4F-EPH as a white powder where the reported effects were euphoria, increased energy, increased confidence, empathy, breathlessness, and insomnia. Such effects reported by users can be helpfully summarized employing the categories utilized by PsychonautWiki (5): physical, cognitive, and aftereffects. The main ones are described below; further details are available in the Supplementary Material.
Physical Effects
Most user reports or comments compare 4F-EPH with ethylphenidate and its related analogs, especially 4F-MPH, in terms of its stimulatory properties. Although some users report it as being more euphoric than 4F-EPH others claim the contrary. It appears to be less stimulating than ethylphenidate but more recreational than 4F-MPH. Some claim to have experienced a “smooth” sensation with 4F-EPH, while others claim it made them feel “jittery.”
A sense of physical comfort and pleasant well-being are reported at moderate doses. This is similar in nature to but weaker than the feeling of euphoria generated by methamphetamine or MDMA (5). The initial “rush” caused by 4F-EPH, lasting 5 to 15 minutes, at moderate to high doses taken via intranasal, intravenous, or sublingual routes is reported to feel like a general “tingling” sensation akin to that experienced during and after orgasm. The rush may also be accompanied by a feeling that the body is lighter or floating; the sensation may continue for some time at moderate to high doses.
There are occasional comments regarding dehydration, especially regarding undissolved powder in the mouth following oral administration, especially when rubbed on the gums or taken sublingually. Regular water intake is suggested. It also can cause damage to such soft tissue. This aspect is much more commonly mentioned in terms of the nasal route of consumption. The term “caustic” is frequently used in this context but is felt to be less caustic than snorting ethylphenidate. Saline irrigation of the nasal passages is recommended.
The cardiac and circulatory systems are subject to effects including vasoconstriction in the lower limbs, a moderate increase in blood pressure compared to 4F-MPH, and increased heart rate, especially at higher doses. Pupils may dilate, especially at higher doses; tightness around the eyes has been reported.
An increase in libido is reported at low to moderate doses, but a decrease can be noticed, especially at higher doses. Higher doses may also induce suppression of orgasm, mainly in male consumers; the latter may also experience temporary erectile dysfunction.
Cognitive Effects
A range of effects can be included under this heading. A lower “cognitive boost” is reported compared to other “phenidates.” Some users just report an experience of wakefulness, but this is considered less strong than “methylphenidate, amphetamine, and most other dopaminergic stimulants” (5).
“Thought acceleration” is regarded as mild to moderate, tending “towards semantic, emotional, social, and abstract thought patterns” (5). Occasionally users report some degree of confusion, with a noticeable decline in understanding basic concepts and remembering simple sequences. Analytical ability may be enhanced by 4F-EPH but only for routine administrative tasks rather than logical activities such as mathematics.
Some users of 4F-EPH report being energized and able to tackle routine household chores or mundane tasks without losing focus or becoming distracted. In this sense, it is regarded as a “functional” stimulant. Some argue that it may be useful for studying, writing papers, and so on. It also appears to enhance abstract and creative tasks and listening to music.
Motivation does not appear to be generated by this molecule. However, empathy, affection for others, social engagement, and emotional positivity are promoted. This may be, in part, due to the mood lift and euphoria it causes.
Aftereffects
As with other stimulants, there is a sense of “comedown” when the effects of 4F-EPH wear off due to the lower levels of neurotransmitter activity. Typical “comedown” effects experienced with stimulants include anxiety, cognitive fatigue, depression, irritability, motivation suppression, thought deceleration, and wakefulness (5). An additional effect noted by several 4F-EPH users was paranoia; one individual stated it lasted one hour.
First-hand or anecdotal reports can be further examined for information regarding routes of administration and dosages, onset and duration of effects, redosing, desired psychoactive effects, acute physical effects, other side effects, and coingested substances. These are examined in the following subsections.
Routes of Administration, Dosage, and Duration of Effects
According to user reports, 4F-EPH is typically taken orally (swallowed) or insufflated (snorted/sniffed). Other routes of administration include rectal (“plugging”), intravenous injection, vaping, sublingual, and rubbing on gums. Some (would be) users have asked their peers for advice about taking the molecule in other ways, for example, vaping in an e-cigarette; suggestions made include “parachuting” (wrapping in a cigarette paper and swallowing); “oral in a Pg (propylene glycol) solution”; “Get a 0.001 mg scale and gel cap your doses. Either you can take them as pills or open the capsule again later and snort straight from the capsule…”; “make a g-damn nasal spray with a water solution.”
Oral dosages are given at the following levels: threshold < 5 mg; light 5 to 10 mg; common 10 to 30 mg; strong 15 to 50+ mg; and heavy > 30 mg. Some cite a dosage of 65 mg over four hours. Nasal dosages are typically described as “bumps,” starting with 2.5 mg up each nostril and building up to 25 mg. A “large bump” is described as 30 mg.
The onset of effects when taken orally takes between 10 and 25 minutes; nasal administration can lead to effects being experienced within 3 minutes. The effects are reported to last four to six hours, with a peak around two to three hours. Residual or aftereffects can last from 1 to 12 hours.
Toxicity and Harm Potential
As there has been no scientific study of the long-term health effects or toxicity of 4F-EPH, combined with only a probably very limited consumption by humans, it is not possible to state what these might be.
Tolerance and Addiction Potential
Users report the development of tolerance to 4F-EPH being more rapid than with ethylphenidate and methylphenidate. They report a need to redose (even at low doses) or the use of higher doses to attain the same level of experience as with their first experimentation with 4F-EPH. Such measures result in feeling jittery, paranoid, and uneasy. One user claims it is “extremely dangerously addictive.” PsychonautWiki (2020) suggests that this molecule produces cross-tolerance with other dopaminergic stimulants.
Dangerous Interactions
There is little or no information specifically mentioned with regard to interactions between 4F-EPH and other substances. One Reddit contributor notes that taking a combination of 150 mg of 4F-EPH with 300 mg of magnesium citrate leads to “mania.” The general advice offered is to start with small doses and increment slowly.
Hospital and Emergency Room Presentations
The introduction of controls on ethylphenidate within the United Kingdom in 2015 (Temporary Drug Class Order) and 2016 (Psychoactive Substances Act) led to a significant reduction in hospital admissions in Scotland (24). It is likely that this may have also impacted on the use of ethylphenidate analogs such as 4-fluoromethylpheniate (4F-MPH) and 4F-EPH. The subsequent control of these analogs may also have restricted their appeal and use.
There are no documented cases in the academic literature of intoxications, overdoses, poisonings, presentations or admissions to hospitals, emergency departments (EDs) or rooms, or calls to poisoning centers involving 4F-EPH. However, there are reports of cases for methylphenidate and several of its analogs (see Discussion section below). TOXBASE suggests that due its structural similarity with ethylphenidate and 4-fluoromethylphenidate, clinical features for 4F-EPH are likely to be consistent with those of amphetamines and similar stimulants (4). However, there is a nonfatal case report concerning 4-fluoromethylphenidate (4F-MPH) a closely related analog (25). There are also case reports of deaths involving 4F-MPH and ethylphenidate (26,27).
No definitive concentrations of 4F-EPH have been established previously for toxic effects or death in animals or humans. There have been no previously reported deaths involving this molecule.
Fatality Case Report
The latest list of molecules regarded as NPS by the Office for National Statistics does not include 4F-EPH, see Box 4 in Definition sheet of their latest dataset (28). This same list is also used in Northern Ireland (29). The specific omission from these lists of 4F-EPH indicates that it has not yet been mentioned in the cause of death fields on medical death certificates in England, Wales, and Northern Ireland. To date, there is also no mention of this molecule in any of the publications from the National Programme on Substance Abuse Deaths based at St George’s University of London.
As part of the European Commission funded EU-MADNESS project (30), led by the University of Hertfordshire (Principal Investigator = FS), the leader (JC) of the work stream on drug-related deaths established a formal arrangement with the NRS, among other agencies, for the quarterly receipt of anonymized data on drug-poisoning deaths. Data for deaths registered in Q1-3 2016, received by JC in December 2016, included a death due to “Complications of MDMA/MDA and 4F-EPH toxicity.” Through the good offices of the NRS, contact was made by JC with the pathologist in the case, and with the appropriate consent of the Crown Office and Procurator Fiscal Service in Scotland, as the decedent had died “whilst in police custody.” Material published following a Fatal Accident Inquiry held into the death and in response to Freedom of Information [FOI] requests by the lead author (JC; i.e., FOI 2020 168 and FOI R024116), together with additional information provided by the pathologist provide the basis for the following case report (31).
Extensive searches of academic literature and the Internet reveal no other fatalities involving 4F-EPH.
Circumstances of Death
The deceased had a history of substance misuse. He had consumed a number of (unspecified) legal and illegal substances in the days prior to his death. These substances had a detrimental effect on his health and well-being. On July 11, he left [home?] under the influence of these substances. As a consequence of his behavior, members of the public contacted the police to report his actions, that is, causing a disturbance in a town center.
At about 16:45 that day police attended the locus where he had been detained by members of the public and arrested him. He was not charged with any offence. He was taken to a police station where he was assessed as unsuitable for detention in a police station.
He had reportedly been aggressive and kicking. The pathologist considered that such a situation suggested elements of stress that could well be associated with various physiological processes, generally causing further stimulatory effects, thus potentially increasing the risk of complications such as increased temperature and cardiac dysrhythmias. Elements of “excited delirium” could be present albeit this is a clinical diagnosis, which was not documented in the information available to the pathologist. In addition, the position of the decedent in the police vehicle prior to his collapse was not clear, albeit it would appear that he was likely sitting down while awaiting his turn to be triaged in the hospital. Although a sitting position would not be generally associated with significant impairment of his lung functions, it could arguably, depending on his body habitus, cause at least some degree of restriction of his breathing abilities that could in part impact on his risks of fatal complications. Taking all this into consideration along with the history and findings at autopsy and in view of the mechanisms of death noted above, there appeared to be no sufficient pathological evidence to implement with certainty the role, if any, of the restraint and body position in his death, although this could not be fully ruled out.
He collapsed and was, therefore, taken to a hospital in the same town and became unresponsive. He exhibited hypothermia, rhabdomyolysis, hepatic, and renal failure. Life was pronounced extinct, in the Intensive Treatment/Care Unit, at 17:25 on July 15 (three days after admission).
An inquiry was conducted into his death by the Police Investigations and Review Commissioner (PIRC) on behalf of the Crown, but no charges were brought against any individual (32). A FOI request was made by the lead author (JC) to PIRC, but no further information about events leading up the decedent’s arrest and detention was released (personal communication to lead author by PIRC, April 29, 2021).
Autopsy Investigations
An autopsy took place on July 20, 2016 (six days after death). The deceased was a White male aged 24 years. He was 1.90 m in height, weighed 103 kg, and had a body mass index of 28.5.
External findings of note were multiple red marks; hematoma; bruises on face, trunk, and upper and lower limbs. The deceased had areas of skin mottling and patchy hemorrhage, likely to be in part the result of his multiorgan failure and coagulopathy. There were, however, localized blunt-force injuries identified in particular on the forearms and the lower legs that were further explored and were found to be associated with underlying soft tissue injury. These would correlate with, but were not associated with, any significant damage or injury to structures or organs that would have directly caused or led to his death. A number of other blunt-force injuries including bruising and abrasions were noted on the body that could have occurred as a result of various mechanisms such as falls, impact with blunt objects or surfaces or other also potentially in part occurring during the restraint procedure. The deceased was reportedly handling a knife at various points. There were no findings at autopsy to suggest that sharp force injury has played a role in his death.
There was evidence of liver failure as well as kidney damage associated with collections of breakdown products of his skeletal muscles as noted clinically. Moderate autolysis of liver with evidence of prominent zone 3 necrosis with a background of mild to moderate preexisting hepatic steatosis was found. The proximal jejunum and spleen were congested.
The postmortem and histology results revealed congested lungs with localized marked edema. Histology also demonstrated patchy intra-alveolar hemorrhage and established bronchopneumonia; the latter was likely a terminal event.
The aorta showed mild atheroma. Neuropathology and extensive heart sampling showed no significant pathology. The latter would not preclude the possibility of a preexisting cardiac disease, albeit there were no suggestions on pathology grounds that this played a role in his death.
In view of the history of drug use over some time before the decedent’s collapse, it could be that the various terminally fatal mechanisms such as muscular, liver, and kidney insults could have arguably been initiated in the days or weeks prior to his hospital admission.
Toxicological Findings and Interpretation
Antemortem toxicology findings were: blood—alcohol 21 mg/dL, 4F-EPH 0.36 mg/L, MDMA < 0.4 mg/L, MDA < 0.4 mg/L; serum—4F-EPH 0.27 mg/L. Postmortem femoral blood, urine, and hair samples were taken for toxicology. These yielded the following results: blood—morphine 0.17mg/L, total morphine 0.21mg/L, 4F-EPH 0.078 mg/L, MDMA 0.15 mg/L, MDA < 0.1 mg/L, lignocaine 0.17 mg/L; urine—4F-EPH detected, morphine 0.36 mg/L. Unknown samples taken from the locus where he was arrested showed 4F-EPH in the form of white powder in a clear plastic packet and also in a clear plastic tub; two yellow amitriptyline tablets in a part blister pack; two plastic packets containing MDMA crystals; residue in five plastic packets contained combinations of MDMA, cocaine, and delta-9-THC; MDMA and delta-9-THC; and 4F-EPH, MDMA, and delta-9-THC.
Interpretation of toxicology results: The traces of alcohol in the hospital blood could well represent postmortem production, albeit this could reflect the use of alcohol particularly in view of the history provided. The postmortem morphine levels are consistent with its administration in hospital. The lignocaine level detected is consistent with use as a local anesthetic.
The most significant toxicological findings were the presence of MDMA, MDA, and 4F-EPH. MDMA and its metabolite MDA were noted at subfatal levels in the postmortem and antemortem hospital samples, albeit these were not possible to fully quantify in the hospital sample.
MDMA/ecstasy is thought to cause death through a number of mechanisms. These include the increased risk of hyperthermia/pyrexia and associated complications (such as organ failure and breakdown of skeletal muscle) as noted in the well-documented clinical history in this case. Other side effects such as neurotoxicity (including serotonin syndrome) and effects on the cardiovascular system have also been documented. Indeed, coadministration of two stimulants (i.e., amphetamine-type stimulants, such as MDMA or MDA, and/or cocaine) might increase both serotonergic and dopaminergic stimulation, through a synergic effect, so that serotonin syndrome is more likely to happen (33).
The antemortem and postmortem levels for 4F-EPH are the first recorded in the academic literature, or indeed reported anywhere, so cannot be interpreted by direct comparison with other cases; the toxicology laboratory, therefore, did not attempt to interpret them. As noted above, little is known about the effects of 4F-EPH.
On the other hand, ethylphenidate, the close analog of 4F-EPH, has been found to produce similar side effects to the amphetamines including the increased risk of pyrexia, tachycardia, paranoia, and tremor, which in turn would have complemented the effects of MDMA/MDA, thus potentially also playing a major role in this decedent’s fatality.
Cause of Death
Based on the evidence outlined above, the pathologist concluded that the cause of death “would appear to be the result of the complications of psychostimulant drug use in particular MDMA/MDA and potentially 4F-EPH with the possibility of concurrent ingestion of alcohol. These led to an increase in…body temperature associated with symptoms of paranoia and hallucinations and complicated by muscular breakdown, kidney and liver failure and pneumonia. The cause of death should therefore be…1a. Complications of MDMA/MDA and 4F-EPH toxicity.” Cause 2 was given as “drug abuse.”
This cause of death was formally handed down by a Fatal Accident Inquiry concluded on August 28, 2018. This was the wording subsequently recorded in the NRS database, where the event was coded and registered as an accidental drug poisoning.
Discussion
Effects of MDMA
Acute consumption of ecstasy (MDMA, MDA) may give rise to a spectrum of physical complications including tachycardia, arrhythmias, asystole, hypertension, cerebral hemorrhage, thrombocytopenia, convulsions, coma, mydriasis, vomiting, diarrhea, metabolic acidosis, rhabdomyolysis, disseminated intravascular coagulation, and acute kidney failure. Increased core body temperature may be induced both by amphetamine-type stimulant consumption and by environmental factors. This can lead to excessive consumption of hypotonic fluids, which in turn can lead to hyponatremia (34).
It is also argued that acute ecstasy consumption leads to serotonin syndrome (35). This appears to be caused, in ecstasy users, by 5-HT1-like and 5-HT2 receptor stimulation induced by MDMA, leading to heightened central serotonin levels (36). Symptoms of this syndrome include mental confusion; agitation; hyperreflexia; incoordination; bruxism; trismus; enhanced physical activity; hypotension, hyperthermia; sweating; shivering; seizures, tremor, and nystagmus; rhabdomyolysis; metabolic acidosis; and myoclonus (34,37,38).
Serotonin syndrome is a possible factor in this combined MDMA/MDA and 4F-EPH-related death. Some of these symptoms are noted to have occurred in respect of the decedent. This suggests, therefore, that an increase in serotonin had occurred especially as no other drugs were detected that could have triggered such an event.
General noradrenergic/dopaminergic receptor stimulation may be responsible for symptoms such as hypertension, overarousal, and tachycardia (39). Concomitant administration of dopaminergic stimulants can enhance release of serotonin (40). More serious adverse consequences, such as delirium, seizures, and coma, are more likely to be experienced if MDMA is coingested with amphetamines and/or cocaine (41). Ecstasy-related deaths are more likely than amphetamine/methamphetamine-related deaths to have alcohol and cocaine detected than the latter, which are more likely to be found in combination with opiates/opioids (42).
Lethal blood levels of MDMA vary considerably where death is due to polydrug use: mean 2.90, median 0.76, range 0.04 to 41.5 mg/L. In fatalities where MDMA is associated with trauma, the average and range were somewhat lower: mean 0.862, median 0.483, range 0.035 to 4.81 mg/L. As might be expected, where MDMA is the sole drug implicated in death, these parameters are higher: mean 8.43, median 3.49, range 0.478 to 53.9 mg/L (43). The considerable overlap in the ranges of these concentrations indicates that as with other stimulants, an MDMA level on its own is insufficient to determine the cause of death. Drug use history, full circumstances of the death, and autopsy findings need to be considered (43).
The postmortem blood level of MDMA in the present case was 0.15 mg/L. This is toward the lower end of the ranges reported by Milroy for polydrug use and trauma deaths, but well below that seen for sole MDMA toxicity fatalities (43).
Effects of “Phenidate” Drugs
Generally, the stimulatory effects of 4F-EPH are considered to be weaker than those of ethylphenidate, although some users feel it is smoother. On the other hand, some individuals found themselves developing tolerance more quickly and having to redose or use higher doses to achieve the same effect.
At the time of writing, no poisonings or deaths, other than the case described here, involving 4F-EPH had been reported in the scientific literature, although Carlier et al. do refer to it (44). Indeed, this article is believed to be the first such article to describe a fatality in which the presence of 4F-EPH was recorded, let alone implicated. There is no relevant pharmacological information on this drug in terms of lethal dosage, LD50, half-life, volume of distribution, and so on for either animal or human subjects. It is not possible to determine from a single blood sample either when a substance was consumed or the exact amount taken. Because of the lack of comparative data, it was neither possible to relate the 4F-EPH concentrations reported here to “recreational” or excessive use, nor to comment as to the toxicological significance of the concentrations. This article now provides some insights into the potentially fatal toxicity of 4F-EPH.
Methylphenidate Intoxications and Deaths
The undesirable effects of 4F-EPH appear similar to those produced by methylphenidate (MPH) (44,45). The main adverse effects in acute poisonings include: (1) central nervous system—anxiety, confusion, delirium, and hallucinations; (2) cardiovascular system—arrhythmia (atrial and ventricular), chest pains, hypertension, palpitations, and tachycardia—these may lead to coma, intracranial hemorrhage, and myocardial infarction/stroke; (3) effects on muscles—contraction, bruxism, and trismus; (4) fasciculation and rhabdomyolysis can lead to renal failure; and (5) hyperpyrexia (> 40 °C) leading to disseminated intravascular coagulation, sweating, and flushing. These groups of effects are commonly noted in poisonings involving amphetamine-type stimulants. Long-term use leads to the development of tolerance and the need to redose. Psychosis may also develop; symptoms typically include delusions, hallucinations, and paranoia. In turn, this may lead the chronic user to become anxious and suspicious generating aggression, hostility, and violent behavior. There are no user reports of 4F-MPH being used with MDMA.
Deaths involving methylphenidate have been ascribed to several mechanisms: intracranial hemorrhage, acute heart failure or arrhythmia, hyperpyrexia, rhabdomyolysis and consequent hyperkalemia or renal failure, and to violence related to its psychiatric effects (46). The ingestion of doses as low as 1.3 mg/kg has caused death (45). Methylphenidate blood levels >50 ng/mL are considered toxic (47,48). The first death involving the ingestion of methylphenidate reported central and peripheral blood levels of 980 and 1100 ng/mL, respectively (49).
Ethylphenidate Poisonings and Deaths
There are few academic papers dealing with poisonings and deaths involving ethylphenidate and analogs related to 4F-EPH; all of these relate to Europe. Three U.K. cases of acute ethylphenidate toxicity were reported by Bailey et al (50). Analytical confirmation was only available for one case, a 21-year-old male who had taken a cumulative total of 500 mg; the blood concentration 20 hours later was 0.24 mg/L. The authors highlighted the following symptoms similar to amphetamine-type stimulant use: agitation, bruxism, chest pain and palpitations, nasal irritation, and pain. Tachycardia was noted in two of these cases. Hypertension, dilated pupils, fever, anxiety, paranoia, and tremor were also noted.
The first death involving ethylphenidate was reported from Germany (51). However, the deceased had mitral valve endocarditis and pneumonia. The postmortem femoral ethylphenidate blood level of 0.1 mg/L may have been sufficient to affect the cardiovascular system.
Parks et al. documented 19 deaths in Scotland that occurred between July 2013 and December 2014 (52). Mean age at death was 37 (range 20-54) years; 14 cases were male; 16 cases came from Edinburgh and its environs. Average postmortem ethylphenidate femoral blood levels were: mean 0.39, median 0.25 (range 0.008-2+) mg/L. Other common substances identified were benzodiazepines (15), opiates (11), methadone (8), antidepressants (6), and antipsychotics (2). Drug toxicity was the sole or a potentially contributory factors in ten deaths, ethylphenidate being specifically cited in five cases. In no instance was ethylphenidate the sole drug detected.
Maskell et al. reported seven deaths from England that occurred between February 2013 and January 2015 where ethylphenidate was detected and quantitated (27). Four cases were ascribed to mixed drug toxicity and one to ethylphenidate toxicity; two were hangings. All decedents were male; median age was 25 (range 23-49) years. Opiates/opioids were present in three of the four mixed drug deaths. Other stimulants were present in three cases.
Ethylphenidate blood concentrations in the four mixed drug fatalities ranged from 0.03 to 0.14 mg/L; the two hanging cases have concentrations of 0.87 and 1.37 mg/L; the case involving only ethylphenidate had a concentration of 2.18 mg/L. When taken in the context of the overdose reported by Bailey et al (50), there may well be an overlap between “therapeutic”/“recreational” and toxic levels of ethylphenidate (27).
4-Fluoromethylphenidate Intoxications and Deaths
Papa et al. note that online user reports suggest that 4-fluoromethylphenidate (4F-MPH; 4F-TMP) is more potent than both methylphenidate and ethylphenidate but with less common adverse side effects such as anxiety, compulsive redosing, and muscle spasms. There are few documented cases of intoxications or deaths involving 4F-MPH (25).
The first intoxication involving 4F-MPH involved a female aged 26 years (25). She was admitted to an ED with the following symptoms: confabulation, confusion, crying, disorientation, incoherent speech, severe psychomotor agitation, palpitations, and tachycardia (100 bpm). With the addition of insomnia, these symptoms persisted for a week while she had been insufflating powder 4F-MPH acquired online. Normal admission vital signs included body temperature (36 °C) and blood pressure (120/75). Intravenous diazepam was given for sedation, with agitation subsiding within a day of admission; tachycardia persisted for two days. Discharge was allowed after two days with a prescription for quetiapine and promazine. Emergency department admission levels of 4F-MPH were: blood 32 ng/mL, urine 827 ng/mL.
4F-MPH was detected in the blood of three individuals intoxicated with NPS tested between 2015 and 2018 in a Polish department of clinical and forensic toxicology (53). One occurred in 2016 and two in 2018. Unfortunately, no further details are provided. A case report from another Polish institution indicates the probable consumption of 4F-MPH by a 28-year-old male with a ten-year history of drug abuse (54). According to his parents, he admitted the use of several drugs before losing consciousness and collapsing on the floor. On the way to hospital, his condition worsened with progressive respiratory failure and vomiting. On arrival, he was unconscious and had to be mechanically ventilated for three days. Tachycardia (120 bpm) was present; blood pressure was within the normal range (120/80). Aspiration pneumonia was diagnosed, along with mild acidosis, anemia, rhabdomyolysis, leukocytosis, high levels of C-reactive protein, and creatine kinase; in time, these all improved. During his first day in hospital, the patient was “partially disoriented, had formal thought disorders, inappropriate affect and cenesthetic hallucinations.” The emergency responders found a few pouches with tablets and herbs at the scene; these had labels like “4F-MPH,” “Bromazepam,” “Flunitrazolam,” and “mild cuddler” (probably synthetic cannabinoids). Toxicological examination of the male’s blood revealed traces of methylphenidate and methamphetamine; blood serum was positive for benzodiazepines (140.5 ng/mL). His pneumonia was successfully treated with antibiotics, and he received tranquilizers for his psychiatric issues. He was discharged after ten days, with further ambulatory therapy.
The only death involving 4F-MPH is reported by Shoff et al (26). They report that although this molecule had first been detected in their area in drug paraphernalia, the first death involving it occurred in 2018. A White male aged 25 years was experiencing paranoia, hearing voices, and seeing imaginary people. When his mother went to check on him the next morning, she found him unresponsive in bed; he was pronounced dead at the scene. No drugs paraphernalia were found, although the decedent was a known methamphetamine user. Key features noted at autopsy were edematous lungs, swollen brain, and distended bladder. Postmortem urine was positive for amphetamine and methamphetamine; cannabinoids; morphine, monoacetylmorphine (6-MAM), codeine, norcodeine; naproxen; diphenhydramine; and 3-methoxyphencyclidine (3-MeO-PCP). Gastric contents were positive for amphetamine and methamphetamine; 6-MAM, codeine, acetylcodeine; naproxen; diphenhydramine; and threo-4F-MPH (0.795 mg). Femoral blood was positive for amphetamine (0.135 mg/L) and methamphetamine (0.850 mg/L); morphine (0.268 mg/L), codeine (0.022 mg/L), papaverine, 6-MAM (1.835 ng/mL); diphenhydramine; 3-MeO-PCP; and threo-4F-MPH (0.012-0.049 mg/L). It is likely that the decedent suffered an acute heroin overdose. However, the effects of codeine, methamphetamine, 3-MeO-PCP, and 4F-MPH may have contributed to the death.
Comparison of Effects of 4F-EPH With Selected Amphetamine-Type Stimulants
Based on the user reports presented above, 4F-EPH has effects which are like those experienced by users of methylphenidate, ethylphenidate, 4-fluoromethylphenidate, as well as MDMA.
Treatment of individuals presenting to EDs who believe they have or are known to have consumed 4F-EPH should be in line with standard practice/protocols for dealing with amphetamine-type stimulants.
Comparative Toxicity of 4F-EPH
The blood level of 4F-EPH in the death described here was 4F-EPH 0.078 mg/L. This compares with methylphenidate blood levels >0.050 mg/L being considered toxic, and levels of central and peripheral blood levels of 0.980 and 1.100 mg/L reported by Cantrell et al (49). The present case is higher than the toxic level for methylphenidate but lower than lethal levels.
Lethal ethylphenidate (EPH) blood levels have been reported as follows: 0.1 mg/L (51); mean 0.39, median 0.25 (range 0.008-2+) mg/L (52); mixed drug fatalities 0.03 to 0.14 mg/L; hangings 0.87 and 1.37 mg/L; ethylphenidate alone 2.18 mg/L (27). The present case is within the levels for mixed drug fatalities involving ethylphenidate.
4-Fluoromethylphenidate (4F-MPH) levels of 0.012 to 0.049 mg/L in mixed drug toxicity deaths were reported by Shoff et al (26). The present case is higher than the 4F-MPH levels.
Given that the MDMA blood level in this case was comparatively low (0.15 mg/L), it is safe to conclude that 4F-EPH did contribute to this death. Moreover, there was no natural disease present to cause or contribute to the death.
Information Needs
Even with 27 documented cases of ethylphenidate-related deaths outlined above, more information is needed about its pharmacology and toxicology (27). How much more so for its analogs such as 4-MPH and 4F-EPH!
Fully appreciating the toxicological ramifications of a single novel NPS is problematic but the potential contribution of such molecules to fatalities cannot be overlooked (26).
Since analogs of methylphenidate are not included in regular screens for drugs of abuse, the number of intoxications, poisoning, and deaths involving such molecules is likely to be underestimated (25). It is important that ED staff and forensic toxicologists do not overlook the possibility of the ingestion of ethylphenidate analogs. Reference standards are available for many of these, including 4F-EPH, facilitating quick identification.
It is essential that, wherever feasible, full and detailed information regarding the circumstances leading to hospitalization or death is gathered. This will enable appropriate toxicological investigations and, if necessary, medical interventions to be undertaken.
Strengths and Limitations of This Study
This article has reviewed the scanty information available from online user reports about the experiences of those consuming 4F-EPH and the effects it engenders. Together with the detailed presentation of a case report of the first death involving this molecule, our research puts into the public domain the best information available at present. It can provide a basis for future in vivo, in vitro, and in silico research on the abuse potential, pharmacology, toxicity, and so on of a little known and understood NPS.
Due to legal restrictions in this case, only limited information is available on the circumstances of the decedent’s behavior leading up to his detention and subsequent events leading up to his death. A more detailed account of this period could provide insight into his behavior and whether it demonstrated the effects of drug consumption.
Conclusions
There is a significant lack of information on 4F-EPH in terms of its pharmacology, pharmacokinetics, dose, acute toxicity, and harms caused by long-term use. This article has outlined what is currently known about the toxicity and lethality of this molecule. There is anecdotal evidence now emerging of serious clinical issues arising from its use; we have described what is believed to be the first death anywhere involving 4F-EPH. Based on the limited reliable evidence currently available, management of patients presenting with acute toxic effects from 4F-EPH should remain pragmatic and be in line with treatments employed for stimulants such as methylphenidate/ethylphenidate, amphetamine, cocaine, and MDMA.
Due to the relatively recent emergence of this chemical on the recreational drug use scene, its lack of widespread use, unknown toxicity, and lack of detection through routine toxicological screens in many forensic toxicology laboratories, clinicians could miss use of this substance. Similarly, fatalities caused by its use may not be identified by those investigating sudden deaths with no apparent cause(s). Determining the role and significance must be provisional and delivered with caution, qualifications, and reservations, as done in this case. Even if its use may be limited, as appears the case from user reports, 4F-EPH should not be ignored—least of all because of its potential to cause and/or contribute to (fatal) toxicity.
This molecule has been detected by forensic providers in the last year and is still available (November 2022) to buy online, so still has the opportunity to cause harm. The information presented here may help with future interpretation of toxicological interpretation of 4F-EPH levels.
Supplemental Material
Supplemental Material, sj-xlsx-1-afp-10.1177_19253621221142480 - First Death Involving 4-Fluoroethylphenidate (4F-EPH): Case Report, User Experiences, and Review of the Related Literature
Supplemental Material, sj-xlsx-1-afp-10.1177_19253621221142480 for First Death Involving 4-Fluoroethylphenidate (4F-EPH): Case Report, User Experiences, and Review of the Related Literature by John Martin Corkery and Fabrizio Schifano in Academic Forensic Pathology
Footnotes
Acknowledgments
The authors thank the recently retired statistician Frank Dixon and his colleagues in the Vital Events Branch, National Records of Scotland (NRS), for the provision of data on drug-related poisoning deaths to the lead author as part of the European Commission-funded EU-MADNESS project (Drug Prevention and 2 Information Programme 2014 16; contract no. JUST/2013/DPIP/AG/4823). Without this collaboration, the significance of the death involving 4F-EPH described here may not have been recognized. Special thanks are due to the pathologist in the case report, Dr. Ralph Bouhaidar for liaising with the Crown Office and Procurator Fiscal Service regarding provision of detailed toxicological and postmortem information. The Crown Office and Procurator Fiscal Service provided access to redacted copies of autopsy and toxicology reports, and the Scottish Courts and Tribunal Service provided information on a Fatal Accident Inquiry. Access to the European Database on New Drugs (EDND) was provided by the European Monitoring Centre for Drugs and Drug Addiction in Lisbon, prior to Brexit in early 2021.
Data Availability Statement
All available data are presented in Supplementary Material.
Ethical Approval
The University of Hertfordshire’s Health, Science, Engineering and Technology Ethics Committee with Delegated Authority confirmed on August 7, 2018 that such research forming part of the lead author’s PhD research does not require ethical approval.
Statement of Human and Animal Rights
N/A.
Statement of Informed Consent
N/A.
Disclosures & Declaration of Conflicts of Interest
This work has not been previously published and has not been submitted for publication elsewhere. Publication is approved by all authors and the responsible authorities where the research was undertaken. If accepted, the paper will not be published elsewhere in the same form, in English or in any other language, without the written consent of the copyright holder. No conflicts of interest are declared here that may have influenced the interpretation of present data. Please note the following: FS was a full member of the U.K. Advisory Council on the Misuse of Drugs (ACMD) during the period when 4F-EPH was recommended for control under the Misuse of Drugs Act 1971 by the Council; JC is a member of the ACMD’s Novel Psychoactive Substances and Technical Committees. Both FS and JC are former members of the National Programme on Substance Abuse Deaths (NPSAD); JC was cofounder of NPSAD in 1997 and Programme Manager from 2005 to 2010). The views expressed here reflect only the authors’ views and not necessarily those of the Home Office, the ACMD, the authors’ employers, or the relevant judicial authorities and officers in Scotland.
Financial Disclosure
The authors have indicated that they do not have financial relationships to disclose that are relevant to this manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
