Abstract
Importance
There is a need to culturally adapt this commonly used patient-reported outcome measure, the Reflux Symptom Index (RSI), for use in Canada and Quebec’s French population.
Objective
To translate and validate the RSI for use in Quebec French speakers, ensuring reliability and validity within this population.
Design
Prospective case-control study.
Setting
Voice and Dysphagia Laboratory at the McGill University Health Centre in Montreal, Quebec, Canada.
Participants
Patients with laryngopharyngeal reflux (LPR) and healthy controls were recruited during outpatient clinical laryngology visits. Participants were eligible if they were at least 18 years old and spoke French as their native language.
Intervention or Exposures
A Quebec-French version of the RSI (Q-Fr-RSI) was validated using the “translation-back-translation” method. Participants completed the Q-Fr-RSI, Fr-VHI, and their laryngoscopy was examined and assigned a Reflux Finding Score at their initial visit. Participants repeated the Q-Fr-RSI via telephone follow-up.
Main Outcome Measures
Feasibility, internal consistency, test-retest reliability, and construct validity of the Q-Fr-RSI were assessed.
Results
Eighty-nine participants were recruited: 49 patients within the experimental group and 40 subjects within the control group. The feasibility was evaluated as percentage of missing answers in the full sample at the first time point, 0%, and second time point, 1.2%. Internal consistency via Cronbach’s alpha was high (.90). Intra-rater reliability via intraclass correlation demonstrated good level of agreement (.84). Internal validity demonstrated a strongly significant difference between the groups’ answers (mean score of 23.9 vs 6.65, respectively, P < .001).
Conclusions
This data suggest that the Q-Fr-RSI can be used to assess LPR in the French-speaking Canadian population.
Relevance
In the bilingual country of Canada, and especially in the province of Quebec where over 90% of people speak French, an appropriate validation of the Q-Fr-RSI allows all patients to be monitored adequately. A Quebec-French patient-reported outcome measure can accurately represent these patients in future research studies utilizing these metrics.

Key Message
- Quebec French and European French have linguistic and cultural distinctions and these differences lead to a need to validate patient-reported outcome measures specifically into Quebec-French.
- Laryngopharyngeal reflux is a common condition seen and diagnosed by otolaryngologists—head and neck surgeons and the Reflux Symptom Index is used to follow patient symptom burden.
- The Quebec French Reflux Symptom Index (Q-Fr-RSI) is a reliable and valid patient-reported outcome measure for Quebec French-speaking patients with laryngopharyngeal reflux.
Background
Laryngopharyngeal reflux (LPR) is a common condition seen and diagnosed by otolaryngologists—head and neck surgeons. 1 LPR, also known as “silent reflux” or “reflux laryngitis,” may be present in 50% of patients who present with dysphonia, throat clearing, globus, postnasal drip, or dysphagia. 2 LPR symptoms often improve after initiation of antireflux therapy in the majority of patients. LPR, however, is not equivalent to gastroesophageal reflux disease (GERD) as the patterns, mechanisms, and treatment plans are distinct. 3 Clinically, the symptoms of LPR and GERD differ in that LPR is often devoid of heartburn, regurgitation, and other symptoms present on currently available validated GERD questionnaires.4-6
In 2002, the Reflux Symptom Index (RSI) was developed by Belafsky et al as a patient-reported outcome measure to document symptom severity in LPR patients. 2 The RSI continues to be useful today due to low diagnostic yields, high costs, and invasiveness of the currently available objective tests.7-10 Based on these limitations, otolaryngologists—head and neck surgeons rely on clinical signs and symptoms to make a diagnosis of LPR. The RSI is a metric that has been proven to assist in the assessment and follow-up of LPR given its high sensitivity. 11
French is the second official language of Canada and over 90% of people in Quebec speak French. 12 The RSI has previously been validated in Arabic, Chinese, Hindi, Italian, and European French, and other languages.10,11,13-19 However, differences between the European and Quebec French languages’ vocabulary, grammar, and syntax differences are noted clinically by patients and formally by linguists, other research groups, and Quebec’s French Language Office. Therefore, the Quebec French version properly addresses the linguistic and cultural distinctions specific to the French Canadian population.20,21 This study has been conducted in order to effectively utilize this instrument in our bilingual clinics in Quebec and Canada as a whole.
Methods
The objective of this cross-sectional study was to translate and validate the RSI for use as a diagnostic and follow-up instrument in Quebec French speakers, ensuring its reliability and validity within this population. This prospective study was conducted in the McGill University Health Centre Voice and Dysphagia Laboratory. Dr Belafsky, the developer of the RSI, has approved this validation study into Quebec French.
Questionnaire Development
The RSI is a self-administered, psychometrically validated and reliable LPR symptom assessment questionnaire developed by Belafsky et al. 2 The RSI had been proven to be easily administered, reproducible, and exhibit excellent construct-based and criterion-based validity. This survey is composed of 9 statements with a Likert-response scale ranging from 0 (no problem) to 5 (severe problem), with an abnormal score if greater than 13 out of a maximum 45 points.
A protocol was followed to develop an ideal Quebec French translation of the RSI (Q-Fr-RSI). The original English version was first translated into French by an independent interpretation service and translated back into English by separate independent interpreters to ensure consistency. A bilingual multidisciplinary team composed of 4 otolaryngologists, 2 speech language pathologists, and 2 otolaryngology nurses then reviewed the Quebec French adaptation independently. Next, comments or feedback were discussed as a group until a consensus was reached and the Q-Fr-RSI was finalized. The final questionnaire is presented in Figure 1.

Indice des symptômes de reflux (Q-Fr-RSI).
Patient Recruitment
Between March 2023 and July 2023, participants were recruited from the Voice Laboratory clinics of the Royal Victoria Hospital in Montreal, Canada. All subjects were invited to participate after signing an informed consent explaining the objectives of our work. A minimum of 36 patients with LPR and 36 controls were recruited during outpatient clinical laryngology visits, as determined by a power analysis prior to recruitment. Participants were eligible if they were at least 18 years old and spoke French as their native language. Symptoms of LPR were objectified by means of a laryngoscopy by their otolaryngologist—head and neck surgeon during the clinic visit. During the laryngoscopy, physical examination signs of reflux were noted using the Reflux Finding Score (RFS). 22 The RFS is a validated metric to document the physical findings and severity of LPR with excellent inter- and intra-observer reproducibility. Patients with a RFS > 7 and a RSI > 13 were within the experimental group while participants with a RFS of 7 or lower and an RSI of 13 or lower comprised the control group.
Data Collection
Clinical data were collected on 2 occasions. For their first assessment while in clinic, participants completed the Quebec French RSI as well as the previously validated Voice Handicap Index (VHI-10).20,23 Participants were called 2 weeks after their clinic visit to complete the RSI survey for a second time. All phone calls were completed by 1 author (J.P.).
Statistical Analysis
Feasibility, internal consistency, test-retest reliability, and construct validity were assessed. Feasibility was evaluated according to the percentage of missing answers. The sample size required to obtain a minimum internal consistency of 80% was calculated using the Cronbach’s alpha method. The test-retest reliability involved a comparison of the scores of the 2 assessments, for which the intraclass correlation coefficient was calculated. The statistical analysis was performed using SAS Studio Release 3.8 (Basic Edition). Cary, NC: SAS Institute Inc.
Results
The Q-Fr-RSI is presented in Figure 1.
Demographics
The study population consisted of 90 participants recruited from the Voice and Dysphagia Laboratory (Table 1): 49 cases were included, 23 patients (46.0%) were males. The mean age (SD) of the reflux cases in the cohort was 61.9 ± 16.4 years. The control group included 40 healthy volunteers, 21 of which (52.5%) were males. The control group consisted of a population with a mean age (SD) of 59.4 ± 21.1 years. None of the control subjects presented with reflux-related concerns. At the time of their laryngology evaluation, the primary complaints of the control group included dysphagia (n = 22), dysphonia (n = 7), nonlaryngology-related concerns (n = 6), vocal fold lesions (n = 4), and airway-related conditions (n = 1).
Demographic Information of the Patient Sample.

Feasibility
This survey was feasible as all surveys were completed at the first time point (100%) and 79/80 (98.8%) surveys were completed at the second time point.
Reliability
The internal consistency (Cronbach’s alpha) for the Q-Fr-RSI total score was high (.90) (Table 2). Test-retest reliability via the intraclass correlation demonstrated good level of agreement (.84).
Cronbach’s Alpha of Q-Fr-RSI Total Score.

Abbreviation: Q-Fr-RSI, Quebec-French Reflux Symptom Index.
Validity
Internal validity was demonstrated by comparing responses of the experimental group patients versus those in the control group, yielding a strongly significant difference between the groups’ answers (mean score of 23.9 vs 6.65, respectively, P < .001; Table 3). External validity was demonstrated by assessing the correlation between VHI-10 and RSI scores (Pearson’s correlation .528, P < .001). Construct validity was demonstrated by assessing the correlation between patients’ RFS score on flexible laryngoscopy and RSI scores (0.748, P < .001; Table 4).
Difference Between Control and Experimental Group on Q-Fr-RSI, RFS, and VHI Scores.
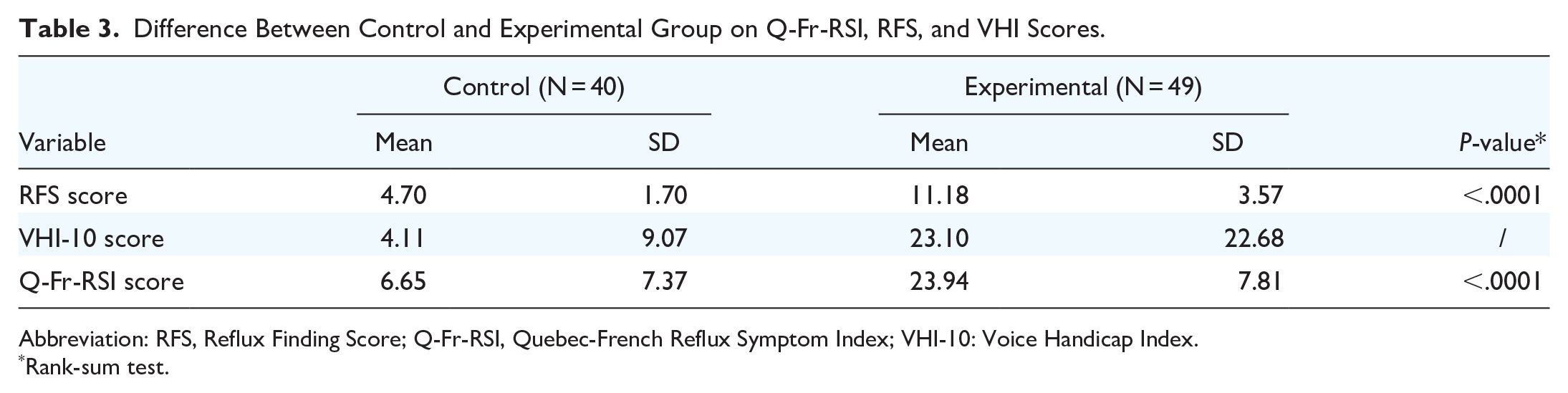
Abbreviation: RFS, Reflux Finding Score; Q-Fr-RSI, Quebec-French Reflux Symptom Index; VHI-10: Voice Handicap Index.
Rank-sum test.
Pearson’s Correlations Between Q-Fr-RSI score with VHI and RFS.

Abbreviation: RFS, Reflux Finding Score; Q-Fr-RSI, Quebec-French Reflux Symptom Index; VHI-10: Voice Handicap Index.
Point biserial correlation.
Discussion
LPR is a highly prevalent and inflammatory condition among patients who consult for dysphonia, chronic cough, and throat clearing. 2 Although ambulatory 24-hour double-probe pH monitoring and/or multichannel impedance testing is considered the gold standard in the diagnosis of LPR, it remains a costly and invasive diagnostic procedure that may not be well tolerated by all patients. 24 In 2002, the RSI was developed by Belafsky et al as a standardized, efficient, and inexpensive method in diagnosing LPR with multiple studies having validated its effectiveness as a diagnostic tool. 2
Since its introduction, the RSI has been translated and validated into more than ten languages.2,11,13-19 To ensure its applicability to their targeted population, these translated RSI questionnaires required not only a linguistic translation but also a cultural adaptation. There are important distinctions between different French-speaking populations’ vocabularies, grammar, syntax, formality, and informality of speech.20,21,25,26 Therefore, a previous European French validation cannot be assumed valid within the Quebec population. 27 The importance of having validated questionnaires in French with the correct syntax, grammar, and vocabulary employed in Canada has been emphasized by another group in Quebec during translation of the VHI-10. 20 This current study assessed feasibility, internal consistency, test-retest reliability, and construct validity of the Q-Fr-RSI in Quebec’s francophone population.
The results demonstrated that the Q-Fr-RSI was feasible, with only 1.2% of participants unable to complete the follow-up questionnaire. The internal consistency analysis yielded high reliability. These results are consistent with other studies, as the Chinese, Greek, and Italian version of the RSI reported a Cronbach’s alpha values ranging from .71 to .99, with the current results within this range at .90.11,14,16
The test-retest reliability results for the Q-Fr-RSI are consistent with the various previously translated versions of the RSI and its value of .84 represents strong reliability. Despite this, some factors may have limited the interclass correlation as the current study had longer follow-up times. This study targeted a follow-up in 14 days, but truly revealed an average follow-up duration of 17.41 days (SD 6.17 days; range 10-48 days) due to failure of patient response. This is longer compared to the follow-up periods observed in similar studies. For instance, the France-French, Italian, Hindi, Spanish, Chinese, and Kannada validation studies mandated participants to complete the questionnaire twice within a 7-day timeframe.13,14,16,19,27,28 The Kazakh validation study contacted participants 10 to 14 days after completing the first questionnaire for retesting. 18
This study exhibits certain limitations to note. The experimental cohort was recruited based on consultations, indicating a propensity for seeking treatment for LPR symptoms. This inclination toward seeking medical attention, coupled with a more extended follow-up interval compared to other studies, may have impacted the test-retest reliability observed in our study. Patients diagnosed and starting LPR treatment following their consultation may exhibit a reduction in their symptoms within 2 weeks which may explain some variability in the test-retest scores. Another limitation to note is that the experimental patients were those with LPR symptoms, RSI scores greater than 13 and RFS scores greater than 7. The experimental patients did not undergo hypopharyngeal-esophageal multichannel intraluminal impedance-pH testing. 29 While it would provide an objective evaluation, it is invasive, expensive, and not easily available to mass numbers of patients in the public healthcare system of Quebec, Canada. Additionally, we did not assess the responsiveness of the Q-Fr-RSI to changes in symptoms over time following treatment. While our primary objective was to validate the instrument through feasibility, reliability, and validity assessments, future research should investigate its sensitivity to symptom improvement over time through longitudinal studies. This would further establish its clinical utility in monitoring treatment outcomes in patients with LPR.
This study features a distinct demographic profile compared to previous studies, notably comprising an older patient cohort with a mean age of 61.9 (±16.4) and 59.4 (±21.1) for the experimental and control groups, respectively. In contrast, other studies reported mean ages ranging from approximately 37 to 51 in the experimental group and 40 to 54 in the control cohort.
Conclusions
The Q-Fr-RSI is a reliable and valid patient-reported outcome measure for Quebec French-speaking patients with LPR.
Footnotes
Author contributions
J.A.S.: Conception and design, acquisition of data, analysis and interpretation of data, drafting and approval of manuscript. A.L.-V.: Conception and design, acquisition of data, drafting and approval of manuscript. G.L.B.: Conception and design, analysis and interpretation of data, drafting and approval of manuscript. J.P.: Acquisition of data, drafting and approval of manuscript. R.C.: Acquisition of data, drafting and approval of manuscript. J.Y.: Acquisition of data, drafting and approval of manuscript. K.M.K.: Conception and design, acquisition of data, drafting and approval of manuscript.
Availability of Data and Materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Karen M. Kost discloses she is a consultant for Pentax Medical.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study was approved by the ethics review board at the McGill University Health Centre (#2023-9343).
