Abstract
Importance
Chronic refractory cough (CRC) has a negative impact on the quality of life of sufferers and is difficult to treat. Although previous studies have documented various treatment methods for CRC, no treatment method has proven fully effective.
Objective
This study investigated the treatment efficacy and adverse effects of neuromodulators (NM), Amitriptyline and Gabapentin, in the treatment of CRC, and the factors that predicted outcomes.
Design
Prospective data review.
Setting
Private otolaryngology clinic.
Participants
This study included 103 successive patients (72 female, 31 male) during the period between 2013 and 2023. Mean age of patients was 56.6 years (standard deviation: 13.5; range: 21 to 83).
Intervention
All patients received treatment using NM, including Amitriptyline and Gabapentin.
Main Outcome Measures
Cough Severity Index (CSI) and the Newcastle Laryngeal Hypersensitivity Questionnaire (NLHQ). The minimal important difference (MID) in CSI was calculated and used as the threshold to determine response to NM treatment.
Results
A MID of 7 or more in CSI was regarded as a response to NM χ(1)2 = 4.813; P = .028. Using this criterion, 55.6% responded to NM therapy. Chemical triggers significantly predicted responses to NM [χ(1)2 = 4.813; P = .028] while thermal, mechanical, and abnormal throat sensation triggers were not significant predictors of response. Pretreatment CSI was a significant predictor of response to NM [χ(1)2 = 5.603; P = .018]. Pretreatment NLHQ, citric acid cough threshold result, and response to superior laryngeal nerve blocks did not predict response to NM. Seventy percent of patients undergoing NM therapy experienced at least one adverse effect.
Conclusion and Relevance
The findings appeared to imply the therapeutic effects of neuromodulators on CRC, especially in patients presenting with chemical triggers. However, the adverse effect was a factor that may impact on application of this treatment method.

Introduction
Chronic refractory cough (CRC) is defined as a cough that persists beyond 8 weeks, despite adequate evidence-based investigation and management of possible underlying causes.1-3 This common but debilitating condition is estimated to affect up to 10% of the adult population and can have significant negative impacts on the day-to-day function and psychosocial well-being of affected patients.4-10 Despite advancements in our understanding of the etiology and underlying pathophysiology of CRC, as well as a widening number of treatment options available, it remains challenging for the treating clinician to determine which patients will benefit from which management option. This can result in multiple failed interventions, treatment fatigue, and considerable financial burden for both the patient and the health care system.
Laryngeal sensory dysfunction describes a vagal afferent sensory neuropathy, resulting in laryngeal sensory symptoms such as abnormal throat sensation and CRC.8,11-14 The majority of patients with CRC report accompanying laryngeal sensory symptoms. 12 Prolonged peripheral sensitization of the larynx is thought to lead to sensitization of central sensory pathways, resulting in persistent heightened laryngeal sensitivity and a lower threshold of response to triggers.15,16 Numerous triggers of cough have been described and can broadly be classified into mechanical (eg, talking, chewing), chemical (eg, perfumes and fumes), thermal (hot or cold), and abnormal throat sensation (eg, tickle and itch).3,17
Neuromodulator therapy is a well-established management option for patients with CRC, primarily targeting sensitized central cough pathways.8,17-20 Although this has been shown to work most effectively in combination with behavioral interventions such as cough desensitization therapy, there is limited literature on factors that predict a positive response to neuromodulator therapy.21-23 In addition, side effect profiles (0%-80%) and efficacy rates vary significantly in the literature, with most studies to date limited by small sample size.24-28 Defining factors that are predictive of successful neuromodulator therapy is important to facilitate customization of patient care within a CRC treatment algorithm, focusing on selecting the correct intervention for each patient. 18 Ultimately, this will enable clinicians to deliver timely and effective interventions for CRC while minimizing morbidity.
The aims of this study were 2-fold. First, to evaluate the treatment efficacy and adverse effects of neuromodulators in patients with CRC, and to identify factors predicting treatment outcomes. The predictive factors investigated include history and investigations which are routinely assessed in our specialized CRC clinic: patient-reported cough triggers, citric acid cough threshold test (CACTT), and response to superior laryngeal nerve (SLN) blocks. CACTT has been used as a measure of treatment responsiveness to Gefapixant, although this did not demonstrate a change. 29 SLN blocks have previously been demonstrated to predict response to peripheral treatments for CRC such as supraglottic Botulinum A toxin, although we hypothesize that this will not be the case for centrally acting neuromodulators. 13 Second, we aimed to calculate the minimal important difference (MID) in the Cough Severity Index (CSI) for CRC patients treated using neuromodulators.
Materials and Methods
Ethical Approval
Ethics approval was obtained from the Sydney Local Health District (RPAH Zone) Human Resources and Ethics Committee (HREC) [Approval LNR/18/RPAH/185].
Participants
This was a retrospective study of patients presenting to a CRC clinic at a single private institution who were managed by an otolaryngologist with subspecialty training in Laryngology. Successive patients who underwent neuromodulator therapy for the treatment of their CRC between 2012 and 2022 were included in this study. The inclusion criteria were: Age >16 years and <83 years; cough duration >3 months; normal chest X-ray or no changes to chest X-ray since cough started; cough persisting despite standard evidence-based management of other conditions, including asthma, respiratory disease, gastro-esophageal reflux disease, sinonasal disease, and obstructive sleep apnea; English speaking, and compliant with follow up.
Data Collection
Data was collected retrospectively from the medical records, including demographic data such as age, gender, and symptoms related to chronic cough, including duration and triggers of cough, which are collected on a routine standard intake form. Details of investigations for chronic cough, such as citric acid cough threshold testing, were also collected. Citric acid cough threshold testing was performed in accordance with the European Respiratory Society Guidelines. 30 Cough response was considered positive if 2 or more coughs were triggered (C2 response threshold). Patients were grouped by C2 threshold into nonsensitive (C2 threshold ≥0.08 mol/L) and hypersensitive (C2 threshold ≤0.06 mol/L), based on previous research by our group (unpublished data). Treatment information collected included details of neuromodulator therapy (type of medication, dose, duration of treatment, side effects, and response to treatment), as well as alternative treatments trialed for chronic cough (speech therapy, laryngeal Botulinum A toxin injection, and SLN steroid block).
Patient-reported outcome measures (PROMs) were collected at regular intervals to assess response to therapy, including the CSI, Reflux Symptom Index (RSI), and the Newcastle Laryngeal Hypersensitivity Questionnaire (NLHQ). These PROMs are outcome measures that take into account symptom severity and the impact of symptoms on quality of life; and have all been previously validated with abnormal scores being: CSI >3 9 ; RSI >12 31 and NLHQ <17.1. 32
Global Self-Ratings of Response:
All patients were asked to complete a global rating scale to self-evaluate the level of improvement posttreatment. The scale had 3 points: no response (1), partial response (2), and complete response (3).
Statistical Analysis
Data was managed using Microsoft Excel 365 and analyzed using the statistical software package IBM SPSS v28.0 (Windows). Descriptive statistics [mean, standard deviation (SD), and 95% confidence interval for continuous variables and frequency and percentage for categorical variables] were used to characterize groups. Treatment effects were examined using a sample t-test with Cohen’s d as effect size. Two-way repeated-measures analysis of variance (ANOVA) was used to evaluate the change in PROMs posttreatment. The effect size for ANOVA was calculated using partial Eta squared (η2), in which effect size of 0.01, 0.1, and 0.25 indicated small, medium, and large effects, respectively. 33 The chi-square test was used to test the association between risk factors and the outcomes for categorical variables. In all statistical analyses, a significance level of 2-sided P = .05 was used.
To determine the MID in CSI for chronic cough patients treated using neuromodulators, 2 methods were applied, which are described below.
Anchor-based method
The global rating scale was used as an anchor to determine the level of improvement. Mean difference of the CSI was calculated from the pre-post CSI data. Then the mean difference of CSI was calculated for each of the response categories using mean and SD. Nonparametric (Spearman’s) correlation coefficients were calculated to find the correlation between pretreatment CSI and global change, between posttreatment CSI and global change, and between CSI difference score and global change. These correlations allowed validation of whether the global change was reflected in the post-CSI score and CSI difference.
Distribution-based (effect size) method
This method examined the effect size (Cohen’s d) for the difference in CSI difference in each response category. The effect size was small, moderate, or large (0.2, 0.5, and 0.8, respectively).
Results
Participant and Cough Characteristics
One hundred and three patients were included in this study. The mean age was 56.6 years, [SD = 13.5, range 21-83 years]. The majority of patients were female (n = 72, 69.9%). The mean duration of chronic cough was 8.6 years (range 0.5-30 years).
Tables 1 and 2 outline the cough characteristics of our patient cohort. The most commonly reported antecedent factor at the onset of cough was an upper respiratory tract infection (n = 45, 44.1%). A significant proportion of patients (n = 41, 40.2%) had no known preceding factor at the onset of their chronic cough. Mechanical stimuli (eg, talking, laughing, and neck position) were the most commonly reported trigger for cough in our patient cohort with many patients reporting multiple triggers.
Preceding Factors of Chronic Refractory Cough in Patient Cohort (n = 103)

Abbreviation: URTI, upper respiratory tract infection.
Triggers of Chronic Refractory Cough in Patient cohort (n = 103)

Some patients report multiple triggers.
Abbreviation: ATS, abnormal throat sensation.
Neuromodulator Therapy: Dose and Side Effect Profiles
84 patients (81.6%) trialed Amitriptyline, 48 patients (46.6%) trialed Gabapentin, and 3 patients (2.9%) trialed Pregabalin. Twenty-nine patients (28.1%) trialed both Amitriptyline and Gabapentin, 1 patient (0.9%) trialed both Amitriptyline and Pregabalin, and 2 patients (1.9%) trialed all 3 medications. Forty percent of patients (n = 34) ceased Amitriptyline, and 36% of patients (n = 18) ceased Gabapentin due to side effects.
The reported side effects of both Amitriptyline and Gabapentin are listed in Table 3. Sedative and anticholinergic side effects were among the most common with both medications, including drowsiness, dry mouth, tremor, and gastrointestinal symptoms. Multiple side effects were common, with 17 patients (20%) taking Amitriptyline and 10 patients (21%) taking Gabapentin reporting more than one side effect. Thirty-one percent (n = 26) of patients taking Amitriptyline and 42% (n = 20) patients taking Gabapentin did not report any side effects.
Side Effect Profile Reported with Neuromodulator Therapy.

The dose of Amitriptyline ranged from 10 to 100 mg daily, and the dose of Gabapentin ranged from 200 to 1800 mg daily in divided doses. Patients were generally started on either 25 mg at night of Amitriptyline or 100 mg 3 times a day of Gabapentin, and the dose was titrated based upon treatment response and side effects. The median dose of Amitriptyline was 30 mg, and the median dose of Gabapentin was 600 mg.
Minimal Important Difference of CSI
Figure 1 shows the change in CSI score after treatment for the 3 response categories.

Changes in CSI in 3 categories of response. CR, complete response; CSI, Cough Severity Index; NR, no response; PR, partial response.
Table 4 shows the change in CSI posttreatment for each category of neuromodulator response. The “no response” group had a small magnitude of change in CSI posttreatment. The “partial response” group had a larger magnitude of change, and the “complete response” group had the largest magnitude of change in CSI posttreatment.
Change in Cough Severity Index for Each Neuromodulator Response Category.

Abbreviation: SD, standard deviation.
Correlations were calculated across the variables of interest. Findings showed that Pearson’s r = .533 (P < .001) between pre- and posttreatment CSI.
Rank correlations were calculated between pretreatment CSI, posttreatment CSI, CSI difference, and global change rating. Rank correlation was used because the global change rating used an ordinal scale (1-3). Spearman’s r = −.083 (P = .4119) between pretreatment CSI and global change rating; Spearman’s r = −.564 (P < .001) between posttreatment CSI and global change rating; and Spearman’s r = .541 (P < .001) between CSI difference and global change rating.
Table 1 shows that the effect size was small for the “no response group,” moderate for the “partial response” group, and large for the “complete response group.” From the anchor-based and distribution-based/effect size approaches, it is reasonable to conclude that a change in the CSI of mean (SD) of 6.9 (6.1) could represent the MID.
Response to Neuromodulator Therapy
Response to neuromodulator (NM) therapy was defined as a change in CSI of at least 7. The response was evaluated for 99 patients as 4 had missing data for CSI (either pre- and/or posttreatment). Figure 2 shows the percentage of patients who responded to NM treatment. This figure shows that 55.6% responded and 44.4% did not respond to NM therapy.

Response to NM as indicated by change in CSI of ≥7 posttreatment. CSI, Cough Severity Index; NM, neuromodulators.
Cough Severity Index
Two-way repeated-measures ANOVA [Response (Yes/No) × CSI (Pre/Post)] with the variable “Response” based on improvement in CSI of ≥7 posttreatment was performed. The model showed significant main effects of treatment: F(1, 97) = 199.897, P < .001, partial η2 = 0.673. There was a significant interaction effect between the variable “CSI” and response group: F(1, 97) = 138.650, P < .001, partial η2 = 0.588, meaning that changes in CSI scores were larger for the response group than for nonresponse group (Figure 3). In the response group, CSI scores dropped by 15.2 posttreatment (Sidak-adjusted P < .001), while in the nonresponse group, CSI only dropped by 1.4 (Sidak-adjusted P = .116).

Changes in CSI scores in response groups post NM treatment. Error bars represent 95% CI. CI, confidence interval; CSI, Cough Severity Index; NM, neuromodulators.
Newcastle Laryngeal Hypersensitivity Questionnaire
Figure 4 shows changes in NLHQ scores after NM treatment. There were also significant main effects of treatment on NLHQ scores: F(1, 64) = 4.977, P = .029, partial η2 = 0.072. There was no significant interaction between the response group and NLHQ score: F(1, 64) = 0.393, P = .533, partial η2 = 0.006. In the response group, NLHQ increased by 2.4 posttreatment (Sidak-adjusted P = .047). Meanwhile, the nonresponse group only showed an increase of 1.4 posttreatment (Sidak-adjusted P = .261).

Changes in NLHQ scores in response groups post NM treatment. Error bars represent 95% CI. CI, confidence interval; NLHQ, Newcastle Laryngeal Hypersensitivity Questionnaire; NM, neuromodulator.
Reflux Symptom Index
Significant main effects of treatment were found for RSI: F(1, 97) = 129.856, P < .001, partial η2 = 0.572. There was also a significant interaction effect between the response to NM and RSI score: F(1, 97) = 30.301, P < .001, partial η2 = 0.238, demonstrating different levels of change in RSI posttreatment between the 2 groups (Figure 5). In the response group, RSI score decreased by 11.7 posttreatment (Sidak-adjusted P < .001), and in the nonresponse group, RSI decreased by 4.1 posttreatment (Sidak-adjusted P < .01).
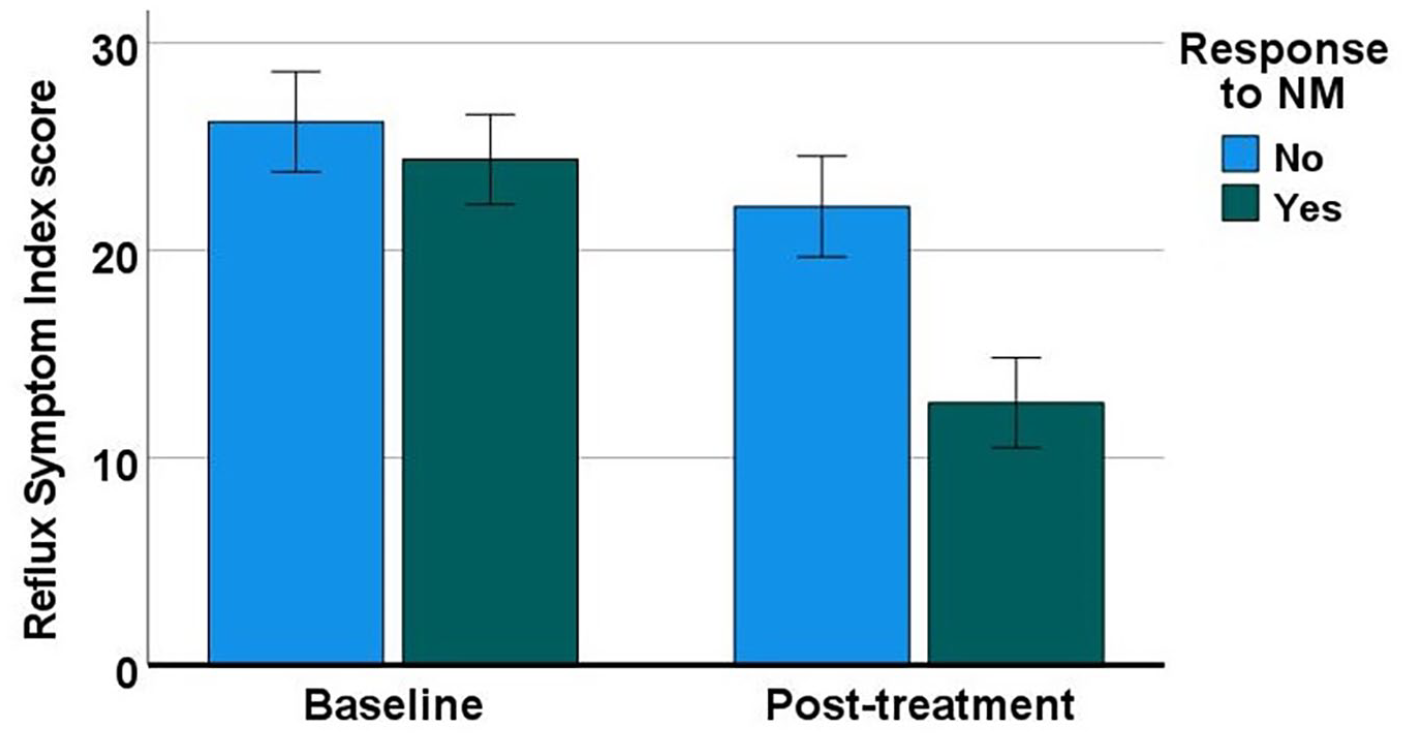
Changes in RSI scores in response groups post NM treatment. Error bars represent 95% CI. CI, confidence interval; NM, neuromodulator; RSI, Reflux Symptom Index.
Predictive Factors for Treatment Outcomes
Cough triggers
Chemical triggers significantly predicted response to NM using MID of 7: χ(1)2 = 4.813; P = .028. Using the MID of 7, the thermal trigger did not predict response to NM [χ(1)2 = 0.849; P = .357] nor did the mechanical trigger (χ(1)2 = 0.003; P = .957) or the abnormal throat sensation trigger [χ(1)2 = 1.573; P = .210].
Citric acid cough threshold test
Cough sensitivity using CACTT did not significantly predict response to NM therapy: χ(1)2 = 0.040; P = .842. Logistic regression was calculated with the predictor being cough threshold testing scores, and the dependent variable being cough response using MID of 7 for CSI. Results showed that the model was not statistically significant: χ(1)2 = 0.266, P = .606. Only 0.6% of the variance in the dependent variable was explained by the predictor, and 56.3% of cases were correctly classified: Wald test = 0.265; P = .607; Exp(B) = 0.467. These results demonstrated that cough threshold testing scores did not predict response using the MID of 7 for CSI.
NLHQ scores
To clarify whether NLHQ predicted response to NM, demonstrated by a decrease in CSI using an MID of 7, the logistic regression equation was calculated with pre-NLHQ as the predictor. The model was not statistically significant: χ(1)2 = 0.0; P = .984. Zero percentage of variance in the dependent variable was explained by pre-NLHQ, and 39.7% of cases were correctly classified. Wald test = 0.0; P = .984; Exp(B) = 1.001. These showed that pretreatment scores of NLHQ did not predict cough response using MID of 7 for CSI.
CSI scores
To evaluate whether CSI scores predicted response to NM therapy using MID of 7, logistic regression equation was calculated with pretreatment CSI scores as the predictor. The model was significant: χ(1)2 = 5.603; P = .018 and 7.4% of the variance in the dependent variable was explained by pretreatment CSI. 62.6% of cases were correctly classified. Wald test = 5.278; P = .022; Exp(B) = 1.065. This demonstrates that pretreatment CSI was a significant predictor of response to NM using an MID of 7 for CSI.
Response to SLN block
Response to SLN block (unilateral or bilateral) did not predict response to NM therapy: χ(1)2 = 0.599; P = .439. A response to the SLN block was defined as = >3 points of improvement in at least one symptom on the previously validated 7-point MYCaW questionnaire 34 , which was adapted for use in this population.
Discussion
This research is among the first to investigate predictive factors for the success of neuromodulator therapy in patients with unexplained or refractory chronic cough.21,23 Of significance, we have demonstrated that patients with chemical triggers for their chronic cough, such as scents, odors, and fumes, are more likely to respond to neuromodulator therapy than those with other triggers (thermal, mechanical, abnormal throat sensation). To our knowledge, this has not been previously reported.
There have been 2 additional studies to date that have investigated factors that predict responsiveness to neuromodulator therapy. Zhang et al found that coughing with movement or eating (ie mechanical triggers) and the presence of abnormal throat sensation on questionnaires were independent predictors of Gabapentin efficacy. 23 Although this is contradictory to our finding that mechanical, thermal, and abnormal throat sensation triggers were not predictive of neuromodulator therapy success, we did find that patients who responded to neuromodulator therapy had significant improvements in PROMs, which evaluate symptoms of abnormal throat sensation (NLHQ and RSI). We did not evaluate individual questions from these PROMs as Zhang et al 23 have due to limitations in sample size, however, we may have had a similar finding given the overall improvement seen on these questionnaires in patients who responded to neuromodulator therapy. More recently, a group evaluating Pregabalin found that patients who responded were more symptomatic at baseline on PROMs. 21 Although we have used different PROMs in this study, this is consistent with our finding that CSI pretreatment was higher in the group of patients who responded to neuromodulator therapy, and that pretreatment CSI was predictive of neuromodulator treatment response.
Overall, based on the calculated MID for CSI of 7, we had a response rate to neuromodulator therapy of 55%. Two randomized controlled clinical trials of Gabapentin or Amitriptyline versus placebo have demonstrated higher efficacy rates of these medications of approximately 70% each.25,27 However, there is significant variation in the response rate reported in the literature, with less stringent outcome criteria than we have utilized here, varying from 10 to 70%.24,28,35,36 This is likely due to differing outcome measures used, differing criteria for what constitutes a response, and wide variation (2-62 participants) in sample size in these studies.24,28
Our cohort had a relatively high side effect profile with neuromodulator medication, with 70% of our patient cohort reporting at least 1 side effect, and approximately 20% of patients reporting at least 2 side effects. There was a high discontinuation rate due to intolerable side effects of 35% overall. We also found a slightly higher side effect rate with the use of Amitriptyline, which has been described previously. 37 Once again, there is significant variation in the literature for reported side effect rates with neuromodulator therapy, ranging from 17% to 80%,24,28 with some additional studies reporting no side effects.18,26 Previous randomized controlled clinical trials of Gabapentin or Amitriptyline versus placebo reported side effect rates of 30% and 66%, respectively.25,27 The side effects reported by our patients are consistent with what has previously been reported, with anticholinergic effects and drowsiness the most commonly reported adverse effects.24,25,27,28,37
There are several limitations of this study. Although the sample size of 103 patients is one of the largest to date in the literature, it was not large enough for subgroup analysis of response rate for individual medications, and the purpose of the study was to evaluate our neuromodulator protocol overall. Furthermore, because the global change rating scale only had 3 levels, smaller changes in CSI may not have been detected; thus, further studies would be helpful to further validate the MID of 7 for CSI that we have identified in CRC patients.
Conclusion
This retrospective series demonstrates in a cohort of patients with CRC that, although there is a high rate of discontinuation due to the side effect profile of neuromodulator medications, this treatment does have utility, particularly in patients who report chemical triggers of their cough.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This Research was funded by the Dr Liang Voice Program at the University of Sydney.
Ethical Approval and Informed Consent Statements
Ethics approval was obtained from the Sydney Local Health District (RPAH Zone) Human Resources and Ethics Committee (HREC) [Approval LNR/18/RPAH/185]. Patient consent was waived due to it being impractical to seek consent for patients seen in the past and it was considered a threat to patient privacy to implement a process to locate and contact each individual participant to seek their consent. This waiver was approved by the Human Research Ethics Committee approval provided above.
