Abstract
Importance
In patients with head and neck squamous cell carcinoma (HNSCC), the discovery of a second synchronous primary cancer of the aerodigestive tract (SSPCA) significantly impacts management and prognosis. Recent advances in imaging have increasingly allowed for identifying SSPCA before performing panendoscopy, raising questions about the latter’s role.
Objective
To establish the incidence of SSPCA and panendoscopy’s impact on management. Complications and costs associated with panendoscopy were also assessed.
Design
Systematic review following the preferred reporting items for systematic reviews and meta-analysis guidelines.
Setting
Operating room panendoscopy.
Participants
Identifiable HNSCC undergoing initial staging workup.
Intervention
Panendoscopy under general anesthesia for SSPCA detection.
Main Outcome Measures
Incidence of SSPCA in HNSCC, change in management caused by panendoscopy, incidence of panendoscopy complications, costs for panendoscopy.
Results
51 studies were included (n = 19,914 patients). SSPCA was present in 6.4% (n = 467/7262) of all panendoscopies. Among patients who had a prior computed tomography (CT) of the neck and chest, a change in management resulting from SSPCA detected through panendoscopy occurred in only 1.1% of cases (n = 3/268), and in 0% of cases for those who had a positron-emission tomography-computed tomography (PET) (n = 0/544). The rate of major complications of panendoscopy was 0.7% (n = 58/8386). Only two recent studies in a private healthcare system reported panendoscopy costs ranging from $3802 USD to $17,296 USD.
Conclusions
The role of panendoscopy in the initial workup of HNSCC should be limited to confirming suspicious findings from initial CT or PET. The incidence of major complications for panendoscopy is low but carries a significant financial burden for patients in the private American healthcare system. More studies are needed to assess the cost-effectiveness of panendoscopies for SSPCA detection in a public healthcare system.
Relevance
Confirms the lack of benefit for systematic panendoscopy for SSPCA detection in HNSCC patients when initial workup includes a CT of the neck and chest or PET.

Keywords
Introduction
Panendoscopy, also known as triple endoscopy, refers to the use of direct laryngoscopy, esophagoscopy and bronchoscopy for the workup of patients with head and neck squamous cell carcinoma (HNSCC). Mostly performed under general anesthesia, panendoscopy is used to characterize the extension of the primary tumor and to detect a potential synchronous second primary cancer of the aerodigestive tract (SSPCA). The relevance of investigating synchronous mucosal cancers arises from the concept of field cancerization, which entails chronic exposure of surface mucosa by carcinogens such as tobacco and alcohol, resulting in multifocal cellular changes predisposing a patient to neoplastic transformation. 1
By definition, a synchronous second primary cancer is detected within 6 months of the diagnosis of another primary cancer. On the other hand, tumors detected more than 6 months after the primary malignancy are considered metachronous. 2 The finding of mucosal SSPCA dramatically and negatively alters a patient’s prognosis, potentially impacting decision making, surgical approaches, postoperative morbidity and therapeutic deintensification strategies. Conversely, financial burdens felt by the healthcare system create the need for diagnostic yield efficiency. 3 Thus, a balanced approach should be adopted.
The reported incidence of SSPCA in the literature is variable, ranging from 0% to 18%.1,4–8 Contemporary initial workup of HNSCC includes a complete history and physical exam, in-office flexible endoscopy and precision imaging (CT of neck and chest and/or PET). Since these investigations often detect SSPCAs before performing panendoscopy, 9 several clinicians have questioned the efficiency of the procedure given its procedural and anesthetic risks, potential treatment delays and costs.1,10–13 Thus, we sought to conduct a systematic review with the aim of elucidating the incidence of panendoscopy-detected SSPCA and its effect on management as well as the procedure’s complication rate and cost.
Methods
We conducted a systematic review in accordance with the 2020 Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA).
Included studies
To ensure that all relevant publications were included, any study that discussed panendoscopy in the context of HNSCC was analyzed. We included all studies that reported the incidence of SSPCAs on panendoscopy as well as complications related to the procedure. Either direct laryngoscopy, esophagoscopy, bronchoscopy or a combination of these had to be performed for the detection of SSPCA to be included. Endoscopic procedures were to be performed in an operative room setting under general anesthesia or sedation. Any article that addressed the costs associated with panendoscopy was included. French- and English-language publications were included.
Excluded studies
We excluded studies with endoscopies performed for unknown primary cancers, non-HNSCCs, or for follow-up of a previous HNSCC. We have also excluded procedures performed under local anesthesia, in the office setting or with special tumor detection protocols such as narrowband imaging or staining. Studies with multiple diagnostic modalities that did not stratify findings specific to panendoscopy were excluded. We excluded abstracts, case reports, reviews, letters to the editor, scientific posters, podium presentations, animal studies, and non–peer-reviewed publications.
Search Strategy
An optimized search strategy within Medline, Embase, EBM, and Google Scholar between 1932 and 2022 was employed (see Supplemental Appendix 1). Bibliographies of the included articles were also individually revised for other potential studies meeting inclusion criteria. This protocol was registered in PROSPERO (CRD42023408810).
The first screening of studies was conducted by two main independent reviewers (S.B., M.K.), based on the previously mentioned inclusion criteria. In the advent of a disagreement, resolution was ensured by a third independent reviewer (P.T.). An identical process was applied to full-text screening of included abstracts for their eligibility for data extraction.
Data Extraction and Analysis
The following data was collected from all included studies: Study design, year of publication, number of participants, age, gender, location, and TNM stage of primary and secondary tumors, types of endoscopy used, types of esophagoscopes used (flexible vs rigid), preoperative imaging used, incidence of panendoscopy-detected SSPCA, change in management due to panendoscopy-detected SSPCA, panendoscopy-related complications and costs.
Qualitative Appraisal of Studies
For qualitative appraisal of included studies, the National Institutes of Health Quality Assessment Tool for Case Series Studies, a 9-question scale was used. Given the absence of patient follow-up and statistical tests (all collected data were descriptive), questions 7 and 8 were omitted; the critical appraisal score was therefore out of 7. Scores of 6 or 7 were considered “good” quality studies, a score of 5 was considered “fair” and a score of 4 or less was considered “poor.”
Synthesis
Statistical analysis was conducted using Microsoft Excel® 2208 version. SSPCA and complication incidences in all studies were pooled and compared according to primary tumor subsite and type of endoscope used. Procedural costs were also compared.
Results
Figure 1 illustrates the article selection process of our review. An inclusion disagreement between the two main reviewers (S.B., M.K.) was noted in 32 of 521 screened abstracts, all of which were resolved by a third independent reviewer (PT).
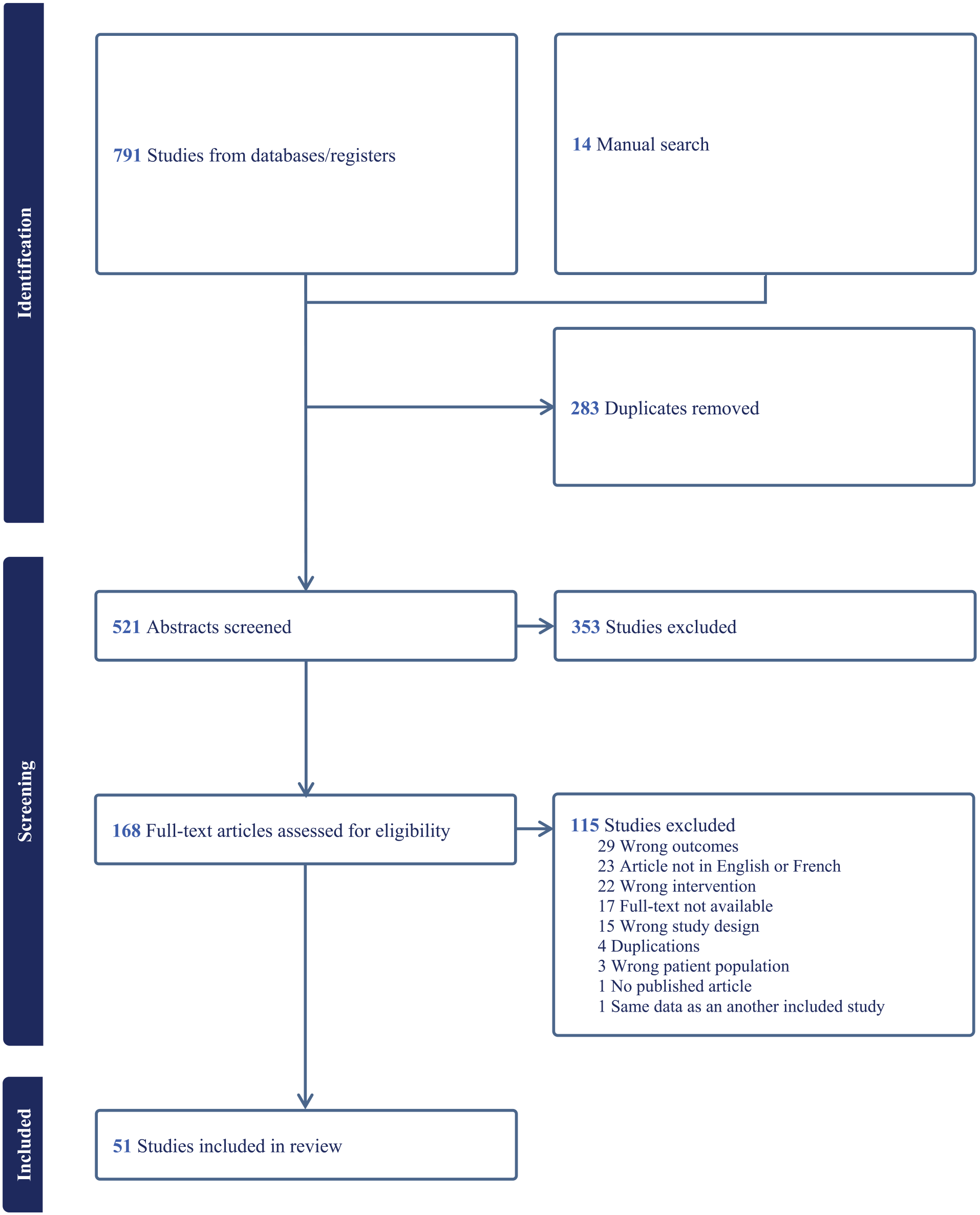
PRISMA flow chart of the selection process.
Fifty-one studies were included in our review, 13 (25%) of which were prospective and 48 (94%) of “good” quality. None of the studies showed a “poor” quality rating. Forty-eight studies described SSPCA seen on panendoscopy (n = 15,929); 24 studies described panendoscopy-related complications (n = 8386) and 5 studies described costs. Study characteristics, type of endoscopy, and pre-endoscopic imaging as well as the rate of endoscopy-discovered SSPCA are shown in Table 1. Table 2 presents the characteristics of the included studies used to determine the incidence of complications with panendoscopy. Studies mentioning panendoscopy costs are described in Table 6 and will be discussed later. Data on age, sex distribution, and TNM stage of primary tumors were insufficient in most studies and are not reported here. We found some heterogeneity between studies for the inclusion or exclusion of in situ SSPCAs. When possible, in situ SSPCAs were included in our review.
Studies Included for Incidence of SSPCA of the Aerodigestive Tract.

Sources: References 1, 2, 4–6, 8–48.
Abbreviations: B, bronchoscopy; BS, barium-swallow; CT, CT-scan; CXR, chest X-ray; E, esophagoscopy; L, laryngoscopy; MRI, magnetic resonance imaging; NS, not specified; Re, retrospective; P, panendoscopy; PET, positron-emission tomography scan; Pr, prospective; SSPCA, second synchronous primary cancer of the aerodigestive tract; US, ultrasound.
Studies Included for Panendoscopy Complications.

Sources: References 5–8, 13–17, 22–26, 32–34, 39, 41, 45, 48–51.
Abbreviations: B, bronchoscopy; E, esophagoscopy; L, laryngoscopy; NS, not specified; P, panendoscopy; Pr, prospective; Re, retrospective.
PRISMA flow chart of the selection process. The incidence of SSPCAs according to the primary tumor subsite is presented in Table 3 (whether found by imaging or endoscopy). In this table, no distinction is made between SSPCAs initially detected by imaging and confirmed by endoscopy, and those specifically detected by endoscopy after being missed by imaging. To address this issue, we further stratified these data for SSPCAs specifically detected only by endoscopy based on the type of imaging used during the initial workup of HNSCC. When this included a neck and chest CT, 2.2% of patients had additional SSPCAs (n = 33/1518) found on panendoscopy only. Conversely, if a PET was used, only 0.5% of patients had additional SSPCAs (n = 3/639) discovered by panendoscopy. While panendoscopy may uncover some SSPCAs missed by imaging, these findings do not always alter management. We summarized in Table 4 the management changes resulting from the discovery of additional SSPCAs on endoscopy.
Incidence of SSPCA Detected by Combined Imaging and Endoscopy, Stratified by Primary Cancer Subsite and Endoscopy Type.

Abbreviation: SSPCA, second synchronous primary cancer of the aerodigestive tract.
Change in Management by Endoscopy According to Pre-Endoscopic Imaging Used.

Abbreviation: PET-scan, positron-emission tomography scan.
Table 5 presents the overall complications of panendoscopy. Specifically for esophageal perforations, we also investigated whether they occurred more frequently with rigid or flexible esophagoscopes. The rate of esophageal perforation during esophagoscopy for HNSCC patients stands at 0.1% with the rigid (n=2/1,937) and 0% with the flexible esophagoscope (n=0/891). When including studies where the type of esophagoscope used was not specified, the rate of esophageal perforation is at 0.1% (n=4/3,705).
Panendoscopy Complications.

Abbreviations: ETT, Endotracheal tube; TMJ, Temporomandibular joint.
Studies describing panendoscopy costs are shown in Table 6.
Panendoscopy Costs.

Abbreviation: OR, operating room.
Discussion
The initial workup of a patient with an HNSCC often detects SSPCA prior to performing panendoscopy. Furthermore, this added modality entails procedural and anesthetic risks, subsequent treatment delays and increased costs. Several clinicians are questioning whether or not it has become obsolete when it is solely used to discover incidental SSPCA. Thus, we conducted a systematic review with the aim of elucidating the incidence of SSPCA, complications and costs related to the use of a panendoscopy for patients with HNSCC.
Incidence of SSPCA
Our review found diverging conclusions for the added benefit of panendoscopy for SSPCA detection. This is due to the discrepancy in reported SSPCA incidence across studies, ranging from 0% to 18%.1,4–8 A pooled analysis of all the included studies revealed an overall detection rate of 6.4% (n = 7262) for SSPCA when the initial workup included a panendoscopy. A previous systematic review completed in 1992 by Haughey et al. revealed a similar incidence of 4% (n = 1677). 52
Given the advent of improved imaging modalities, panendoscopy might not be providing as much of an added screening benefit as it once did. Previously, pre-endoscopic imaging consisted of a chest X-ray with or without barium-swallow, which has low sensitivity and specificity.53,54 Nowadays, pre-endoscopic imaging includes a high-resolution CT of the neck and chest and/or a PET. As shown by our pooled analysis, a PET was able to detect 99.5% of SSPCAs, missing only 3 lesions. When comparing PET to panendoscopy, Hearle et al. showed a detection rate for SSPCA of 6.1% versus 4.5%, respectively. 9 Furthermore, the sensitivity of PET and panendoscopy were 100% and 74%, respectively, reinforcing the superiority of this imaging modality compared to panendoscopy for SSPCA detection. Moreover, of the 5 additional SSPCA revealed by the PET, 4 were in the periphery of the bronchial system, which cannot be visualized during the panendoscopy.
Another concept to consider when assessing the utility of a screening exam is its impact on patient management. Our analysis showed that only 1.1% of patients with preoperative CT had a change in their management following panendoscopy. Furthermore, this number drops to 0% if a PET was done prior to panendoscopy.
SSPCA incidence was also stratified according to the primary HNSCC subsite as well as the endoscopic modality used. The overall incidence of endoscopy-detected SSPCA ranged from 3.3% for primary hypopharyngeal cancer to 9.0% for primary oropharyngeal cancer. However, the incidence of SSPCA for primary oropharyngeal cancers does not stratify for HPV p16 status. Given the high prevalence of risk factors such as smoking tobacco and alcohol which lead to field cancerization in the p16 negative group, we hypothesize that these patients may have a higher incidence of SSPCA when compared to their p16 positive counterparts. In parallel, Xu et al. observed a 2.8% incidence of SSPCA discovered by PET and panendoscopy for oropharyngeal p16 positive patients and 10.2% for p16 negative patients (P = .02). 55 However, the authors did not identify whether SSCPA was specifically detected by PET or panendoscopy. More studies are needed to clarify the role of panendoscopy in the detection of SSPCA in primary oropharyngeal cancers according to their HPV p16 status.
Panendoscopy Complications
As with every surgical procedure, a risk/benefit analysis must be considered. With a major complication rate of 0.7%, we consider that panendoscopy has a low prevalence of serious adverse events. Given the nature of the included articles, stratification of complication rate according to the primary tumor’s subsite, TNM stage and patient’s comorbidities was not feasible. Future studies analyzing predisposing risk factors leading to a heightened risk of adverse events are warranted to improve pre-procedural counseling.
One of the most significant complications during panendoscopy is respiratory distress requiring unplanned tracheostomy. Eissner et al. revealed that this procedure was required in 1.6% of all panendoscopies in the context of HNSCC (32/2059). 49 In their patient series, all unplanned tracheostomies occurred in patients with primary oropharyngeal, hypopharyngeal or laryngeal HNSCC. The 32 patients with unplanned tracheostomy were compared with 180 control patients without tracheostomy. Patients’ characteristics were compared in terms of sex, age, tumor location/staging, blood clotting disorder, body mass index, pulmonary disease and radiotherapy exposure. They revealed that the only statistically significant risk for unplanned tracheostomies was a primary laryngeal tumor and an elevated partial thromboplastin time (PTT). Hence, pre-endoscopic consent of these patients should include the disclosure of their increased risk of tracheostomy to ensure informed consent.
For esophageal perforations during panendoscopy, both flexible and rigid esophagoscopy groups reported low incidence with respectively 0 (n = 891) and 2 (n = 1937) perforations (P = .34). However, certain patients with limited cervical extension resulting from conditions such as cervical spondylarthrosis or obesity may not be suitable for rigid esophagoscopy.7,50 Also, out of the 5 included studies that reported dental trauma (12/1125 endoscopies), only McGarey et al. reported dental trauma specific to esophagoscopy during panendoscopy, which occurred in 4 out of 551 (0.7%) rigid esophagoscopies.7,14–16,56 As no data on the difference in efficacy of detection of SSPCA by rigid versus flexible oesophagoscopy exists, we encourage clinicians to use the modality they are most comfortable with to decrease operator-dependent complications.
Panendoscopy costs
With mounting systemic healthcare pressures, it is imperative to conduct cost-benefit analyses on procedures to avoid unnecessary spending. This is especially true for a publicly funded system. In this review, 5 studies analyzed costs related to panendoscopy and were all conducted in the United States, thus reflecting the costs associated with a private healthcare system. Muller et al. reports that the average panendoscopy fee in their hospital is $17,296 USD. Commercial insurance could reimburse up to $29,854 USD while Medicare/Medicaid would reimburse up to $6430 USD. 1 This contrasts with Sawaf et al. which yields much lower costs ($3802 USD), although both studies don’t specify the exact procedures included in their fees. 17 Given the important costs HNSCC patients face and the known association between HNSCC and low socioeconomic status, 57 the addition of panendoscopy fees for patients lacking medical insurance can considerably affect their financial situation and possibly deter them from undergoing eventual treatment.
Understanding the financial burden of panendoscopy in the context of a public health care system was impossible given the paucity of data in the literature. Given a public health system is regulated by a budget, particular attention is paid to the quality of life impact of certain procedures, with measurements often reported in terms of Quality-Adjusted Life Years. 3 Moreover some hospitals lack easy access to the operating room and are constrained by significant delays to perform certain procedures. Thus, the addition of panendoscopy to the initial workup of HNSCC does not only imply significant costs, but may also impose delays in initiating treatment and may clog the surgical waiting list. Therefore, studies are needed to assess the financial and organizational impact of panendoscopy on a public health system.
Clinical Application
Here, we question the systematic use of panendoscopy for detecting SSPCAs in the initial workup of HNSCC. Our research reveals that only 1.1% of patients undergoing a pre-panendoscopy CT of the neck and chest see a change in their management with panendoscopy. Hence, the potential complications and additional delays associated with panendoscopy for initiating primary cancer treatment may outweigh the benefits of detecting a rare subclinical SSPCA. Furthermore, for those who have had a PET, there does not appear to be any added benefit to support the inclusion of a panendoscopy to detect subclinical SSPCAs in the initial investigation of HNSCC. Therefore, we advocate against routine panendoscopy in HNSCC’s initial workup. Instead, we propose limiting its use to three key indications: (1) investigating an unknown primary HNSCC, (2) assessing the extent of a primary HNSCC for future surgery, and (3) confirming suspicious findings on initial imaging workup of HNSCC.
Limitations
We are aware that this review has limitations. First, several studies could not be included because of a lack of stratification, for example, studies reporting the combined incidence of synchronous and metachronous cancers, inclusion of unknown primary HNSCC patients and the combined incidence of SSPCA detected by radiology and panendoscopy. In addition, some studies did not stratify or describe the type of imaging done at the initial workup. Correlation between the TNM stage and the incidence of SSPCA was unfortunately inconstant and under-reported as well. For panendoscopic complications, almost no study specified which specific type of endoscopy had caused the complication (eg, dental trauma). Finally, cost studies of panendoscopy in a public health system setting are absent from the literature, thus no conclusion on its financial burden can be made.
Conclusion
The addition of panendoscopy for SSPCA detection in the initial investigation of HNSCC rarely changes the management of patients who have undergone a CT of the neck and chest prior to panendoscopy. Furthermore, change in management is null if a PET was done before panendoscopy. Therefore, we discourage routine panendoscopy for subclinical SSPCA detection in identifiable HNSCC cases. We believe panendoscopy should be reserved for confirming suspicious findings from initial CT or PET. While panendoscopy carries a low incidence of major complications, its cost can be significant in private American healthcare settings without adequate insurance coverage. In public healthcare systems with limited financial resources, further studies are needed to define the cost-effectiveness of panendoscopies for SSPCA detection.
Supplemental Material
sj-docx-1-ohn-10.1177_19160216251316215 – Supplemental material for Panendoscopy for Head and Neck Cancers: Detection of Synchronous Second Primary Cancers, Complications and Cost-Benefit Analysis: A Systematic Review
Supplemental material, sj-docx-1-ohn-10.1177_19160216251316215 for Panendoscopy for Head and Neck Cancers: Detection of Synchronous Second Primary Cancers, Complications and Cost-Benefit Analysis: A Systematic Review by Samuel Bellavance, Michel Khoury, Eric Bissada, Tareck Ayad, Apostolos Christopoulos, Jean-Claude Tabet, Louis Guertin and Paul Tabet in Journal of Otolaryngology - Head & Neck Surgery
Footnotes
Author Note
This study was presented as podium presentation at the Canadian Society of Otolaryngology-Head & Neck Surgery 2024 Annual Meeting, Montreal, Canada, in June 2024.
Author Contributions
Samuel Bellavance: Conceptualization, data curation, formal analysis, methodology, investigation, data curation, writing-original draft, writing-review & editing, visualization, supervision, project administration. Michel Khoury: Conceptualization, methodology, investigation, data curation, writing-original draft, writing-review & editing. Eric Bissada: Writing-review & editing. Tareck Ayad: Writing-review & editing. Apostolos Christopoulos: Writing-review & editing. Jean-Claude Tabet: Writing-review & editing. Louis Guertin: Writing-review & editing. Paul Tabet: Conceptualization, methodology, investigation, writing-original draft, writing-review & editing, visualization, supervision, project administration.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Additional supporting information is available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
