Abstract
Background
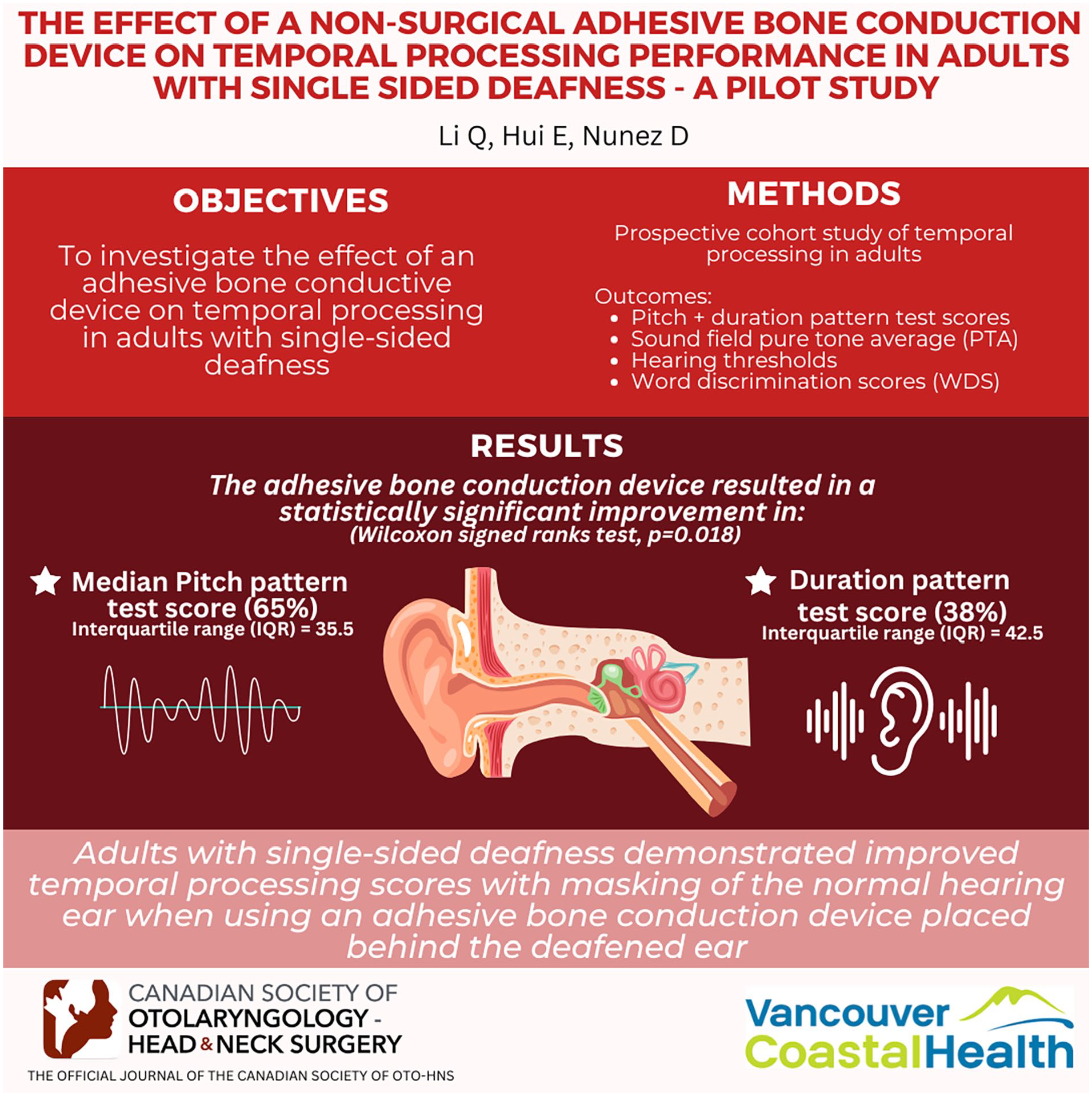
There is a dearth of information on the effects of bone conductive devices on temporal processing in individuals with single-sided deafness (SSD). This study investigates the effect of an adhesive bone conductive device on temporal processing in adults with SSD.
Methods
A prospective cohort study of temporal processing in adults with SSD was undertaken. Outcome measures were the pitch pattern test (PPT) and the duration pattern test (DPT) scores, sound field pure tone audiometric (PTA) averaged hearing thresholds, and word recognition scores (WRS). These were measured under 3 conditions: unaided in quiet, unaided in noise, and aided in noise. Eight SSD novice hearing amplification users were recruited.
Results
The adhesive bone conduction device (BCD) resulted in a statistically-significant improvement (Wilcoxon signed ranks test, P = .018) in median [interquartile range (IQR)] PPT score, 65% (IQR = 35.5), and DPT score, 38% (IQR = 42.5) in noise.
Conclusions
Adults with SSD demonstrated improved temporal processing scores with masking of the normal-hearing ear when using an adhesive BCD placed behind the deafened ear.
Background
A bone conduction hearing device transfers sounds through the skull to the inner ear. Therefore, bone conduction devices (BCDs) were initially introduced to provide a hearing solution for patients with conductive hearing losses (CHL) and normal inner ear function. These devices are also used to manage single-sided deafness (SSD). SSD is defined as a severe-to-profound unilateral hearing loss resulting in an unaidable ear. 1 BCDs are believed to work in individuals with SSD because they overcome the head shadow effect by transmitting sound incident on the side of head with the poorer hearing ear through the skull to the contralateral ear. Cross-skull attenuation of a bone conduction stimulus while often assumed in clinical practice to be 0 dB for speech frequency sound varies across individuals and frequencies and can be as high as 8.6 dB for specific frequencies. However, cross-skull attenuation does not explain the variation in benefit from BCDs in SSD. 2 The development of more powerful BCDs has allowed individuals with purely CHL and those with mixed hearing losses associated with elevated bone conduction thresholds, to also benefit from BCDs.
BCDs are broadly categorized into 2 types based on the transmission pathway of the sound transducer output to the cochlea, namely, through intact skin (skin drive) devices or bypassing the skin (direct drive) devices. 3 Skin-drive devices consist of the classical noninvasive device in which the sound transducer is fixed by a closely-applied external soft head band to the scalp, or newer invasive and noninvasive fixation options. Current invasive options also labeled as passive transcutaneous devices fix the externally-applied sound transducer by magnetic coupling to a metallic component implanted into the skull below the skin, that is, Sophono® (Medtronic, Dublin, Ireland) and Baha Attract® (Cochlear Ltd., Sydney, Australia). A noninvasive fixation solution is provided by Adhear® (MED-EL Inc, Innsbruck, Austria) whereby the sound transducer is fixed to the scalp via an adhesive pad. Direct-drive BCDs are all invasive, and the earliest solution delivered the output of an externally-positioned transducer through a rigid metal percutaneous abutment fixed to a titanium screw Osseo-integrated into the skull as in the Oticon Ponto® (Oticon LLC, Somerset, NJ, USA). Newer direct-drive solutions implant the sound transducer under the skin and into the skull bone. These implants that are also known as active transcutaneous BCDs include the Bonebridge® (MED-EL Inc) and the Osia® (Cochlear Ltd.). Electromagnetic sound information is transferred trans-cutaneously through the skin from an external processer coupled magnetically to an implanted package, that is, relayed to the transducer. 3
A noninvasive skin-drive device (BCD attached with a headband) is suitable for individuals in whom an invasive BCD is contraindicated: for example, individuals with thin skull bones such as young children or those undergoing serial radiological imaging to monitor an intracranial lesion where image quality would be degraded by the implant. A skin-drive trial is also a gold standard for preoperative assessment of candidacy for an implanted BCD. It can be fitted as soon as the hearing loss has been diagnosed, reducing prolonged auditory deprivation. Skin-drive devices provide consistent improved speech reception and speech discrimination, especially in noisy environments for individuals with mimicked hearing loss. 4 Additionally, skin-drive devices improve sound localization abilities but aided thresholds were unsatisfactory. 5 Park et al 4 showed that hearing thresholds and speech recognition in noise using 2 commercially-available BCDs were significantly improved compared with unaided conditions in 30 participants with simulated CHL. Although the tightness of the head band plays an important role in skin-drive BCD effectiveness, using a tight head band is uncomfortable. 6
The FDA and Health Canada recently approved a new type of skin-drive BCD, Adhear (MED-EL Inc). The device consists of a sound processor and an adhesive adapter pad. The adhesive adapter is placed onto the skin behind the ear, and the sound processor is simply clipped on and off of a snap coupling on the lateral surface of the adhesive adapter pad. 6 The sound processor amplifies and transduces sound into vibrations that are transmitted via the adhesive adaptor pad through skin to the underlying skull bone, and subsequently to the cochlea. This device overcomes the cosmetic disadvantage of obtrusive headbands and the discomfort issue. 6 It also provides a solution for patients who are reluctant to undergo surgery or who wish to have a trial before undergoing implant surgery. A number of studies have shown that the adhesive BCD is a valid treatment for unilateral and bilateral CHL patients. Dahm et al 7 reported significant improvements in word and sentence recognitions in quiet and noisy situations, sound quality, and quality of life (Assessment of Quality of Life-8 Dimensions questionnaire, AQol-8D) in 12 participants with CHL with the adhesive BCD. Similarly, Mertens et al 8 found that 70% of participants with SSD reported that the adhesive BCD was partially useful or better than contralateral routing of signal (CROS) hearing aids. There was however no noticeable improvement in speech perception in noise.
A common complaint of BCD and traditional air conduction hearing aid users is that even with their devices they continue to experience difficulty understanding speech in background noise. Studies have shown that speech-in-noise intelligibility is strongly related to auditory temporal processing capacity for both normal-hearing and hearing-impaired listeners. 9 Identification of speech consonants relies largely on rapid temporal cues. 10 In English, voiced consonants can be differentiated from the unvoiced consonants based on the difference in the duration of vowels sounds preceding the consonant. The impacts of traditional air conduction hearing aids on temporal processing have been widely investigated 9 ; however, similar studies of BCDs are rare. Bance et al 11 compared duration and gap discrimination in 13 CHL participants with direct-drive BCDs and conventional hearing aids. These groups’ responses were compared with those of normal-hearing control participants. The authors concluded that BCDs and conventional hearing aids achieved comparable levels of temporal processing function in CHL participants.
There are limited clinical data, and no published studies assess temporal processing function in participants with hearing loss using an adhesive BCD, to the best of our knowledge. Hence, the primary aim of this study was to determine the effect of the adhesive BCD on temporal processing function in adults with SSD under 3 test conditions: (1) unaided in quiet; (2) unaided in noise; and (3) aided in noise. Secondarily, its effect on free field pure tone average (PTA) audiometric hearing thresholds and word recognition scores (WRS) was observed in noise.
Methods
This prospective single-subject repeated-measure study with each participant serving as their own control was approved by the institutional ethics committee. Participants were evaluated in a single session with a minimum of 2 breaks. All subjects were tested with a commercially-available adhesive BCD Adhear (MED-EL Inc) in the neuro-otology clinic at Vancouver General Hospital.
Participants
Individuals 18 years and older with SSD who had not previously used hearing amplification were recruited. SSD was defined as a severe-to-profound unilateral hearing loss resulting in an unaidable ear. 1
Eight adult English first language speakers participated in the study (3 males and 5 females; mean age = 34.4 years, standard deviation = 11.4 years, range = 19-55 years). Table 1 summarizes the participants’ demographic information and otological findings.
Participants Demographic Information and Otological Findings.

Abbreviations: F, female; M, male; SSD, single-sided deafness.
Test Environment
All tests were performed in a sound-treated booth. A sound level meter was used to identify a position in the booth where the sound pressure level originating from speakers positioned at 1 m distance to the right and left at 45° azimuth was equal. This position was used for all tests. At least 1 training round was undertaken to familiarize participants with the tests before collecting test responses. The practice round consisted of 5 test items for each ear, totaling 10 practice testing items (testing round approximately a duration of 10 minutes). A GSI 61 audiometer with an attached compact disc (CD) player was used for audiometric and temporal processing assessments.
Temporal Processing Assessment
The duration pattern test (DPT) and the pitch pattern test (PPT) were adopted for temporal processing assessment. Commercially-available recorded CDs (Auditec Inc, St. Louis, MO, USA) were used as described by Musiek et al. 12 DPT and PPT tones were presented at 50 dB. In the DPT, participants were presented with three 1 kHz tones in succession that were separated by a 300 ms silent interval. One of the tones in each sequence differed from the other 2 by being either of long duration (500 ms) or of short duration (250 ms). Participants were asked to report the order and duration of the 3 tones. There were 6 possible permutations. 12 Similarly, in the PPT, participants were presented with 3 tones in succession. Each tone was 200 ms in duration, and one of the tones in each sequence differed from the other 2 by being either of a high pitch (1122 Hz) or of a low pitch (880 Hz). The tones were separated by a silent interval of 150 ms. Participants were asked to report the order and pitch of the 3 tones that they heard, for example, “high, high, low.” There were 6 possible permutations. Thirty runs, each of the DPT and PPT, were assessed under each set of test conditions.
Sound Field Pure Tone and Speech Hearing Assessment
Pure tones were presented through a speaker facing the participant’s poorer hearing ear at the same time as 50 dB masking noise was presented through an insert earphone to the better hearing ear to record Sound field (PTA4) 13 responses. Similarly, in speech tests, 50 dB masking noise was applied to the participants’ better hearing ears while word lists were presented to the poorer hearing ear. Participants were asked to repeat the words they had heard and the percentage of words correctly identified by each participant was calculated as the WRS.
Test Conditions
The temporal processing and hearing tests were administered under 3 test conditions. The order of the tests performed under each test condition was at random. A minimum of 2 breaks was given to participants during the assessment appointment to reduce fatigue and inattention.
Condition 1: Unaided sound field tests with no masking noise and no BCD
Condition 1 tests collected, DPT and PPT scores, WRS and sound field hearing threshold test (PTA4) data without BCD aiding and in the absence of contralateral ear masking noise.
Condition 2: Unaided sound field tests with 50 dB hearing level noise presented to the better hearing ear and an adhesive adapter without a BCD applied to the skin immediately behind the participants’ poorer hearing ear
Condition 2 collected test data when undertaken with noise masking of the better hearing ear. The non-hair-bearing skin behind the pinna of the poor hearing ear was cleaned with rubbing alcohol and allowed to dry. The Adhear (MED-EL Inc) adhesive adapter was then positioned on the post-aural skin and held firmly in place for 60 seconds to achieve adherence. A 50 dB hearing level (HL) test–specific masking noise was applied to participants’ better hearing ears through insert earphones. White noise generated by a GSI 61 audiometer was used for unaided PTA4 hearing thresholds, DPT and PPT scores. Speech noise was used to evaluate WRS.
Condition 3: Aided sound field tests with 50 dB HL noise presented to the better hearing ear and an adhesive BCD fixed to the skin behind the poorer hearing ear
In condition 3, the Adhear (MED-EL Inc) BCD was attached to the adhesive adaptor. The preprogramed program 1 setting was selected for all tests. Aided PTA4 thresholds, WRS, DPT, and PPT scores were recorded while applying masking noise to the better hearing ear as described in condition 2.
Statistical Analysis
PPT and DPT scores recorded in noise with the poorer hearing ear unaided were compared with those obtained in noise with the poorer hearing ear aided using Wilcoxon matched pairs signed-rank tests. This pilot study’s focus was to determine whether the adhesive BCD affected temporal processing; therefore, to reduce the risk of a type 2 error no statistical comparison of sound field PTA4 or WRS was undertaken. The latter study outcomes are presented descriptively. Statistical significance was set at P < .05. Analyses were performed with the Statistical Program for the Social Sciences version 24.0 (IBM Corp., Armonk, NY, USA).
Results
Temporal Processing Performance on the PPT and the DPT
Median PPT and DPT scores were 92% [interquartile range (IQR) = 12] and 96% (IQR = 13) respectively, when unaided in quiet (condition 1); 0% (IQR = 0) for both the PPT and the DPT when unaided in noise (condition 2); 65% (IQR = 26.5) for the PPT and 38% (IQR = 27.5) for the DPT when aided with an activated adhesive BCD in noise (condition 3). PPT and DPT scores improved significantly when participants were aided in noise (condition 3) compared with when they were unaided in noise (condition 2; Wilcoxon Ranks Sum, P = .018) (Figure 1).

Median PPT and DPT scores across test conditions in adults with SSD. Box plots indicate the median (centered horizontal line) with 25%- and 75%-quartiles (upper and lower horizontal lines), and lower boundary of data set (Q1 − 1.5 × IQR) and upper boundary of data set (Q3 + 1.5 × IQR) as minimum and maximum (whisker) values. The cross “X” indicates the mean value. See text for details of the 3 test conditions.
PTA4 Thresholds
When participants were tested unaided in quiet (condition 1), the median (IQR) unaided sound field averaged (PTA4) was 21.9 dB HL (IQR = 4.4), which reflects the hearing threshold of the better hearing ear under ideal sound field test conditions. When participants were tested unaided with masking noise (condition 2), the median unaided PTA4 threshold was 36.9 dB HL (IQR = 10.9). A median threshold PTA4 of 28.1 dB HL (IQR = 6.6) was obtained when participants were tested with masking noise and an activated adhesive BCD applied behind the poorer hearing ear (condition 3).
Word Recognition Score
When participants’ word recognition was assessed, a median score of 96% (IQR = 4) was obtained when tested unaided in quiet (condition 1); 5% (IQR = 24) unaided with masking noise (condition 2); and 58% (IQR = 58) aided with an adhesive BCD applied behind the poorer hearing ear and masking noise (condition 3) (Table 2).
Participants’ Sound Field Pure Tone Threshold Average (PTA4) in dB Corrected to 1 Decimal Place by Test Condition.
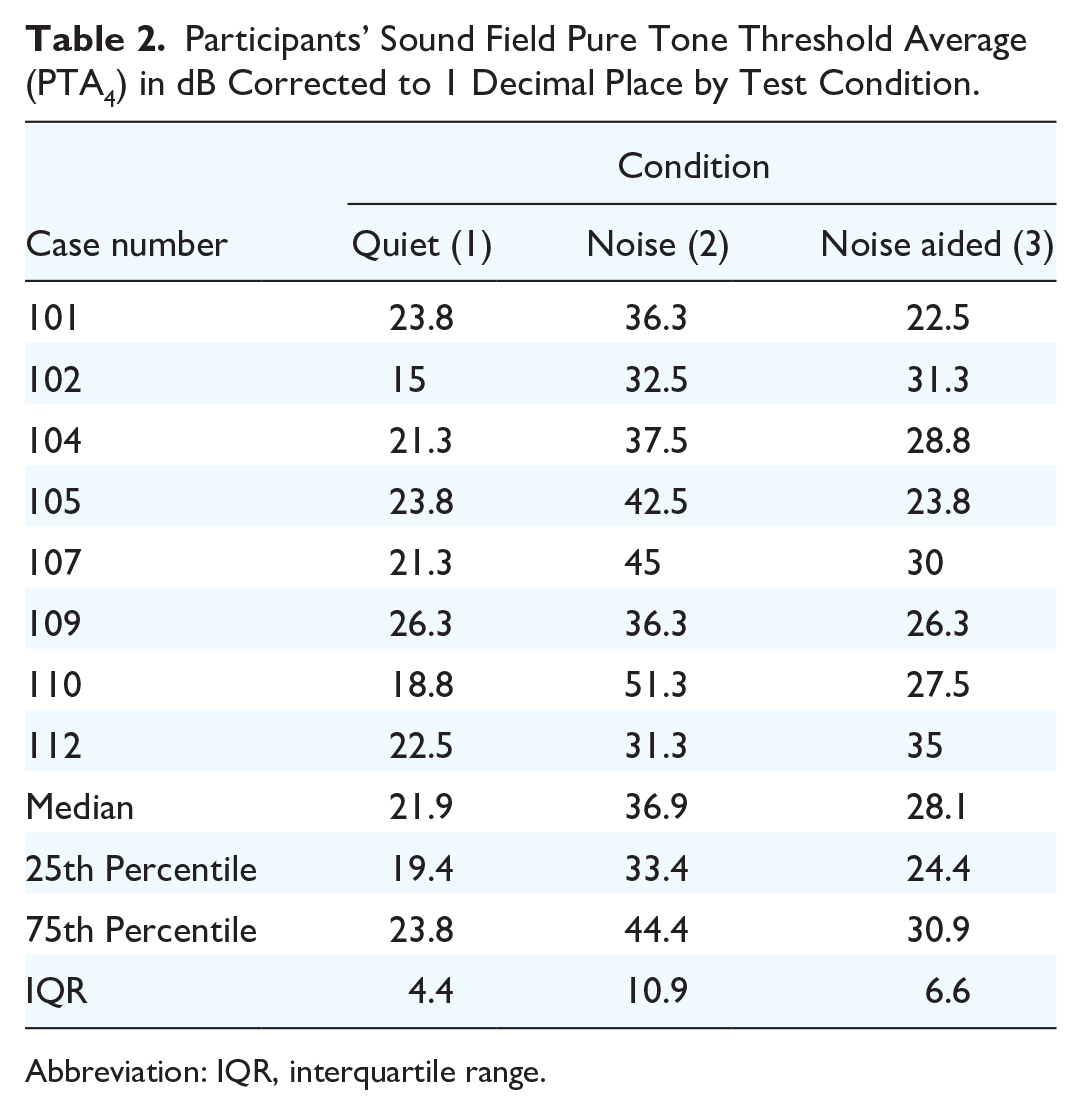
Abbreviation: IQR, interquartile range.
Discussion
A number of studies used behavioral audiological tests and subjective questionnaires to conclude that both children and adults with SSD or CHL derive significant benefit from adhesive BCDs. 8 In this study, temporal processing of sounds was assessed in adults with SSD unaided and adhesive BCD aided. This study focused on temporal processing of sound because it affects speech perception, 14 and previous studies on BCDs have not reported their effect on temporal processing.
Temporal processing deficits affect speech intelligibility, especially in noisy surroundings. 14 In this study, temporal processing evaluations PPT and DPT scores in noise improved significantly when using the adhesive BCD (Figure 1). These results illustrate that in noisy conditions, median PPT score trended higher than median DPT score, the reverse of that in quiet. These trends were not statistically analyzed, but it is established that normal-hearing individuals perceive frequency change cues better than duration change cues. 10 Musiek et al 12 concluded that the DPT is a less valid test of cochlear hearing loss than the PPT. Median PPT and DPT scores in participants with SSD in quiet in the current study are consistent with published scores for normal-hearing adults in quiet. 12 When noise was introduced into the participants better hearing ears, their temporal processing deteriorated to near to 0. This suggests that these participants experienced significant difficulties in detecting temporal changes in speech stimuli in background noise consistent with a reduction in speech intelligibility. Aided in noise, median PPT and DPT scores improved to 65% (IQR = 35) and 38% (IQR = 42.5), respectively. Chowsilpa et al’s 15 meta-analysis of temporal processing tests determined that changes of 31% in the PPT and 22% in the DPT are clinically important. This is consistent with the adhesive BCD achieving a clinically-important improvement in participants’ temporal processing in noise; however, that improvement did not result in a level of temporal processing that matched that in quiet conditions unaided, median PPT and DPT scores of 92% (IQR = 12) and 96% (IQR = 13) respectively.
Speech intelligibility increases with enhancement of consonant duration in hearing-impaired and normal-hearing individuals. Furthermore, Bance et al 11 reported improvements in temporal processing scores in chronic otitis media sufferers with CHL when aided with a unilateral direct-drive BCD. The current study identifies that the adhesive BCD leads to significant improvements in temporal processing scores in adults with SSD and suggests that the central auditory processing deficit in these individuals can be mitigated.
This improvement in temporal processing is mirrored in these participants’ WRS performances. Study participants’ median WRS in noise without aiding was 5% (IQR = 24) and, with adhesive BCD aiding, it was 58% (IQR = 58). This is consistent with an improvement in temporal processing and is in agreement with den Besten et al’s 16 report of a Cochlear Attract BCD aided mean WRS in quiet of 69.5% with a 50 dB speech stimulus and occlusion of the better hearing ear occlusion in 15 SSD participants 6 months postfitting. SSD patients fitted with the direct-drive percutaneous Ponto device achieved mean aided WRS in quiet of 90.8% 3 months postfitting. 17 Direct-drive BCDs including active transcutaneous devices bypass the sound attenuation effect of the skin, and so hearing outcomes are expected to be superior to passive transcutaneous BCDs. However, studies to date do not separately report the effect of active transcutaneous BCDs on WRS in the adult SSD subgroup of patients, most likely because individual studies report too few patients with SSD to robustly interpret their effect. Although it is not yet possible to definitively compare all available BCD options in SSD patients, available evidence supports direct-drive BCDs achieving superior WRS outcomes compared with passive transcutaneous devices.
The present study’s limitations include the inherent selection bias related to recruitment from individuals presenting to a tertiary hospital service, and the failure to assess aided sound field responses in quiet with the activated adhesive BCD fixed to the skin behind the poorer hearing ear. Additionally, pure tones can cause standing waves in sound field test environments, thus reducing the reliability of the PTA4 threshold findings. Some investigators used 4 kHz rather than 3 kHz thresholds in PTA4 analyses. The ideal PTA frequency thresholds for study is controversial. The hearing and equilibrium expert committee of the American Academy of Otolaryngology Head and Neck Surgery recommends that the PTA thresholds at 0.5, 1, 2, and 3 kHz be used for the evaluation and reporting of CHL treatment. 13 These guidelines were adopted in the current study to facilitate comparison with the literature on surgical treatments of CHL. Only the immediate effect of the adhesive BCD on temporal processing was assessed here, Hearing aid use in patients with sensorineural hearing loss leads to immediate improvements in pitch and temporal processing. Furthermore, temporal processing continues to improve with ongoing hearing aid use over time. 18 There are conflicting reports concerning improvement in pitch processing with continued hearing aid use in sensorineural hearing loss subjects.18,19 Further work is required to determine whether continued adhesive BCD use in SSD subjects will lead to a greater improvement in temporal processing ability.
Conclusions
This study shows that an adhesive BCD substantially improved temporal processing ability in background noise in individuals with SSD. It therefore provides a viable alternative treatment for these individuals. In future work, the effect of the adhesive BCD on temporal processing performance of adults with unilateral CHL and the effect of prolonged adhesive BCD use can be investigated. Additionally, these findings will allow for future comparisons of the temporal processing function achieved with other hearing solutions such as direct-drive BCDs and CROS hearing aids in individuals with SSD.
Footnotes
Acknowledgements
None.
Author Contributions
L.Q.: conceptualized the study and acquired the data; L.Q. and D.A.N.: co-designed the study; L.Q., D.A.N., and E.H.: analyzed and interpreted the data, and drafted and revised the manuscript critically for important intellectual content. All authors read and approved the final manuscript.
Availability of Data and Materials
The datasets analyzed in the current study are available from the corresponding author upon reasonable request.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The senior author (D.A.N.) received research funding for a previous study from NexGen Hearing and is currently supported by research grants from the Rotary Club of Vancouver Hearing Foundation, Pacific Otolaryngology Foundation, and Vancouver Coastal Health Research Foundation, and the University of British Columbia.
Ethics Approval and Consent to Participate
This study has been approved by the University of British Columbia Office of Research Ethics (Clinical Research Ethics Board; UBC CREB number: H19-00269), granted to Dr Li Qi, Principal Investigator.
