Abstract
Importance
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a highly infectious virus that has been reported to be transmitted via aerosolization and other aerosol-generating activities. However, viral plumes from CO2 lasers have yet to be investigated.
Objective
Evaluate the viability of SARS-CoV-2 in the CO2 laser plume in a porcine model for laryngeal surgery.
Design
Experimental studies.
Setting
Laboratory.
Participants
Wild-type SARS-CoV-2 (Wt), Omicron SARS-CoV-2 (Omicron), and fluorescent SARS-CoV-2 (GFP).
Intervention
Three viral strains on porcine larynx exposed to CO2 laser setting—1.5, 7, and 15 W.
Main Outcome Measures
The laser plume was collected during ablation. Dilutant of the plume was then applied to Vero E6 cells for culture; quantitative real-time reverse transcription polymerase chain reaction (qRT-PCR) (in duplicate) and fluoroscopy were used to establish the presence of viable virus.
Results
Wt, Omicron, and GFP strains were found to be present on the larynx before and after the application of the laser by qPCR copies (cp) per microliter (µl). After ablation with 1.5, 7.0, and 15 W with Wt, the plume samples were shown to be 0, 0, and <10 cp/µl, respectively. For GFP, at all intensity levels, the copies were less than 10 cp/µl. Omicron did not show any copies at any of the power levels. Cultures did not show the presence of viable Wt, Omicron, and GFP strains in any of the collected laser plumes.
Conclusion
The CO2 laser plume of SARS-CoV-2 aerosolized minimal viral particles. None of the tested experimental conditions demonstrated a viable virus after laryngeal ablation with the CO2 laser.
Relevance
CO2 laser use in the context of virus SARS-CoV-2 suggests minimal transmission risks associated with CO2 laser procedures when appropriate safety protocols are followed.

Key Message
- CO2 laser aerosolized minimal SARS-CoV-2 virus particles.
- Aerosolized virus was not viable to infect cell lines.
Introduction
In the spring of 2020, the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) outbreak was declared a global pandemic by the WHO. In the field of otolaryngology, it was particularly concerning due to the anatomic reservoir of the virus in the upper respiratory tract.1,2 During the global pandemic, concerns of intubation of patients with that were infected with SARS-CoV-2 would cause aerosolization of the virus and the term “aerosol generating medical procedures” was coined. 3 Precautions and recommendations have been devised to protect healthcare staff while treating patients with SARS-CoV-2 in the critical care 4 and otolaryngology setting.5,6 Increased risks of performing surgery on patients with SARS-CoV-2 have been examined. Previous reports in the medical literature have shown that surgery in an infected field can aerosolize the virus and cause transmission to surgical team members if proper precautions are not taken.1,7 These reports described aerosolization potentially via electrocauterization in an infected surgical field.8,9 Other studies have shown previously that SARS-CoV-2 was not detectable in aerosol electrocautery plumes. 10 However, viral plumes from other heat sources, like CO2 lasers, have yet to be elucidated.
Use of CO2 lasers in otolaryngology was first described in 1971.11,12 Since then, its use has been expanded to excision of oral mucosal and laryngeal lesions, otologic surgery, and facial plastic surgery. 13 It is a particularly useful tool in the subspecialty of laryngology for precise excision of lesions with reduced thermal injury. One specific application where a laser has proven quite useful is in the treatment of Recurrent Respiratory Papillomatosis. This condition is caused by human papilloma virus (HPV), a small non-enveloped DNA virus, and previous reports have shown that HPV can be aerosolized via laser. 14 This places surgical team members at risk for exposure and possible infection, and safety steps for protection during laser use have been implemented. 15 While HPV aerosolization from a CO2 laser has been documented previously, to our knowledge, no reports have been published for aerosolization of SARS-CoV-2 using a CO2 laser. Our group set out to examine the potential viability of aerosolized SARS-CoV-2 using a CO2 laser in an ex vivo porcine model. The primary outcome was to determine the SARS-CoV-2 gene copy number in the surgical plume from a CO2 laser. The secondary outcome was to evaluate the viability of the aerosolized virus using the tissue culture infectious dose (TCID) assay.
Materials and Methods
All experiments were performed in a Public Health Agency of Canada (PHAC) licensed containment level 3 (CL3) lab after institutional Ethics and Biohazard committee approval. Porcine larynges were obtained from a local meat processor in our area. The porcine larynges were divided midline to establish a hemi-larynx.
Wild-type SARS-CoV-2 (Wt), Omicron SARS-CoV-2 (Omicron), and fluorescent SARS-CoV-2 (GFP) were used during the experiments. All 3 strains were mixed with Dulbecco’s Modified Eagle Medium (DMEM) in a 1:1 ratio. The resulting viral suspensions were applied to the laryngeal surface of the porcine hemi-larynges with a total of 5 ml applied to the false cords, ventricle, true cords and sub-glottis, followed immediately by laryngeal ablation. Each virus strain was tested in triplicate for each of the CO2 laser settings of 1.5, 7, and 15 W. The laryngeal surface was ablated for 1 minute after application for each strain in an enclosed workspace within a fume hood. Laser plume was collected using Sartorius MD8 AirScan onto gelatin filters and dissolved using DMEM. The experiments were performed in a closed container inside a fume hood. The distance from the Sartorius MD8 AirScan filter and the lasered specimen was approximately 20 cm with AirScan airflow set to 80 l/min. Positive controls were collected from the laryngeal surface after ablation and pipetted onto gelatin filters with DMEM. Another positive control, named “Hood Control,” was collected from the fume hood ambient air with a gelatin filter placed on the working surface of the fume hood adjacent to the experimental set-up. Negative control was a gelatin filter alone. The dilutant was then applied to Vero E6 cells for culture. After 1 week of growth, media was then transferred to 1.5 ml microcentrifuge tubes and immediately heat inactivated at 56°C for 30 minutes before transfer to lower containment for viral RNA isolation. RNA extraction was performed from 140 ml of each undiluted sample using a QIAamp® Viral RNA Mini Kit (Qiagen, Toronto, Canada) according to the manufacturer’s instructions. Quantitative RT-qPCR was performed in technical duplicate on a QuantStudio™ 5 Real-Time PCR System (ThermoFisher, Mississauga, Canada) using TaqMan® Fast Virus One Step RT-PCR Master Mix (ThermoFisher) with primer/probe sets against SARS-CoV-2 nucleocapsid gene (N1 and N2) from Applied Biosystems (ThermoFisher). A viral gene copy number and the virulence of a virus can vary from species to species, that is, a more virulent virus can infect cell lines with fewer gene copies. Therefore, a viral gene copy number per sample was calculated using standard curves of known SARS-CoV-2 copy numbers. The tissue culture infectious dose 50/ml (TCID50/ml) was calculated based on the percentage of positive vs. negative wells at each dilution using the Spearman and Karber algorithm. Fluoroscopy with an EVOS M7000 (Thermofisher) microscope was used to image and quantify viable SARS-CoV-2.
Results
To validate the experimental model, the presence of Wt mRNA was measured before and after ablation of the larynx. Wt was measured from the test liquid prior to application to the larynx. The quantity of mRNA was 88,859.48 cp/μl (Figure 1). Then, after ablation of the larynx with 15 W, RT-qPCR was run on the supernatant from the porcine larynx and demonstrated 8669.34 cp/μl of mRNA. The “Hood Control” interestingly produced 32,119.4 cp/μl of mRNA. Our negative control or “Media Only” had 0 cp/μl. Furthermore, we wanted to measure the infectious viral titer of Wt. We calculated the TCI50 (50% TCID) from our qPCR data. We used N1 as the primary measurement, and the N2 target gene was used as the internal control. TCI50 for the Wt after laser treatment for N1 and N2 was 12,022.64 cp/μl for both (Figure 2). Our positive control was 10 cp/μl, and our negative control was 0 cp/μl.
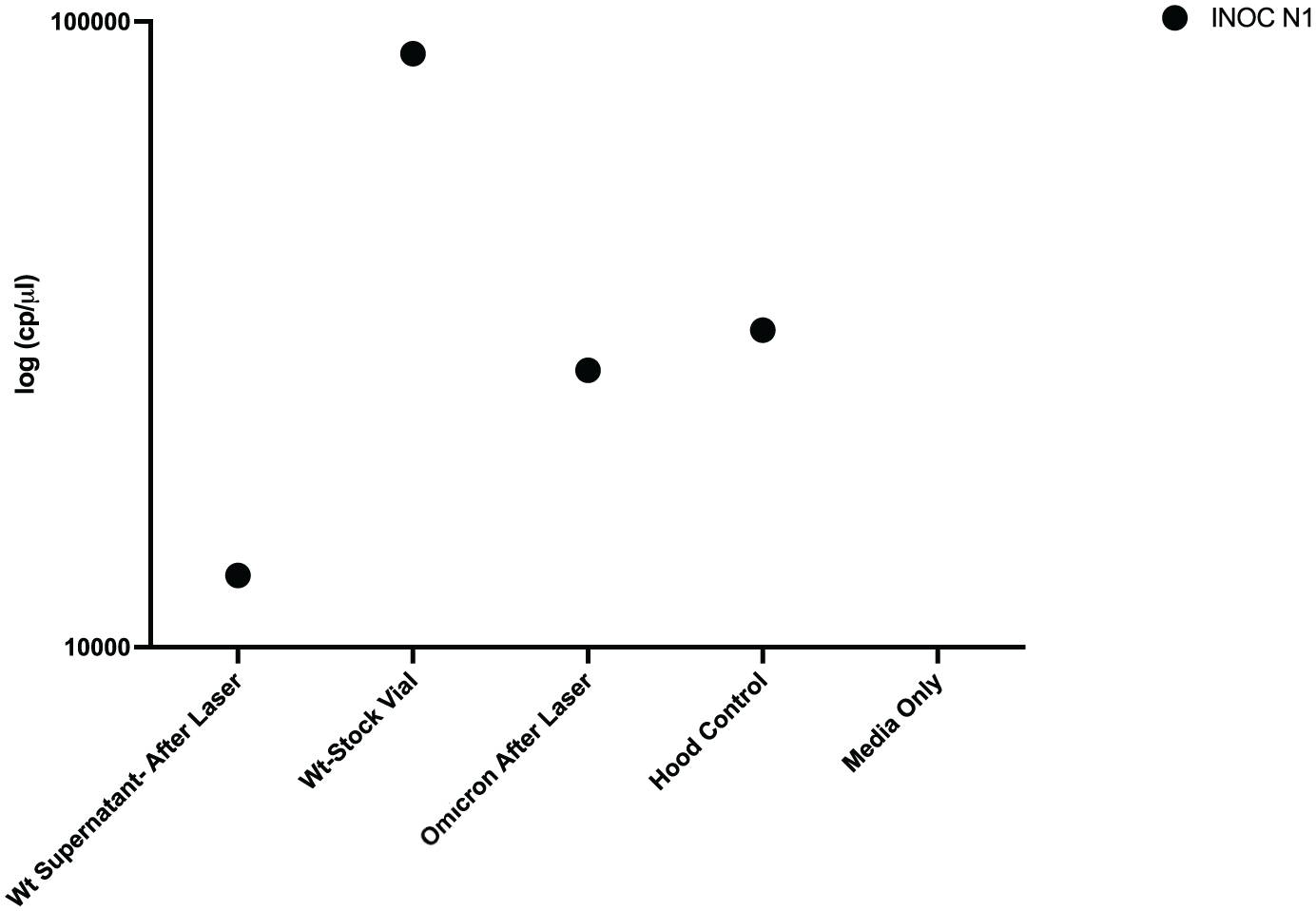
Validation of wild-type SARS-CoV-2 (Wt) aerosolization of viral mRNA in experimental model.

Validation of wild-type SARS-CoV-2 (Wt) infectivity with TCID50 calculation. TCID50, tissue culture infectious dose 50/ml.
Cauterization with CO2 laser of Wt, Omicron and GFP strains yielded aerosolized copies of viral mRNA but were not virulent.
Viral mRNA of aerosolized Wt strain at 1.5, 7.0, and 15 W produced 4501.94, 2584.38, and 5498.41 cp/μl, respectively (Figure 3). The aerosolized data were then used to calculate the TCI50 for N1 and N2, and all 3 power settings yielded 0 cp/μl at the N1 and N2 mRNA sequence (Figure 4). Viral mRNA of aerosolized Omicron strain at 1.5, 7.0, and 15 W produced 422.33, 960.55, and 61.01 cp/μl, respectively (Figure 3). For all 3 settings, power settings yielded 0 cp/ml at the N1 and N2 mRNA sequence (Figure 4). Finally, viral mRNA of aerosolized GFP strain at 1.5, 7.0, and 15 W produced 23,650.08, 4520.1, and 2200.98 cp/ml, respectively (Figure 3). For GFP 1.5 W, N1 and N2 TCID50 is 0.4 and 0.63, respectively. For GFP 7W, N1 and N2 TCID50 were 0.78 and 1.41, respectively. Finally, GFP 15 W for N1 and N2 TCID50 were 2.95 and 3.39, respectively (Figure 4).

Aerosolization of wild-type SARS-CoV-2 (Wt), Omicron SARS-CoV-2, and fluorescent SARS-CoV-2 (GFP) mRNA at CO2 laser settings of 1.5, 7.0, and 15 W.
Wild-type SARS-CoV-2 (Wt), Omicron SARS-CoV-2, and fluorescent SARS-CoV-2 (GFP) estimated infectivity (TCI50).
The GFP strain did not infect Vero E6 cells, as measured by fluoroscopy. Aerosolized GFP dilutant was applied to Vero E6 cells after cauterization using power levels 1.5, 7, and 15 W. In Figure 5, Brightfield and 488 nm demonstrated no active infection from the GFP strain at all 3 laser power levels. Positive control of application of the dilutant from the larynx surface prior to laser cauterization demonstrated viable virus growth.

Fluorescent SARS-CoV-2 (GFP) did not infect Vero E6 cells after cauterization treatment.
Discussion
When the SARS-CoV-2 pandemic was developing in the spring of 2020, there were many reports regarding the viability and infectability of the virus. Van Doremalen et al reported that an aerosolized virus can remain viable up to 72 hours on different surfaces. 16 With previous reports 8 of SARS-CoV-2 infection of operating room (OR) staff from exposures to SARS-CoV-2 positive patients, a better understanding of possible occupational exposure risks to surgeons and other operating room personnel was required. Our results demonstrate that SARS-CoV-2 mRNA can be aerosolized by CO2 laser use; however, this viral material did not generate viable, quantifiable virus in cell cultures.
SARS-CoV-2 is an enveloped RNA virus that has been shown to be heat sensitive in other publications.7,17,18 Chin et al found that SARS-CoV-2 can be inactivated by heat as low as 70°C. Our group has previously demonstrated that electrocautery can damage SARS-CoV-2 and inactivate the virus. 10 Electrocautery tip temperatures ranges from 100°C to 1200°C, well beyond the 70oC threshold. CO2 laser has lower thermal temperatures than electrocautery; it can also dissipate the heat transfer to surrounding tissues using a pulsed function. 19 Reports of other enveloped viruses, such as Human immunodeficiency virus (HIV), have shown that they can be aerosolized and remain viable to infect cell lines. 20 This study also demonstrated, however, that HIV did not maintain its viability beyond 28 days post-infection, which led the authors to believe that the CO2 laser still had the ability to damage the virus to a point where it became nonviable. Johnson et al found that use of electrocautery produced nonviable HIV. 21 Interestingly, in this same study, it was noted that when using a bone saw, aerosolized HIV maintained its viability and infectability. Previous studies have shown a variable amount of virus viability and infectability after the use of the CO2 laser. In our study, we were still able to detect SARS-CoV-2 mRNA in aerosolized samples. Interestingly, the quantity of viral gene copy did not show a linear decrease in the total quantity, which may have to do with obliteration of viral elements. Comparatively, Hepatitis B Virus (HBV) has similar features. Kwak et al 22 found that with HBV-positive patients, with laparoscopic electrocautery, smoke plumes were PCR positive for HBV DNA. However, the group did not test the viability of the virus. On the other hand, HPV DNA has been noted to be aerosolized after use of CO2 laser. Sawchuk et al found that use of the CO2 laser was able to aerosolize HPV and Bovine papilloma virus (BPV) DNA on plantar warts. 14 Another study demonstrated that 2 out of 3 cases of large genital condyloma that were ablated with a CO2 laser had HPV DNA detected in the smoke plumes. The reports of the HPV virus being viable after being aerosolized are unfortunately not conclusive.23,24 Kunachak et al cauterized juvenile papilloma specimens with CO2 laser and yielded negative growth in cell lines. 23 On the other hand, Lobraico et al did a retrospective survey study of various specialties that used CO2 laser for ablation of HPV associated lesions. The survey polled 824 respondents across multiple specialties and found that the overall incidence of HPV associated lesions was 3.2%. The highest incidence of acquired lesions was dermatologists with 15.2%. Dermatologists were responsible for 65.4% of positive results from the survey. However, the authors concluded it was impossible to determine whether the causation of the HPV associated lesions was due to the CO2 laser or direct contact with the lesion itself.
SARS-CoV-2 is a highly infectious virus that is transmitted via aerosolization and other aerosol-generating activities, such as intubation and jet ventilation, in the OR and thus may place OR staff at risk for infection. Use of the CO2 laser is an aerosol-generating activity in the OR, and its use increases risk of aerosolizing SARS-CoV-2. In one study, CO2 laser has been shown to aerosolize the virus in the OR and it was shown that jet ventilation was not sufficient in aerosolizing the virus. 25 Also, Sanmark et al showed that use of a microdebrider and cold steel produces less aerosolization of SARS-CoV-2 compared to CO2 laser. 26 Interestingly, the CO2 laser created more aerosolization of SARS-CoV-2 than a patient coughing. In light of this, multiple recommendations have been made for protection of OR staff, including the use of personnel protective equipment such as properly fitting N-95 and negative pressure ORs in SARS-CoV-2 positive cases.27,28 Other protective adjuncts,29,30 such as a protective curtain or “Aerosol box,” have been suggested, but the overall utility of these is still debatable. With other viruses, previous studies have found that a surgical mask was able to filter out the DNA vapors. The viability of the HPV virus after cautery or CO2 laser remains unclear; as such, it is recommended that PPE and exhaust ventilation be used.15,31 When dealing with any type of surgical intervention, the surgical team must not only understand the transmission properties of an associated virus as related to the pathology, but also the type of precautions needed to protect OR staff.
While our study is novel in the viability of SARS-CoV-2 in CO2 laser plumes, our study is not without limitations. The power of the CO2 laser was set for 1.5, 7, and 15 W to try to account for all possible powers of laser use in the aerodigestive tract, which is not the lower powers generally used in laryngeal laser surgery. These higher powers are, however, reflective of power levels used elsewhere in the body and provide evidence for the lack of viable virus aerosolization at these levels. The current laboratory set-up is limited to the use of a CO2 laser and does not include other laser sources. KTP laser or blue light laser has been used in treating HPV lesions and examined for aerosolization.32,33 While difficult to say that our study would be translatable to KTP lasers, a future study would be to examine different laser types to see aerosolization and viability of SARS-CoV-2 or other coronaviruses. Furthermore, we are planning to run experiments in settings such as an office and a full OR set-up and will have the ability to create very realistic scenarios to test other lasers and other hypotheses.
Conclusion
This study contributes valuable insights into the safety considerations surrounding CO2 laser procedures in laryngology, particularly in the context of viral transmission risks of SARS-CoV-2. While our findings indicate the presence of viral RNA in laser plumes, the lack of virus viability suggests minimal transmission risks associated with CO2 laser procedures when appropriate safety protocols are followed.
Footnotes
Authors’ Note
Presented at the Canadian Society of Otolaryngology Annual Meeting, Montreal, Quebec, June 3rd, 2024. This material has never been published and is not currently under evaluation in any other peer-reviewed publication.
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available within the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CIHR COVID-19 May 2020 Rapid Research Funding Opportunity grant number 0624001749.
Ethical Approval and Informed Consent
The authors confirm that the Institutional Ethics and Biohazard committee approved this study.
