Abstract
Importance
Septoplasty, turbinoplasty, and septorhinoplasty are common and generally-safe procedures. However, their effects on the olfactory system, despite its anatomical proximity, are not well documented. Given the importance of olfaction for quality of life, evaluating the olfactory impact of these surgeries is warranted.
Objective
This study aimed to assess the olfactory sequelae of nasal surgeries.
Design
Systematic review with meta-analysis
Setting and Participants
A search equation used in PubMed/Medline, Cochrane Library, Web of Science, and Embase identified 7780 articles published since 1990, evaluating the adverse events of nasal surgeries.
Exposure
The studies included evaluated adult patients exposed to one of the following nasal surgeries: septoplasty, septorhinoplasty, and/or inferior turbinoplasty.
Main outcome measures
Olfactory assessment results and number of patients presenting with preoperative or postoperative hyposmia were extracted, as well as the number of patients presenting with surgery-induced hyposmia.
A meta-analysis was conducted to compare different surgical subgroups.
Results
One hundred and two studies were ultimately included in the analysis, centralizing data from 16,072 patients. Surgery-induced hyposmia was identified in 2.7% of cases, regardless of the surgical intervention performed. Overall, nasal surgeries led to a postoperative improvement in hyposmia scores [3.73 (±1.58) vs 2.39 (±1.45), P < .001] and a reduction in the number of patients with hyposmia [22.2% (945/4254) vs 14.5% (380/2620), P < .001]. The meta-analysis revealed no significant difference between the surgical subgroups.
Conclusion and Relevance
The present systematic review and meta-analysis suggest that nasal surgery may impair olfaction in a non-negligible number of cases and highlight the need for objective and systematic olfactory assessment in nasal surgery studies.

Key messages
Olfaction should be assessed pre- and postoperatively in nasal surgery using psychophysical assessment.
The present study confirms that nasal surgery improves the sense of smell for a majority of patients.
The present study also suggests that nasal surgery may impair olfaction in a non-negligible number of cases [2.7%] and that patients should be warned.
Introduction
Nasal surgery, whether only functional or associated with an esthetic procedure, aims to optimize nasal airflow; it is considered safe with an approximate 3% complication rate following septoplasty. 1 Theoretically, improving nasal patency should not induce olfactory dysfunction [OD], as olfaction capabilities depend on the proper functioning of the olfactory epithelium, which covers the upper parts of the nasal septum, the superior turbinate, and the middle turbinate, and remains untouched during nasal surgery. 2 Yet, nasal surgery is performed near the olfactory epithelium, and there is some uncertainty regarding the precise distribution of olfactory filaments within the nasal fossa. 3 OD impacts not only quality of life and well-being but also the ability to detect dangers such as smoke or spoiled food,4 -6 and is thereby associated with increased mortality. 7 OD is common, affecting 16% of the general population 8 and at least 2.7 millions of Americans,9,10 and several tests have been developed for its assessment. Subjective evaluations include questionnaires or visual analog scales [VAS], and standardized assessments include tests like the Sniffin’ Sticks Test [SST],11,12 the University of Pennsylvania Smell Identification Test [UPSIT], 13 and the European Test of Olfactory Capabilities [ETOC]. 14 One of the mechanisms of OD is conductive dysosmia, which can be caused by nasal obstructions due to septal deviation or turbinate hypertrophy.15,16 Thus, septorhinoplasty, septoplasty, or turbinoplasty 17 may either improve olfaction by removing these obstructions—thereby enhancing particle access to the olfactory cleft18,19—or degrade it by affecting receptor areas because of the proximity of the olfactory epithelium.
Current literature on the postoperative impact of nasal surgeries on olfaction remains inconsistent. Some studies report postoperative improvement in olfaction,20,21 others find no significant change, 22 and others describe a deterioration in olfactory function.23 -27 Moreover, there is a lack of data regarding the onset and duration of OD: hyposmia is sometimes described as transient, with olfaction improving spontaneously within 2 to 12 months,23,24 while other authors report persistent postoperative anosmia in 1% to 2% of cases.25 -27
The primary objective of this systematic review and meta-analysis was to assess the olfactory sequelae of nasal surgeries, including septorhinoplasty, septoplasty, and inferior turbinoplasty.
Methods
The systematic literature review was conducted and reported following the Preferred Reporting Items for Systematic Review and Meta-Analyses [PRISMA] guidelines 28 to ensure a reproducible research strategy and statistical analysis. The detailed protocol for the systematic review is registered on PROSPERO [CRD42023487678].
Information Sources
Studies were identified using the PubMed/Medline [US National Library of Medicine], Cochrane Library [Wiley], Web of Science [Clarivate], and Embase [Elsevier] databases, with searches conducted on November 28, 2023. Additional references were sourced from the reference lists of relevant articles. Automated alerts were set for each database to monitor the publication of potentially-relevant studies during the drafting and analysis phases. These alerts were discontinued on February 18, 2025.
Search Strategy
The search equations were developed based on Medical Subject Headings [MeSH], abbreviations, and free-text terms related to olfactory disorders following nasal surgery. Keywords included, but were not limited to, terms such as “anosmia,” “hyposmia,” “septorhinoplasty,” “septoplasty,” and “turbinoplasty.” Only studies published in English or French since 1990 were included. The search equations were created with assistance from a research librarian at the University Hospitals of Lyon [C.G.] and are detailed in Supplemental Material 1.
Selection Process
Results from the queried databases were imported into the Covidence systematic review software [Veritas Health Innovation, Melbourne, Australia], which automatically removed duplicates. Screening was conducted by 2 investigators [M.F. and A.F.] in 2 phases: initially by title and abstract, followed by full-text review. Case series with fewer than 10 patients and systematic reviews were excluded. In case of disagreement, a meeting was held to achieve consensus [M.F. and A.F.]. Inclusion criteria required an olfactory assessment or a reported proportion of hyposmic patients, both preoperatively and postoperatively in adult patients undergoing nasal surgery, or the mention of the number of patients in whom hyposmia was induced by nasal surgery. Interventions involving middle turbinate surgery, sinus surgery or patients with chronic rhinosinusitis, nasal valve treatment with LATERA® insertion [Stryker, Amsterdam, the Netherlands], sleep apnea treatment, or septal perforation repair were excluded. Studies in which postoperative olfactory measurement was performed less than 1 month after surgery were also excluded. The PRISMA flowchart created with Covidence (Figure 1) details the transparent phases of the selection process.

Flow chart representing the identification, screening, and inclusion of studies.
Interventions
While some studies only focused on 1 type of surgery, others included different groups based on the type of surgery. For the present systematic review and meta-analysis, we chose to analyze the data according to intervention group rather than study. To that end, studies were divided into intervention groups according to the surgery type: septorhinoplasty, septoplasty, and inferior turbinoplasty. For septorhinoplasty and septoplasty combined with an adjunctive inferior turbinoplasty, only septorhinoplasty or septoplasty procedure was considered for consistency.
Data Collection Process and Outcomes
When available, the following raw data were extracted from studies: pre- and postoperative olfactory measurements [regardless of the type of assessment used], pre- and postoperative number of patients with hyposmia, and number of patients who developed surgery-induced hyposmia. In studies with multiple olfactory assessments, only the final evaluation was considered to reflect long-term outcomes, and the olfactometry data were preferred over those from subjective scales.
The primary outcome was the proportion of patients who developed surgery-induced hyposmia after nasal surgery regardless of the olfactory assessment used [olfactometry or subjective scale]. Surgery-induced hyposmia was defined as a postoperative decline in olfactory function, irrespective of baseline status. The secondary outcomes were the pre- and postoperative olfactory assessment results and the number of patients with hyposmia. Hyposmia was defined in each study according to the thresholds specific to each olfactory assessment tool used: for example, <31/48 on the SST, <34/40 on the UPSIT [with an MCID of 4], 29 <9/12 on the Brief Smell Identification Test [BSIT; with an MCID of 1], 30 and <6/8 on the Connecticut Chemosensory Clinical Research Center [CCCRC] test. 31 Since continuous olfactory measurements were obtained using various scales, the results were then prepared for synthesis 32 using a standardization on a 10-point hyposmia scale [0 = no hyposmia, 10 = complete anosmia].
The analysis was conducted for all intervention groups and then by subgroup: first by surgical subgroup [septorhinoplasties, septoplasties, and inferior turbinoplasties], then by olfactory assessment type [olfactometry and subjective scales], and finally by timing of assessment postsurgery [less than 3 months or at least 3 months].
Meta-Analysis
Effect size estimates were based on standardized mean differences [SMDs] between groups. For binary outcomes, pooled risk ratios [RRs] were estimated using the number of patients with hyposmia in each intervention group. For the meta-analysis, the intervention groups considered were those mentioned above but with an additional level of precision: Turbinoplasties were categorized as surgical or instrumental while septoplasties and septorhinoplasties were categorized as either performed alone or combined with turbinate procedures. Meta-analyses were run when at least 2 studies were available. Subgroup and meta-regression were run if at least 10 studies were available. Data were analyzed using R version 4.3.3, with the “meta” package, version 7.0-0 [R Foundation]. Restricted maximum likelihood estimator for τ 2 was used. To compute the summary effects’ confidence intervals [Cis], both the conventional method for random effects and the Hartung-Knapp modification were used, 33 and the most conservative method [ie, the method that produced the largest CI] was kept. 34 The I 2 with its 95% CI was used to assess the heterogeneity of effect sizes, and the Egger test and funnel plots were used to estimate publication biases.
Risk-of-Bias Assessment
The risk of bias was assessed using the Office of Health Assessment and Translation adapted for clinical studies for randomized controlled trials [RCTs], 35 and the Methodological Index for Non-Randomized Studies [MINORS] for the other ones. 36 The MINORS index is a bias assessment tool that provides, for cohort studies, a total score out of 16 based on 8 criteria, and for non-randomized controlled trials [NRCTs], a total score out of 24 based on 12 criteria. A study was considered to be at moderate risk of bias for scores ranging from 9 to 16 for NRCTs and from 6 to 11 for cohort studies. Below these ranges, studies were considered at high risk of bias, and above, at low risk of bias. Bias assessment was conducted by A.F. and M.F.
Results
Descriptive Results of the Population Studied
Study Selection
A total of 7780 studies were initially identified using the MeSH-based search equation. After full-text evaluation of 264 articles, 99 studies were selected for analysis. Three additional studies were included during publication monitoring,37 -39 resulting in a total of 102 studies (Figure 1). These included 56 prospective cohorts, 17 retrospective cohorts, 14 NRCTs, and 15 RCTs. Twenty-six of the 102 studies included more than 1 surgical group, resulting in 129 intervention groups analyzed, totaling 16,712 patients with a mean age of 33.39 [±6.62] years and a sex ratio of 0.57 (±0.18; Table 1).
Characteristics of the Intervention Groups Included in the Systematic Review.
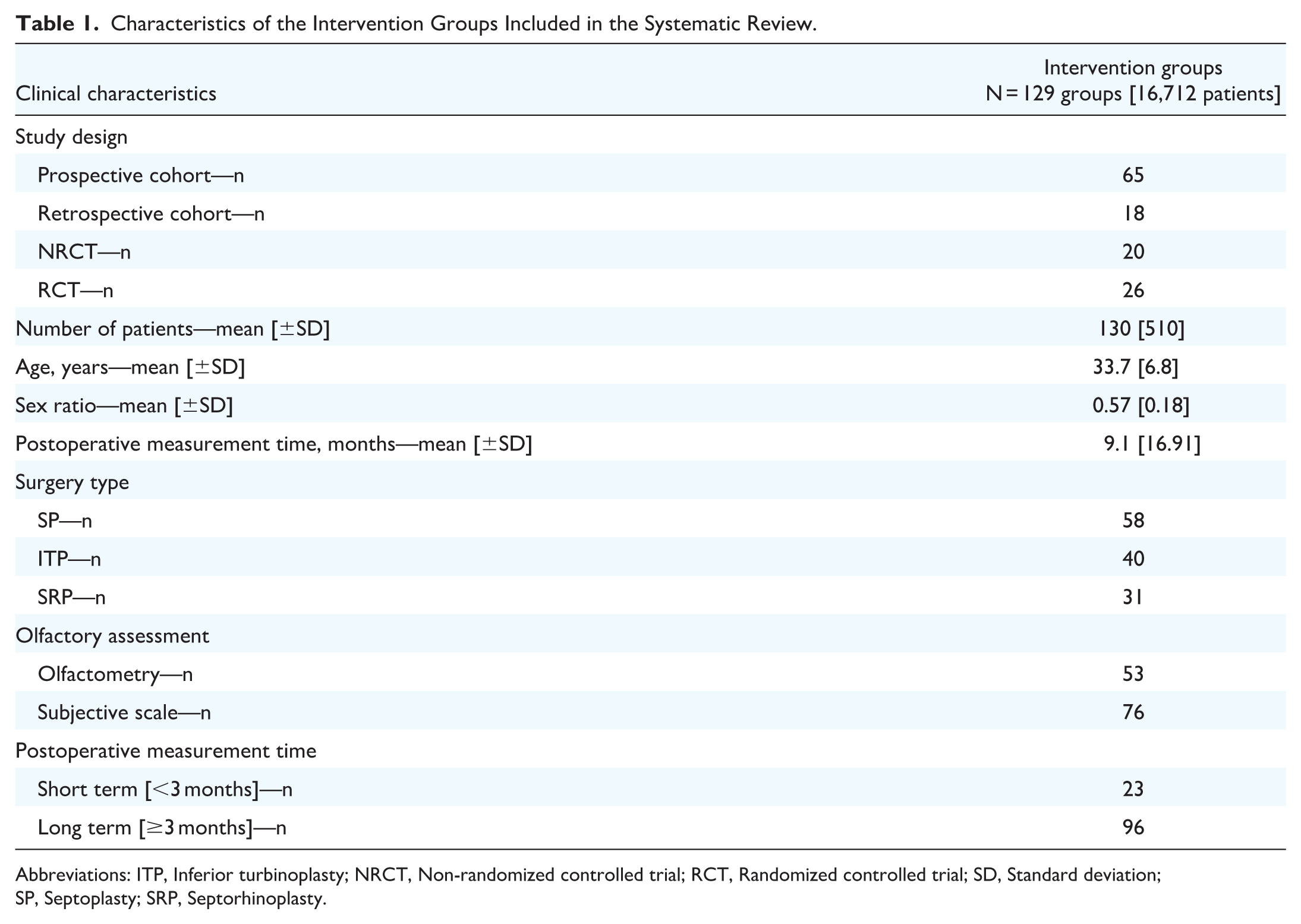
Abbreviations: ITP, Inferior turbinoplasty; NRCT, Non-randomized controlled trial; RCT, Randomized controlled trial; SD, Standard deviation; SP, Septoplasty; SRP, Septorhinoplasty.
Intervention
Of the surgical groups, 45.0% [58/129] underwent septoplasty, 31.0% [40/129] had inferior turbinoplasty, while 24.0% [31/129] had septorhinoplasty. Among septorhinoplasties, 12.9% [4/31] were performed with turbinoplasty and 87.1% [27/31] without additional procedures.
Olfactory Assessment
Olfaction was evaluated at a mean of 9.1 [±17] months postsurgery. Olfactometry was employed in 41.1% [53/129] of the groups, while the remaining 76 groups utilized various subjective scales (Table 1). Among the 53 groups that used olfactometry, 41.5% [22/53] used SST, 15.1% [8/53] used the UPSIT or its shorter version [BSIT], 26.4% [14/53] used the CCCRC, and 17.0% [9/53] used other tests, including the Elsberg method, Cain olfactometry, olfactory threshold determination, and Smell Diskettes Test. SST data could not be analyzed as among the 22 studies reporting its use, 7 groups only underwent a threshold analysis [/16], 1 group had no numeric data available and measurement times were different for all (Supplemental Table 1). Among the groups using subjective scales, 46.0% [35/76] used a binary criterion, 14.5% [11/76] used a locally-validated olfaction questionnaire, and 39.5% [30/76] used a VAS for hyposmia ranging from 0 [perfect olfaction] to 10 [total anosmia].
Descriptive Olfactory Results
Results Across all Studies
For the 80 groups reporting hyposmia scores, the average preoperative score was 3.73 [±1.58] on a 10-point scale, while the postoperative score was 2.39 [±1.45] (Table 2). This difference was statistically significant [P < .001]. Postoperatively, a significantly-lower proportion of patients reported hyposmia than preoperatively [14.5% (380/2620) vs 22.2% (945/4254), P < .001; Table 3]. Surgery-induced hyposmia was present in 2.7% [326/12,032] of patients for the 50 groups reporting this data.
Mean Preoperative and Postoperative Hyposmia Scores According to Surgery Type and Olfactory Assessment.
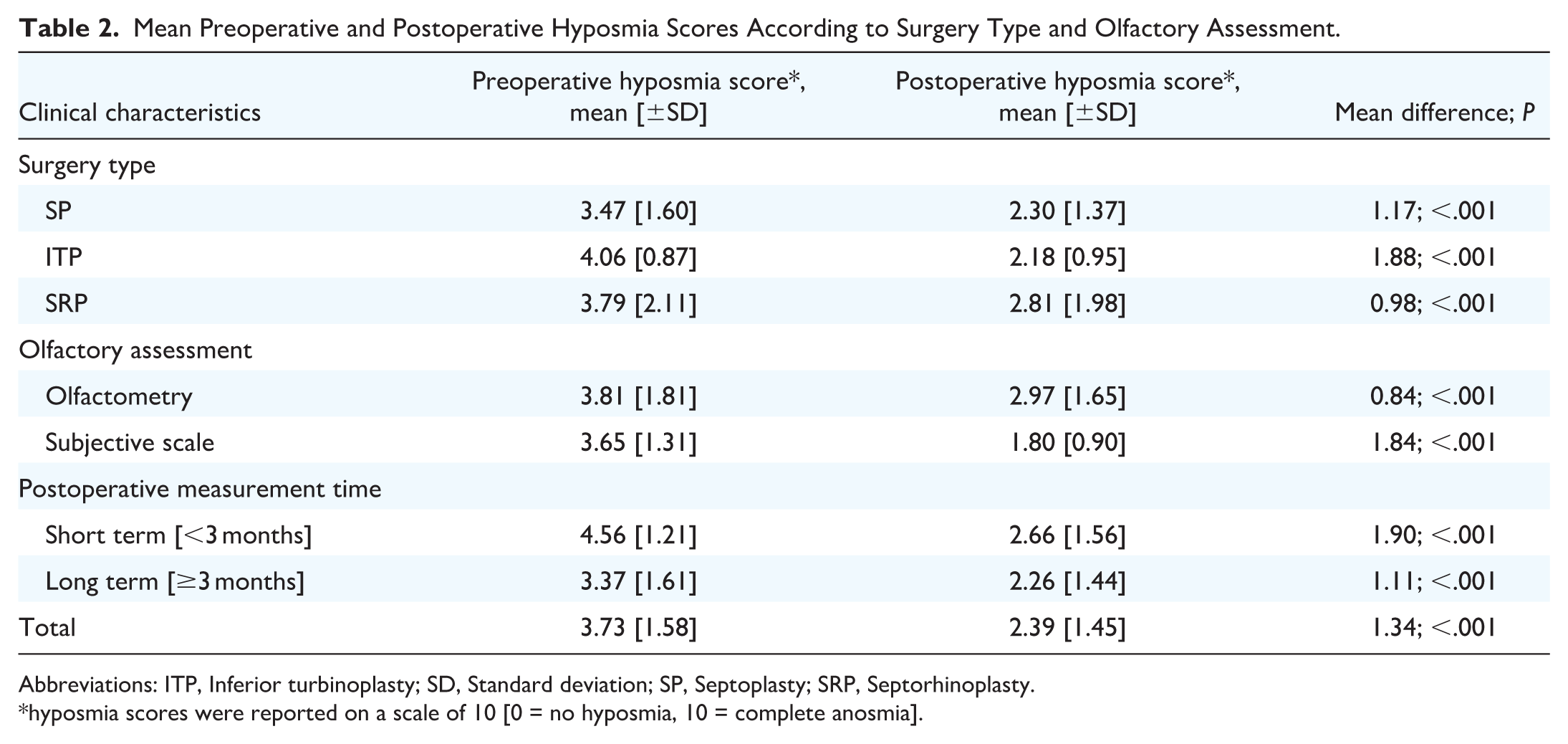
Abbreviations: ITP, Inferior turbinoplasty; SD, Standard deviation; SP, Septoplasty; SRP, Septorhinoplasty.
hyposmia scores were reported on a scale of 10 [0 = no hyposmia, 10 = complete anosmia].
Preoperative and Postoperative Proportion of Patients with Hyposmia According to Surgery Type and Olfactory Assessment.

Abbreviations: ITP, Inferior turbinoplasty; SP, Septoplasty; SRP, Septorhinoplasty.
Results by Surgery Type
Among the 58 groups undergoing septoplasty, the mean preoperative hyposmia score was significantly higher than the postoperative score [3.47 ± 1.60 vs 2.30 ± 1.37, mean difference = 1.17, P < .001; Table 2]. A significantly-lower proportion of patients were hyposmic postoperatively than preoperatively [18.1%, 209/1155 vs 23.1%, 450/1947, P < .001; Table 3]. Surgery-induced hyposmia was observed in 2.7% [240/8877] of patients.
Similar trends were observed across other surgical subgroups, with systematic postoperative improvements in olfactory scores and reductions in the number of patients with hyposmia (Tables 2 and 3). Surgery-induced hyposmia was also present in each surgical subgroup: 3.3% [23/702] after inferior turbinoplasty and 2.6% [63/2453] after septorhinoplasty.
Results by Type of Olfactory Assessment
Among the 53 groups that used olfactometry, the mean preoperative hyposmia score was significantly higher [indicating poorer olfaction] than the postoperative score [3.81 ± 1.81 vs 2.97 ± 1.65, mean difference = 0.84, P < .001; Table 2]. The percentage of patients with hyposmia did not differ significantly between preoperative and postoperative assessments [15.3%, 400/2611 vs 14.4%, 216/1499, P = .440; Table 3]. Postoperatively, 3.8% [103/2742] of patients exhibited surgery-induced hyposmia.
In the 76 groups that used subjective scales, the mean preoperative hyposmia score was significantly higher than the postoperative score [3.65 ± 1.31 vs 1.80 ± 0.90, mean difference = 1.84, P < .001; Table 2]. The percentage of patients with hyposmia was significantly lower postoperatively than preoperatively [12.0%, 164/1370 vs 33.2%, 545/1643, P < .001; Table 3]. Postoperatively, 2.0% [223/9253] of patients exhibited surgery-induced hyposmia. The rate of surgery-induced hyposmia was significantly higher in the olfactometry group than in the subjective scale group [3.7% vs 2.5%, P < .001].
Results by Timing of Assessment Postsurgery
When considering only the 96 groups for whom olfaction was measured at least 3 months after surgery [mean measurement = 10.8 ± 18.4 months], a significant improvement in the postoperative olfactory score compared with the preoperative score was observed [2.26 ± 1.44 vs 3.37 ± 1.61, P < .001]. The percentage of hyposmic patients was significantly lower postoperatively than preoperatively [14.4%, 326/2269 vs 19.5%, 767/3938, P < .001; Table 3]. Surgery-induced hyposmia was reported in 2.9% [310/10,750] of patients. Twenty-three groups were considered short term [<3 months], and 10 groups had no data available regarding the timing of assessment postsurgery.
Meta-Analysis
Among the 102 studies included, only 21 featured a comparison between 2 distinct intervention groups and were therefore eligible for meta-analysis; among these, only 11 had similar designs that could be compared.21,40 -51 The studies available allowed for the comparison between septoplasty alone and septorhinoplasty alone as well as between instrumental and surgical turbinoplasties for inferior turbinoplasty.
When comparing septoplasty with septorhinoplasty, regarding observational studies,21,41 -45 the SMD for olfaction score was not significant [−0.21 (−0.67; 0.25); 4 studies; Figure 2A]. There was no significant difference in the proportion of surgery-induced hyposmia between groups [relative risk (RR) = 0.64 (0.14; 2.89); 3 studies; Figure 2B]. Regarding RCTs,46 -48 data were not sufficient for group comparison [no continuous data for olfaction score and no data regarding the proportion of surgery-induced hyposmia].
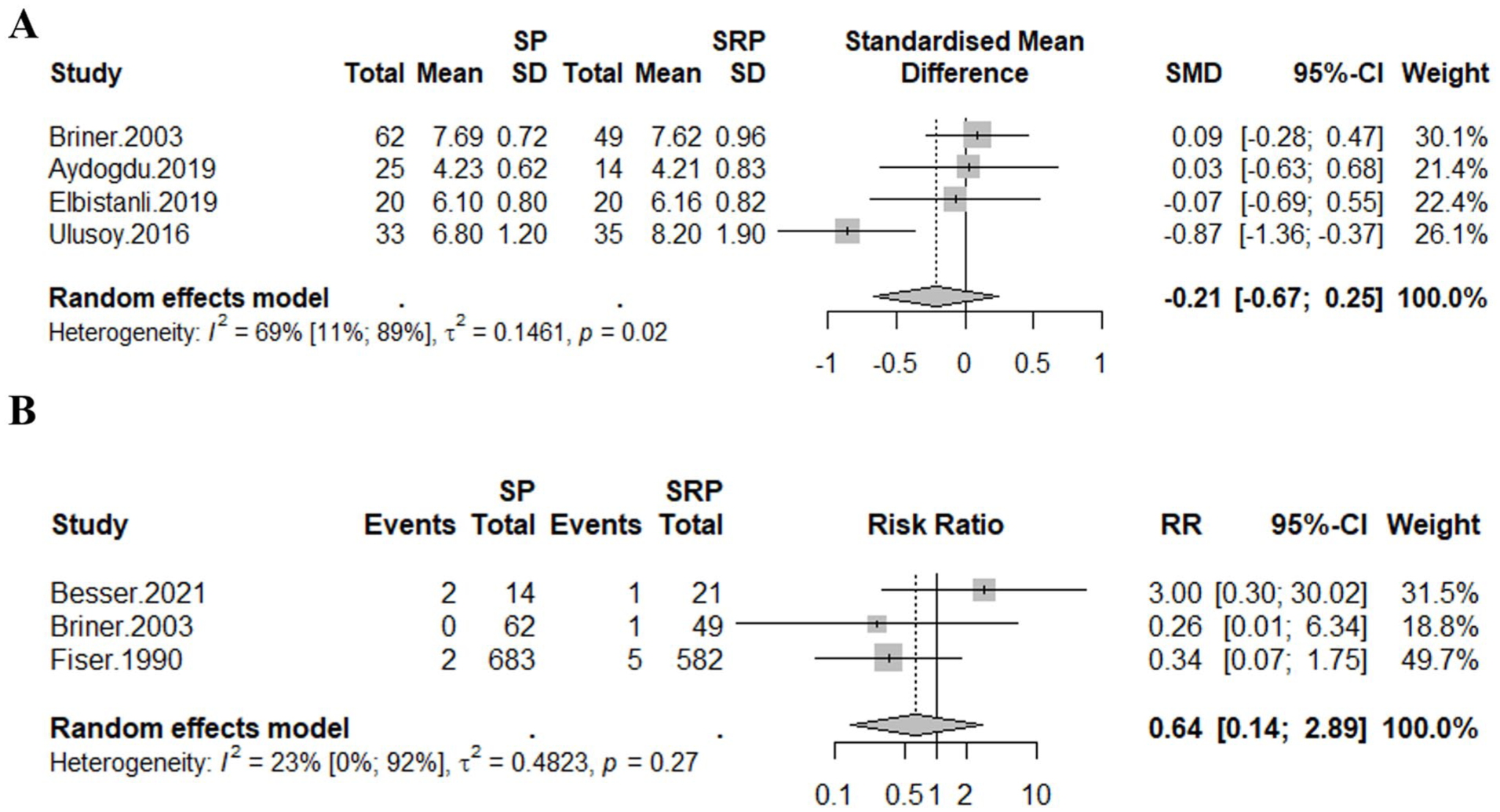
Forest plot of observational studies comparing septoplasties to septorhinoplasties. (A) Olfaction score; (B) Surgery-induced hyposmia. SP, Septoplasty; SRP, Septorhinoplasty; SMD, Standard mean difference; SD, Standard deviation; CI, Confidence interval; RR, Risk ratio.
When comparing instrumental with surgical inferior turbinoplasty, regarding RCTs,49,50 the SMD for olfaction score was not significant [−0.16 (−0.45; 0.13); 2 studies; Figure 3]. No data regarding the proportion of surgery-induced hyposmia were available.

Forest plot of randomized controlled trials comparing olfaction score between instrumental and surgical inferior turbinoplasties. Instrumental ITP, Instrumental inferior turbinoplasty; Surgical ITP, Surgical inferior turbinoplasty; SMD, Standard mean difference; SD, Standard deviation; CI, Confidence interval.
Risk of Bias in Included Studies
Among the 102 studies included, 29.4% [30/102] were at low risk of bias, 68.6% [70/102] had a moderate risk, and 2.0% [2/102] had a high risk. A moderate risk of bias was observed in 71% [53/73] of the cohort studies, 4% [6/14] of the NRCTs, and 73% [11/15] of the RCTs. A high risk of bias was identified in 1% [1/73] of the cohort studies and 7% [1/15] of the RCTs. For the cohort studies, the mean MINORS score was 10.3 [±2.3] out of 16, while for the NRCTs, it was 15.6 [±3.7] out of 24.
Discussion
The present systematic review, which included 102 studies and 16,712 patients, identified a non-negligible proportion of surgery-induced hyposmia [2.7%] across all different nasal surgeries. Furthermore, the meta-analysis revealed no significant difference in olfactory sequelae between septoplasty and septorhinoplasty. However, this lack of difference might be explained by study heterogeneity and the scarcity of quantitative, objective data.
In the present review, olfactory impairment was identified as a potential adverse event of nasal surgical procedure based on data from over 10,000 patients. Postoperatively, nearly 3% of patients experienced a decline in olfaction, a finding consistent with prior reports of persistent anosmia following functional nasal surgery [0.3%-3%].26,45,52 A major factor in olfactory assessment is the timing of the postoperative measurement. Several authors 23,24,53 describe a marked initial olfactory decline postsurgery, with partial or complete recovery by 6 months. However, the data presented here were obtained after an average follow-up of more than 6 months, suggesting that the surgery-induced hyposmias identified in this review may be long-lasting and warrant clinical vigilance. This raises questions regarding follow-up duration, as some researchers suggest that follow-up beyond 1 month for septoplasty patients is unnecessary due to the stable nature of symptoms. 54
Beyond the hypothesis of surgical trauma to the neuroepithelium due to persistent uncertainty regarding its extent, 3 the results of the present review may be consistent with recent studies on the characteristics and consequences of nasal airflow. 55 Asama et al 56 described conductive olfactory dysfunction as resulting from mouth breathing in case of significantly-increased nasal resistance, leading to a reduction in airflow through the olfactory cleft. The removal of the obstruction would therefore allow an increase in airflow within the olfactory cleft, thereby improving the sense of smell. This mechanism is consistent with the overall improvement in olfaction observed in the present study and by several authors18,19. However, beyond a certain threshold, this reduction in nasal resistance has been shown to alter airflow distribution, particularly by significantly decreasing airflow to the upper regions of the nasal cavities, 57 which could contribute to olfactory impairment. Nevertheless, the underlying pathophysiological mechanisms remain uncertain to date.
A rigorous screening of 7780 articles on septoplasty, turbinoplasty, and septorhinoplasty ultimately resulted in only 102 [1.3%] studies reporting the olfactory sequelae of nasal surgeries. Fewer than 50% of these studies employed validated olfactometry tests, highlighting a critical oversight in evaluating this risk, despite the documented impact of hyposmia on quality of life.4 -6 The present analysis found a significant improvement in the proportion of patients with hyposmia between preoperative and postoperative assessments, suggesting a positive olfactory outcome from the studied surgical interventions [septorhinoplasty, septoplasty, and/or turbinoplasty] with improved mean hyposmia scores postoperatively. This may be explained by an increase in the olfactory cleft airflow. 58 Importantly, none of the 102 included studies reported a significant long-term decline in olfactory function after surgery, a reassuring finding consistent with previous studies 27 and other literature reviews.18,59 However, while the scientific community broadly agrees on the positive olfactory effects of these surgeries, it often overlooks the onset and persistence of surgery-induced hyposmia in a minority of patients. This risk nevertheless does not appear to be negligible and should therefore be included in the routine preoperative information provided to patients.
Only less than half of the groups in this review used objective psychophysical testing, which affects the review’s external validity. This disparity in olfactory assessment methods appears to underestimate the actual incidence of surgery-induced hyposmia. Indeed, the proportion of surgery-induced hyposmia observed with olfactometry was significantly higher than that reported with subjective scales, regardless of the type of surgical procedure performed. As highlighted by the meta-analysis by Desiato et al, 60 which found 28.8% hyposmia on psychophysical testing compared with 9.5% on subjective measures in the general population, olfactometry appears considerably-more sensitive than subjective questionnaires. This finding underscores the importance of standardized psychophysical olfactory assessments in both clinical practice and research to reduce bias in olfactory outcomes.
Finally, several limitations need to be considered. Firstly, there was heterogeneity of the surgical interventions. The grouping of similar surgical indications and/or interventions, in order to summarize results from an extensive literature search, prevented us from assessing the difference that could exist between a septoplasty alone, a septorhinoplasty alone, or a septoplasty with turbinoplasty. This is even more relevant as turbinoplasty appears to have an additional olfactory impact, as suggested by Dąbrowska-Bień et al 1 for inferior turbinoplasty, and studies have shown that olfactory filaments are present in the middle turbinate mucosa.3,61,62 Furthermore, inferior turbinate hypertrophy may coexist with disease states that are well established to impair olfactory function; nonetheless, the postoperative improvement observed following nasal surgery challenges the notion that the underlying inflammatory process alone accounts for the olfactory deficit, and separating these different groups in future studies would be valuable. Furthermore, several methodological limitations affect the strength of the evidence. Only 18 of the included studies were prospective, double-blind RCTs, and among them, only 27.8% [5/18] had a low risk of bias. Overall, only 29.4% [30/102] of the studies analyzed were at low risk of bias, 68.6% [70/102] had a moderate risk, and 2.0% [2/102] had a high risk. This risk is attributed to study designs and measurement methods. Also, olfactory scores were converted into a hyposmia scale, thus effect size was lost. However, the considerable variability in olfactory assessment across studies required methodological harmonization to enable a meaningful summary of the available evidence. The other limitation of the review is the relatively-small number of studies, which used standardized olfaction assessments [41.0%; (53/129)]. SST data could not be analyzed as among the 22 studies reporting its use, 7 groups only underwent a threshold analysis [/16], 1 group had no numeric data available and measurement times were different for all.
Conclusion
The present systematic review and meta-analysis suggest that nasal surgery improves the sense of smell for a majority of patients, but may nevertheless impair olfaction in a non-negligible number of cases. This highlights the need to systematically inform patients about this risk. However, this conclusion is based on a heterogeneous group of studies, as evidenced by the low number of studies included in the meta-analysis. This review also highlights the need for objective and systematic olfactory assessment in nasal surgery studies using validated and standardized tools.
Supplemental Material
sj-docx-1-ohn-10.1177_19160216261416371 – Supplemental material for Olfactory Sequelae of Nasal Surgeries: A Systematic Review with Meta-Analysis
Supplemental material, sj-docx-1-ohn-10.1177_19160216261416371 for Olfactory Sequelae of Nasal Surgeries: A Systematic Review with Meta-Analysis by Apolline Favreau, Valentin Favier, Florent Carsuzaa, Gerold Besser, Caroline Giroudon, Margaux Legré, Stéphane Tringali, Nazim Benzerdjeb, Mikail Nourredine and Maxime Fieux in Journal of Otolaryngology - Head & Neck Surgery
Supplemental Material
sj-docx-2-ohn-10.1177_19160216261416371 – Supplemental material for Olfactory Sequelae of Nasal Surgeries: A Systematic Review with Meta-Analysis
Supplemental material, sj-docx-2-ohn-10.1177_19160216261416371 for Olfactory Sequelae of Nasal Surgeries: A Systematic Review with Meta-Analysis by Apolline Favreau, Valentin Favier, Florent Carsuzaa, Gerold Besser, Caroline Giroudon, Margaux Legré, Stéphane Tringali, Nazim Benzerdjeb, Mikail Nourredine and Maxime Fieux in Journal of Otolaryngology - Head & Neck Surgery
Footnotes
Acknowledgements
We thank Véréna Landel (Direction de le Recherche en Santé, Hospices Civils de Lyon) for her help in manuscript preparation.
Data Availability Statement
Data supporting reported results are available on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Consent for Publication
No subjects were involved in the study. All authors have read and agreed to the published version of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
