Abstract
Importance
Onabotulinumtoxin A (BTX) is the first-line treatment for laryngeal dystonia (LD). The US FDA designated BTX as a pregnancy category C drug because there are no well-controlled studies in pregnant women.
Objectives
To describe pregnancy and neonatal outcomes in pregnant LD patients treated with BTX injections and to assess the current practice and comfort level of Canadian laryngologists in treating these patients.
Design
Case Series and Survey.
Setting
Academic, tertiary care, laryngology center.
Participants
Pregnant women who received BTX for LD from 2010 to 2020.
Intervention
A retrospective chart review was conducted for eligible patients. An anonymous, online, cross-sectional survey was distributed to Canadian laryngologists, according to Dillman’s Total Design method.
Main outcome measures
Pregnancy and neonatal outcomes.
Results
Four adductor LD patients received BTX during each of their 2 pregnancies; 8 pregnancies were examined. Patients received a median of 4.5 treatments (IQR 3) with a median cumulative dose of 5.12 U (IQR 2.93 U) over the whole pregnancy. One pregnancy was pre-term and the median APGAR score was 9 (IQR 0.5). All mothers breastfed their infants. There were no adverse events (AE) and all children were healthy, with a median age of 7 (IQR 2). Twenty-three Canadian laryngologists responded to the survey, for an 85.2% response rate. Three laryngologists (13.0%) have previously injected BTX in a total of 5 pregnant women. All 5 pregnancies went to term with no AE. The majority (68.4%) of respondents indicated that they would offer BTX injections to pregnant LD patients after obtaining informed consent.
Conclusion
This case series was the largest in the literature of pregnant LD patients undergoing BTX treatment. The majority of Canadian laryngologists would offer the procedure to pregnant women with informed consent.
Relevance
This study added to the growing literature on the safety of BTX injections in LD patients during pregnancy.

Key Messages
Onabotulinumtoxin A (BTX) for the treatment of laryngeal dystonia (LD) is a widely accepted first-line treatment. The US Food and Drug Administration designated BTX as a pregnancy category C drug because there are no well-controlled studies in pregnant women.
This case series was the largest in the literature of pregnant LD patients undergoing BTX treatment. Four adductor LD patients each had 2 pregnancies. All 8 children were healthy with no adverse events during pregnancy or the neonatal period.
A cross-sectional survey of Canadian laryngologists reported that the majority would offer pregnant LD patients BTX injections with informed consent.
Introduction
Onabotulinumtoxin A (BTX) (BOTOX®/BOTOX® Cosmetic, Allergan, Inc., Irvine, CA, USA) is approved by the United States Food and Drug Administration (FDA) for the treatment of urinary incontinence from detrusor overactivity, migraine headaches, upper limb spasticity, cervical dystonia, axillary hyperhidrosis, blepharospasm, strabismus, and cosmetic indications for treatment of glabellar and lateral canthal lines.1 -3 The use of BTX for the treatment of laryngeal dystonia (also known as spasmodic dysphonia) is an off-label indication, but it has become a widely accepted first-line treatment.4,5 In fact, the American Academy of Otolaryngology—Head & Neck Surgery recommended its use for laryngeal dystonia patients in the Clinical Practice Guidelines on Hoarseness. 6
Currently, the FDA designates Onabotulinumtoxin A as a pregnancy category C drug, which means that “Animal reproduction studies have shown an adverse effect on the fetus and there are no adequate and well-controlled studies in humans, but potential benefits may warrant use of the drug in pregnant women despite potential risks”.1,2,7 There are practical and ethical challenges with recruiting pregnant women for research studies to determine drug safety in pregnancy. Numerous clinical trials routinely exclude pregnant women. In fact, in 97.5% of drugs approved by the FDA from 2000 to 2012, the teratogenic risk in human pregnancy was “undetermined”. 8
Many women of reproductive age are treated with BTX for the indications listed above. 9 There are case reports of pregnant patients receiving BTX for cosmetic reasons, migraines, cervical dystonia, achalasia, and strabismus.9 -22 Allergan published a 24-year retrospective review of BTX administration during pregnancy in their Global Safety Database. Three cases of pregnant laryngeal dystonia patients were noted, but the results were aggregated with the 232 pregnancies from all indications. 9
Laryngeal dystonia (LD) is a rare neurological disorder of unknown etiology that is characterized by focal involuntary spasms of intrinsic laryngeal muscles during specific speech items.23 -25 LD is an enigmatic and devastating voice disorder that impairs communication.26 -28 Depending on the muscles involved, LD is categorized into adductor, abductor, and mixed; adductor LD is the most common.5,29 The most common decade of LD onset is 40 to 50 years of age, with a reported female to male ratio of up to 4:1.29 -33 As LD can occur in women of reproductive years, it is important to explore whether the mainstay treatment is considered safe during pregnancy.
The primary objective of this study was to evaluate pregnancy and neonatal outcomes of LD patients treated with BTX injections during pregnancy at a tertiary, academic practice. The secondary objectives were to determine the current practice of Canadian laryngologists in treating pregnant patients with LD and to assess their comfort level with administering BTX during pregnancy. Based on previous case reports and the physiologic understanding of BTX, we hypothesized that there would be no adverse events (AE) reported in our patient population.
Methods and Materials
Retrospective Chart Review
After receiving Research Ethics Approval from the University of British Columbia (UBC) (REB # H20-03577), a retrospective chart review was conducted from January 1, 2010 to December 30, 2020 at an academic tertiary care center. The principal investigator’s patients with LD were identified by billing codes (n = 131). This patient roster was then further examined to identify women of childbearing age (18-43). Charts were reviewed to identify which patients received BTX injections during pregnancy. Patients were then identified and contacted to obtain consent.
A patient survey was administered when the participants visited the principal investigator for their next regular BTX injection from January 1, 2020 to December 31, 2021. Written permission was obtained from patients to access obstetrical records which confirmed the details of their past pregnancies. Each pregnancy was treated as an independent unit of analysis. Otolaryngology and Obstetrical charts were reviewed. Children’s medical records were not explicitly reviewed. Instead, the self-reported, subjective perceptions of the mothers concerning their children’s health were collected at the time of follow-up.
A single fellowship-trained laryngologist performed all the BTX injections. Laryngeal electromyography (EMG) (AccuGuide Injection Monitor, Medtronic Xomed, Jacksonville, FL, USA) was used to guide the BTX injections with the technique described by Blizter et al. 5 Onabotulinumtoxin A (BOTOX®, Allergan Inc) was reconstituted in preservative-free saline to a final dose concentration of 2 U/0.1 ml. BTX was injected unilaterally or bilaterally into an active area of the thyroarytenoid muscle for adductor SD patients. The PI’s practice was to start a new patient with a 1 U bilateral thyroarytenoid dose. If a patient developed side effects of breathiness or dysphagia that were unacceptable to the patient, then a unilateral dose was offered. Doses were then titrated to maximize benefits and minimize side effects. The technique was not changed because the LD patients were pregnant.
The primary outcome of this study was to evaluate pregnancy and neonatal outcomes after BTX injection(s) were administered for LD during pregnancy. Specific data extracted from the medical records included: Fetal outcomes of gestational age, genetic abnormality, fetal defects, appearance, pulse, grimace, activity, and respiration (APGAR) scores, birth complications, significant AEs to mother and/or fetus, maternal age, timing of injection, injection dose, and breastfeeding status.
Statistical analysis was completed using Microsoft Excel. Due to the small sample size and non-normal distribution of the data, median and interquartile ranges were calculated.
Survey of Treating Physicians
The second part of the study was also approved by the Research Ethics Board at UBC (REB # H22-02774), and the consent was obtained from each participant prior to survey initiation. An online, anonymous, cross-sectional survey was sent between November 8 and December 28, 2022 to all the Canadian laryngologists who are members of the laryngology committee of the Canadian Society of Otolaryngology—Head & Neck Surgery. For centers without a laryngologist (eg, University of Saskatchewan and Memorial University), the general otolaryngologist who performed BTX injections on LD patients were included. The total denominator of potential participants was 27. The survey was offered in French and in English.
The survey was distributed according to Dillman’s Total Design Method, 34 since it has been proven to enhance survey response rates. Respondents were contacted the first time in week 1 (November 8) with the invitation to participate and the survey link. In weeks 2, 4, and 7, reminders and survey links were sent to potential participants who did not answer the survey at that time. After week 8 (December 28), survey enrollment was closed. The survey data were collected on UBC’s secure Qualtrics Survey Program (Qualtrics, Version November 2022, Copyright © 2020, Provo, UT, USA).
The survey was composed of 13 questions (Supplemental Appendix 1) and adapted from Morgan et al’s original survey of neurologists’ and physiatrists’ practice patterns for treating pregnant patients who required BTX. 21 The survey first screened whether participants offered BTX injections to LD patients. Details about the BTX practice were elicited, for example, the size of the LD practice. Participants were asked whether they had previously injected BTX in pregnant LD patients and, if so, what their patients’ pregnancy outcomes had been. The survey also evaluated the level of comfort of each physician and their preferred management option for BTX injections in pregnant patients.
Results
Retrospective Chart Review
Four patients with adductor LD were identified to meet the inclusion criteria. Each patient had 2 separate pregnancies while receiving BTX treatment for LD. Therefore, we analyzed 8 separate singleton pregnancies which received BTX treatment for LD.
Six of 8 pregnancies received BTX treatment in all 3 trimesters. Six of 8 pregnancies received BTX treatment in the first trimester. Patients received a median of 4.5 treatments (IQR 3) during their pregnancies, with a median dose of 1.0 U (IQR 0.5 U) per vocal fold per treatment. The median cumulative dose in the whole pregnancy was of 5.12 U (IQR 2.92 U). Median maternal age at the time of birth was 33.5 (IQR 2.75) years. Pregnancy and Botox characteristics are listed in Table 1.
Pregnancy and Botulinum Toxin A Characteristics of the Study Population.

T1,T2, and T3 BTX = First, Second, and Third Trimester Frequency of Onabotulinumtoxin A Injections.
Total BTX = Total Frequency of Onabotulinumtoxin A injections during pregnancy.
T1, T2, and T3 dose = First, Second, and Third Trimester Total Dose of Botulinum toxin A for each treatment in units.
Total dose = Total Cumulative Dose of Botulinum toxin A injections during each pregnancy in units.
Abbreviations: GPA, Gravida, Para and Abortis; Pt., patient.
Of 8 deliveries, 1 was preterm and 7 were term. There were no fetal losses. Six were cesarean sections and 2 were vaginal. Median APGAR score was 9 (IQR 0.5). All the infants were breastfed while the patient received laryngeal BTX injections. No significant morbidity or mortality was noted to mother or child, except for the 1 preterm birth. All children are now alive and well, with a median age of 7 (IQR 2). Overall, no AEs were noted. Please refer to Table 2 for these results.
Delivery and Infant Characteristics of the Study Population.

Total dose = Total Cumulative Dose of Botulinum toxin A injections during each pregnancy in units.
APGAR 1 and 2 = Appearance, Pulse, Heart Rate, Grimace, Activity (10/10 Possible). First APGAR score is performed 1 minute after birth, and the second APGAR score is performed 5 minutes after birth.
Abbreviations: BRF, did the patient breastfeed; C/S, cesarean section; GA, gestational age in weeks; GPA, Gravida, Para and Abortis; SVD, spontaneous vaginal delivery; TOB, type of birth; Pt., patient.
Survey of Canadian Laryngologists
Twenty-three responses were received from 27 Canadian laryngologists, for a response rate of 85.2%. Two respondents were excluded because they did not treat LD patients: one was a pediatric otolaryngologist on the laryngology committee, and another was a new laryngologist with no LD patients in the practice. Twenty-one out of 23 responders (91.3%) were treating LD with BTX injections, with a median of 36 (IQR 27.5) patients in their practice. Other indications for BTX injections were cricopharyngeal dysfunction (n = 18), muscle tension dysphonia (n = 10), paradoxical vocal fold dysfunction (n = 9) and sialorrhea (n = 8).
Only 3 of the 23 respondents (13.0%) had previously injected BTX in 1 to 2 pregnant patients each, for a total of 5 women. The indications were laryngeal dystonia (n = 4) and palatal myoclonus (n = 1). Of the 5 women, 2 were injected during their first trimester, while 1 was during the second and 2 during the last trimester. The 5 pregnancies went to term with no AEs in the postnatal period. All 3 laryngologists used Botox® as the type of botulinum toxin A, with a median dosage of 1.5 U (IQR 0.125) for LD patients.
The majority (82.6% or 19/23) of the survey respondents answered the last 2 questions of the survey. About half (47.4% or 9/19) of them reported being uncomfortable with injecting BTX in pregnant LD patients (Figure 1). About one-fifth (21.1% or 4/19) mentioned being comfortable, but none were very comfortable with this treatment in pregnant patients.

Canadian laryngologist’s level of comfort in injecting botulinum toxin in pregnant laryngeal dystonia patients.
The respondents were also questioned about their preferred management of BTX injections in pregnant LD patients (Figure 2). The majority (68.4% or 13/19) of the respondents would discuss the risks/benefits/alternatives and offer the BTX injections after obtaining informed consent. About one-quarter (26.3% or 5/19) would advise the patient to wait until after her pregnancy to resume BTX injections.

Preferred management options by Canadian laryngologists for botulinum toxin injections in pregnant laryngeal dystonia patients.
Discussion
BTX is a neurotoxin produced by an anerobic, gram-positive bacterium, Clostridium botulinum. It causes a paralytic effect by blocking the release of acetylcholine at the presynaptic motor nerve terminal. 35 BTX exerts its effect by causing a selective chemodenervation. Within laryngology, BTX can be used to manage vocal tremor, chronic cough, vocal process granuloma, cricopharyngeal muscle dysfunction, muscle tension dysphonia, paradoxical vocal fold dysfunction, palatal myoclonus, and sialorrhea. The safety of BTX injections for LD has been established in several large studies.5,36,37 Possible side effects of BTX injections for LD include throat pain, dysphagia, aspiration, breathy voice, allergic reaction, and stridor for abductor patients.5,36,37 Contraindications to BTX treatment include neuromuscular disorders (eg, myasthenia gravis), concomitant aminoglycoside use, allergies to constituents of the botulinum toxin product, pregnancy, and breastfeeding. 38 This paper challenges the traditional teaching that pregnancy and breastfeeding are contraindications to BTX treatment for LD.
A feared complication of BTX is botulism, a neuroparalytic illness characterized by symmetric, descending flaccid paralysis of motor and autonomic nerves. 35 It is a rare but potentially fatal illness as it can paralyze the respiratory muscles and cause severe respiratory distress. Botulism can be classified as foodborne, wound, iatrogenic, and infant. Iatrogenic botulism is rare and caused by systemic spread of a BTX injection. Infant botulism occurs when the bacteria’s spores grow in an infant’s intestines and produce the toxin, which subsequently causes the illness. None of the babies or breast-fed infants in this study had botulism, respiratory, or neurological complications. The median APGAR score was 9, and none required NICU care or intubation. In 6 of the 8 pregnancies, patients had BTX treatment during the first trimester of pregnancy. This is relevant as most fetal loss is experienced in the first trimester. 39
In LD treatment, BTX is injected intramuscularly in very small doses (eg, 1.25 U). In contrast, the doses for other indications are much higher, for example, 20 to 85 U for cosmetic purposes in the glabellar and 100 to 300 U for cervical dystonia. 40 There should be little systemic distribution of BTX from intramuscular or intradermal injection, and BTX is not measurable in peripheral blood when injected at recommended dosages.1,2,9,11 One study reported that BTX can diffuse from distant sites such as facial musculature to the arm as detected by single-fiber EMG, especially at high doses.41,42 However, the effects appeared to be subclinical in this study.41,42 If AEs are dose dependent,41,42 there should be a very low risk of expected AEs with laryngeal BTX injections. Hildebrand et al measured the concentration of BTX in different body fluids of pregnant rabbits that were injected intravenously with a lethal dose of BTX. 43 There was no detectable level of BTX in the placenta or fetal blood. Based on this study’s results and the known molecular size limitations for placental transfer, BTX is believed to be too large to cross the human placenta barrier.11,43
Many studies have been performed to establish the safety of BTX in both therapeutics and cosmetic treatments.44 -55 In 2009, the FDA required black box warnings on labeling of BTX products about the possible risk of botulism-like symptoms associated with systemic spread of the toxin. These botulism-like symptoms included muscle weakness, difficulty speaking, loss of bladder control, difficulty breathing or swallowing, blurry or double vision, and drooping eyelids. There were reports of children with cerebral palsy developing these symptoms after receiving off-label and high-dose injections to treat muscle spasticity. 44 Several retrospective studies and systematic reviews in neurology have established the short- and long-term safety of BTX for movement disorders.45 -47 For example, a retrospective review of 235 patients who received BTX for movement disorders was conducted over 10 years. AEs were mostly minor and occurred in 27% of patients, occurring over 4.5% of treatment cycles. The AEs were intolerable in only 1.3% of patients who discontinued therapy. 45 Many studies in dermatology, facial plastic surgery, and oral maxillofacial surgery have established the long-term safety of BTX for cosmetic indications.48 -55 A publication on the AEs reported to the FDA over 13.5 years for all indications of BTX revealed 1437 incidents; 406 followed therapeutic use, and 1031 followed cosmetic use. AEs predominantly occurred in women, and serious AEs were 33-fold more common for therapeutic than cosmetic indications. This difference was attributed to higher BTX doses used for therapeutic indications and sicker patients with more medical comorbidities. 49
Focusing on pregnant patients, we continue to have minimal data to provide patient counseling with regard to safety. Table 3 shows a summary of the literature on BTX injections administered intramuscularly during pregnancy. Allergan published an open-access 29-year retrospective study of their global safety database. 9 Overall, the prevalence of fetal defects was comparable in both the BTX-treated and general populations. Indications for BTX were heterogeneous, and there was variability in treatment sites and BTX doses used. Cardon et al published a case series involving 3 LD patients who received BTX treatment while pregnant with no AEs noted. 56 This current study had a larger sample size of 4 LD patients and 8 pregnancies. There were several publications on cervical dystonia, which has a female predisposition and is exacerbated during pregnancy. 9 , 12 - 15 , 21 One noteworthy case reported a patient who received BTX during all 4 of her singleton pregnancies. She had a total cumulative dose of 1200 U during her entire pregnancy, which was the highest dose in Table 3. 12 There were no AEs observed in mother or fetus for this patient. 12 Further case reports for cosmetics, 16 achalasia,17-18 strabismus, 19 and migraines20,57 are listed in Table 3.
Literature Review of Botulinum Toxin A Injections Administered Intramuscularly During Pregnancy
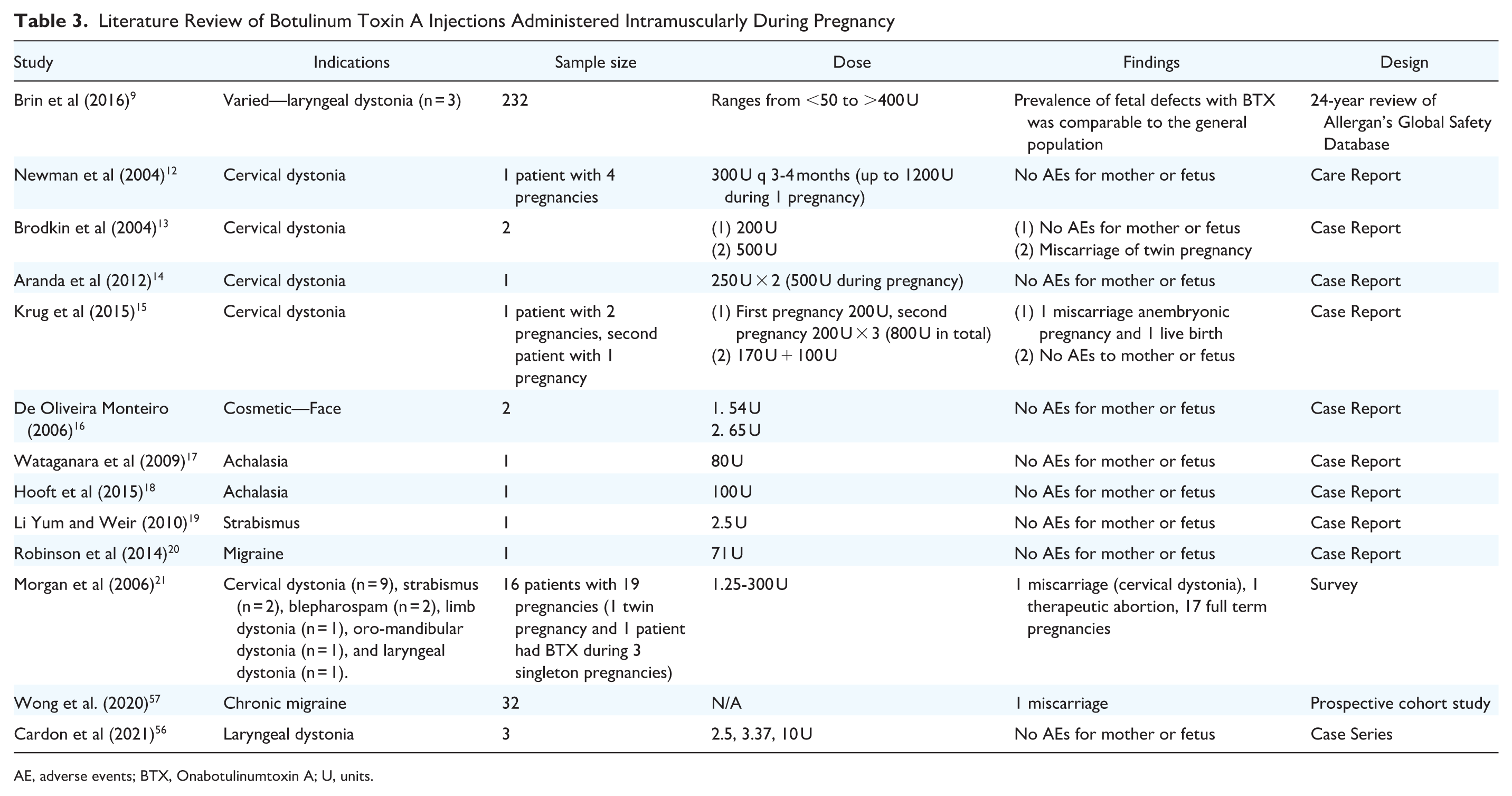
AE, adverse events; BTX, Onabotulinumtoxin A; U, units.
All 4 patients included in this retrospective chart review underwent the informed consent process, and the risks, benefits, and alternatives of intramuscular BTX injection into the larynx during pregnancy were discussed. All 4 of these patients had a good therapeutic response to BTX injection prior to pregnancy, and all had established care with the laryngologist for years. Two of these patients were healthcare professionals—one was a family physician, and one was a labor and delivery room nurse. Consultations with the patients’ obstetrical team were performed in all cases. All 4 patients made the informed decision to proceed with the BTX injection during all 8 pregnancies. They chose to breastfeed all 8 of their babies and receive BTX treatments; there were no AEs observed in the mothers or babies.
Morgan et al surveyed 396 physicians (mostly neurologists and physiatrists) who use commercially available BTX. 21 The survey responses showed that only 12 of the 396 physicians had knowingly or unknowingly injected pregnant women with BTX. Of the 12 physicians who injected pregnant patients, only one physician reported being “very comfortable” using BTX during pregnancy. A total of 16 pregnant women received BTX injections, only one of which had SD. In the total of 19 pregnancies that were examined, 1 pregnancy was medically terminated and 1 woman miscarried. This latter had received BTX injections for cervical dystonia. The remaining 17 pregnancies went to full term without complication.
The response rate to this survey was 85.2%, which was above the accepted average of online surveys (34%) 58 and superior to the “guaranteed” 80% return rate with the Dillman’s Total Design method. 34 This allows an excellent external validity of the survey results. This study showed that only a few Canadian laryngologists had knowingly or unknowingly injected BTX in pregnant patients for either LD or palatal myoclonus, for a total of 5 women. This small number can be explained by the low prevalence of LD [50,000 people in North America according to Dysphonia International (formerly the National Spasmodic Dysphonia Association)], 59 but also by the variable comfort level of physicians in injecting BTX in pregnant patients. In fact, the survey results reported that only 21.1% of Canadian laryngologists were “comfortable” with injecting BTX in pregnant SD patients, and none were “very comfortable.” These results differed from those obtained by Morgan et al where 41.7% of the respondents were “somewhat comfortable” and 8.3% were “very comfortable” with the procedure. As per our survey’s preferred management option for pregnant LD patients, respondents mainly chose to discuss the risks/benefices/alternatives with the patient and offer the treatment with informed consent.
This study should be interpreted in the context of its strengths and limitations. With 4 patients and 8 pregnancies in the retrospective chart review, the sample size was small. Patients were recruited from one academic center, which may limit the external validity of this part of the study. Because of the retrospective nature of the study, with no randomization or an established control group, the data collected were subject to recall bias. The survey was also limited by recall bias from the respondents. Furthermore, although obstetrical records were obtained, access to the children’s health records was not available. We relied on self-reported data from the mothers. The survey that was distributed to the laryngologists was not validated. Our thorough literature review did not uncover any validated questionnaire on this topic; instead, we adapted a previous survey from the literature. 21 In this survey, there was also a risk of nonresponse bias. Physicians with good results may have been more likely to complete the survey; those with AEs may have preferred not to disclose those results.
We cannot claim that these results are causal or definitive proof that BTX injections are safe in pregnancy for LD patients. Multicenter research with larger and more diverse patient populations needs to be done to explore BTX treatment in pregnant LD patients to facilitate safe and effective care. We hope that this case series, survey, and literature review on the current practice of Canadian laryngologists will help guide the informed consent process with future pregnant LD patients.
Conclusion
This case series was the largest in the literature of pregnant LD patients undergoing BTX treatment. This study was the first survey distributed to laryngologists to evaluate the current practice in treating pregnant LD patients in Canada. This report adds to the growing literature on the safety of BTX during pregnancy. BTX was designated as an FDA pregnancy class C drug, meaning there are no adequate and well-controlled studies in humans, but potential benefits may warrant use of the drug in pregnant women despite potential risks. In reviewing the clinical literature and pharmacology of intramuscular BTX injections, there were very low risks of systemic distribution, and BTX was unlikely to cross the human placental barrier to the fetus. Few adverse events were reported in the literature. Close monitoring of the fetus and collaborative care with the patient’s obstetrician were encouraged. This study was not causational or definitive; however, it provided more data for clinicians to reference when discussing the implications of BTX treatment for pregnant LD patients. It may give the opportunity to certain pregnant patients to continue receiving adequate treatment for their LD during their pregnancy and consequently maintain a good quality of life. The results from this study also form a basis for more definitive future research on BTX treatments in pregnant women.
Supplemental Material
sj-docx-1-ohn-10.1177_19160216251392559 – Supplemental material for Botulinum Toxin Treatment in Pregnant Laryngeal Dystonia Patients: Case Series and Survey of Current Practice of Canadian Laryngologists
Supplemental material, sj-docx-1-ohn-10.1177_19160216251392559 for Botulinum Toxin Treatment in Pregnant Laryngeal Dystonia Patients: Case Series and Survey of Current Practice of Canadian Laryngologists by Kaishan Aravinthan, Mélissa Patry and Amanda Hu in Journal of Otolaryngology - Head & Neck Surgery
Footnotes
Acknowledgements
We would like to acknowledge Simran Gill (research coordinator, Division of Otolaryngology Vancouver General Hospital) in assisting with the creation and distribution of the survey.
Author Contributions
KA conducted the retrospective chart review, data collection, analysis, and interpretation. He also did a literature review and was an important contributor in writing the manuscript. AH conceived the survey, designed, and supervised this study. She was a major contributor in writing and editing the manuscript. MP conducted the survey, analyzed, and interpreted the survey data, and was a significant contributor in writing the manuscript.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Informed Consent
This study was approved by the University of British Columbia Research Ethics Board (REB # H20-03577 and #H22-02774). Consent was obtained from each participant before consulting their obstetrical records and prior to the survey initiation.
Supplemental Material
Additional supporting information is available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
