Abstract
Importance
In different types of carcinoma, sex has a strong impact on risk factors, diagnosis, treatment, and outcomes. Previous studies have reported that female patients with head and neck carcinoma (HNC) have better survival.
Objective
This study aimed to analyze sex differences in tumor regression and survival, as well as host- and disease-related factors.
Design
A retrospective cohort study.
Setting
Tertiary Care, University Hospital.
Participants
1115 patients registered in the Head and Neck Tumor Registry of the Department of Otorhinolaryngology, Head and Neck Surgery, Medical University of Innsbruck were included.
Main Outcome Measures
Several host- and disease-related factors were analyzed to determine possible sex differences.
Results
This study included 1115 patients with HNC, of whom 78.6% (n = 876) were men and 21.4% (n = 239) were women. There were no differences in tumor regression or incident cases per year in our department between male and female patients with HNC. Statistically significant sex differences were found for tumor site (P < .001), Union for International Cancer Control (UICC) stage (P = .04), treatment modality (P = .027), alcohol consumption (P = .005), and alcohol cessation (P < .001).
Conclusions and Relevance
This study revealed significant sex differences in patients with HNC. Female patients with HNC were characterized by a higher percentage of oral carcinoma, lower UICC tumor stage, single treatment modality, and less alcohol consumption than men.

Introduction
Sex exerts a strong impact on health and illness and affects disease onset and progression, diagnosis, therapy, and therapeutic outcomes. 1 Among distinct tumor types, women are reported to have better survival than men. In an observational population-based study, women exhibited a lower risk of mortality due to lung, stomach, and head and neck carcinomas (HNC) than that in men. 2
Literature on sex differences among patients with HNC regarding survival and host- and disease-related factors is scarce, although better survival among female patients with HNC has been reported in multiple studies. Leoncini et al., 3 in a multicenter study including 4005 patients with HNC, described a reduced risk of recurrence in women with laryngeal carcinoma than in men (hazard ratio [HR] = 0.39, 95% confidence interval [CI]: 0.24-0.74). Fakhry et al. 4 evaluated the prognostic value of sex in patients with HNC; in a cohort of patients diagnosed with oropharyngeal cancer (OPC), the mortality risk was lower in women than in men (adjusted HR: 0.55; P = .04). 4
In addition to sex differences in survival, differences in host-related factors have also been evaluated in patients with HNC. Majszyk et al. reported differences in smoking and alcohol consumption among 512 patients with laryngeal carcinoma. The percentages of women and men who smoked were similar (97.1% vs. 98%), and 81.1% of women and 94.6% of men smoked more than 20 cigarettes per day. Heavy alcohol consumption was reported more frequently among men (69.3%) than among women (20.3%). 5
In this study, we assessed sex differences in survival and recurrence and evaluated factors such as treatment, tumor stage, tumor site, and patient habits (eg, smoking and alcohol consumption) in patients with HNC who were treated at the Department of Otorhinolaryngology, Head and Neck Surgery, Medical University of Innsbruck between 2008 and 2019.
Materials and Methods
This retrospective study aimed to evaluate possible sex differences in several host- and disease-related factors in patients with HNC. The study was approved by the Institutional Board of the Ethics Committee of the Medical University Innsbruck. The reference number was 1374/2022.
Inclusion and Exclusion Criteria
All patients registered in the Head and Neck tumor registry were included in this study. The tumor registry consecutively included all patients with histologically confirmed HNCs who were treated as inpatients in our department from January 2008. The exclusion criteria were patients with HNC who were not in an inpatient setting at our department and patients with carcinoma of the eye, thyroid, esophagus, brain, or spinal cord. As well as patients with melanomas or nonmelanoma skin cancers, sarcomas, and lymphomas. Patients with benign tumors, head and neck metastases from distant primary tumors, or precancerous or dysplastic lesions in the head and neck without invasion were also excluded.
Evaluated Factors
All patients suspected of having HNC underwent routine examination, including a CT scan of the head, neck, chest, and abdomen, followed by panendoscopy under anesthesia during diagnostic workup to define the tumor site, TNM classification, and treatment possibilities. The findings were recorded on a tumor evaluation sheet by an investigator. Tumor biopsies were obtained and sent to the Department of Pathology for routine histopathological examination. An additional tumor biopsy specimen was kept in a cell culture medium and sent to the Laboratory for Molecular Biology and Oncology to detect tumor markers such as p16, PD-L1, and EGFR. During panendoscopy, the anesthetist was asked to provide the American Society of Anesthesiologists (ASA) score; this information was also documented on the tumor evaluation sheet. Each patient was then discussed with the interdisciplinary head and neck tumor board, where the recommended treatment modality and remission state (complete tumor regression) after treatment were defined. The waiting time from diagnosis to therapy was 3 weeks for surgical treatment and 6 weeks for radiochemotherapy (RCT) or radiotherapy. Considering a recent extended review, we have the lowest treatment delay for both surgical and non-surgical therapies. 6
The remission state after treatment was performed 10-12 weeks after treatment using our restaging protocol, including clinical examination and an additional CT scan. Clinical follow-up consisted of a clinical examination every 6 weeks for the first 3 follow-ups, every 3 months for the following 3 follow-ups, and then every 6 months until 5 years after diagnosis, a CT scan was performed once a year. Data on remission status and death were regularly updated in the Head and Neck tumor registry by the scientific staff. Data on sex, age, smoking, and alcohol consumption were recorded in the patient files during the first hospital stay for the diagnostic workup. The treating physician asked each patient about the number of pack-years and whether they had discontinued smoking. If the patient discontinued smoking, the year of smoking cessation was recorded. Nurses documented alcohol consumption (≥1 unit per day, <1 unit per day) and the date of alcohol cessation (currently <1 year or cessation >1 year). One unit of alcohol per day was defined as the consumption of 10-12 g of pure alcohol. For example, a small glass of beer or an eighth of wine.
Data Analysis
Data were collected in the Head and Neck Tyrolean Tumor Register and analyzed using SPSS® (IBM Corp. Released 2016. IBM SPSS Statistics for Windows, Version 24.0. Armonk, NY, USA). Frequency data were tabulated and analyzed using Pearson’s Chi-square and Fisher’s exact tests. Statistical significance was set at P < .05. The impact of sex differences on disease-related outcome parameters was investigated using univariate analysis if the outcomes were metric, ordinal logistic regression if the outcomes were categorical, or binary logistic regression if the outcomes were dichotomous. For the combined effects, we utilized a generalized mixed model using Tweedie with log link, the estimation method of Fisher and Wald Chi-square statistics, and identified the main effects of sex, tumor localization, alcohol consumption, alcohol cessation, and their combined effects.
Results
Disease-Related Factors
This study included 1115 patients with HNC, of whom 79.6% (n = 876) were men and 21.4% (n = 239) were women. There were no differences between male and female patients for age at first diagnosis, in univariate analysis the age difference was not related to sex (estimated marginal means: men: 62.55 ± 1.28; women: 64.86 ± 1.25; P = .13)
The most common tumor site in both sexes was the oropharynx (30.8% in men, 32.2% in women). Interestingly, oral (23.8% vs. 14.3%), hypopharyngeal (9.4% vs. 5.4%), and laryngeal carcinomas (25.1% vs. 15.9%) were more common in women than those in men (P < .001). Ordinal logistic regression also revealed a significant relationship between sex and tumor site localization (P = .011). Histologically, the most common tumor was squamous cell carcinoma, followed by adenocarcinoma. There were no significant differences in tumor histology between the male and female patients (P = .996).
Women received primary radiotherapy (RT) (11.4% vs. 6.3%) and surgery only (36.0% vs. 32.3%) more often than men. While surgery with postoperative radiation (15.8% vs. 19.3 %) and surgery with postoperative RCT or radioimmunotherapy (RIT) (6.6% vs. 10.5 %) was more often indicated in men. There was nearly no difference in the application of primary RCT or RIT between women and men (30.3% vs. 31.7%) (P = .027) (Table 1).
Disease-Related Factors.

Abbreviations: PORT, postoperative radiation; RCT, radiochemotherapy; RIT, radioimmunotherapy; RT, radiotherapy.
Host-Related Factors
Data on alcohol consumption, smoking, and therapy compliance were collected as host-related factors. Less alcohol consumption (<1 unit per day) was observed in the female population (24.6% vs. 36.6%) (P = .005); in addition, alcohol cessation > 1 year was more frequent in women than that in men (90.3% vs. 75.2%), (P < .001; binary logistic regression odds ratio [OR], 2.346; 95% CI: 1.24-4.43). Smoking habits were not associated with sex (P = .25). Therapy compliance was approximately 90% for both sexes and did not show any significant difference (P = .45) (Table 2).
Host-Related Factors.

Abbreviation: PY, pack years.
Clinical Factors and the Indirect Effect of Sex
Clinical factors such as tumor regression, death, the Union for International Cancer Control (UICC) stage, and the ASA score may be sex-related. The complete tumor regression to therapy was approximately 76% for both sexes; sex did not have a significant impact on complete tumor regression in the first restaging 10 to 12 weeks after treatment (P = .788 with Wald Chi-square test in binary logistic regression). The survival time was 79.4 months (95% CI: 74.5-84.3) in men and 81.1 months (95% CI: 72.7-88.6 months) in women (Log-rank [Mantel-Cox] test, P = .231).
Although UICC stage IV was the most frequent stage, the proportion of stage IV cancer was higher in men than that in women (54.1% vs. 44.8%); in women, a higher proportion of UICC stages II (14.2% vs. 10.8%) and III (21.3% vs. 15.9%) was observed (P = .04). Ordinal logistic regression also revealed a significant relation of sex with the UICC stage (OR: 1.34; 95% CI: 1.07-1.74; P = .026). This was a complex clinical factor that was significantly sex-associated. We investigated whether sex affects the UICC stage directly or if the UICC stage is influenced by other variables that are sex-associated, such as tumor site or alcohol consumption. Indeed, other factors related to sex, such as tumor site, alcohol consumption, and alcohol cessation, determined the UICC score, but not sex directly. We used a generalized mixed model to validate the main effects of sex, tumor site, alcohol consumption, alcohol cessation, and their combined effects. In this model, sex did not have a significant independent effect on UICC staging (P = .393); however, tumor site (P = .015), alcohol consumption (P < .001), and alcohol cessation (P = .003) had significant effects. ASA classification was not related to sex (Table 3).
Clinical Factors Indirectly Related to Sex.
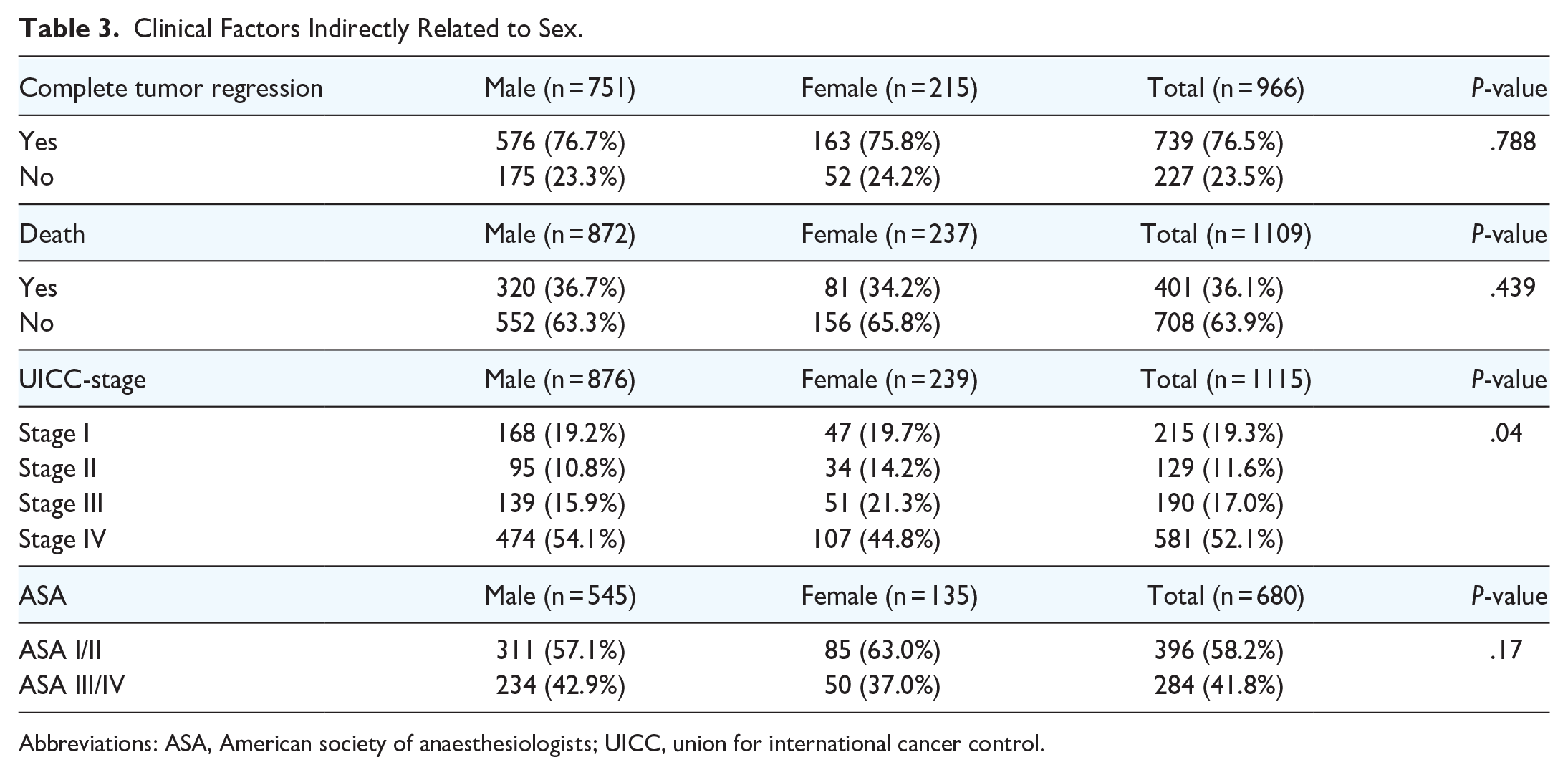
Abbreviations: ASA, American society of anaesthesiologists; UICC, union for international cancer control.
Tumor Markers
Several molecular markers potentially related to carcinoma development, therapy outcome, and survival have also been investigated, including p16, the surrogate marker of HPV, 7 EGFR, and PD-L1. EGFR was positive in 39.2% of the women and 45.6% of the men, and PD-L1 was positive in 53.8% of the women and 48.4% of the men. P16 positivity was detected in 28.2% of women and 23.7% of male patients with HNC (Table 4). These parameter distributions were not significantly affected by sex (EGFR, P = .65; PD-L1, P = .71; p16, P = .19; Wald Chi-square statistics in ordinal logistic regression) (Table 4).
Tumor Markers.

Abbreviations: EGFR, epidermal growth factor receptor; PD-L1, programmed cell death ligand 1.
Number of Newly Diagnosed Patients in Our Department Per Year
The annual number of patients newly diagnosed with HNC since 2008 is summarized in Figure 1. The highest number of female patients with HNC was observed in 2015 (n = 25) and 2016 (n = 28); during this period, the highest number of male patients was also observed (n = 84 in 2015 and n = 85 in 2017). The lowest number of female patients was observed in 2011 and 2018 (n = 13). In the last decade, there has been neither a steady increase nor a steady decrease in the numbers of men and women.

Number of newly diagnosed patients per year.
Discussion
In this study, sex differences in 1115 patients with HNC were evaluated; most patients were from the Tyrol region, Austria, and all patients were Caucasians.
In this study, 78.6% (n = 876) of the patients with HNC were men and 21.4% (n = 239) were women. In the USA, HNC is approximately 2 to 3-fold more common among men than that among women. 8 This ratio varies by geographic region; however, in most countries, men have a 2 to 5-fold greater risk of HNC than women. In our region, the number of male patients was 3.7 times higher than the number of female patients, which is within the above-mentioned range. The authors attributed this finding to higher rates of alcohol and tobacco use among men. 9 Smoking is a strong risk factor for HNC in both men and women. 10 Decreased smoking-related mortality in men and increased mortality in women have also been reported in Western countries. The Swedish population was screened over 25 years for smoking-related mortality, based on lung carcinoma mortality between 1997 and 2016. The authors described a sex gap in life expectancy of 5.0 years in 1997 and 3.4 years in 2016. 11 We did not observe sex differences in our study group for smoking; however, fewer women than men reported drinking alcohol (P = .005), and alcohol cessation for >1 year was more frequent in women (P = .001).
In previous studies, men were more likely to be drinkers than women, and men drank more heavily than women. 12 However, the sex gap is narrowing. 13 In a meta-analysis, Slade et al. 14 described a linear decrease in the sex ratio over time. Among the cohort born in the early 1900s, men were 2.2 (95% CI: 1.9-2.5) times more likely than women to consume alcohol and among the cohort born in the late 1900s, men were 1.1 (95% CI: 1.1-1.2) times more likely to consume alcohol. Williams et al. 15 reported an increase in the frequency of heavy drinking among younger Caucasian and Hispanic women, in contrast to a decline in heavy drinking among younger men. The authors suggested that the increased frequency of heavy drinking among younger white and Hispanic women was connected with changes in women’s social roles and norms in recent decades. In Tyrol, both alcohol consumption and cessation differences were moderate but significant.
Another risk factor for HNC, particularly for the development of an OPC, is HPV, which was detected in this study by p16 immunohistochemistry (IHC). No significant differences in p16 IHC detection were observed between male and female patients.
Additional important clinical variables that could have been indirectly sex-related were also considered. Nevertheless, we did not observe differences in complete tumor regression (P = .785) and survival time (79.4 vs. 81.1 months). In previous studies, better survival in female patients was described for distinct subtypes of OPC 4 and HNC in general.2,16 However, Roberts et al. 17 described a prospective epidemiologic study on 286 female and 286 matched male patients with HNC and found no significant differences between women and men in terms of recurrence-free, disease-specific, and overall survival. When the analysis was restricted to individual sites (oral cavity, oropharynx, or larynx/hypopharynx), there was no evidence of a sex-associated disparity in survival associated with sex. The lack of differences between male and female patients in complete tumor regression after therapy and survival might also be related to the lowest possible time delay between tumor diagnosis and the start of therapy, in particular, 3 weeks in the case of surgical treatment and 6 weeks in the case of RCT or radiotherapy. These time delays were considered the lowest based on a recent extended review article by Hanna et al. 6
Another important finding was the higher proportion of UICC stages II and III and the lower proportion of UICC stage IV in women (P = .04), as well as differences in therapy (P = .027). Women underwent surgery or radiotherapy alone more often, which may be related to the lower proportion of female patients with UICC stage IV disease. The UICC stage differences in patients with HNC were related to the tumor site, alcohol consumption, and alcohol cessation, which are direct sex-related variables; however, UICC staging was not directly affected by sex in our analysis.
Park et al. 18 described in their study with 884 patients with locoregionally advanced HNC that women were less likely to receive cisplatin concurrently with radiation (adjusted OR: 0.69; 95% CI: 0.48-0.99) and women had also a lower odd of receiving any radiation (adjusted OR: 0.79; 95% CI: 0.56-1.12). The authors described women in their study as undertreated and potentially missing the opportunity for adequate therapy. Similar sex disparities in therapy have also been reported for other cancer types, including bladder, pancreatic, kidney, and colon cancers, as well as lymphomas.19-21
As aforementioned, tumor site was associated with sex in patients with HNC in our study. In Tyrol, a higher proportion of oral carcinomas was observed in the female population, and a higher proportion of hypopharyngeal and laryngeal carcinomas was observed in the male population (P < .001). As a certain proportion of patients with oral carcinoma were also treated in other departments, such as the Department of Maxillofacial Surgery, this result is not representative. Kruse et al. 22 described a higher proportion of women with oral carcinoma, particularly in the population without the risk factors of alcohol and tobacco use and in patients older than 70 years. In a case-control study of 250 women with oral carcinoma and a control group of 996 age-matched female participants without oral carcinoma, the influence of education and oral hygiene on diagnosis was evaluated. Wearing dentures had an increased risk in less educated women only (OR: 2.23; 95% CI: 1.14-4.34), whereas an inverse relation to the risk of carcinoma was described for tooth-brushing twice a day or more in women with high school education or above (OR: 0.50; 95% CI: 0.25-0.98). Furthermore, oral ulceration and loss of more than 5 teeth are associated with an increased risk of oral carcinoma. The authors concluded that oral hygiene may be a risk factor for oral carcinoma in women; however, this influence can be modified by educational standards. 23
In this study, no differences were found between men and women in the number of newly diagnosed patients in our department per year. In a retrospective study by the U.S. National Cancer Institute’s Surveillance, Epidemiology, and End Results, a decreased HNC incidence of 0.29% per year for men and 0.28% for women was observed in 149.301 patients with HNC between 2002 and 2012. However, in patients with OPC, an increased rate of 2.89% per year was observed in men and 0.57% in women. 24
The limitations of this study are that, as reported previously, not all patients with HNC of the Tyrol region were treated at our department; a small portion of patients were also treated in a baseline hospital, and some patients with oral cancer were treated at the Department of Maxillofacial Surgery and therefore not included in our tumor registry. Furthermore, the number of female patients was lower than the number of male patients (239 vs. 876), especially in patients with hypopharyngeal cancer, the number of female patients was low (13 vs. 82). This may have influenced the results. Furthermore, in this study, a large patient population with different histotypes was analyzed, including all patients with HNCs.
This study lacks generalizability due to the limited multicultural representation in our population.
Conclusions
Several sex-related differences were observed in patients with HNC in this study. Female patients with HNC were associated with a higher proportion of oral carcinoma, lower UICC for the International Cancer Control stage, therapy with a single treatment modality, and lower alcohol consumption than those in men. There was no significant difference in the number of newly diagnosed patients per year in our department. This study supports the significance of sex-related differences in patients with carcinoma of head and neck.
Footnotes
Acknowledgements
We thank Maria Anegg for collecting the data from our Head and Neck tumor registry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent Statements
The study was approved by the Institutional Board of the Ethics Committee of the Medical University Innsbruck. The reference number was 1374/2022.
