Abstract
Importance
Hypopharyngeal squamous cell carcinoma (HPSCC) is an aggressive cancer with poor outcomes. Neoadjuvant therapy (NAT) may allow organ preservation, but determining safe surgical margins after NAT is a critical challenge.
Objective
To evaluate the safety of reduced surgical margins after neoadjuvant tislelizumab and afatinib in locally advanced HPSCC by comparing pathological margins to an upfront surgery control group.
Design
Prospective, single-center pilot cohort study.
Setting
Tertiary referral center in Western China.
Participants
Forty-one patients with locally advanced HPSCC; 32 received NAT (treatment group), and 9 served as an upfront surgery control group.
Intervention or Exposures
Treatment group: 2 cycles of neoadjuvant tislelizumab plus 6 weeks of afatinib. Control group: upfront surgery. Both groups underwent resection with a 1.5 cm naked-eye surgical margin.
Main Outcome Measures
Primary outcomes were minimal peripheral surgical margin (MPSM) and minimal deep surgical margin (MDSM). Secondary outcomes included radiological (RECIST v1.1) and pathological tumor response.
Results
The NAT group had a 20/32 (62.5%) radiological objective response rate and a 16/32 (50.0%) major pathological response rate [including 9/32 (28.13%) pathological complete response]. The mean MPSM was significantly greater in the treatment group versus control [3.38 mm vs 1.71 mm; 95% Confidence interval (CI): 0.10-3.24; P = .038]. The mean MDSM was also significantly greater in the treatment group versus control (2.09 mm vs 1.04 mm; 95% CI: 0.12-1.97; P = .029).
Conclusions
Combined immune-targeted NAT effectively downstages HPSCC. The significantly larger pathological margins observed support that reducing clinical surgical margins after this regimen is generally safe.
Relevance
This NAT regimen may allow for less extensive resections, facilitating laryngeal preservation and improving quality of life without compromising oncologic safety. Larger validation studies are needed.

Key Messages
A neoadjuvant regimen of tislelizumab and afatinib achieves high pathological response rates (50.0% MPR) in patients with locally advanced hypopharyngeal squamous cell carcinoma.
Patients receiving this neoadjuvant therapy had significantly larger minimal peripheral and deep surgical margins compared to an upfront surgery group, despite an identical 1.5 cm clinical resection margin being applied to both.
These findings support the safety of reducing surgical margins following this specific immunotargeted neoadjuvant therapy, which may enable more organ-preservation surgeries.
Introduction
Hypopharyngeal squamous cell carcinoma (HPSCC) is a relatively uncommon malignancy, comprising approximately 3% to 5% of all head and neck cancers, with a substantial proportion of HPSCC patients, between 60% and 85%, presenting with advanced-stage disease (III-IV) at diagnosis. 1 Unfortunately, the reported 5-year survival rates for stages III and IV HPSCC are only 36% and 24%, respectively. 2 Moreover, locally advanced HPSCC frequently necessitates total laryngectomy as a curative procedure, resulting in a significant impairment of patients’ overall quality of life due to the loss of both speech and olfactory function. While oncologic outcomes remain the primary objective of treatment for HPSCC patients, the preservation of patients’ quality of life is equally important.
Neoadjuvant therapy (NAT) can effectively reduce tumor volume prior to surgical intervention, potentially converting initially unresectable tumors into operable ones. Consequently, the preservation of the larynx and pharynx may become feasible. Furthermore, NAT has demonstrated superior long-term oncologic and functional outcomes in multiple clinical studies.3,4
Immune checkpoint inhibitors (ICIs) have demonstrated efficacy in patients with advanced head and neck squamous cell carcinoma (HNSCC), leading to significant tumor regression and improved survival. 5 While single-agent neoadjuvant ICI therapy has shown limited success in HNSCC with a relatively low major pathological response (MPR) rate (4.3%-8%),6,7 the integration of ICIs with existing therapeutic approaches is crucial to accelerate and intensify tumor regression during the neoadjuvant phase. Considering the fact that the epidermal growth factor receptor (EGFR) is overexpressed in the majority of HNSCC cases (80%-100%) and is associated with poor treatment responses and unfavorable clinical outcomes, 8 combining anti-EGFR monoclonal antibodies or small molecule tyrosine kinase inhibitors (TKIs) with ICIs can probably demonstrate an ideal therapeutic effect as NAT.
Despite the improved NAT design, it remains uncertain whether the surgical margin for patients undergoing NAT should be based on the visible tumor residual foci before therapy. The generally recommended surgical margin for the primary tumor of HPSCC is 1 to 1.5 cm to achieve R0 resection.9,10 Previous studies have revealed discrepancies between macroscopic and microscopic regression, often characterized by distant intramural dissemination of fragmented tumor cells that are only visible under microscopic examination. This suggests the persistence of residual tumor foci, even when the tumor appears to have regressed macroscopically. 11 If the surgical margin is based solely on the tumor extent before NAT, the preservation of organ function may be compromised.
To address the controversy surrounding surgical margins after NAT, we conducted a novel NAT regimen combining a PD-1 inhibitor (tislelizumab) with an EGFR-TKI (afatinib) in patients with locally advanced HNSCC within the registered neoCHANCE-1 trial (NCT05517330). As a control group, we also included hypopharyngeal cancer patients who declined NAT prior to surgery. Following tumor resection with a 1.5 cm clinical mucosal surgical margin for both groups, we assessed and compared the tumor margins in each group.
Materials and Methods
This is a prospective, single-center cohort study done at a tertiary referral center in Western China, starting in August 2022 and ended at December 2024. Ethics approval was obtained from the Ethics Committee of the institutional review board. All patients were required to provide informed consent prior to their enrollment. The work has been reported in line with the STROCSS criteria. 12
Patients and Drug Administration
Patient inclusion and exclusion criteria adhered to those established in the previously concluded phase II clinical trial conducted by our group. 13 A total of 32 patients were included in the treatment group, comprising 12 patients whose tumor samples were derived directly from the trial and 20 patients recruited after trial conclusion. Nine patients who declined NAT due to non-medical considerations (eg, financial constraints or personal preference) but otherwise met criteria for larynx-preserving surgery comprised the control cohort.
Patients in the treatment group received intravenous tislelizumab at a dose of 200 mg on the first day of every 3-week cycle, for a total of 2 cycles. Afatinib was administered orally at a dose of 30 mg from the first treatment day until the day before surgery, spanning a total of 6 weeks.
Treatment Efficacy Evaluation
The radiological outcomes of each patient were evaluated based on Response Evaluation Criteria In Solid Tumors version 1.1 (RECIST v1.1). The evaluation includes a head and neck contrast CT or MRI scan, done prior to treatment initiation and prior to surgical intervention.
All patients who underwent surgery were evaluated by pathologists blinded to patient information. Pathological response was determined by the percentage of residual viable tumor (RVT) in resected specimens, using previously reported methods.14,15 Pathological analysis was conducted on both the complete primary tumor and all sampled lymph nodes. RVT% was calculated by summing the areas of the tumor and dividing by the total area of the tumor bed across all slides. The tumor bed was defined as the region involving RVT, necrosis, and regression bed. Pathological responses were categorized as follows: pathological complete response (pCR) (%RVT = 0), MPR (%RVT ≤ 10%), partial pathological response (PPR) (10% < %RVT ≤ 50%), and no pathological response (NPR) (%RVT > 50%).
Surgical Resection and Contraction Rate Calculation
The partial hypopharyngectomy is conducted by a senior head and neck surgeon to reduce inter-operator variability. After visualization of the tumor, the surgical margin was determined using the soft ruler and labeled using the suture line, a roughly 1.5 cm surgical margin under naked eye was applied to all the patients in both treatment and control group (Figure 1A), for patients with complete response (CR), the remaining scar tissue or the estimated center of the cancer before treatment were deemed as the tumor site. The circumferential frozen pathology examination was applied for detecting positive surgical margins during the surgery, and the margin positivity was based solely on the frozen pathology results. The circumferential frozen pathology examination was applied for detecting positive surgical margins. The complex 3-dimensional anatomy of the hypopharynx means that achieving a uniform 1.5 cm surgical margin in all directions is often not surgically feasible or appropriate. For certain samples, for which the 1.5 cm surgical margin can’t be achieved in all directions, the samples were also included if there is at least 2 surgical margins measuring 1.5 cm on diametrically opposite sites of the tumor. Our study’s aim was to specifically measure the resulting pathological margin when a deliberate 1.5 cm clinical margin was applied. To do this in a standardized way, we had to ensure we were measuring specimens where this 1.5 cm margin was successfully and completely resected along at least 1 valid axis. And the slicing direction is perpendicular to this axis.

Illustration of sampling and measurement of contraction rate. (A) Measuring a 1.5 cm surgical margin after visualizing the tumor. (B) Measuring the knot-to-knot distance after resection. (C) Measuring the knot-to-knot distance after fixation. (D, E) Slicing strategy.
Following tumor resection, the knot-to-knot distance of the resected tissue was remeasured using a vernier caliper with a resolution of 0.1 mm (Figure 1B). After fixation, the knot-to-knot distance was measured again (Figure 1C). The final sample-to-section contraction rate was calculated using the formula: 1—(distance under microscopy / 1.5).
Slicing Protocol and Section Selection
Following 24 hours of fixation in formalin, fixed specimens were sectioned radially to capture the bilateral mucosal margins and the deep margin at a 3 mm interval (Figure 1D, E). For samples where a 1.5 cm surgical margin could not be achieved in all directions but had at least 2 surgical margins measuring 1.5 cm on diametrically opposite sides of the tumor, the slicing was performed parallel to the margin connection line. After slicing, sections were processed routinely, sectioned at 4 μm, stained with hematoxylin and eosin, and coverslipped. Sections with pathologically observable tumor lesions were selected for inclusion in the statistical analysis; 1 tumor sample can have multiple sections included in the analysis. For patients with a pCR, the samples were excluded from analysis.
Measurement of the Minimal Peripheral and Deep Surgical Margin
The minimal peripheral surgical margins (MPSMs) were measured for both the mucosal margin and the deep tissue margin. The measurement of MPSMs follows the standard protocol described by Kortekaas et al 16 in the practical guideline for measuring margins in vulvar squamous cell carcinoma. The detailed measurement technique is detailed in Figure 2.

Schematic illustration of MPSM and MDSM measurement. (A) An auxiliary line was drawn perpendicular to the mucosa from the edge of the submucosal lesion. The distance between the intersection of this line with the mucosa and the margin was defined as the MPSM. The closest distance between the cancer lesion and the deep margin was defined as the MDSM. (B) For samples with mild mucosal curvatures, the curvature was disregarded. (C) For samples with protrusive lesions, the distance was measured within the sample, not outside of it. (D) For samples with marked curvature, the summation of 2 or more lines was defined as the MPSM. (E) Illustration of MPSM measurement in a patient’s sample. MPSM, Minimal peripheral surgical margin; MDSM, Minimal deep surgical margin.
After documenting the MPSMs for each pathological section, each section can generate 1 mucosal margin data and 1 deep margin data, for each sample with multiple included sections, the smallest MPSM and minimal deep surgical margin (MDSM) from all the included sections of this sample were deemed as the final MPSM and MDSM of this sample, respectively.
Statistical Analysis
Categorical variables were presented as frequencies or percentages, while continuous variables were reported as mean (standard deviation). Mann-Whitney U was used for ordinal data comparison in basic demographics, while the Welch t-test was used to compare the mean of continuous data in basic demographics. The Welch t-test was used to compare the mean MPSM between the treatment and control groups. All statistical analyses were performed using IBM SPSS Statistics for Windows, version 26 (IBM Corp., Armonk, NY). Two-sided P < .05 indicated statistical significance.
Results
Demographics
A total of 32 patients with locally advanced HPSCC were enrolled from the clinical trial, and another 9 patients were included in the control group (Figure 3), all of whom were male patients, with an average age of 63.42 (47-83) years. The general characteristics of the patients are presented in Table 1. There were 16 (39.02%) and 25 (60.98%) patients with stages III and IV disease, respectively, with 34 (82.93%) primary lesions from the pyriform sinus and 7 (17.07%) from the posterior pharyngeal wall. There were no significant differences in age, primary lesion, or tumor, node and metastasis (TNM)staging, American Joint Committee on Cancer staging, and other basic demographic data between the 2 groups.

Patient enrollment and treatment flowchart.
Basic Demographics of the Patients.

Abbreviations: AJCC, American Joint Committee on Cancer.
Treatment Outcomes of NAT
A total of 32 patients received the NAT, 6 (18.8%) of them achieved radiological CR, 14 (43.8%) achieved partial response, and 12 (37.5%) had stable disease (Figure 4A). The objective response rate (ORR) was 62.5%, and the disease control rate (DCR) was 100%. Under pathological examination, 16 (50.0%) achieved MPR, among which, 9 (28.13%) patients achieved pCR, 9 (28.13%) patients achieved PPR, and 7 (21.88%) had NPR (Figure 4B, C).

The treatment outcome of the neoadjuvant therapy. (A) The pathological outcomes of the treatment. (B) The radiological outcomes of the treatment. (C) The percent RVT in the treatment group. RVT, Residual viable tumor.
Following NAT, 8 out of 9 patients with T4a disease exhibited a favorable response, allowing the surgical plan to be de-escalated from total laryngectomy to larynx-preserving surgery. While 4 of them didn’t require the use of flap reconstruction. Although the remaining 1 patient showed a suboptimal response to NAT, the patient also underwent organ-preservation surgery in accordance with strong personal preference, using a well-established surgical approach previously described by our surgical team. 17
Among the 32 patients, 10 (31.25%) of the patients had extensive lesion that required flap reconstruction of the pharyngeal/esophageal defect after surgery, 3 used lateral arm free flap, 2 used pedicled supraclavicular island flap, 1 used posterior tibial artery perforator flap, 1 used anterolateral thigh flap, 1 pectoralis major flap, 1 jejunal free flap and 1 thyroid gland flap. Twenty-four (75%) patients had pathologically confirmed lymph node metastasis, and 1 patient had internal jugular vein infiltration. Only 3 patients had mild atypical hyperplasia during intraoperative frozen pathology, which weren’t further intervened; all the other patients had clean surgical margins. Only 1 patient developed a major postoperative complication; he developed gastric bleeding after surgery, which was successfully managed through gastrointestinal decompression and omeprazole. Three (9.38%) patients developed mild dysphagia after removing the nasogastric tube on the 1-month follow-up visit, which was tolerable. All patients in the NAT group were successfully weaned off their nasogastric tubes weeks after the conclusion of the surgery. In addition, all patients in the treatment group were successfully decannulated after weeks or a few months post-discharge.
Treatment Outcomes of the Control Group
A total of 9 patients underwent resection surgery for the hypopharynx and partial larynx without NAT. Two patients had positive surgical margins during the first frozen pathology; 1 patient still had a positive surgical margin during the repeated frozen pathology, who underwent a more extensive resection. Seven (77.78%) out of 9 patients had confirmed lymph node metastasis, and 5 (55.56%) had major blood vessel invasion. Six (66.67%) patients required flap reconstruction due to a large defect after resection surgery. Three used the lateral arm free flap, 2 used the anterolateral thigh flap, while 1 used the pectoralis major flap. All patients in the control group were successfully weaned off their nasogastric tubes and tracheostomy tubes weeks after the conclusion of the surgery, identical to the treatment group.
Sample Contraction Rate
A total of 35 out of 41 patients were deemed as having an optically evaluable lesion and were included for the sample contraction rate calculation. The in vivo peripheral surgical margin (PSM) was deemed as 15.00 mm, the mean immediate ex vivo PSM was 12.52 ± 2.12 mm, and the mean PSM after formalin fixation was 11.57 ± 2.06 mm. The calculated contraction rate from in vivo to immediate ex vivo was 17% [95% Confidence interval (CI) 11%-21%], and the contraction rate from in vivo to formalin fixation was 23% (95% CI 17%-29%).
Minimal Peripheral and Deep Surgical Margin Evaluation
Among the 41 samples, there were 240 sections with identified cancer lesions and evaluable mucosal surgical margin, 173 sections of 23 patients from the treatment group, and 67 sections of 9 patients from the control group were included in the final analysis. For patients with multiple evaluable sections, the section with the smallest PSM was deemed the MPSM of this patient. The resulting mean measured MPSM was 3.38 ± 2.19 mm for the treatment group, and 1.71 ± 1.66 mm for the control group before adjusting for contraction. The mean MPSM of the treatment group is statistically greater than that of control group (t = 2.23, 95% CI: 0.10-3.24; P = .038) (Figure 5A).
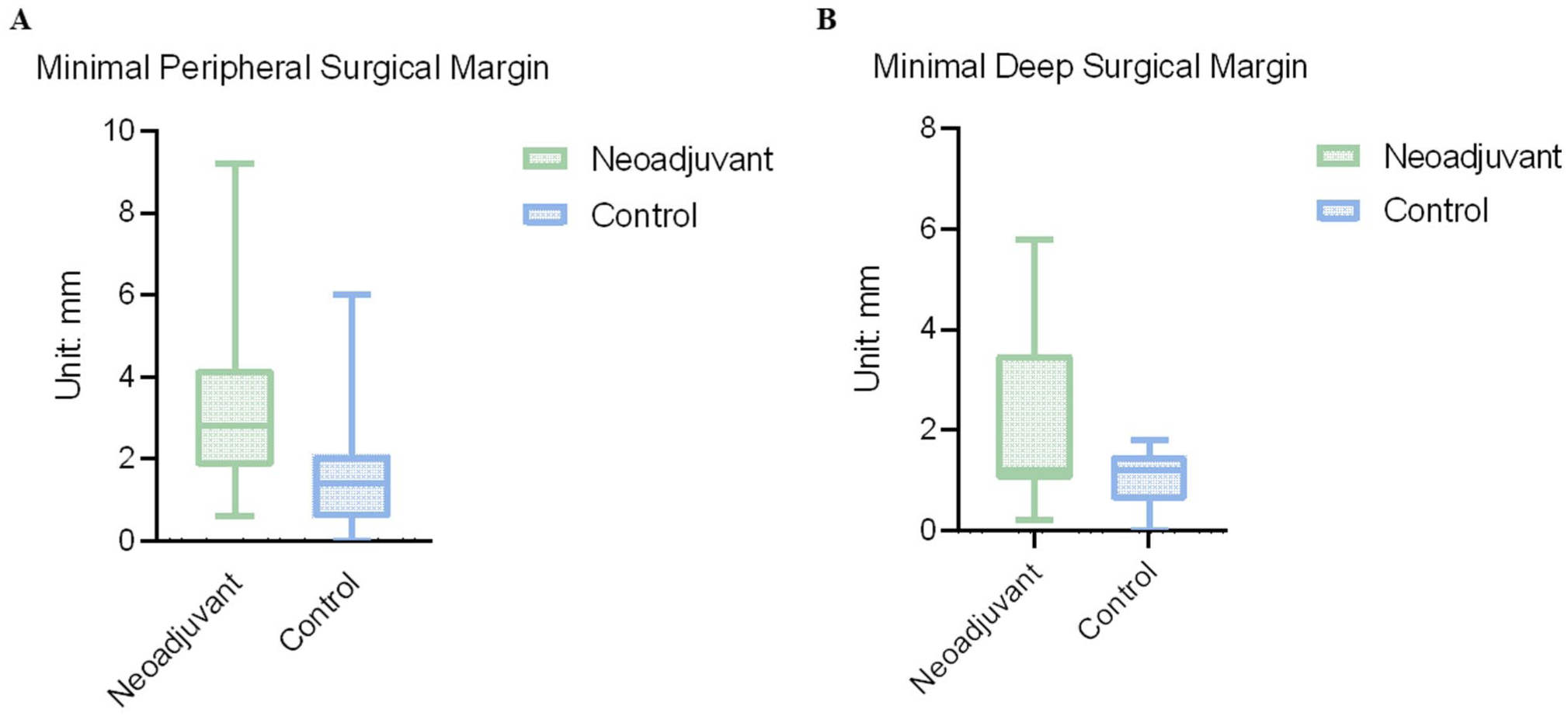
The comparison of MPSM and MDSM between the treatment and control group. (A) Comparing MPSM between the treatment and control group. (B) Comparing MDSM between the treatment and control group. MPSM, Minimal peripheral surgical margin; MDSM, Minimal deep surgical margin.
There were 213 sections with evaluable deep surgical margins from 32 patients, 21 patients from the treatment group, and 9 from the control group. The resulting mean measured MDSM was 2.09 ± 1.81 mm for the treatment group and 1.04 ± 0.56 mm for the control group. The mean MDSM of treatment group is statistically greater than that of control group (t = 2.31, 95% CI: 0.12-1.97; P = .029) (Figure 5B).
Discussions
The findings of our study indicated that the combination of EGFR-TKI and PD-1 inhibitor NAT is effective, with more than half of the patients achieving either pCR or MPR. The reduction of surgical margin under naked eyes is generally safe after the NAT, with the minimal peripheral and deep surgical margin larger in the treatment group than in the control group.
HPSCC generally has the worst prognosis among all HNSCCs, with a 5-year overall survival (OS) rate at about 30% to 35%, 18 the HPSCC is relatively less treatment-responsive than other types of HNSCCs; the ORR was only 52% in a prior study with combination induction chemotherapy (Taxol, cisplatin, 5-FU) and cetuximab. 19 The efficacy of NAT in HPSCC has been indicated in prior studies,7,20 for patients with locally advanced HPSCC, the need for total laryngectomy can significantly impair the function and quality of life, while the downstaging of the tumor can potentially lead to laryngeal preservation surgery.
Studies consistently show that positive surgical margins are associated with a significantly higher risk of local tumor recurrence, while close margins can also pose a potential risk of tumor recurrence. The submucosal spread patterns of HPSCC predispose to inadequate margins in clinical practice. 21 Prior study in buccal mucosa cancer has demonstrated decreased positive/close margin rate in neoadjuvant chemotherapy patients than patients with upfront surgery; however, no statistical significance was generated. 22 Zhang et al 23 had suggested that for pancreatic cancer patients undergoing neoadjuvant chemoradiation, even when patients had positive surgical margins, the survival was not significantly affected, significantly longer than that of patients with upfront surgery who had positive surgical margins. For patients with HPSCC, if the tumor is large, the surgery may result in a relatively large defect that requires flap reconstruction, or the patient may need total laryngectomy to achieve R0 resection of the tumor. Flap reconstruction or total laryngectomy can impact the normal function of the larynx and pharynx, thereby impacting the quality of life. The objective of our study was to evaluate whether it’s safe to decrease the resection range of HPSCC after NAT, to decrease the necessity of flap reconstruction or total laryngectomy.
Our study demonstrated evidence that combined immune-targeted NAT prior to surgery significantly impacts the surgical margins achieved during HPSCC resection, with measurable differences in both the MPSM and the MDSM between treatment and control groups. The observed increase in MPSM in the treatment group (mean: 3.08 ± 1.87 mm) compared to the control group (mean: 1.62 ± 1.67 mm, P = .031) under the similar surgical circumstances with naked-eye surgical margin of 1.5 cm suggests that NAT may facilitate decreasing the resection range, thereby enabling surgeons to achieve primary closure or laryngeal preservation without compromising oncologic principles. Similarly, the MDSM, reflecting the proximity of deep margins to residual tumor, showed a statistically significant improvement in the treatment group (mean: 2.17 ± 2.14 mm) over the control group (mean: 1.02 ± 0.59 mm, P = .036). Though normally the deep margin of HPSCC is considered not as important as the mucosal margin, especially when the tumor resides in the piriform fossa or posterior pharyngeal wall, due to the natural separation by the prevertebral fascia, the deep margin can sometimes lead to tumor recurrence when it’s too close to the margin. Especially for postcricoid lesions, a close deep margin may necessitate total laryngectomy.
The combination therapy of ICI plus targeted therapy has been indicated to yield promising results in HNSCC patients, 24 we hypothesized the potential efficacy of combining PD-1 inhibitor tislelizumab and EGFR-TKI afatinib in patients with advanced resectable HPSCC. The high ORR and DCR rates observed in this study indicate the potential benefit of this treatment strategy in HPSCC. In addition, the significant proportion of patients achieving pCR and MPR suggests that this combinatory NAT can effectively downstage tumors, potentially improving surgical outcomes by enabling laryngeal preservation. In contrast, the patients who underwent surgery alone had a poorer prognosis, with a higher rate of lymph node metastasis, major vessel invasion, and positive intraoperative surgical margins. These factors are known to be associated with poor survival outcomes. The relatively smaller sample size of the control group is attributed to the fact that most patients prefer to undergo NAT prior to surgery to minimize the resection extent and potential surgical complications. Consequently, only a minority of patients whose diseases were eligible for laryngeal preservation surgery opted for surgery alone.
The implications of these findings extend beyond margin measurements. Wider surgical margins are strongly associated with reduced local recurrence rates and improved disease-free survival in various malignancies. Therefore, the observed enhancements in MPSM and MDSM in the neoadjuvant group may translate into potential long-term benefits for patients. Furthermore, the consistent statistical significance across both margin types reinforces the efficacy of combined immune-targeted NAT as a critical component of multimodal treatment strategies for this patient population.
In terms of preliminary survival outcomes, till the time of drafting this article, 21 out of 32 patients were available for 1-year disease-free survival (DFS) and OS 1-year follow-up time. Average follow-up time was 16.88 ± 3.82 months. Five (27.78%) patients experienced tumor recurrence/metastasis, and 4 (22.22%) patients died. Among patients with tumor recurrence/metastasis, 2 patients initially achieved pCR, and 1 patient achieved PPR. Patient 03 had local recurrence along with kidney and lung metastasis; the patient eventually died at the 12th postoperative month (POM). Patient 04 was found to have local recurrence at the 10th POM and soon died at home. Patient 05 had lung metastasis diagnosed at 8th POM and died of severe hemoptysis at 13th POM. Patient P06 didn’t have recurrence or metastasis; the patient died of fulminant COVID-19 pneumonia at 13th POM. The mean DFS of the included 21 patients was 15.05 ± 4.44 months (95% CI: 10.6-19.5 months). The 1-year OS was 80.9%. Further survival data remain to be fully disclosed in the subsequent reports of this trial, as this article focused mainly on the surgical margin.
However, several limitations should be considered when interpreting these results. First, the sample size, while sufficient for statistical analysis, is relatively modest and may limit the generalizability of the findings. Patients nowadays are more willing to undergo NAT prior to surgery to achieve better functional preservation. Second, the influence of contraction-related factors on margin measurements, although accounted for in this study, requires further investigation to standardize margin assessment methodologies. Contraction rates may vary among normal mucosa, dysplastic mucosa, and cancerous regions. Lastly, the treatment group’s higher number of evaluable sections raises concerns about potential selection biases or differences in tumor biology between groups. Moreover, in the present study, though 1.5 cm margins are seemingly adequate in histology and frozen section assessments, the final proof of adequacy depends on locoregional control and survival, which is at present not adequate. Longer-term follow-up of the present cohort would be required to confirm the validity of such an approach. In addition, future studies with larger cohorts and longer follow-up periods are necessary to validate these findings and explore their prognostic implications.
Conclusions
Our findings suggest that combined immune-targeted NAT for HPSCC patients can not only downstage tumors but also facilitate primary closure of the defect or laryngeal preservation surgery, potentially improving oncologic and quality-of-life outcomes. These data support that reducing the surgical margin is generally safe after immune-targeted NAT.
Footnotes
Author Contributions
D.W. and J.L. conceived and designed the study; D.W., J.W., Z.J., Z.W., M.A., P.Z., L.F., L.W., and L.L. performed the investigation and data collection; Z.J. and P.Z. performed formal analysis; D.W., J.W., and Z.J. wrote the original draft; D.L., F.C., H.C., X.P., and J.L. acquired funding and supervised the project; all authors contributed to reviewing and editing the manuscript. D.W., J.W., Z.J. and Z.W. contributed equally as co-first authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from the Strategic Cooperation Special Fund of Sichuan University-Dazhou Municipal People’s Government (2021CDDZ-25, 2022CDDZ-16).
Ethical Considerations
This study was approved by the Ethics Committee of West China Hospital (Ethics Code: No. 2022-268) on June 16, 2022. This research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki.
Consent to Participate
All participants provided written informed consent prior to enrollment in the study.
