Abstract
Background
Intracochlear fibrosis and inflammation remain important limitations in cochlear implantation (CI). Glucocorticoids are routinely used to ameliorate the inflammatory response following CI. This study investigates the long-term effects of an intratympanically-applied triamcinolone-acetonide suspension on intracochlear impedance changes in CI recipients and investigates differences in drug concentrations and timepoints of injection.
Methods
A total of 87 patients were included in the study, of whom 39 received an intratympanically-applied triamcinolone-acetonide suspension at either 10 or 40 mg/ml, 1 hour or 24 hours prior to cochlear implantation, while 48 patients served as an untreated control group. Electrode impedances were measured and compared over a period of 3 years following cochlear implantation.
Results
The preoperative intratympanic application of a triamcinolone-acetonide suspension resulted in significantly lower mean impedances following cochlear implantation compared with an untreated control group at first fitting (4.66 ± 1.3 kΩ to 5.90 ± 1.4 kΩ, P = .0001), with sustained reduction observed over 3 months. A sustained reduction was observed after spatial grouping of the electrode contacts, with significant improvements in both the middle cochlear region over a 24 month period (from 3.97 ± 1.3 kΩ to 5.85 ± 1.3 kΩ, P = .049) and the basal region over a 6 month period (from 5.02 ± 1.3 kΩ to 5.85 ± 1.3 kΩ, P = .008). The injection of 10 mg/ml of triamcinolone-acetonide 1 hour prior to cochlear implantation resulted in higher impedances compared with 40 mg/ml and 24 hour time interval until surgery.
Conclusion
A single preoperative intratympanic injection of triamcinolone-acetonide significantly reduces electrode impedances across the entire cochlea. This effect is sustained for up to 2 years, after which impedances gradually equalize between the groups. A preoperative triamcinolone-acetonide injection could therefore be a favorable approach to attenuate the immediate tissue response following cochlear implantation.

Background
Cochlear implantation (CI) is considered the most successful treatment option for profoundly-hearing-impaired and deaf patients, with over 1 million patients having been fitted with a CI by 2022. 1 Cochlear trauma during surgery and subsequent inflammation remain important limitations that can significantly affect implant performance.2-4 Glucocorticoids are routinely used to ameliorate the inflammatory response following CI. However, systemic application has not found to be beneficial in counteracting the immediate trauma and long-term increase in impedances.5,6 By following a previously-reported patient population, this study investigates the long-term effects of an intratympanically (IT) applied triamcinolone-acetonide (TAAc) on electrode impedances after CI. 7
Insertion of the electrode array during CI surgery leads to an immediate mechanical trauma and the initiation of an inflammatory response, resulting in the disruption of cochlear homeostasis, loss of residual hearing, and fibrosis.2,8 The acute inflammation causes permeation of proteins into the perilymph, the adsorption of monocytes and macrophages on the electrode array, and the buildup of collagen. Over time, this results in fibrosis and ossification around the electrode array. 2 Fibrosis and ossification have been associated with the loss of residual hearing in electric acoustic stimulation CI recipients while ossification has been associated with poorer word-recognition scores.2,3 The fibrous sheath might further act as an insulator, resulting in broader spread of current and a decrease in dynamic range. 9 In addition, the increased current required to overcome the insulating layer might lead to higher power consumption of the implant, affecting battery life and potential duration of use.
Reduction in tissue response as measured by the preservation of residual hearing is partially accomplished by the utilization of soft surgery techniques and the use of shorter electrodes.10,11 Pharmacological treatment strategies using the potent anti-inflammatory glucocorticoids have additionally been shown to counteract the above-mentioned processes and attenuate the immediate inflammatory response in CI, in both human and animal studies.5,12,13 However, recent studies only found a limited effect of systemically-applied glucocorticoids in the human cochlea, possibly due to low blood flow and the restrictiveness of the blood-perilymph barrier.5,6,14 Local drug application strategies therefore emerged as a promising alternative, enabling significantly-higher and more consistent drug concentrations in the perilymph.7,15,16 Several studies investigated the effects of intraoperatively-applied glucocorticoids prior to electrode insertion on impedances, yielding conflicting results.17-21 Glucocorticoids act via immediate non-genomic effects and elicit a delayed modulation of the transcription of inflammatory genes.22,23 A longer duration between the application of glucocorticoids and CI could therefore be beneficial in preparing the cochlea for the electrode insertion trauma and decreasing the subsequent tissue response.
Electrical impedance telemetry is a routinely-performed measurement in CI recipients to evaluate the resistance between the electrode and the surrounding tissue. Impedances are affected by a variety of factors including cochlear perilymph composition, inflammation, and tissue growth around the electrode.18,24 In preclinical animal studies, a clear correlation between tissue response during CI and an increase in impedances could be established.25,26 More recently, elaborate measurement protocols have established impedances to be a clinically-feasible biomarker for cochlear health, such as predicting changes in speech-recognition before noticeable in monosyllabic tests.27,28 Routine impedances measurements during a patient’s outpatient visit can therefore be utilized to gain temporal information on the cochlear homeostasis following CI.
In a previous clinical trial, we evaluated the perilymph drug concentration of the glucocorticoid TAAc after IT application 1 hour and 24 hours prior to CI. 7 By following up this patient population over a time span of 3 years, we aimed to assess the long-term effects of 2 different IT-applied TAAc concentrations and the influence of the injection timepoint on postoperative impedances in CI recipients and compare these results to an untreated control group.
Materials and Methods
Study Design and Patients
In this case-control study, longitudinal impedance data from subjects of a previous clinical trial, 7 as well as from all other cochlear implant recipients at our institution during the same inclusion period (March 1, 2017-May 31, 2020), were retrospectively analyzed.
Inclusion criteria were age between 18 and 90 years, normal intraoperative cochlear anatomy without ossification, and complete electrode insertion confirmed by postoperative X-ray. Exclusion criteria consisted of patients who regularly took oral steroids or who had received local steroids before or during CI surgery. This study was approved by the ethics committee of the Medical University of Vienna (EK1456/2017). Demographic data such as age, sex, date of surgery, preexisting illnesses and medication were analyzed from the medical records of the Vienna General Hospital database system.
Patients were divided into 5 groups, of which 4 received a preoperative IT injection of TAAc 7 and 1 acted as an untreated control. In 2 groups, TAAc was applied approximately 24 hours prior to CI at a concentration of 10 mg/ml (24 hour-10 mg) or 40 mg/ml (24 hour-40 mg). In 2 other groups, TAAc was administered 1 hour prior to electrode insertion, also at a concentration of 10 mg/ml (1 hour-10 mg) or 40 mg/ml (1 hour-40 mg). These 4 treatment groups stem from a prospective clinical trial, in which Dahm et al previously quantified perilymph TAAc concentration prior to CI and observed comparable drug levels in the 40 mg groups as well as the 1 hour-10 mg group, however, lower median concentrations in the 10 mg-24 hour group. 7 The control group included all patients who met the inclusion criteria and were implanted with a CI during the same time period, but did not receive any pre- or intraoperative IT glucocorticoids.
All included subjects were implanted with one of three MED-EL cochlear implants (MED-EL GmbH, Innsbruck, Austria). The FLEXsoft electrode is 31.5 mm long with a basal diameter of 1.3 mm and apical dimensions of 0.5 × 0.4 mm. The FLEX28 is 28 mm long with a basal diameter of 0.8 mm and apical dimensions of 0.5 × 0.4 mm. The standard electrode is also 31.5 mm long with a basal diameter of 1.3 mm and an apical diameter of 0.5 mm. While the standard electrode comprises of 5 double-wired electrodes at the apical contacts, the FLEX electrode has 5 single-electrode wires to reduce its apical diameter.
Measurement of Impedances
Impedance measurements were carried out by clinical engineers using the standard MED-EL telemetry system (MAX interface box, clinical software MAESTRO 6 and 7). Measurements were performed intraoperatively and at every subsequent fitting appointment. The electrode fitting starts 4 to 5 weeks after surgery and continued at 3, 6, and 12 months in the first year following CI. Afterward, follow-up appointments were scheduled annually. Only data of patients who continued fitting at our institution were available, thus explaining missing data over time.
Impedance measurements of the 12 electrode contacts were analyzed as a mean across all contacts and stratified into 3 groups, corresponding to the covered cochlear regions. To account for differences in electrode design between the FLEX and standard electrodes in the apical 5 contacts, the grouping of the cochlear regions was chosen accordingly. The first 5 contacts (C1-C5) were defined as the low-frequency group. The next 3 (C6-C8) form the mid-frequency group and the last 4 (C9-C12) represent the high-frequency group covering the base of the cochlea.
Application of Triamcinolone-Acetonide
In patients who received TAAc (Volon-A®; Dermapharm Inc., Gruenwald, Germany) 24 hours prior to surgery, IT injection was performed in a supine position with the treated ear oriented upward. Xylocaine spray (10 mg/puff) was applied into the outer ear canal for local anesthesia. A 1 ml suspension containing 10 mg/ml TAAc (group 24 hour-10 mg) or 40 mg/ml TAAc (group 24 hour-40 mg) was then IT-injected using a 25G (0.50 × 90 mm) needle. Individuals remained lying for further 20 minutes to enable sufficient contact time to the inner ear entryways. Patients receiving the IT application 1 hour prior to CI were injected with the suspension immediately after general anesthesia. Group 1 hour-10 mg received 10 mg/ml TAAc, and group 1 hour-40 mg received 40 mg/ml TAAc. TAAc was applied until the middle ear was filled up, and a backflow into the external ear canal was noticed.
Data Evaluation and Statistical Methods
Data are expressed as mean ± standard deviation. Differences in impedances were compared between TAAc treated and control groups by unpaired t-tests. TAAc treated subgroups were analyzed descriptively to assess the effect of dosage and injection timepoints. Two-sided P-values <.05 were considered statistically significant. Statistical analyses were performed using the GraphPad Prism 9.5.1 software.
Results
A total of 87 patients who received a CI between March 1, 2017, and May 31, 2020, were included in the study. Thirtynine subjects received an intratympanic injection of either 10 or 40mg/ml TAAc, 1hour or 24 hours prior to surgery while 48 did not receive any glucocorticoids and were allocated to the control group. Patient characteristics of the study cohort and etiologies of hearing loss are described in detail in Table 1.
Patient Characteristics of the Study Cohort, Grouped into Treatment Group and Control Group.
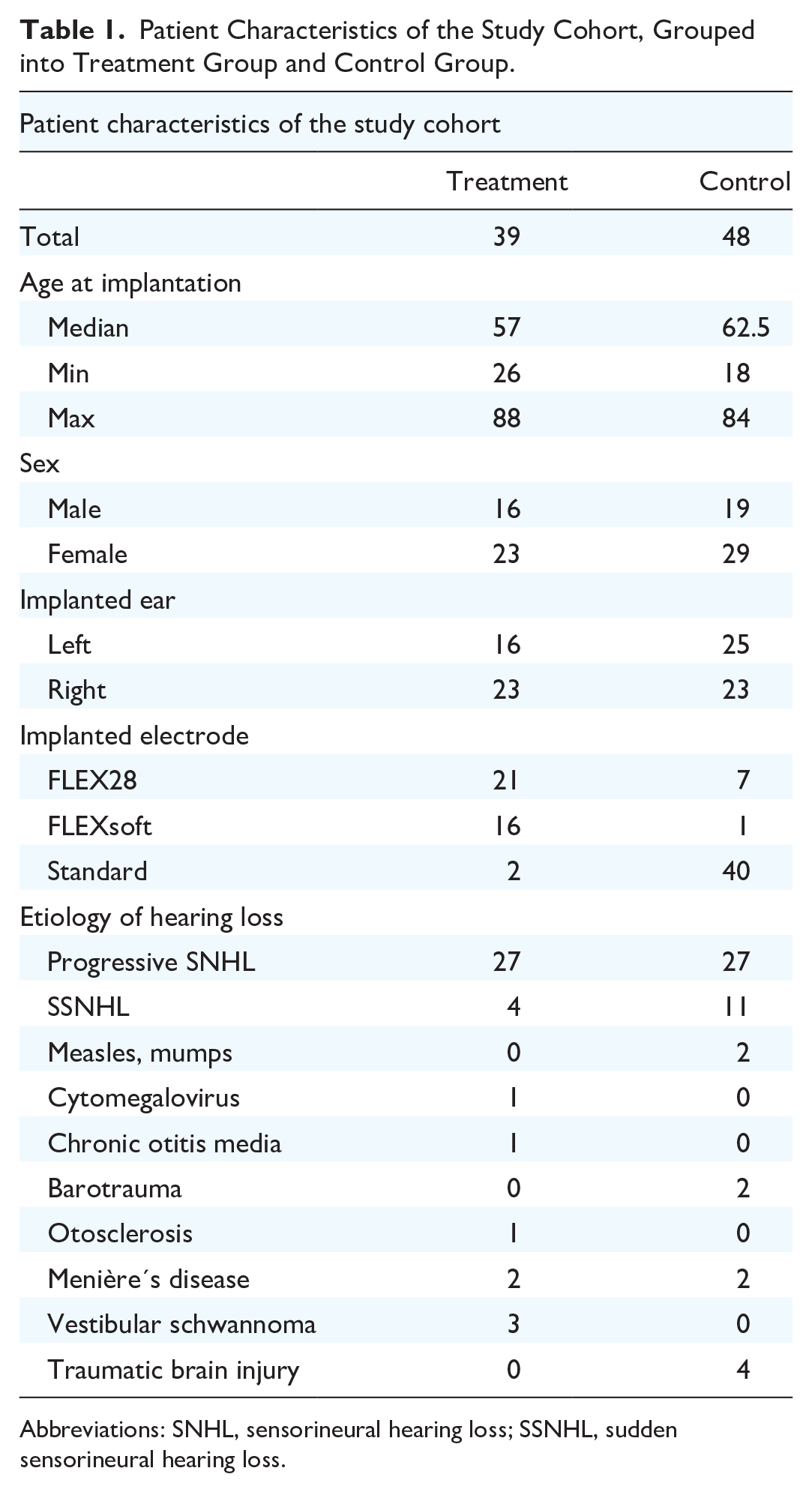
Abbreviations: SNHL, sensorineural hearing loss; SSNHL, sudden sensorineural hearing loss.
Overall, mean impedances across all 12 electrodes showed lower intraoperative values in the TAAc-treated group than in the control group (4.98 ± 1.4 kΩ and 5.48 ± 1.1 kΩ, P > .05). Impedances in the TAAc-treated group decreased on the first fitting appointment, while impedances in the control group increased. Significantly-lower impedances in the TAAc group were observed at first fitting (4.66 ± 1.3 kΩ to 5.90 ± 1.4 kΩ, P = .0001) and 3 months after CI (4.96 ± 1.1 kΩ to 5.77 ± 0.9 kΩ, P = .0005), compared with the control group. Subsequently, impedances in both groups gradually converged, with a slight increase in the TAAc group and a decrease in the control group. These trends remained consistent and nearly parallel until the final measurement timepoint at 36 months after CI (5.49 ± 0.9 kΩ to 5.52 ± 0.9 kΩ, P > .05, Figure 1).

Mean average impedance values of all 12 electrode contacts over the time course of 36 months, divided into the control and TAAc-treated groups. FF, first fitting; TAAc, triamcinolone-acetonide; 3M-36M, 3 months to 36 months.
The division into 3 electrode groups representing the basal, middle, and apical cochlear regions revealed finer differences between the treated and untreated groups. Intraoperatively, impedances were significantly different in the mid-frequency electrodes with 4.41 ± 1.7 kΩ in the TAAc and 5.47 ± 1.5 kΩ in the control group, respectively (P = .0034, Figure 2B). Impedances in the apical area were 5.54 ± 1.5 kΩ and 5.85 ± 1.5 kΩ (Figure 2A) and 4.70 ± 1.6 kΩ to 4.99 ± 1.6 kΩ in the basal area (Figure 2C) for the TAAc and control groups, respectively (P > .05). On the first fitting appointment, apical impedances slightly increased in both groups and were significantly lower in TAAc than in the control group, with 5.88 ± 1.9 kΩ to 6.76 ± 1.6 kΩ (P = .025, Figure 2A). In the middle and basal regions, impedances in the TAAc group were significantly lower than in the control group, with 3.75 ± 1.2 kΩ to 5.42 kΩ ± 1.6 and 3.83 ± 1.6 kΩ to 5.21 ± 1.5 kΩ, respectively (P < .0001, Figure 2B,C).

Mean impedance values over the time course of 36 months for (A) apical, (B) middle, and (C) basal electrode contacts, divided into the control and TAAc-treated groups. FF, first fitting; TAAc, triamcinolone-acetonide; 3M-36M, 3 months to 36 months.
Following the first fitting, impedances of the apical electrodes in the TAAc treated group increased until 12 months, after which a slight reduction was noted until 36 months. In comparison, impedances in the control decreased from the first fitting appointment to 12 months after surgery. Here, an insignificantly-higher impedance of 6.39 ± 1.9 kΩ in the TAAc-treated group compared with that of 5.75 ± 1.3kΩ in the control group was observed (P > .05). From 12 months after surgery, apical electrode impedances stabilized in both groups and slightly declined to 5.90 ± 1.4 kΩ in the TAAc-treated group and 5.46 ± 1.5 kΩ in the control group at 36 months, respectively (P > .05, Figure 2A).
In the middle cochlear region, TAAc treatment resulted in significantly-lower impedances compared with the control group over 6 months, with 3.88 ± 0.9 kΩ to 5.08 ± 1.3 kΩ (P < .0001) at 3 months and 4.09 ± 0.7 kΩ to 5.08 ± 0.9 kΩ (P < .0001) at 6 months, respectively. A decrease in impedances was noted in the control group between the first fitting up until 24 months, after which they continuously increased. In comparison, impedances in the TAAc-treated group increased from the first fitting up to 12 months but were continuously lower than in the control group with a significant difference at 24 months (P = .049). At 36 months following CI, impedances reached 4.09 ± 0.7 kΩ and 4.85 ± 1.9 kΩ in the TAAc-treated and control groups, respectively (P > .05, Figure 2B).
Basal impedances remained significantly lower in the TAAc-treated group than in the control group until 6 months following CI, at 5.02 ± 1.3 kΩ to 5.85 ± 1.3 kΩ respectively (P < .0001-P = .008). Impedances increased almost parallel to each other until 36 months to 5.97 ± 1.9 kΩ and 6.41 ± 1.8 kΩ in the TAAc-treated and control groups, without significant difference (P > .05, Figure 2C).
Further, the influence of different application timepoints (1 hour vs 24 hours prior CI) and TAAc concentrations (10 mg/ml vs 40 mg/ml) on electrode impedances following CI were evaluated. The patient characteristics of the subgroups are described in Table 2. Intraoperatively, no pronounced difference in impedances was observed at any region between the different treatment groups (Figure 3A). At the first fitting appointment, apical impedances in the 1 hour-10 mg group were larger at 7.29 ± 1.9 kΩ than at 5.24 ± 0.1 kΩ in the 1 hour-40 mg group, 5.02 ± 1.6 kΩ in the 24 hour-10 mg and 5.78 ± 2.1 kΩ in the 24 hour-40 mg group. In the middle and basal regions, only minor differences between the treatment groups were noticed (Figure 3B). Impedances in the 1 hour-10 mg group remained elevated in the apical region compared with the other 3 groups (Figure 3C) until 6 months after CI. Subsequently, no major differences were observed over 12 months after surgery (Figure 3D and E).
Patient Details of the 4 Subgroups in the Treatment Group.

Abbreviation: TAAc, triamcinolone-acetonide.

Subgroup analysis of different TAAc concentrations and application timepoints over the course of 12 months, grouped into apical, middle, and basal electrode contacts. TAAc, triamcinolone-acetonide.
Discussion
In the present study, we investigated the effects of a preoperatively IT-applied TAAc suspension on postoperative electrode impedances following CI over the course of 3 years. Impedances measure the resistance around the cochlear implant and are proposed as a biomarker to predict residual hearing loss, the presence of inflammation as well as fibrous growth and bone formation around the cochlear implant.19,27,29 We demonstrate that a single preoperative IT application of TAAc significantly and immediately reduces electrode impedances following CI.
When averaging impedance values across all 12 electrodes, we noted a significant decrease in impedances in the TAAc group for the first 3 months when compared to the control group. Subsequently, there was a nearly-parallel and consistent trend. However, when differentiating the changes in impedances into 3 electrode groups based on their cochlear region (apical, middle, basal), finer differences were observed. Here, the effects were greatest in the middle and basal cochlear areas in which a sustained reduction for up to 24 months was observed. Apical impedances however were only decreased up to 1 month. Multiple previous studies confirmed the inflammatory and fibrotic response following CI to have a basal-to-apical gradient, most pronounced near the site of electrode insertion, with a gradual decrease in severity with increased distance from the insertion site, 2 explaining the more pronounced increase in impedances in the basal cochlear regions, and the increased protective effects of TAAc in these regions.
To the best of our knowledge, the effects on impedances by an IT injection of TAAc have not been reported previously. Our results, however, follow a similar time course to studies that applied glucocorticoids intraoperatively during CI.17,18,20 Paasche et al found significantly-lower impedances across all electrodes after an intracochlear injection of 20 mg/ml TAAc, most pronounced in the basal cochlear areas, persisting for over 3 years. 17 Prenzler et al utilized an intracochlear catheter to deliver TAAc and observed significantly-reduced impedances in middle contacts from day 10, which increased to the level of the control group after 4 weeks. 21 In a follow-up study, they observed a sustained reduction in impedances over 3 months using higher drug concentrations of 20 mg/ml TAAc. 20 The differences in longevity of effect between these studies might relate to the choice of implanted electrodes. Prenzler et al used lateral-wall electrodes, similar to our study, which have been shown to have lower postoperative impedances than peri-modiolar electrodes, possibly due to lower insertion trauma during CI. 28 However, Paasche et al implanted peri-modiolar electrodes, which may cause a more pronounced tissue response due to an initial higher trauma, after which an intracochlear TAAc injection could elicit greater protective effects. In lateral-wall electrodes, however, other treatment modifications, such as increasing the applied drug concentrations or changing the application timepoint, may be necessary to further attenuate postoperative impedances.
Compared to these studies, we applied TAAc intratympanically at 1 hour or 24 hours prior to surgery into the middle ear, from where it continuously diffuses through the entryways of the inner ear, for example, round window, oval window, and bony canals, into the perilymph.30-32 A benefit of the preoperative application of glucocorticoids might lie in the preparation of the cochlea to the upcoming insult. Following the IT administration of TAAc in our study, we observed a significant reduction in impedances in the intraoperative measurement, suggesting an immediate effect on cochlear homeostasis. In contrast, intracochlear administration just prior to the insertion of the electrode array did not result in significant intraoperative differences in terms of impedance. 20 Glucocorticoids elicit immediate effects by acting on inflammatory and apoptotic pathways. 33 However, they further exert delayed genomic effects by modulation of the transcription of pro-inflammatory cytokines and immunoregulatory genes. 34 By extending the time period between TAAc administration and electrode insertion, the implanted ear could be further conditioned for the upcoming insertion trauma, taking advantage of both genomic and nongenomic effects. Another benefit of a preoperative IT injection might be the longer cochlear distribution time of the injected drug prior to CI. We speculated that impedances in our study will only be modulated in the basal-to-middle cochlear areas, given that drug distribution in perilymph toward the cochlear apex solely rely on diffusion. 35 However, we observed very similar effects in the cochlear apex after the preoperative IT injection compared with intracochlear injections of others. 20 The continuous diffusion of TAAc from the middle ear into the perilymph might facilitate apical drug transport despite its rapid clearance, allowing for the reduction in impedances in the cochlear apex.35-37
To further elucidate on the importance of time and drug concentration, we performed a subgroup analysis of the 4 different TAAc-treated groups. In our study, 40 mg/ml of TAAc attenuated insertion trauma when administered just before and 24 hours prior to CI. This fits well into the current literature, observing high glucocorticoid concentrations to be an important factor in improving postoperative impedances.18,20 However, in the 1 hour-10 mg group, but not in the 24 hour-10 mg group, we observed higher impedances from 1 month to 6 months in the apical cochlear region, suggesting that time is another important factor on the tissue response following CI.38,39 The time course of gene expression after dexamethasone treatment shows a significant activation and repression beginning 1 hour after treatment with an increasing effect for up to 4 hours. 23 While high concentrations of TAAc sufficiently diffuse toward the cochlear apex and attenuate insertion trauma when administered just before injection, 18 low drug concentrations may require support from a longer period between application and CI to achieve a similar protective effect. 39
Overall, these data suggest that an IT-applied TAAc immediately attenuates the inflammatory response following CI and results in a sustained reduction in impedances, comparable to intracochlear injection. However, a slow and progressive increase in impedances suggests a chronic cochlear response, such as a foreign body reaction or chronic electrical stimulation, which cannot be alleviated by a single glucocorticoid injection.
Our study has a few limitations. First, this retrospective study design did not allow for meticulous scheduling of the measurement timepoints. Data were extracted from the routine clinical outpatient visits, and as such, the timepoints varied and therefore had to be grouped into intervals. Secondly, the retrospective design did not allow for the matching of patient characteristics across the groups, such as distribution of the implanted electrodes. While the treatment group primarily received FLEX28 and FLEXsoft electrodes, the control group underwent implantation with standard electrodes, which are double-wired in the apical contacts. We consider this potential bias negligible, and it may even reinforce our results, as previous studies reported lower impedances in standard electrodes than in FLEX electrodes, 40 suggesting that the impact of TAAc treatment might be even more pronounced. Despite promising findings on the effects of TAAc following CI, prospective trials are required to clarify its effects on postoperative impedances given the multitude of influencing factors.
Conclusions
This is the first study to evaluate long-term impedance changes in CI recipients following the IT application of TAAc at varying concentrations and application timepoints. We demonstrate that a significant reduction in electrode impedances throughout the entire cochlea can be achieved by a single preoperative IT application of TAAc. This effect is sustained for up to 2 years, after which impedances gradually equalize between the groups over a span of 3 years. A preoperative TAAc injection therefore might be a promising strategy to prepare the inner ear for the upcoming implantation and mitigate the immediate tissue response over the whole cochlear area.
Footnotes
Acknowledgements
None.
Author Contributions
M.N., study concept and design; acquisition of data; analysis and interpretation of data; drafting of manuscript; and critical revision; R.L., acquisition of data and critical revision of manuscript; C.H., A.B.A., and V.D., study concept and design; analysis and interpretation of data; and critical revision of manuscript; J.C.G., drafting of manuscript, study concept and design; analysis and interpretation of data; and critical revision of manuscript; W.D.B. and D.R., study concept and design and critical revision of manuscript; C.A., study concept and design; interpretation of data; and critical revision of manuscript. All authors read and approved the final manuscript.
Availability of Data and Materials
The analyzed data of the study are available from the corresponding author upon reasonable request.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.A., C.H., and D.R. receive funding from MED-EL Corporation, Innsbruck, Austria, unrelated to this study. No influence was made on the interpretation of the data. The other authors have no conflict of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This study was approved by the ethics committee of the Medical University of Vienna (EK1456/2017).
