Abstract
Importance
Head and neck cancers (HNC) impose a significant economic burden on healthcare systems. Understanding the direct medical costs across different phases of care is crucial for resource allocation and cost-effectiveness evaluations, particularly in universal healthcare settings.
Objective
To quantify the direct medical costs of HNC over 60 months postdiagnosis and examine cost variations by cancer subsite, stage, and treatment modality.
Design
Population-based, matched case–control study using administrative healthcare data.
Setting
Ontario, Canada, a province with a publicly funded universal healthcare system.
Participants
We included 19,832 adults diagnosed with HNC between 2007 and 2020. Each case was matched with 5 noncancer controls based on age, sex, and comorbidity.
Exposures
HNC diagnosis, categorized by cancer subsite, stage, and treatment modality.
Main Outcome Measures
Mean per-person direct medical costs attributable to HNC over a 63-month period, analyzed by phase of care, cancer subsite, stage, and treatment modality.
Results
The mean per-person cost attributable to HNC over 63 months was $53,812.9 ± $762.2. Costs peaked in the first 3 months postdiagnosis ($9709.7 ± $36.1 per month) and declined over time. Larynx/hypopharynx cancers incurred the highest costs across most phases. Advanced-stage cancers were associated with increased costs, with stage IV cancers nearly doubling the costs of stage I. Multimodal treatments, particularly surgery combined with chemoradiation, resulted in the highest costs across all phases (P < .01).
Conclusions
HNC results in substantial healthcare costs, with significant variations by subsite, stage, and treatment modality. The highest costs occur in the early treatment phase and remain elevated for patients requiring multimodal therapies.
Relevance
These findings provide critical data for policymakers and health system authorities to optimize resource allocation and assess cost-effectiveness. Future research should explore indirect costs and the impact of early detection strategies to reduce the economic burden of HNC.
Level of evidence
3

Graphical Abstract
Introduction
Head and neck cancers (HNCs) include malignancies localized within the upper aerodigestive tract, ranking as the seventh most prevalent cancer type globally. 1 Recent decades have witnessed a notable increase in HNC survivors, primarily due to the surge in Human Papillomavirus (HPV)-associated oropharyngeal cancers and improvements in survival rates.2 -4 Furthermore, the aggressive nature of HNC often necessitates a multimodal treatment approach and frequently results in persistent morbidity and prolonged medical care, contributing to substantial healthcare costs.5,6
Efforts to estimate the economic burden of HNC have highlighted substantial healthcare costs associated with these malignancies.3,6 -12 Despite numerous studies on the clinical advancements in HNC, the direct economic burden of HNC for healthcare systems remains inadequately studied. 6 A comprehensive understanding of the direct medical costs attributable to HNC, along with the trajectory of these costs following diagnosis, is critical for helping health system authorities plan for rising costs as well as enabling cost-effectiveness analyses for health economists and policymakers. The objectives of this study were to: (1) quantify the direct medical costs attributable to HNC during the first 5 years after diagnosis and describe the monthly cost trajectory by cancer subsite, stage, and treatment modality; and (2) compare these costs to those incurred by noncancer controls.
Methods
Study Setting and Design
This retrospective cohort study with matched controls was conducted at IC/ES in Ontario, Canada, a nonprofit organization authorized to collect and analyze health care and demographic data without consent. As such, this study does not require review by a Research Ethics Board. Ontario provides universally accessible physician services to 14.6 million population under the Ontario Health Insurance Plan (OHIP). 13 For HNC care, surgeries and radiotherapy are reimbursed through the OHIP. Systemic therapy that is intravenous and takes place at a hospital are funded through Ontario Health—Cancer Care Ontario’s New Drug Funding Program (NDFP); the Ontario Drug Benefit (ODB) provides coverage for oral chemotherapy drugs and other take-home drugs for older adults (age 65 or above) and those living in a special care home or receiving social assistance.
Data Sources
Administrative datasets were linked using unique encoded identifiers and analyzed at IC/ES (Supplemental Appendix I). The Ontario Cancer Registry (OCR) captures 96% of cancer diagnoses across the province.14,15 Client Agency Program Enrollment (CAPE) identifies primary care providers and their rostered patients. 16 The Registered Persons Database (RPDB) maintains the demographic information of OHIP-eligible individuals. Statistics Canada’s Postal Code Conversion File (PCCF) was used to create neighborhood rurality status. 17 The Immigration, Refugees and Citizenship Canada (IRCC) Permanent Resident Database (with data from January 1985 to May 2017) includes records of individuals who immigrated to Ontario during this period. The Ontario Marginalization Index (ON-MARG) database reports neighborhood-level access to material resources and ethnic diversity.18,19 To identify healthcare resources use, at the provincial level, the OHIP claims database holds all physician billings; the NDFP database identifies patients receiving injectable cancer drugs at a hospital; the ODB database records oral drug use for the eligible population; and the Assistance Devices Program (ADP) database identifies individuals with physical disabilities who require a medical device. At the national level, the Canadian Institute for Health Information (CIHI) collects data from local health facilities and authorities on hospital discharges, day surgeries, emergency department visits, outpatient clinic visits, rehabilitation, continuing and complex care, and home and long-term care.
Study Cohorts and Matching (Case-Control)
We created the primary study cohort (case) to comprise OHIP-eligible residents of Ontario who received a diagnosis of HNC (index event) at age 18 or above between January 1, 2007 and October 31, 2020. HNC diagnoses were identified using the International Classification of Diseases for Oncology, version 3 (ICD-O-3) codes from the OCR (Supplemental Appendix II). We included patients with new diagnoses of squamous cell carcinoma of the HNC of either oral cavity, oropharynx or larynx/hypopharynx anatomic sites. Following our previous work,20,21 we excluded patients who received radiotherapy within 180 days before the index or Cetuximab 180 days after the index. We further excluded patients with any cancer diagnosis 5 years before the index (see flow diagram in Supplemental Figure 1). We followed all case patients from the index date for 60 months, or to the date of death, of a new cancer diagnosis, or to October 31, 2021, or to the end of OHIP registration (due to moving out of Ontario), whichever occurred first.
Using the RPDB database, we constructed a control cohort by hard matching each case patient with 5 adults from the general population who did not have a HNC diagnosis (control) up to October 31, 2021, in addition to having no cancer diagnosis 10 years before the assigned index date. Each control was assigned the same index date as their matched case, and their healthcare utilization was followed over the same time window, beginning 3 months before and extending up to 60 months after the index date. The matching was based on age (within 1 year), sex, and the Elixhauser comorbidity index calculated using hospitalization records 5 years before the index date 22 ; these variables were chosen as they were believed to strongly influence the use of healthcare services.23 -26 Controls matched to a case patient who later died were censored on that death date. If a control died, we reweighted the remaining controls to maintain the matching ratio. 27
Calculating Monthly Costs
We used the GETCOST, a patient-level case costing macro available at IC/ES, to calculate individual’s direct medical costs from the perspective of the Ontario public healthcare payer. 28 Details of the GETCOST can be found elsewhere.28,29 In brief, the costs of visit/claim-based encounters are determined at utilization, and the costs of episodic services are estimated using a cost-per-weighted-case method that involves a resource utilization weight. These costs cover a range of healthcare components, including physician services, outpatient visits, hospital admissions, pharmaceuticals, medical devices, laboratory testing, and complex as well as home care services. 30 All costs were adjusted to 2020 Canadian dollars. For each individual, we calculated their monthly costs by running the GETCOST macro for each 30-day window during which they were fully observed. To estimate phase-specific monthly costs, we created 5 clinically meaningful HNC care phases over a 63-month period, 3 including a 3-month prediagnosis phase (3 months before the index), 3 treatment phases (0-3, 4-6, and 7-12 months after the index), and a survivorship phase (13-60 months after the index). HNC-attributable monthly costs were defined as the excess healthcare costs in HNC cases compared to matched controls over the same time intervals, calculated as the mean monthly cost difference between groups.
Outcomes
Primary outcome
Our primary outcome was mean per-person monthly costs attributable to HNC, both overall and by phase of care. To estimate the overall HNC-attributable costs, we calculated the average per-person excess monthly costs incurred by the case over the matched control over the entire 63-month period.11,24,26 For each phase of care, we regressed individual’s monthly costs on their cohort type (case vs control) in a mixed-effect linear regression model. We used the estimated coefficient and the associated standard deviation (SD) of cohort type to approximate the mean monthly costs attributable to HNC for that phase of care.
Secondary outcomes
Using only the case cohort (HNC patients), we calculated the mean monthly costs for each HNC subsite, stage, and treatment modality. Specifically, we used the OCR diagnosis records to identify 3 subsites (oral cavity, larynx/hypopharynx, and oropharynx; Supplemental Appendix II). Stage data (0/unknown/missing, I, II, III, IV) derived from pathology reports were also obtained from the OCR. We classified 6 treatment modalities following an established algorithm20,21: surgery alone, radiotherapy alone, chemoradiotherapy, surgery + radiotherapy, surgery + chemoradiotherapy, and no treatment/palliative care.
Statistical Analysis
We compared the distribution of characteristics between the case and the control cohorts using a standardized difference (std diff) >0.10 to indicate a significant imbalance. 31 We plotted the mean and cumulative monthly costs for the 2 cohorts to illustrate the trajectory of their cost differentials over time. Similarly, for the case cohort alone, we plotted the mean and cumulative monthly costs for each HNC subsite, stage, and treatment modality; linear regressions were conducted to confirm an overall difference in mean monthly costs by these variables. Linear regression was chosen over other models for continuous outcomes due to its simplicity, ease of interpretation, and the ability to directly estimate mean differences across groups. Additionally, with the large sample size in our study, linear regression is robust to deviations from normality in residuals, making it a suitable choice for our analysis. Analyses were 2-sided where P-value <.05 signaled statistical significance. We performed all analyses using SAS Enterprise Guide version 7.15 (SAS Institute Inc., Cary, NC, USA).
This study was conducted and reported in accordance with the REporting of studies Conducted using Observational Routinely collected Data (RECORD) guidelines to ensure transparency and completeness in the presentation of our findings. 32
Results
Cohort Characteristics
We included 19,832 adults diagnosed with HNC between January 2007 and October 2020 in the case cohort. Matching each case with 5 adults from the general population yielded a control cohort of size 99,160. In Table 1, we report that the 2 cohorts were indeed balanced in terms of age, sex, and comorbidity at the index. However, when comparing to the controls, patients in the case cohort were less likely to be an immigrant (7.7% vs 14.5%, std diff = 0.22), a resident of Central Ontario (25.0% vs 29.7%, std diff = 0.11), or living in a neighborhood in the most ethnically diverse quintile (17.5% vs 23.3%, std diff = 0.15). HNC case patients had higher mortality and second cancer rates, leading to shorter follow-up durations and lower likelihood of reaching the study end date or completing the full 5-year follow-up compared to controls (Table 1).
Characteristics of the Study Cohort (n = 118,992).

Abbreviations: HNC, head and neck cancer; SD, standard deviation.
The index represents the date of HNC diagnosis for the case cohort. We hard matched each case with 5 controls based on the index month and year, age (±1 year) at index, sex, and the Elixhauser comorbidity groups (excluding cancer) calculated using hospitalization records during the 5 years before the index. Individuals in the control had no diagnosis of any cancer during a 10-year period before the index. Bold values indicate standardized differences > 0.1, suggesting meaningful imbalance between groups.
Variables used for matching.
These variables had missing data with proportions ranging from 0.1% to 0.9%. The distribution of missing data did not differ between the case and the control (all standardized differences <0.03).
Mean Monthly Costs Attributable to HNC
We report the mean monthly costs (in 2020CAD) for the case and the control cohorts separately within each phase of care (Table 2, Figure 1). Mean (±SD) monthly costs attributable to HNC, that is, the excess costs incurred by the case over the controls, were the lowest during the 3-month pre-diagnosis care phase ($148.2 ± $18.1) and peaked over the first 3 months after the diagnosis ($9709.7 ± $36.1). For the next 3 months, mean monthly costs attributable to HNC reduced by 2-folds ($4281.6 ± $29.5) and continued to decrease to $1444.4 ± $22.7 (7-12 months after the diagnosis), and eventually remained at $877.1 ± $20.5 for the survivorship care phase (13-60 months after the diagnosis). Overall, we estimated the mean per-person costs attributable to HNC over the entire 63-month period to be $53,812.9 ± $762.2 in total or $854.2 ± $12.2 for each month.
Mean Monthly Costs in the Case and the Control Cohorts.

Abbreviations: HNC, head and neck cancer; SD, standard deviation.
We calculated the overall mean monthly costs by pooling the costs of patients with a full 63-month observation window.
Case–Control values are mean monthly excess costs attributable to HNC, estimated from a linear mixed-effects model with case–control status as the predictor and a compound symmetry structure for repeated monthly measures. Values are least-squares mean differences (±standard error).

Monthly (A) and cumulative (B) mean costs of the case and the control.
Mean Monthly Costs by HNC Subsite, Stage, and Treatment Modality
Using our case (HNC patients) cohort, we found the mean monthly costs to differ significantly by HNC subsite, stage, and treatment modality (all P-values <.01; Table 3).
Mean Monthly Costs in the Case Cohort Stratified by Cancer Subsite, Stage, and Treatment Modality.

Abbreviation: SD, standard deviation.
We calculated the overall mean monthly costs by pooling the costs of patients with a full 63-month observation window.
When stratified by subsite, patients with larynx/hypopharynx cancer incurred the highest mean (±SD) monthly costs in 4 of the 5 care phases, with costs peaking at $12,489.4 ± $15,496.2 in the first 3 months after diagnosis—a more than 13-fold increase from prediagnosis costs of $986.0 ± $3016.0 (Figure 2). Over the entire 63-month period, larynx/hypopharynx cancer patients had the highest mean monthly costs at $1401.5 ± $4667.8, followed by oropharyngeal cancer ($1329.2 ± $4345.1) and oral cavity cancer ($1134.7 ± $4293.5). For cancer stage, the mean monthly costs increased with advancing stage in every care phase (Figure 3). Estimations of mean monthly costs over the entire 63-month window were $861.1 ± $3171.5, $1287.0 ± $4512.6, $1520.1 ± $4880.8, and $1623.5 ± $5276.0 for stages I, II, III, and IV cancer, respectively, while patients with stage 0 or unknown/missing stage information reported an average cost of $1053.8 ± $3810.9 per month. Over the entire 63-month window, patients with stage IV cancer incurred mean monthly costs nearly double (88%) those of patients with stage I cancer. In terms of treatment modality (Figure 4), patients receiving surgery + radiotherapy reported the highest overall monthly mean costs ($1728.9 ± $5588.3), while patients with palliative or no treatment who incurred the lowest monthly mean costs ($909.7 ± $3371.0). Notably, the costs for surgery + radiotherapy were higher compared to those for chemoradiotherapy, which had a lower mean monthly cost of $1460.5 ± $4809.1 Regardless of the treatment modality, all patients reached the peak of their mean monthly costs during the first 3 months after the diagnosis, although recipients of surgery + chemoradiotherapy ($13,892.0 ± $14,501.7) costed almost twice as much as recipients of palliative care/no treatment ($7151.1 ± $14,315.7).
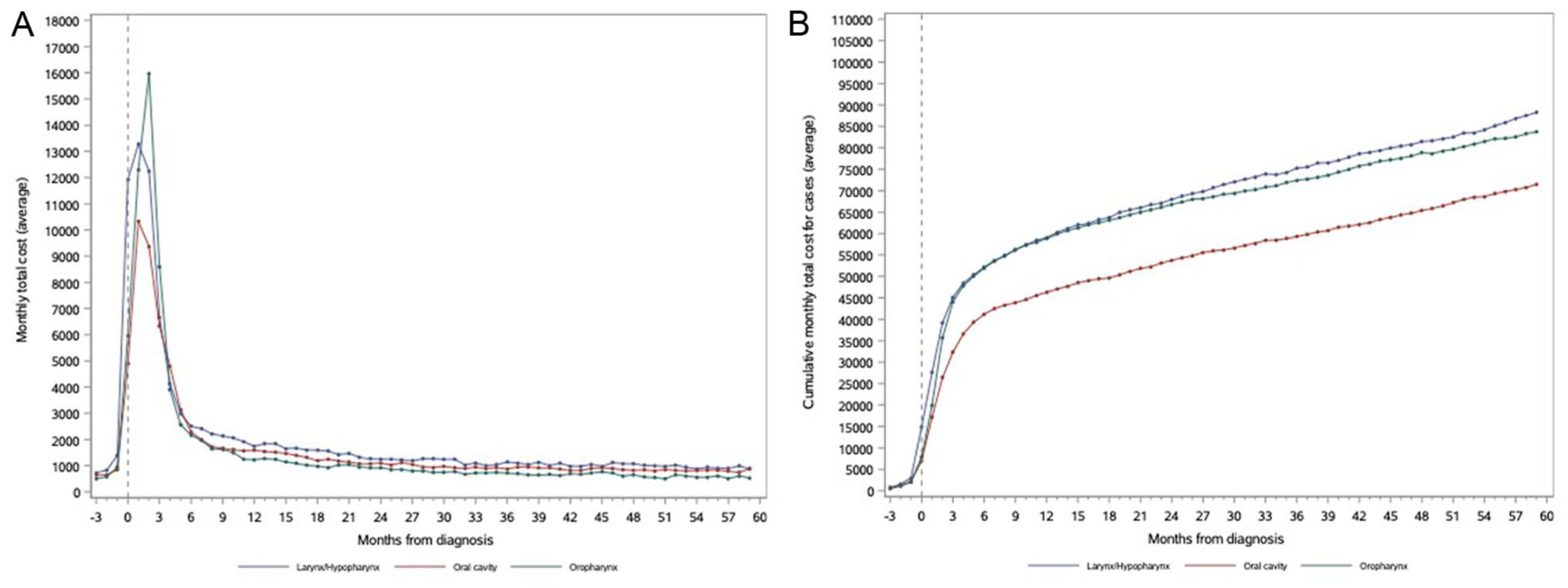
Monthly (A) and cumulative (B) mean costs of the case cohort by cancer site.

Monthly (A) and cumulative (B) mean costs of the case cohort by stage.
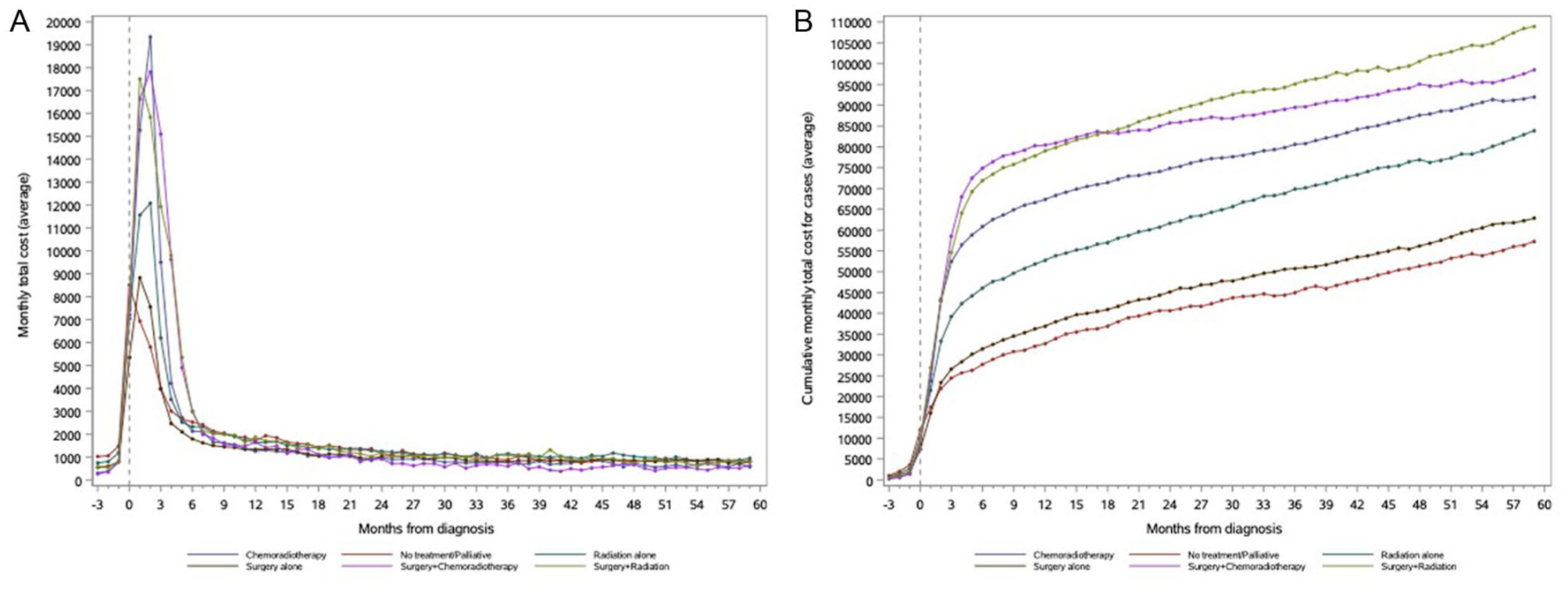
Monthly (A) and cumulative (B) mean costs of the case cohort by treatment modality.
Discussion
This is the first population-based study presenting detailed monthly data on the direct medical costs associated with HNC. We provided a precise estimate of the per-person direct medical costs attributable to HNC over a 63-month period. Our findings show that peak costs occurred in the initial 3 months following diagnosis, likely due to intensive diagnostic and therapeutic interventions. Differences in direct cancer care costs were observed based on both cancer site and treatment characteristics. Advanced stages and cases requiring multiple modalities of treatment, such as surgery combined with chemoradiation, had the highest monthly costs. This direct association between advanced cancer stage, multimodal therapies, and increased costs has been consistently reported in previous studies.6,10,34,35 However, the higher costs observed for patients receiving surgery plus radiotherapy compared to those receiving surgery plus chemoradiotherapy may reflect differences in patient health status. Patients ineligible for chemotherapy often have poorer baseline health or postoperative complications, leading to greater healthcare utilization and, consequently, higher overall costs.
Our study found that larynx/hypopharynx cancers were associated with the highest monthly costs across most phases of care, followed by oral cavity cancers. This corroborates findings from both the Swedish and Italian healthcare systems, which also reported the highest overall direct costs for larynx/hypopharynx cancers, followed by oral cavity cancers.12,36 These higher costs may be attributable to the multimodality treatments typically used for these subsites, such as radiation/chemoradiation followed by salvage surgery for larynx/hypopharynx cancers, which often have high recurrence rates. 7 Furthermore, even larynx cancers that are not treated with surgery often require diagnostic procedures like laryngoscopy with biopsy, further adding to costs.33,34
Higher costs of care for HNC survivors compared to controls remained significant up to 5 years after diagnosis. The highest direct costs during the survivorship period were consistently observed in patients undergoing multiple modalities of treatment. Increased costs in this period may be attributed to late toxicities, sequelae, and ongoing cancer surveillance.8,11,37 This corroborates findings from a recent study by Massa et al, which reported increased overall costs of treatment in the survivorship period for patients undergoing multiple modalities of treatment. 3 Therefore, our study further highlights the critical need for optimal resource allocation with a long-term perspective. Long-term costs are particularly important when assessing the cost-effectiveness of different treatment modalities. For example, surgery may result in higher immediate complications, while radiation is more likely to lead to long-term toxicities.38,39 Understanding these trajectories is crucial for guiding treatment decisions and ensuring optimal resource allocation.
Our study uniquely contributes to HNC cost analysis by providing a detailed monthly breakdown of costs over a 5-year period by subsite, stage, and treatment modality—a level of detail that is rare in existing literature, especially within systems offering universal health coverage. 35 While prior studies have typically focused on overall costs,3,6 our emphasis on monthly costs provides deeper insights into cost trajectories and resource utilization over time. Many previous studies also concentrated on shorter follow-up periods,6,7,36 often excluding the survivorship phase from direct cost evaluations. Additionally, by comparing cases to matched controls, we were able to isolate the excess costs directly attributable to HNC, providing a more accurate estimate of the financial impact of the disease.
Despite the robust methods used in our study, there are limitations to consider. First, administrative databases are not ideal for identifying tumor stage and site; therefore, our cohort accrual was limited to October 2020 to ensure the full maturity of these data from our cancer registry. Additionally, the study used only the Ontario healthcare system as the payer, meaning that other direct medical cost items meaningful to patients and society, such as out-of-pocket expenses for out-of-hospital dental care and most outpatient medications, were not captured. Another limitation of this study is the generalizability of its findings to nonuniversal healthcare systems, where differences in insurance coverage, access to care, and out-of-pocket expenses could impact overall costs. However, the use of a universal healthcare setting allows for a comprehensive capture of real-world costs, providing estimates that reflect true healthcare resource utilization without being influenced by variations in insurance status. Furthermore, we did not consider any indirect costs associated with HNC, such as lost productivity and mobility, job insecurity, and other patient-borne consequences. This necessitates further investigation beyond our current costing method and data sources. Lastly, because the AJCC staging system was revised twice during the study period, staging classifications may have varied by year of diagnosis, although this likely impacted only a small proportion of patients.
Cancer care represents a significant portion of the healthcare budget, and these costs are expected to grow due to increasing survival rates and rising treatment prices. 35 While previous studies have evaluated direct costs associated with various cancers, our study provides a more detailed analysis of HNC costs by offering a monthly breakdown of expenses over a 5-year period. Studying HNC costs specifically is crucial for several reasons. First, new treatment modalities, such as immunotherapy and advanced surgical techniques like robotic surgery, come with higher costs.37 -39 Second, the treatment of these cancers often involves complex and costly interventions, such as extensive surgeries, radiation, and chemoradiotherapy. These treatments not only have distinct cost trajectories but also long-term impacts on patient function and disability, making it critical to understand their financial impact on the healthcare system.3,7,8,40,41 Finally, the increasing survival rates due to the rise in HPV-associated oropharyngeal cancer add to the importance of assessing long-term care costs.42,43 By examining costs across different subsites and stages of HNC, our study provides valuable data for guiding cost-effective strategies within a publicly funded, universal healthcare system, helping policymakers optimize clinical outcomes while ensuring the financial sustainability of care. Although descriptive, our findings may inform future economic evaluations that can compare the costs and benefits of different treatment approaches. Additionally, as higher costs were observed in advanced stages, future studies should explore the cost-effectiveness of early detection strategies, as early diagnosis may help reduce the need for more intensive and expensive treatments, alleviating the overall economic burden on the healthcare system.44 -46
Conclusion
This cost of illness study presents a comprehensive understanding of the direct and long-term cost associated with HNC. Treatment modality seems to be the main driver of direct costs in the treatment of patients with HNC cancers and remain elevated during the survivorship period. Further studies are warranted to explore the indirect costs of HNC, integrate patient-reported outcomes into economic evaluations, and assess the cost-effectiveness of early detection and intervention strategies.
Supplemental Material
sj-docx-1-ohn-10.1177_19160216251406521 – Supplemental material for Head and Neck Cancers Medical Costs in a Universal Health System: A Population-Based Case-Control Study
Supplemental material, sj-docx-1-ohn-10.1177_19160216251406521 for Head and Neck Cancers Medical Costs in a Universal Health System: A Population-Based Case-Control Study by Noémie Villemure-Poliquin, Rui Fu, Kelvin K. W. Chan, Qing Li, Kennedy Ayoo, Frances Wright, Irene Karam, Natalie G. Coburn, Julie Hallet and Antoine Eskander in Journal of Otolaryngology - Head & Neck Surgery
Footnotes
Acknowledgements
ICES Funding Acknowledgment: This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health (MOH) and the Ministry of Long-Term Care (MLTC).
Required PCCF Acknowledgment: This document used data adapted from the Statistics Canada Postal CodeOM Conversion File, which is based on data licensed from Canada Post Corporation, and/or data adapted from the Ontario Ministry of Health Postal Code Conversion File, which contains data copied under license from ©Canada Post Corporation and Statistics Canada.
IRCC Acknowledgment: Parts or whole of this material are based on data and/or information compiled and provided by Immigration, Refugees and Citizenship Canada (IRCC) current to October 31, 2020. However, the analyses, conclusions, opinions and statements expressed in the material are those of the author(s), and not necessarily those of IRCC.
MOH & CIHI Data Provider Acknowledgment: Parts of this material are based on data and/or information compiled and provided by CIHI and the Ontario Ministry of Health. The analyses, conclusions, opinions and statements expressed herein are solely those of the authors and do not reflect those of the funding or data sources; no endorsement is intended or should be inferred.
OH Data Provider Acknowledgment: Parts of this material are based on data and information provided by Ontario Health (OH). The opinions, results, view, and conclusions reported in this paper are those of the authors and do not necessarily reflect those of OH. No endorsement by OH is intended or should be inferred.
Toronto Community Health Profiles Acknowledgment: We thank the Toronto Community Health Profiles Partnership for providing access to the Ontario Marginalization Index.
ORGD Acknowledgment: Parts of this report are based on Ontario Registrar General (ORG) information on deaths, the original source of which is ServiceOntario. The views expressed therein are those of the author and do not necessarily reflect those of ORG or the Ministry of Public and Business Service Delivery.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Ontario Ministry of Health and the Ontario Ministry of Long-Term Care (annual grant is awarded to the Institute for Clinical Evaluative Sciences).
Ethical Approval
This retrospective cohort study with matched controls was conducted at IC/ES in Ontario, Canada, a nonprofit organization authorized to collect and analyze health care and demographic data without consent. As such, this study does not require review by a Research Ethics Board.
Data Availability Statement
Data and codes used in this study are available from the corresponding author upon reasonable request, subject to data sharing agreements and ethical approval requirements.
Supplemental Material
Additional supporting information is available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
