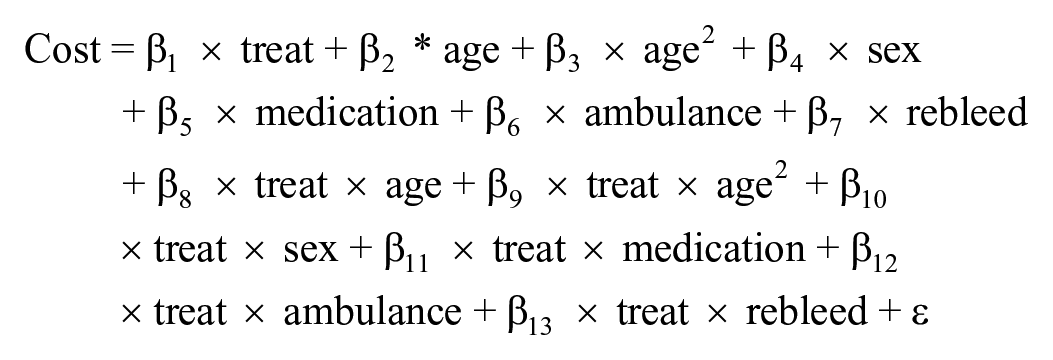Abstract
Importance
Epistaxis affects approximately 60% of the population over their lifetime. When conservative attempts fail, nasal tampons are often required to stop anterior bleeding. Health economics is critical in our publicly funded system. Determination of cost-effective interventions is crucial.
Objective
To compare the total cost of Merocel and Rapid Rhino from the perspective of a provincial payer and an academic hospital for the management of anterior epistaxis.
Design
Retrospective review.
Setting
London Health Sciences Centre emergency department (Victoria and University campus).
Participants
Patients ≥18 years of age presenting with anterior epistaxis. The participants were 67% men and 33% women. Approximately, 63% were on anticoagulant medication, and 35% used an ambulance to arrive at the hospital.
Intervention
Rapid Rhino or Merocele, which was dependent on the site of presentation.
Main Outcome Measures
Rebleed rate.
Results
The rate of rebleeds with Merocel was 42% (26/62), whereas it was 24% (4/17) with Rapid Rhino. The inverse probability weighted regression adjustment results show that patients receiving Rapid Rhino did not have a statistically significant difference in costs per patient ($62.40, 95% CI: −$25.75 to $150.55) from the hospital perspective or the provincial health care payer perspective ($78.25, 95% CI: −$18.38 to $174.89).
Conclusion and Relevance
There was no significant difference in cost between Rapid Rhino and Merocel for anterior epistaxis from a hospital or provincial payer perspective.

Introduction
Epistaxis is a common presenting complaint for patients in the Emergency Department (ED). Approximately, 60% of the population will experience an episode of epistaxis over their lifetime. 1 Many of these episodes are self-limited and resolve with the application of pressure, but 6% require further medical interventions.2,3 In the ED, nondissolvable nasal packing is commonly used for control after first-aid measures fail. Merocel is one of the more popular nondissolvable nasal packs and is a compressed, dehydrated sponge composed of hydroxylated polyvinyl acetate. 4 It is not only effective but also relatively affordable. Rapid Rhino is another nondissolvable nasal pack composed of a single-cuffed inflatable balloon catheter that is coated with carboxymethylcellulose. The inflatable balloon is high volume, but of low pressure, and is designed to uniformly tamponade the entire nasal cavity, while carboxymethylcellulose provides a scaffold for clot formation.
A systematic review from 2017 concluded that Rapid Rhino appeared to be the most comfortable nondissolvable nasal pack, yet Merocel is routinely used in hospitals because of its lower price. 5 Although Merocel is less expensive than Rapid Rhino, it can be more challenging to successfully insert for the first time. 6 A systematic review and meta-analysis found that Rapid Rhino is more comfortable for patients. 5 Currently, few studies have analyzed the cost of anterior epistaxis using Merocel and Rapid Rhino.5,7-10 The purpose of the present study was to compare the total cost of Merocel and Rapid Rhino from a provincial payer and an academic hospital perspective for the management of anterior epistaxis.
Methods
Ethics approval for this study was obtained from both the Lawson Health Research Institute and Western University to retrieve patient medical charts from Victoria and University Hospitals (HSREB: 119264). Victoria and University hospitals were chosen because they are both teaching hospitals located in London, Ontario, Canada, with similar patient demographics (Appendix). Both of these hospitals are under the London Health Sciences Centre (LHSC) allowing for similar billing practices and approaches to handling care. After research ethics board approval was received, a retrospective review was performed for all adult patients presenting to the LHSC (Victoria or University Hospital) ED in London, Ontario, Canada. Patients included in the study were 18 years or older with a primary diagnosis of epistaxis and received either Merocel or Rapid Rhino nasal packing between January 1, 2018 and December 31, 2018 without a history of recent surgery in the head and neck region. This timeframe was chosen because Rapid Rhino was trialed at University Hospital for the first 6 months of the year and was preferentially used for management while under trial. Patient demographics, anticoagulation status, type of epistaxis intervention, recurrence of epistaxis (rebleed event), and use of emergency medical services were retrieved from medical charts. The use of an ambulance was not a previously discussed risk factor in the literature; however, using an ambulance could indicate a more severe bleeding or rebleeding which may incur more costs and was therefore included in the list of explanatory variables. Patient data within physical charts were cross-referenced to the electronic patient records to ensure accuracy and to verify any missing data.
A rebleed event was defined as the recurrence of epistaxis within 14 days of receiving first nasal packing for epistaxis treatment, or if Otolaryngology—Head and Neck Surgery (OHNS) was consulted to provide further intervention during the initial epistaxis treatment. A follow-up period of 2 weeks was chosen to be consistent with a previously published paper by Newton et al. 11 Rebleeds were measured as a dichotomous variable (0 for absence, 1 for presence), and healthcare costing information included only the first rebleed after the patient was discharged from the ED, even if multiple bleeds occurred within 2 weeks. Multiple rebleeds were defined as a return to the ED for a second rebleed after their initial discharge within the 2-week follow-up period. Treatment of rebleed presentation was at the discretion of the treating physician and could range from conservative management to repacking of the nose or consultation with the otolaryngology service.
The cost analysis was conducted from 2 distinct perspectives: the hospital and the provincial healthcare payer. Hospital cost data were obtained from the LHSC case-costing center. Provincial health care costs included all physician fees in addition to total hospital costs. Physician fees were obtained from the “Multiple Systems Assessment Billing” section within the Schedule of Benefits in consultation with ED physicians for codes billed in presented scenarios. 12 Multiple systems assessment billing can occur when an ED physician performs a history and physical examination for more than 1 body part or system. 12 Additional physician fees were included for interventions performed and described in the health records. OHNS billings (base consultation fee + scoping fee + travel fee + first person seen fee) were also taken into account if the patient was seen by OHNS in consult during the encounter.
Inverse probability weighted regression adjustment (IPWRA) was used to calculate the average treatment effect on the treated estimates, after accounting for the differences in observed patient characteristics between the 2 groups.13-15 The IPWRA estimation proceeded in 2 steps. First, we estimated propensity scores using a logistic regression on treatment selection. The variables included in the logistic regression model were patient’s sex, age, age-squared, anticoagulant medication use and the use of an ambulance at the initial visit. The chance of unobservable selection into treatment was negligible in our study as Victoria Hospital never had a Rapid Rhino option for patients. For better balancing of covariate distributions across the 2 groups of patients, we used a covariate balancing propensity score procedure.14,16 After the propensity scores were estimated, we constructed inverse probability weights (ie, assigning the inverse of probability of treatment for patients treated with Rapid Rhino vs Merocel). After applying weights, we find that the standardized difference in means of each covariate is closer to zero, and the variance ratio is closer to 1 (see Appendix), demonstrating the covariate balancing condition required for IPWRA. In the second step, we estimated the outcome model (ie, costs) using these propensity score weights. The outcome model for our IPWRA is specified as follows:
We were interested in the estimated coefficient on “treat” (Rapid Rhino = 1, Merocel = 0) as the treatment effect on the treated estimate. The explanatory variables included in the outcome model above are sex, age, use of anticoagulant medications and use of an ambulance for the initial ED visit. These variables were chosen from taking into consideration expert opinion, as well as previous literature that indicated that these variables were potential risk factors for epistaxis incidence and rebleed events, and therefore could confound the relationship between the nasal pack and total costs.17-19 Sex was coded as a dichotomous variable representing 0 for males and 1 for females. Similarly, data on anticoagulant medication and the use of an ambulance were dichotomous variables, where 1 represented presence and 0 represented absence. Age and age-squared were continuous variables. The inclusion of age and age squared could help provide a potential nonlinear relationship between the age of the patients and the total cost. As the cost data were positively skewed, log-gamma regression models fit the data better and, hence, were used to assess differences in total costs between Rapid Rhino and Merocel.
Several sensitivity analyses were undertaken to ensure that our results were robust to alternative inclusion/exclusion criteria. The first sensitivity analysis was conducted for patients who had multiple bleeding events. The cost data were only included until the first bleed in our main analysis, but some patients experienced multiple bleeds after their first bleed within the 2-week timeframe. For these patients, only the first bleed was included in our main analysis. However, these patients could have potentially presented to the ED with more severe bleeding or different unmeasured characteristics than patients who did not have multiple bleeding events. We excluded patients who experienced multiple bleeding events to determine whether this affected the results. Another sensitivity analysis was conducted by excluding patients who were classified as rebleed and who required an OHNS consult during their initial visit, as a consult might indicate a more severe bleed. Finally, we excluded patients who did not return to the ED for packing removal to understand if this would affect our conclusions, as our main analysis included these patients.
Results
A total of 465 patients who were admitted to the ED at University and Victoria Hospital with a primary diagnosis of epistaxis were identified. Among those, 89 patients had received nasal packing with Merocel or Rapid Rhino, 72 (81%) from the Merocel group, and 17 (19%) from the Rapid Rhino group. Patients with epistaxis as a postoperative complication were excluded.. After verification of case-costing information and cross-referencing of paper and electronic charts, 7 patients from the Merocel group were excluded. A “common support region” restriction based on estimated propensity scores was then imposed to ensure that the propensity score distributions between the Merocel and Rapid Rhino groups overlapped. This analysis found that 3 patients in the Merocel group were not in the common support region and had to be excluded. There were 62 patients remaining in the Merocel group (Figure 1) and 17 patients in the Rapid Rhino group.

Inclusion and exclusion for Merocel.
Rebleed Rate
Table 1 shows the rebleed rates for the study. Patients discharged with a nasal pack, as shown in Table 1, were those patients who left the ED with a nasal pack in place. A small minority of initial patients were treated with nasal packing but had the nasal pack removed before they left the ER (1 in the Rapid Rhino group, 3 in the Merocel group). This was either secondary to failure with subsequent cauterization or due to patient discomfort. There were no patients who required readmission. Overall, patients treated with Merocel had a higher rebleed rate than those treated with Rapid Rhino (42% vs 24%). Merocel also had a higher rate of rebleeds during the 14-day follow-up after the initial treatment (39% vs 19%). However, Rapid Rhino had a similar rate of rebleeds as Merocel when looking at rebleeds that needed an OHNS consult during the initial visit (6% vs 5%). There were no significant differences between Merocel and Rapid Rhino in terms of total rebleed (P = .17) or ENT consultations during the initial visit (P = .86) according to the Wilcoxon rank-sum test.
Rebleed Rates for Merocel and RapidRhino.

Costs
Descriptive data revealed that Rapid Rhino had a higher median cost than Merocel ($367.54 vs $276.80) from the hospital’s perspective. When using the IPWRA, we found a nonsignificant difference in costs per patient, with a significance level of alpha .05 ($62.40, 95% CI: −$25.75 to $150.55).
From the provincial health care payer perspective, Rapid Rhino costs, on average, $24.77 more than Merocel (Table 2). However, the IPWRA estimates resulted in $78.25 (95% CI: −$18.38 to $174.89) difference in costs per patient, which was not statistically significant (P = .11). Therefore, our sample did not provide evidence that the costs of Rapid Rhino and Merocel are different.
Descriptive Statistics on Costs for Merocel and Rapid Rhino.

We also performed several sensitivity tests by excluding patients who could potentially skew the results (Table 3). One patient who received Rapid Rhino and 3 patients who received Merocel were excluded because they received OHNS consultations for initial visits. Additionally, 8 patients who received Merocel and 2 patients who received Rapid Rhino were excluded because they had multiple bleeds. In the Merocel group, 16 patients were excluded, and 2 patients who received Rapid Rhino were excluded as they did not return to the ED for pack removal. After these patients were excluded, there was no significant difference between the costs of Rapid Rhino and Merocel.
Average Treatment Effect Estimates Using IPWRA.
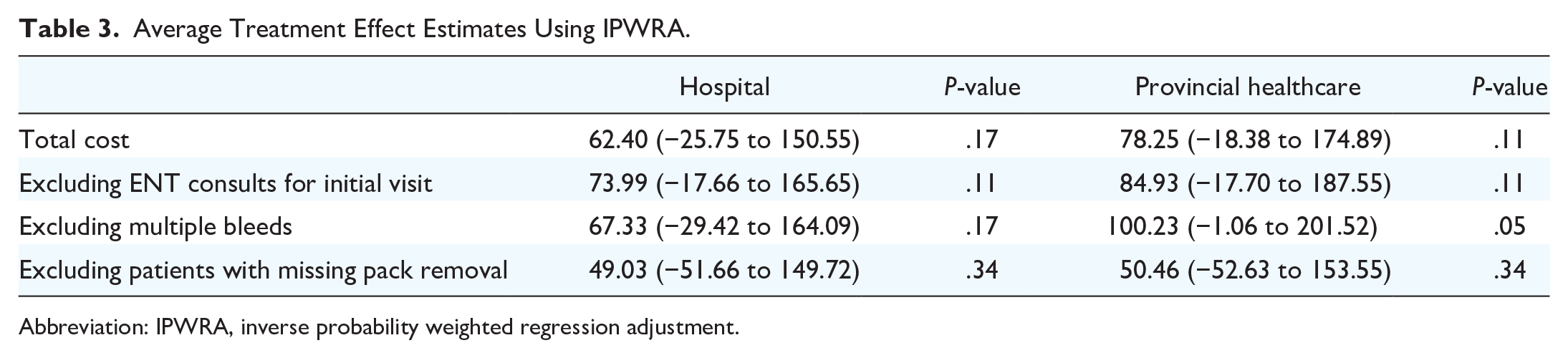
Abbreviation: IPWRA, inverse probability weighted regression adjustment.
We found that 14 of the 28 patients who took an ambulance to their initial visit had a rebleed, in contrast to 16 out of 51 patients that had a rebleed but did not take an ambulance. Similarly, 13 out of 28 patients who took the ambulance to their initial-visit had a follow-up bleed, while 14 out of 51 patients had a follow-up bleed, but did not take the ambulance. This result was significant at the 10% alpha level. From the provincial care perspective, we found that taking an ambulance results in an increase of $99.17 (95% CI: $1.85 to $196.49) per patient compared with patients not taking an ambulance. This result was significant at the 5% level (P = .05).
Discussion
Epistaxis is a very common problem in the ED. For episodes that are not self-limiting, nondissolvable packing is often required. To date, no studies have directly compared the costs of Merocel and Rapid Rhino from the prospectives of hospitals and provincial payers. Our findings suggest that there is no significant difference in health care costs between the 2 nasal packs, despite Rapid Rhino being more expensive per unit.
Rebleeding rates can greatly affect the number of times a patient will present to the ED. Newton et al 11 utilized the same 2-week follow-up as ours and found a 26% (24/92) rate of rebleeding for patients who were discharged from the hospital with Merocel. 11 Our rebleeding rate of 39% (23/59) for patients who left the hospital with a nasal pack inserted was slightly higher than Newton et al. Singer et al 20 reported that the rate of rebleeding was 6% in those treated with Rapid Rhino when the bleeding rate was measured at 3 days after pack insertion. This rate is much lower than our reported rebleed rate of 19% at 2 weeks with Rapid Rhino. The difference is likely due to their follow-up time, which was only 3 days versus 2 weeks in this study.
There is a paucity of cost-analysis studies in the literature concerning nasal tampons. This limits our data for comparison. Nithianandan et al 10 conducted an economic evaluation in Ottawa, Canada, for anterior epistaxis using 30-day total cost. Their analysis found that the median cost for Merocel was $763.98 (IQR: $632.25 to $830.23). This is roughly comparable to the provincial health care costs in this study of $406.02 (IQR: $290.14 to $600.22), since our study evaluated costs for the first rebleed within 2 weeks in contrast to 30 days. The difference between groups may be accounted for by either more complete global costing information or increased healthcare utilization beyond 2 weeks versus this study.
Murray et al 7 conducted a cost-effectiveness analysis where the median cost for their Merocel/Vaseline gauze group for the healthcare perspective (including physicians) using a 30-day time horizon in Edmonton, Canada, was $2704.51 (IQR: $354.71 to $3846.71). This estimate was much higher than our estimates even when taking into consideration the different follow-up periods and relatively higher physician remuneration in Alberta. This difference in cost estimates is likely because Murray et al included inpatient costs, including surgical/medical ward costs.
Our study is not without limitations. Absent or missing data meant that a number of patients had to be excluded. Eighteen patients (16 Merocel, 2 Rapid Rhino) did not return to the ED for packing removal; therefore, the rebleed rates and costs of packing removal could not be calculated. The accuracy of the paper medical charts from the ED were variable in the information documented and the current case costing system used at LHSC does not directly allocate all consumable costs to specific patients. The ED billings were estimated using fees that apply to a community setting, as the remuneration at the LHSC as an academic institution is complex. Since no quality-of-life data are available for these epistaxis patients, a formal cost-effective analysis was not possible. In terms of our statistical analysis, certain risk factors such as smoking status, abuse of drugs, bleeding disorders, warfarin usage, and proper training for epistaxis management, which can affect the risk of rebleeding and may confound the relationship between costs and treatment, could not be adjusted for in this study.21-25 Furthermore, we could not stratify patients based on whether they experienced bilateral bleeding. We could also not stratify patients on the basis of the number of tampons packed until the bleeding was successfully stopped. These variables would have helped in capturing the severity of the patient’s bleeding.
Our findings may not be generalizable to other institutions in different provinces in Canada with different physician remuneration plans, and the specific product cost may also differ across institutions; however, the general trend is that Rapid Rhinos costs more per unit than Merocel.
Conclusions
Anterior epistaxis is a very common condition that affects the vast majority of the population in their lifetime. This study directly compares the costs of Merocel to that of Rapid Rhino. Our analysis revealed that there was no significant difference in cost for Merocel or Rapid Rhino for anterior epistaxis.
Footnotes
Appendix
Weighted Coefficients Before and After IPWRA Weighting.

| Variable | Standardized difference in means (unweighted) | Standardized difference in means (weighted) | Variance ratio (unweighted) | Variance ratio (weighted) |
|---|---|---|---|---|
| Sex | −0.26 | −0.005 | 0.82 | 0.99 |
| Age | −0.30 | −0.01 | 1.80 | 1.06 |
| Age² | −0.27 | −0.002 | 1.32 | 0.99 |
| Ambulance | −0.12 | 0.001 | 1.11 | 1.00 |
| Medication | −0.20 | −0.02 | 0.93 | 0.98 |
Abbreviation: IPWRA, inverse probability weighted regression adjustment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.