Abstract
Importance
Histopathological findings are important to guide our decision between cochlear and auditory brainstem implant in inner ear malformations (IEM) with cochlear hypoplasia (CH), incomplete partition (IP), and common cavity.
Objective
Cochlear implantation (CI) has variable outcome in IEM. Histopathology of IEM is evaluated to understand the causes of this variation.
Design
Evaluation of human temporal bone specimens with IEM.
Participants
Twenty temporal bone specimens from 10 donors who had IEMs were evaluated.
Main outcome measures
Classification of IEM and analysis of cochlear morphology [inner and outer hair cells, spiral ganglion cells, and cochlear nerve (CN)] to understand the functional outcome of implantation.
Results
Among the temporal bones with IEMs, 16 were classified as CH, 2 were IPs type II, and 2 had a normal cochlear anatomy, but absent CNs. The CN was hypoplastic in 3 specimens with CH-III. The nerve was lying adjacent to the bony wall, which would be difficult to demonstrate with current magnetic resonance imaging (MRI). The mean percentage of loss of spiral ganglion cells in CH-III among our specimens was 62%. Some specimens showed asymmetric development, which will play a crucial role in the benefit from CI. Two specimens from the same donor with a fully-developed cochlea demonstrated absent CN.
Conclusion
Better MRI sequences are needed to demonstrate the hypoplastic CN adjacent to the wall of the internal auditory canal. Audiological examination is of paramount importance to demonstrate the presence of neural connection between the cochlea and the brainstem. In asymmetric developments between 2 sides, more developed side should be chosen for CI. Depending on the findings cochlear or auditory brainstem implants may be chosen for the contralateral findings. If there is only one implant available, the side with the better developed CN should be picked.

Introduction
In the first Consensus Meeting on Auditory Brainstem Implantation (ABI) in complex inner ear malformations (IEMs), 1 indications were divided into 2 groups: definite and possible. Definite indications comprise IEM such as complete labyrinthine aplasia, rudimentary otocyst, cochlear aplasia, cochlear aperture (CA) aplasia, and cochlear nerve (CN) aplasia with clear absence of neural tissue necessary for the transmission of electrical impulse from the cochlea to the brainstem, although a recent report indicates benefit from cochlear implantation (CI) in rudimentary otocyst. 2 Possible indications are CN deficiency accompanying cochlear hypoplasia (CH), CA hypoplasia, incomplete partition (IP) type I, and common cavity cases. In these cases, the child may not obtain benefit from CI procedure. In these cases, ABI or CI/ABI become better management options. The latter group continues to be a challenge to the implantation teams. Imaging and audiology are not very clear in possible indications, and human histopathological temporal bone specimens may shed some light on our understanding of this group of IEMs. In the present study, human temporal bone specimens (HTBS) with IEMs at the collection of Minnesota University and Massachusetts Eye and Ear Infirmary were evaluated from this perspective.
Materials and Methods
The study was conducted from May 2018 to April 2020 in the Otopathology Laboratories at the University of Minnesota and Massachusetts Eye and Ear Infirmary. The histopathology of HTBs with various IEMs in the laboratory’s collection was examined. We identified 20 temporal bone specimens from 10 donors who had IEMs. They were classified according to the classification system by Sennaroglu and Bajin. 3 The Institutional Review Board of the University of Minnesota (0206M26181) and Massachusetts Eye and Ear Infirmary (#2019P003272) approved this study.
To analyze cochlear hair cell losses, we individually counted the number of inner and outer hair cells in the midmodiolar and 4 adjacent sections (2 above and 2 below the midmodiolar level). The percentage of hair cell losses was estimated by calculating the ratio between the number of counted hair cells and the total number of expected hair cells in those sections (% of hair cell loss = 1 − [total number of hair cells counted/total number of expected hair cells]). The average loss of inner and outer hair cells was considered mild when less than 33%, moderate when between 34% and 66%, and severe when exceeding 67%. To analyze the population of spiral ganglion cells, we used the methodology proposed by Otte et al 4 : in all sections including the modiolus, spiral ganglion cells were outlined and individually counted with the aid of computer software, which allowed cell labeling. The raw number of spiral ganglion cells were multiplied by 106 to account for unstained sections and then multiplied by a predetermined factor of 0.9 5 to account for doubly-counted cells located at the interface between sections, and the results were compared to previously-reported normative data of spiral ganglion cells according to age. 6
Results
Type of IEM
Among the temporal bones with IEMs, 2 (10%) were classified as cochlear hypoplasia type II (CH-II), 14 (70%) were cochlear hypoplasia type III (CH-III), 2 (10%) were incomplete partitions type II (IP-II), and 2 (10%) had a normal cochlear anatomy, but absent CNs. A summary of the findings is listed in Table 1.
Summary of Inner Ear Findings in Temporal Bones with Inner Ear Malformations.

Abbreviations: A, aplastic; Asy, asymmetric; CH, cochlear hypoplasia, Modiolus classification see attachment; CN, cochlear nerve; Col, collapsed; D, dilated; Dev, still developing, normal for that age; H, hypoplastic; Hyd, hydropic; I-V refers to the degree of modiolar development, graded from I-V; Lat, lateral; N, normal; SCC: Semicircular Canal; Sup, superior.
Cochlear Hypoplasia Type 2
We found 2 human temporal bones (HTB) from 1 donor with CH-II (Figure 1). The cochlea consisted of a basal and an apical turn (without a middle turn). As per previous classifications, 7 this defect was described as partial modiolar defect: The modiolus and interscalar septa (ISS) were defective in the upper half of the cochlea; however, the interscalar septum was present between the basal and apical turns.

Cochlear hypoplasia type II at the midmodiolar level, showing reduced turns and a hypo-developed modiolus. The cochlear nerve is present and is well developed.
The otic capsule was normal with all 3 layers fully developed. Diameters of the internal auditory canal (IAC) and the CA were normal in both specimens. The CN was present and seemed normal. Morphology or width of the vestibular aqueduct were normal.
Quantitative analysis of the cochlear neuroepithelium revealed mild loss of inner and outer hair cells (mean loss of inner hair cells = 16%; mean loss of outer hair cells = 28%) in all available turns. Regarding the number of spiral ganglion neurons, we did not identify a significant difference in the number of those cells between those 2 temporal bones (right side = 26,244; left side = 26,874). As compared with age-matched controls from normative data, the number of spiral ganglion neurons was (in average) 25% lower. In those 2 specimens, the spiral ganglion neurons were proportionally distributed along all 4 segments of Rosenthal’s canal.
Cochlear Hypoplasia Type III
Our study included 14 HTBs from 7 donors who were classified as CH-III. CH-III cochlea consists of one and a half turns (normal = 2.5 turns). In all 14 specimens, the modiolus was shortened but normally developed (type II); additionally, the scala and ISS were smaller, but otherwise normal in development. The cochlear morphology between each ear from donors varied from symmetric (Figure 2) to asymmetric (Figure 3). Among the HTBs that were asymmetric, we observed mild-to-severe morphologic differences between ears (Figures 3 and 4).

Two representative human temporal bone sections from the same donor with cochlear hypoplasia type III at the midmodiolar level. The cochlear morphology between ears is quite similar.

Two representative human temporal bone sections from the same donor with cochlear hypoplasia type III at the midmodiolar level. The cochlear morphology between ears is very different: (A) In the left ear, the cochlea is more developed, and the IAC and cochlear nerves are normal. (B) In the right ear, the cochlea is smaller and has fewer turns; the IAC is narrow, and the cochlear nerve is absent. IAC, internal auditory canal.

Two representative human temporal bone sections from the same donor with cochlear hypoplasia type III at the midmodiolar level. The cochlear morphology between ears is slightly different: (A) In the left ear, the cochlea is less developed, but the IAC and cochlear nerves are normal. (B) The cochlea is slightly better developed in the right ear, and the IAC and cochlear nerves are normal. IAC, internal auditory canal.
Analysis of the bony labyrinth revealed that the 14 specimens had all bone layers (endosteal, enchondral, and periosteal) present. In 2 specimens (14.2%), all 3 layers were fully developed, but in the remaining specimens (85.8%), the middle enchondral and outer periosteal layers were in less developed stages. The IAC diameter was within normal in all specimens. The CN was present in the 14 specimens; however, in 3 specimens (21.4%) CN was hypoplastic (Figure 3B). In these 3 HTBs with the hypoplastic CN, we observed that the nerve was not in the center of the IAC but rather adjacent to the bony wall. Considering the position of the nerve, it would be difficult to correctly evaluate the development of the nerve with the currently-available magnetic resonance imaging (MRI). The vestibular aqueduct was normal in all HTBs with CH-III.
Quantitative analysis of the cochlear neuroepithelium revealed a great variability in the percentage of loss of both inner and outer hair cells. In 4 of the specimens (28.5%), the organs of Corti were rudimentary, and therefore, counting of cells was not feasible. In the remaining specimens, we observed no signs of loss of hair cells in 1 (7.1%), mild loss in 7 (50.0%), moderate in 2 (14.3%), and severe loss of inner and outer cochlear hair cells in 1 (7.1%). The mean loss of both inner and outer hair cells was 33%.
Analysis of the number of spiral ganglion neuron cells was not possible in 3 of the specimens (21.4%) due to artifacts. The results revealed that in all specimens there was at least a mild degree of loss of spiral ganglion cells affecting all the 11 remaining specimens. As compared with age-matched controls from normative data, the mean percentage of loss among our specimens was 62% (mean number of spiral ganglion cells = 13,664; expected mean count from normative data = 35,500). The percentage of loss of spiral ganglion cells varied greatly among the HTBs, and the degree of losses seemed to correlate with the degree of cochlear malformation. For example, in 1 specimen with a less developed cochlea and hypoplastic CA, not a single spiral ganglion neuron was identified (100% loss), but in 1 case that had the cochlear morphology more closely related to a normal one, there was only a 14% loss of those cells. Furthermore, we observed that in 2 HTBs there was a large number of spiral ganglion cells located within the CN in the IAC, in the region adjacent to the base of the cochlea, and not in Rosenthal’s canal. In the HTBs, which had ganglion cells in Rosenthal’s canal, we observed that the main bulk of these cells were located in the upper basal turn and early middle term (segments II and III) and in the early basal turn (segment I), in descending order. In segment IV, the number of spiral ganglion cells also varied greatly, ranging from a complete absence of cells to near-normal (as compared with normative data). As expected, we also identified a positive correlation between the degree of the development of the cochlea and number of spiral ganglion cells in segment IV, which means that cochleae with a less developed middle and upper turns had the lowest number of spiral ganglion cells in segment IV. In 1 specimen, the CN was present but spiral ganglion cells were completely absent (Figure 5).

Representative human temporal bone section from a donor with cochlear hypoplasia type III. The IAC diameter in this specimen is normal, and the cochlear nerve is present. However, the bulk of spiral ganglion neurons was located within the IAC and not in Rosenthal’s canal. IAC, internal auditory canal.
Cochlear Nerve Aplasia
We found in the TB collection of the Massachusetts Eye and Ear Infirmary 2 specimens from the same donor with a fully-developed cochlea; however, in these specimens, the CN was completely absent (Figure 6). In association with the complete aplasia of the CN trunk, it was also observed agenesis of the spiral ganglion neurons and efferent nerve fibers. The remaining bony (modiolus, ISS, scala tympani, and both vestibules) and cellular (organ of Corti, stria vascularis, spiral limbus, and spiral ligament) structures seemed normal. In both specimens, all 3 layers of the otic capsule were normal.
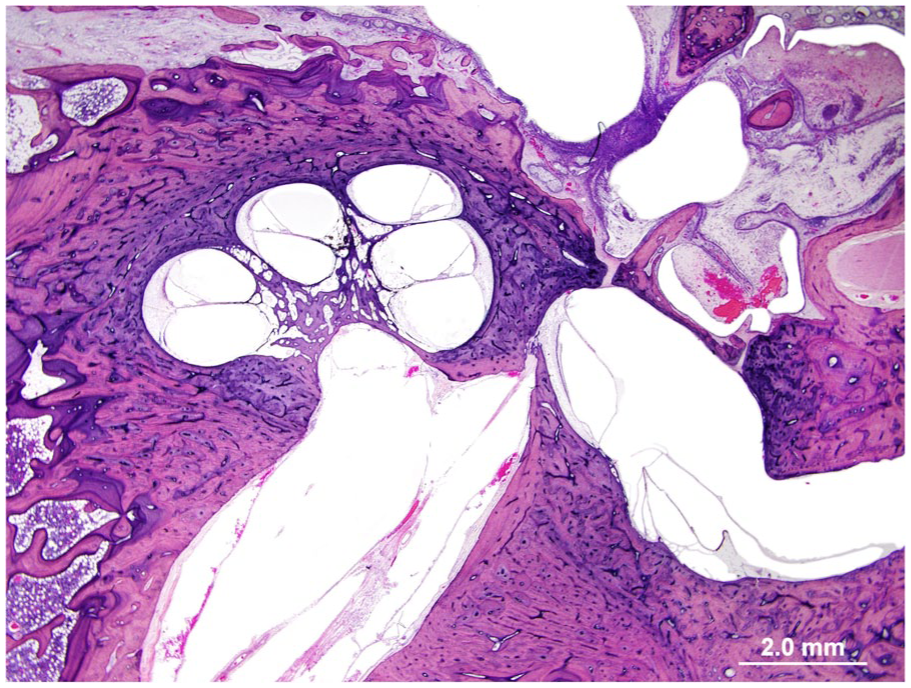
Representative human temporal bone section from a donor with normal cochlear and internal auditory canal anatomy, but the cochlear nerve was aplastic.
Incomplete Partition Type II
The HTB collection at the University of Minnesota comprised 2 HTBs from 1 donor, which had an IP-II. According to her medical records, she had normal hearing by the age of 4, and then, she developed a progressive hearing loss—she was deemed completely deaf by the age of 6. She died of heart failure at the age of 90.
The morphology of the 2 specimens was a characteristic of previous descriptions of IP-II (Figure 7): The apical turn was flattened, and internally, there was a partial upper modiolar defect, with a defective interscalar septum; additionally, there were signs suggesting hydrops of the scala vestibuli. The vestibular aqueduct was enlarged and had a patent opening into the vestibule; the endolymphatic sac was also severely enlarged. All 3 layers of bone of the otic capsule were present and within normal. The IAC appeared normal, and the cochlear, vestibular, and facial nerves were present and normal in size.

Characteristics of IP-II. (A) is the left ear and (B) the right ear. The apical turn was flattened, and internally, there was a partial upper modiolar defect, with a defective interscalar septum; additionally, there were signs suggesting hydrops of the scala vestibuli. IP-II, incomplete partition type II.
Quantitative analysis of the number of cochlear hair cells revealed severe loss in both specimens. The mean loss of outer hair cells was 96.0%, and mean loss of inner hair cells was 87%. The age of the donor at the time of death and the long-standing hearing loss should be taken into account in the advanced degree of loss of cochlear hair cells. Regarding the spiral ganglion cells, we observed a moderate loss in both specimens as compared with expected for the age of the donor (mean number of spiral ganglion cells in the 2 specimens = 11,088; normative data = 18,700; average loss of spiral ganglion cells = 41%). The spiral ganglion cells were distributed in normal proportions among all 4 segments of Rosenthal’s canal.
Discussion
It is well established in the literature that the functional hearing outcomes of CI is, in general, worse in patients with IEMs when compared to the results of patients with normal inner ear anatomy. The present study provides valuable histopathological information, which should be kept in mind when choosing the side and method of implantation (CI vs ABI), in addition to anticipating the outcomes of implantation.
Regarding the symmetry of IEMs between ears from the same donor, IEMs may be symmetric (Figure 2) or asymmetric (Figure 3A and B). In the present study, 10 (55.6%) specimens had symmetric IEMs, while 8 (44.4%) were asymmetric. In a symmetric IEM, it can be expected that similar treatment option may provide similar outcome on both sides. However, in those specimens with asymmetric IEMs, it is possible that 1 ear is better developed than the contralateral ear. This indicates that a specific hearing habilitation strategy, although sufficient to provide better hearing in 1 ear, may not be adequate to the contralateral ear. The patient should be evaluated carefully with hearing and imaging tests to decide which would be the best hearing rehabilitation strategy, including which ear and which device (HA, CI, or ABI) would achieve better functional results.
The asymmetry in cochlear anatomy may be mild or severe. Figure 4 is an example of mild asymmetry. In this specific donor, the right ear consisted of only basal turn, but the left ear had an additional apical turn. If CI was performed on both sides, it would be expected that the left ear had a better functional outcome as compared with the right ear. The relevance of this information is that—in certain countries—insurance may reimburse unilateral implantation only; in these cases, the decision process leading to which ear would obtain optimal hearing rehabilitation results is critical. Therefore, the results of the present study may provide insights to better guide surgeons on parameters that predicted the functional outcomes (the left ear, in this particular example). In addition, the quality of the CT and MRI is also a pivotal factor in this equation. If the slice thickness and reconstruction interval of the CT scans are not appropriate, it may be impossible to detect small abnormalities that could compromise the results of the hearing rehabilitation strategies. An example of severe asymmetry is given in Figure 3. In this situation, choosing the right side for implantation will certainly lead to better outcomes. Conversely, in case of unilateral implantation on the left ear, the outcome may be catastrophic, with very little benefit from CI surgery. Thus, clinicians need to carefully scrutinize CT and MRI in search for these subtle differences that could determine the better developed side, which is most likely to benefit most of CI.
A CA stenosis is diagnosed when the width of the aperture is less than 1.5 mm. This is usually associated with CN deficiency. As can be seen in Figure 3B, CN fibers in the IAC are markedly diminished; they are passing through the ossified aperture area to reach cochlea. It is very difficult to trace these CN fibers with MRI because meatal portion of the hypoplastic CN was located either adjacent or very close to the bony part of the IAC: As there is no fluid between the deficient nerve and the wall of the IAC, it is impossible to differentiate the nerve from the bone. Consequentially, the nerve is usually reported as “aplastic.” In heavily-T2-weighted 3D MRI sequences, both the nerve and the bone appear similar, and to properly differentiate them could be challenging. In this situation, it would be difficult to detect this nerve, which is hypoplastic but adjacent to bony wall. This specific HTB section raises the concern that it may be necessary to develop sequences where neural and bone tissues appear as different structures; otherwise, the CN may be deemed “aplastic” as though it is “hypoplastic.”
Considering the lower amount of neural tissue in the CN in the left ear in this HTB, it is very difficult to expect the same functional outcome of a CI as compared with a patient with a normal CN.7,8 As the contralateral ear had a normal CN, it is expected that the best hearing rehabilitation strategy for this patient would be bilateral CI, as the left would provide additional benefit to the contralateral, better developed ear. If, however, CNs were bilaterally hypoplastic, it would be very difficult to expect the same outcome with bilateral CI. The best management of a patient with bilateral hypoplastic CNs would be CI in 1 ear and an ABI on the contralateral ear.
In 2 specimens (1 donor), we observed normal bony cochlear structures, but both the CN and all spiral ganglion cells were absent bilaterally (Figure 6). Considering the absence of the CN, this donor would certainly not benefit from a CI, thus requiring an ABI for hearing rehabilitation. Therefore, the findings from the present study reveal that the assumption that the MRI would be unnecessary in patients with a normal CT scan is not valid, and therefore, preoperative MRI is mandatory for every case. 9 In this specific situation, the CT may be normal, but the MRI would corroborate the complete absence of CN and spiral ganglion. Therefore, the MRI is more important than the CT for predicting the functional outcome of CI.
One specimen has normal CA and the CN, but the spiral ganglion cells were markedly diminished (Figure 5). In this specific case, the main bulk of spiral ganglion cells were interspersed within the distal portion of the CN in the fundus of the IAC rather than in Rosenthal’s canal, and no fully-differentiated nerve fibers were seen within the internal aspect of the cochlea. To our knowledge, this is the first study to demonstrate the presence of SGCs outside of Rosenthal’s canal, within the CN in the distal part of the IAC. The clinical relevance of the observation of Spiral ganglion cells (SGCs) outside of the cochlea is still obscure. 10 With the current methodology of this study, it is impossible to affirm whether those ganglion neuron cells are functional or whether or not they could be stimulated with the electrical impulse of the cochlear implant, considering they are farther away from the cochlea. We hypothesize that such abnormality in the position of SGCs may clinically correlate with some of the poor functional results following CI observed in patients who have CH and normal CNs. This is one of the cases where speech discrimination and language development may not be satisfactory after CI, and there is no radiological method to predict this preoperatively.
The patient with IP-II had bilateral characteristic progression of hearing loss. In both specimens, we observed a severe loss of cochlear hair cells, which was expected due to the presence of a long-lasting deafness (~85 years). However, the number of SGCs revealed only moderate loss as compared with normative data for age-matched controls. The slight loss of SGCs even in patient with severe loss of cochlear hair cells have also been previously demonstrated in the literature: It has been observed that mature SGCs may survive for prolonged time periods even after complete loss of inner hair cells. 11 Therefore, it seems that the neural structures are resilient, and even prolonged periods of lack of input do not lead to severe degeneration of the SGCs. 12 These findings, in addition to the near-normal cochlear bony morphology, the normal CA and IAC, and the normal CN, would support maximum benefit for CI in this patient. In this regard, it has been demonstrated findings that corroborate our hypothesis: Among patients who have IEMs, cases of IP-II are by far the best hearing performers following CI. 3
In conclusion, in asymmetric IEMs between 2 sides, more developed side should be chosen for CI. Depending on the findings, CI or ABI may be chosen for the contralateral findings. If there is only one implant available, the side with the better developed CN should be preferred.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Institutes of Health / National Institute on Deafness and Other Communication Disorders (U24DC020851-04).
