Abstract
Objective
The surgical strategy of cholesteatomas is still controversial. This study aimed to compare the hearing improvement and determine the prognostic factors between endoscopic and microscopic ear surgery for attic cholesteatoma via a multicenter retrospective study.
Methods
This retrospective study included 169 patients with attic cholesteatoma who received endoscopic ear surgery (EES) or microscopic ear surgery (MES) from 12 otorhinolaryngology centers. Hearing improvement between EES and MES was evaluated, including the postoperative pure tone average (PTA) and air-bone gap (A-B Gap), as well as the hearing threshold across the low-, mid-, and high-frequency. The success rate of grafts was collected. Linear regression was performed to access the prognostic value of preoperative PTA and A-B Gap. Patients were followed up for at least 3 years.
Results
The graft success rate of EES was 89.66% (78/87) versus 80.49% (66/82) for MES. The postoperative PTA and A-B Gap demonstrated significant improvement in EES compared to MES (Post-PTA: t = 3.281, P = .001; Post-A-B Gap: t = 2.197, P = .029). In the EES group, there were 59 ears (67.82%) with a postoperative A-B Gap ≤20 dB HL, which revealed a higher rate of successful hearing outcomes in EES as opposed to MES (χ2 = 9.904, P = .019). There were significantly better hearing improvement, shorter surgical times, and lower hospital stays in EES for epitympanic cholesteatoma without stapes superstructure involvement. The preoperative AC ≤79 dB and/or preoperative A-B Gap ≤52 dB was associated with a better prognosis in EES for epitympanic cholesteatoma with stapes superstructure involvement.
Conclusions
EES showed higher graft success rate, better hearing improvement, shorter surgical times and hospital stays for attic cholesteatoma, particularly without stapes superstructure involvement. The range of preoperative PTA and A-B Gap have shown the prognostic value, which maybe a favorable surgical indication for EES or MES.

Keywords
Introduction
Cholesteatoma is a mass of stratified keratinizing squamous epithelium in the middle ear, resulting in ossicular chain disruption and hearing loss, and leading to severe intracranial and extracranial complications. 1 According to the pathogenesis, cholesteatomas are categorized into congenital, primary, and secondary acquired cholesteatoma. Primary acquired cholesteatoma is caused by selective dysventilation of the attic or eustachian tube dysfunction, while secondary acquired cholesteatoma is associated with persistent infection, tympanic membrane perforation, and the skin migration in the middle ear.2,3 Congenital cholesteatoma is linked to the epithelial rest theory. Surgery was one of the most effective strategies for cholesteatomas.
Cholesteatoma can be completely resected by endoscopic ear surgery (EES) and microscopic ear surgery (MES). However, which measure to choose is still a controversial issue. According to Livio Presutti, the surgical indication of epitympanic cholesteatoma for EES was the extension of cholesteatoma with limited attic extension (I-C1a and I-C1b), antrum (I-C2a), mesotympanic, protympanic and hypotympanic (I-C2b), sinus tympani and/or facial nerve involvement (I-C3), including lesion characteristic about cholesteatoma in pouch shape (S1) or with infiltrative matrix (S2). 4 On the other hand, the congenital cholesteatoma with or without ossicular involvement and without mastoid extension (stages I, II, and III) can be removed by EES, according to William P. Potsic 5 .
MES requires postauricular incision and extensive temporal bone drilling to achieve complete resection of the lesion, and a series of potential postoperative complications, such as taste abnormality and sensation loss of auricle. 6 Compared with MES, EES tends to have more flexible operation angles, a larger maximum visible radius in various directions, a mean gain in an observable distance of 19.18%, and may be beneficial for observing and removing cholesteatoma from hidden sites. 7 There are provided horizon visualizations to observe the middle ear landmarks of the resected temporal bone, which measures only 6.84 mm3 by EES; this is significantly less than 18.16 mm3 observed vis MES. 8 Furthermore, many studies have indicated that the successful graft take rate was 84.0% to 97.2% and the postoperative A-B Gap was 5.3 dB to 13.8 dB after EES, which can achieve the same satisfactory clinical effect compared to MES. 9 Especially for moderate middle ear risk (MER = 4-6), EES has achieved a superior hearing improvement. 10 The anesthesia times, surgical times and costs, hospital stays, postoperative pain Visual Analogue Scale (VAS), and analgesics consumption were shorter in EES than in MES, obviously improving the life quality.11,12 The recurrent and residual rates of cholesteatoma were significantly lower in EES than in MES. 13 Hence, endoscopic surgery has become a safe, minimally invasive approach for cholesteatoma management.
In recent years, the comparison between EES and MES in hearing improvement and its prognosis has become trending topic in cholesteatoma research. 14 On this basis, we launch a large-scale, multicenter study to evaluate the hearing improvement of EES and MES on cholesteatoma. Another purpose of our study is to predict postoperative hearing improvement based on preoperative hearing thresholds. The results of our study also provide the treatment strategies for the selection of endoscopic or microscopic surgery for cholesteatoma, based on the range of preoperative hearing thresholds and the presence/absence of the stapes superstructure.
Materials and Methods
Participants
A total of 169 patients diagnosed with cholesteatoma were included in this study within the otorhinolaryngology department across 12 hospitals between November 2016 and March 2021. The inclusion criteria for cholesteatoma are as follows: (i) hearing loss for a duration of at least 3 months, (ii) the soft tissue-like density shadow filling in the middle ear via high-resolution computed tomography of the temporal bone, (iii) and the confirmation via postoperative pathological diagnosis. The exclusion criteria include (i) the acute stage of middle ear infection, (ii) malformation of the ossicular chain or cochlea, (iii) the history of noise exposure, sudden sensorineural hearing loss, (iv) profound hearing loss, and (v) the lack of clinical, follow-up data and/or postoperative audiological data.
All individuals had baseline data and follow-up visits that were available for a minimum of 3 years. The graft take rate was calculated using the following formula: the number of successful graft take cases/the total cases × 100%. The hospital costs encompassed all expenses related to the execution of the surgical procedure during hospitalization. Hospital length of stay was defined as the length of time patients were admitted to the hospital both before (1-2 days) and after surgery, whereas the surgical times were delineated as the duration between the initiation and conclusion of the operation. This retrospective study series was performed in accordance with the Declaration of Helsinki. The approval of all procedures was obtained by the Ethical and Scientific Committee.
Surgical Technique
All patients were administered general anesthesia and local anesthesia, which was injected subcutaneously into the tragus and external ear canal with 1.0% lidocaine and 1‰ epinephrine. Controlled hypotension, with a systolic blood pressure of 75 to 90 mmHg or average arterial pressure of 50 to 65 mmHg, was used during surgery via MES or EES.
There were surgical instruments, including otology microsurgical instruments, microscopes, rigid endoscopes with 0° and 30° angles, lengths of 11 and 14 cm, and outer diameters of 2.7 and 3.0 mm (Storz, Germany), which were connected to a camera and a high-definition monitor for positioning. The steps of EES or MES include the creation of the tympano-meatal flap with a circular incision of approximately 180° at a distance of approximately 1 to 1.5 cm from the fibrocartilaginous ring of the tympanic membrane in the posterosuperior and posteroinferior portions of the external auditory canal, preservation of chorda tympani, removal of bony in the lateral attic wall, evaluation of the precise location and extent of lesions of cholesteatoma, resection of complete cholesteatoma including tympanic cavity and/or mastoid, ossicular chain reconstruction or not depending on whether ossicles were invaded, myringoplasty, and reconstruction of lateral attic wall.
All microscopic and endoscopic procedures were performed by an ENT surgeon who was the member in Professional Committee of Endoscopic Ear Surgery of Guangdong Clinical Medical Association with nearly 20 years of experience in different hospital. All ENT surgeon have taken the EES and MES training course in order to homogenize surgical operation and data collection.
Audiological Evaluations
The preoperative and postoperative audiological data were collected, consisting of air conduction (AC) for each frequency, bone conduction (BC) for each frequency, pure tone average (PTA), and air-bone gap (A-B Gap). Hearing thresholds of AC were measured by pure-tone audiometry at 0.125, 0.25, 0.5, 1, 2, 4 and 8 kHz, while BC was measured at 0.25, 0.5, 1, 2, and 4 kHz for both ears in a sound-proof booth.
Low-frequency AC (LF-AC), mid-frequency AC (MF-AC), and high-frequency AC (HF-AC) were calculated by computing the mean of AC at frequencies of 0.25 and 0.5, 1.0 and 2.0, 4.0 and 8.0 kHz, respectively. The hearing improvements, including PTA and A-B Gap, were determined by analyzing the preoperative and postoperative hearing thresholds, and are presented as PTA improvement and A-B Gap closure.
Statistical Analysis
Statistical analysis was performed using SPSS (IBM, Armonk, New York, USA, SPSS Statistics, Ver.27.0). Postoperative and preoperative PTA and A-B Gap, LF-, MF- and HF-AC improvement, PTA improvement, A-B Gap closure, surgical time, hospital length of stay, and cost between MES and EES group were assessed to estimate the significance of differences by using independent samples t-test. The paired sample t-test was conducted to assess the preoperative and postoperative PTA and A-B Gap in both EES and MES group. Linear regression was used to assess the effectiveness of preoperative PTA and A-B Gap in predicting postoperative hearing improvement in both EES and MES groups. The Chi-square test was used to assess the significant differences in success for graft rate and hearing between EES and MES group. In this study, P < .05 was statistically significant.
Results
The study included 169 patients, 103 males (60.95%) and 66 females (39.05%), 65 right ear (38.46%), 72 left ear (42.60%), and 32 bilateral cholesteatoma (18.94%). The average age was 33.13 ± 14.59 years. A total of 87 individuals underwent treatment through EES, while 82 individuals received treatment through MES. There were the demographic and clinical characteristics of individuals between the EES and MES groups in terms of age, gender, surgical side, degree of HL, surgical time, hospital stay, and cost (Table 1).
Demographic and Clinical Characteristics of Individuals [Mean ± SD or n (%)].
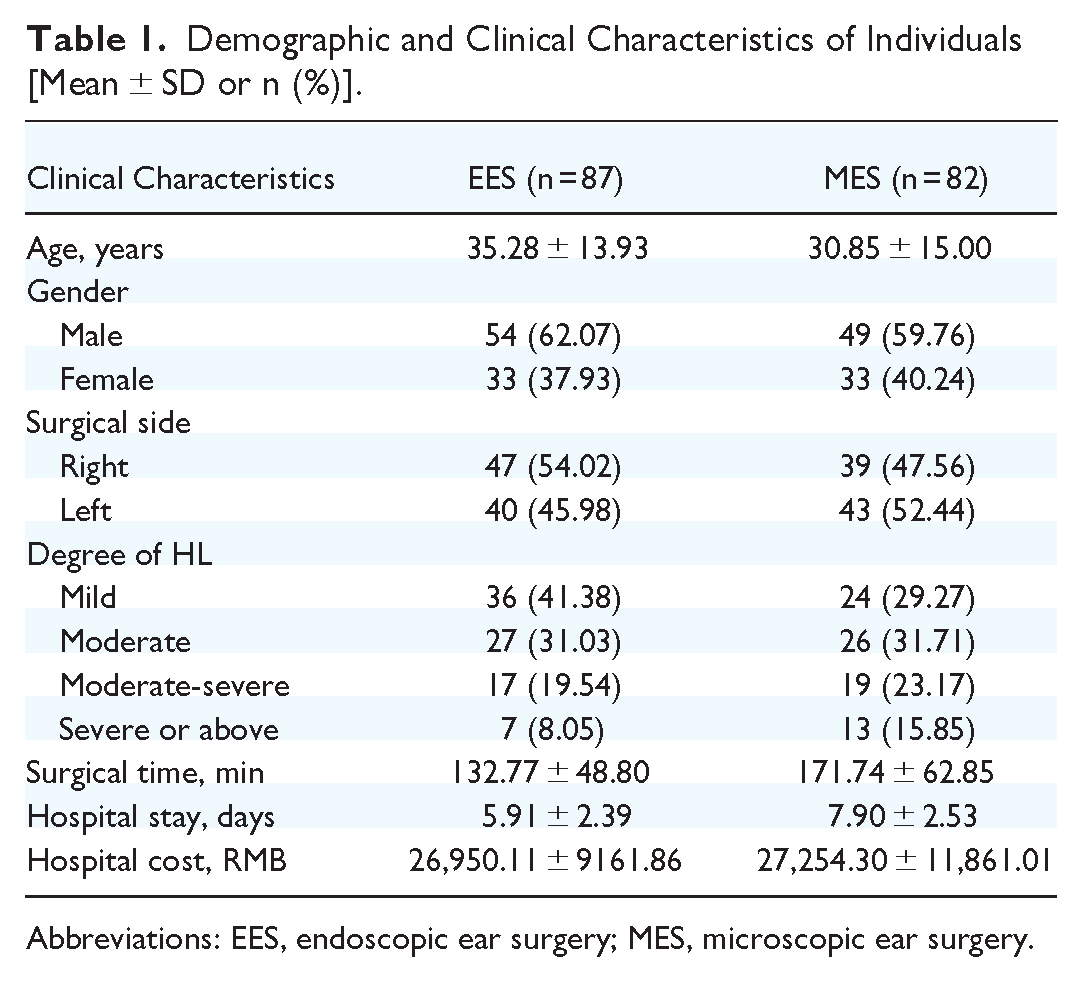
Abbreviations: EES, endoscopic ear surgery; MES, microscopic ear surgery.
In this study, the classification system proposed by Livio Presutti in his book “Endoscopic Ear Surgery: Principles, Indications, and Techniques” was utilized to categorize a total of 3 cases of congenital cholesteatoma, 114 cases of primary acquired cholesteatoma, 37 cases of secondary acquired cholesteatoma, and 15 cases of recurrent cholesteatoma.
A total of 7 cases presented with limited attic cholesteatoma, with 3 cases undergoing MES and 4 cases undergoing EES. Ninety-four patients were diagnosed with epitympanic cholesteatoma, specifically affecting the malleus, incus, incudomalleolar joints, and incudostapedial joints, which required the removal of diseased tissue and subsequent partial ossicular replacement prosthesis reconstruction, with 44 patients undergoing MES and 50 patients undergoing EES. A total of 57 patients presenting with cholesteatoma affecting various anatomical structures including the anterior and posterior crus of the stapes, stapes footplate, promontory, round window membrane, and round window niche, necessitated the implementation of total ossicular replacement prosthesis reconstruction, with 27 patients undergoing MES and 30 patients undergoing EES. Autogenous cartilage was used in 4 patients who had erosion of the incus and/or stapes superstructure. Six patients did not undergo reconstruction because of patient/parent/physician refusal. One patient developed petrous bone cholesteatoma.
The graft success rate of EES was 89.66% (78/87), accompanied by occurrences of tympanic membrane perforation in 3 cases, epithelial loss in 3 cases, granulation in the surgical cavity in 1 case, ossicular prolapse in 1 case, and attic retraction in 1 case. The recurrence rate of EES was 3.45% (3/87). Additional complications observed in the study included external auditory canal stenosis in 1 case, otitis media with effusion (OME) in 5 cases necessitating tympanostomy tubes in 4 cases, with 1 case left untreated. The graft success rate of MES was 80.49% (66/82), and complications were observed in 6 cases of epithelial loss, 3 cases of granulation in the surgical cavity, one case of combined epithelial loss and granulation in the surgical cavity, 2 cases of tympanic membrane perforation, 1 case of combined tympanic membrane perforation and cholesteatoma in the surgical cavity, 1 case of extensive cholesteatoma in the surgical cavity, 1 case of severe attic retraction, and 1 case of combined tympanic membrane retraction and OME. The recurrence rate of MES was 6.10% (5/82), with one case recurring twice (Figure 1).

Cholesteatoma removal in EES. (A-D) Left ear, high-resolution computed tomography of the temporal bone showed that patchy soft tissue density shadows in the bilateral tympanic, measuring 8.7 mm × 8.2 mm, including left malleus and incus invasion, (E) TM perforation and cholesteatoma in the pars flaccida, invagination in the pars tensa, (F) cholesteatoma made inroads on attic, retro- and meso-tympanum, (G, H) cholesteatoma was entirely removed, (I) partial ossicular replacement prosthesis reconstruction, (J) graft of TM and external auditory canal posterior wall reconstruction, (K) well-epithelialized graft of TM 45 days after surgery. TM, tympanic membrane; EES, endoscopic ear surgery.
The postoperative PTA and A-B Gap in the EES group exhibited significant improvement than preoperative (PTA: t = 10.345, P = .000; A-B Gap: t = 7.235, P = .000), as well as the MES group (PTA: t = 7.600, P = .000; A-B Gap: t = 6.682, P = .000). The postoperative PTA and A-B Gap were significantly better in the EES group compared to the MES group (Post-PTA: t = 3.281, P = .001; Post-A-B Gap: t = 2.197, P = .029) (Figure 2). The average hearing improvement is 14.43 dB in EES and 12.70 dB in MES (t = −0.795, P = .428). Both EES and MES demonstrated better hearing improvements in the low- and mid-frequency AC than high frequency (Table 2).
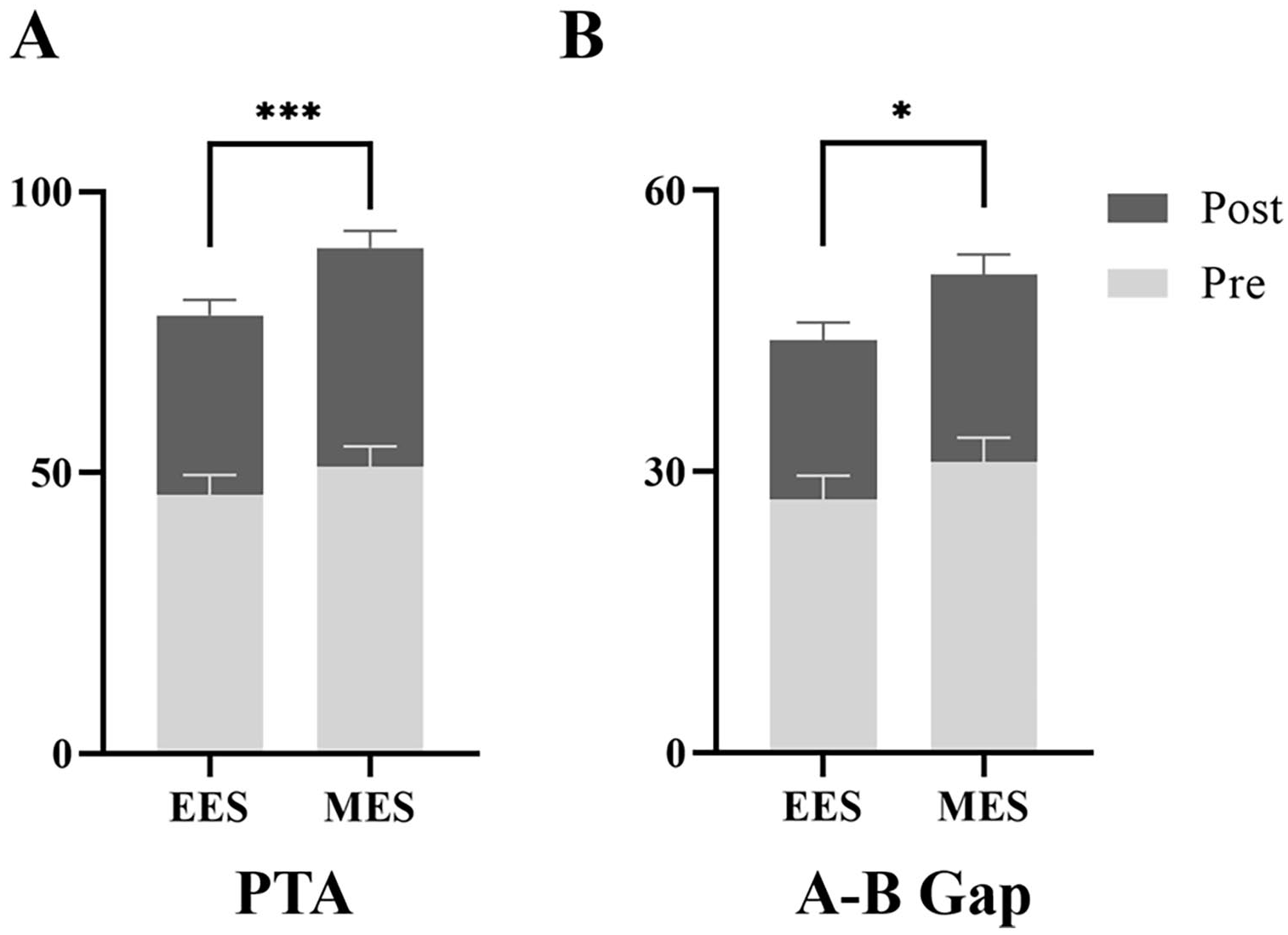
Comparison of preoperative and postoperative (A) PTA and (B) A-B Gap between EES and MES. Pre, preoperative; Post, postoperative; A-B Gap, air-bone gap; PTA, pure tone average; EES, endoscopic ear surgery; MES, microscopic ear surgery.
Hearing Improvement and Graft Rate in EES and MES [Mean (95% CI) or n (%)].

Success for hearing was based on the range of postoperative A-B Gap.
Abbreviations: A-B Gap, air-bone gap; EES, endoscopic ear surgery; HF-AC, high frequency air conduction; LF-AC, low frequency air conduction; MES, microscopic ear surgery; MF-AC, mid frequency air conduction; PTA, pure tone average.
In the EES group, the postoperative A-B Gap ≤20 dB HL was observed in 59 ears (67.82%), whereas in the MES group, this was found in 36 ears (43.9%). In the EES group, a total of 23 ears (26.44%) exhibited a postoperative A-B Gap ≤10 dB HL, whereas the MES group only had 13 ears (15.85%) with the same outcome. The rate of success for hearing was found to be higher in the EES group compared to the MES group (χ2 = 9.904, P = .019). The audiometric results were visualized using Amsterdam hearing evaluation plots (Figure 3).
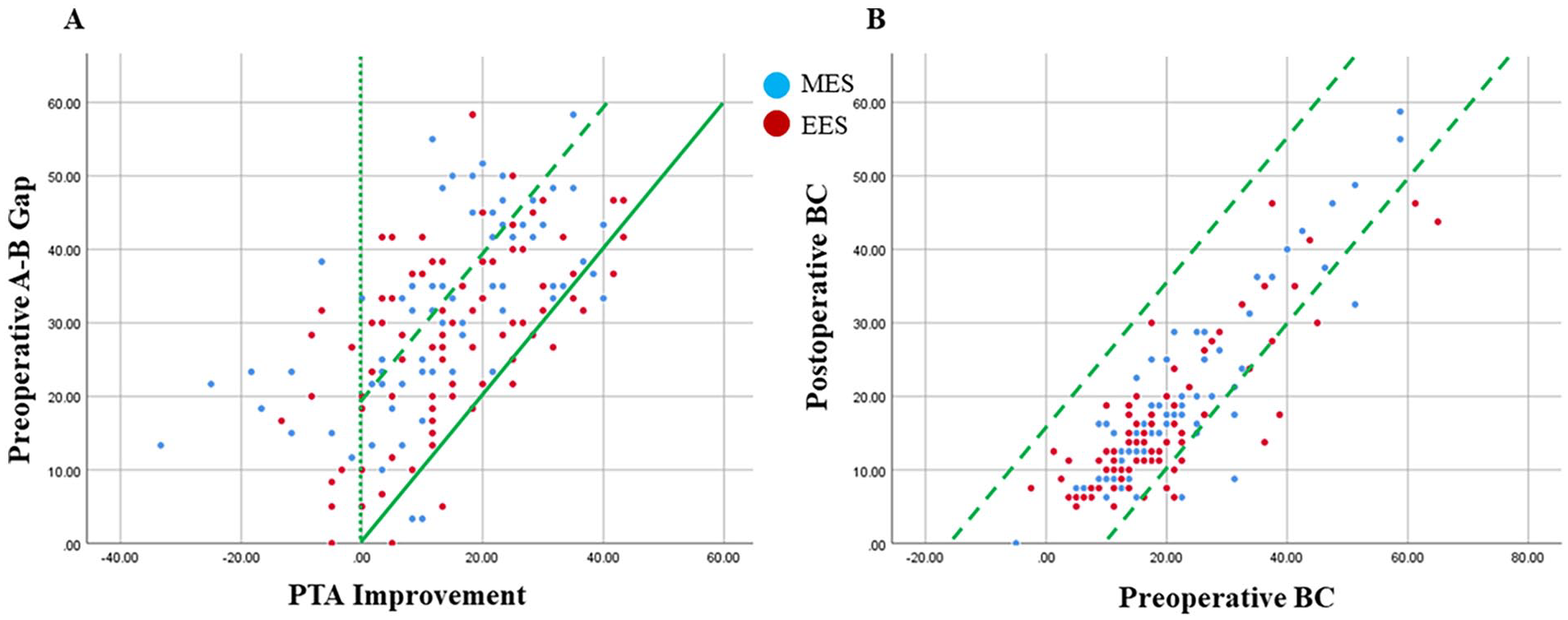
Hearing thresholds evaluated with the Amsterdam hearing evaluation plots. (A) PTA improvement (X-axis) compared with preoperative A-B Gap (Y-axis), the 2 diagonal lines indicated that ΔBC ≤10 dB, (B) Preoperative BC (X-axis) compared with postoperative BC (Y-axis), the solid diagonal line showed complete closure of the gap between preoperative AC and BC. The area between the lines indicated successful surgery with A-B Gap ≤20 dB. PTA, pure tone average; AC, air conduction; BC, bone conduction; A-B Gap, air-bone gap.
In contrast to MES group, EES group resulted in significantly shorter surgical time (t = 4.427, P = .000) and hospital length of stay (t = 5.934, P = .000) with stapes superstructure, as well as better postoperative PTA (t = 2.091, P = .039) (Table 3). MES and EES groups showed no significant differences in surgical time (t = 0.853, P = .397) or hospital length of stay (t = 1.217, P = .229) when cholesteatoma invaded stapes superstructure, but both groups exhibited favorable hearing improvement (Figure 4).
Hearing Improvement, Surgical Time and Hospital Stay With/Without Stapes Superstructure in EES and MES [Mean (95% CI) or n (%)].
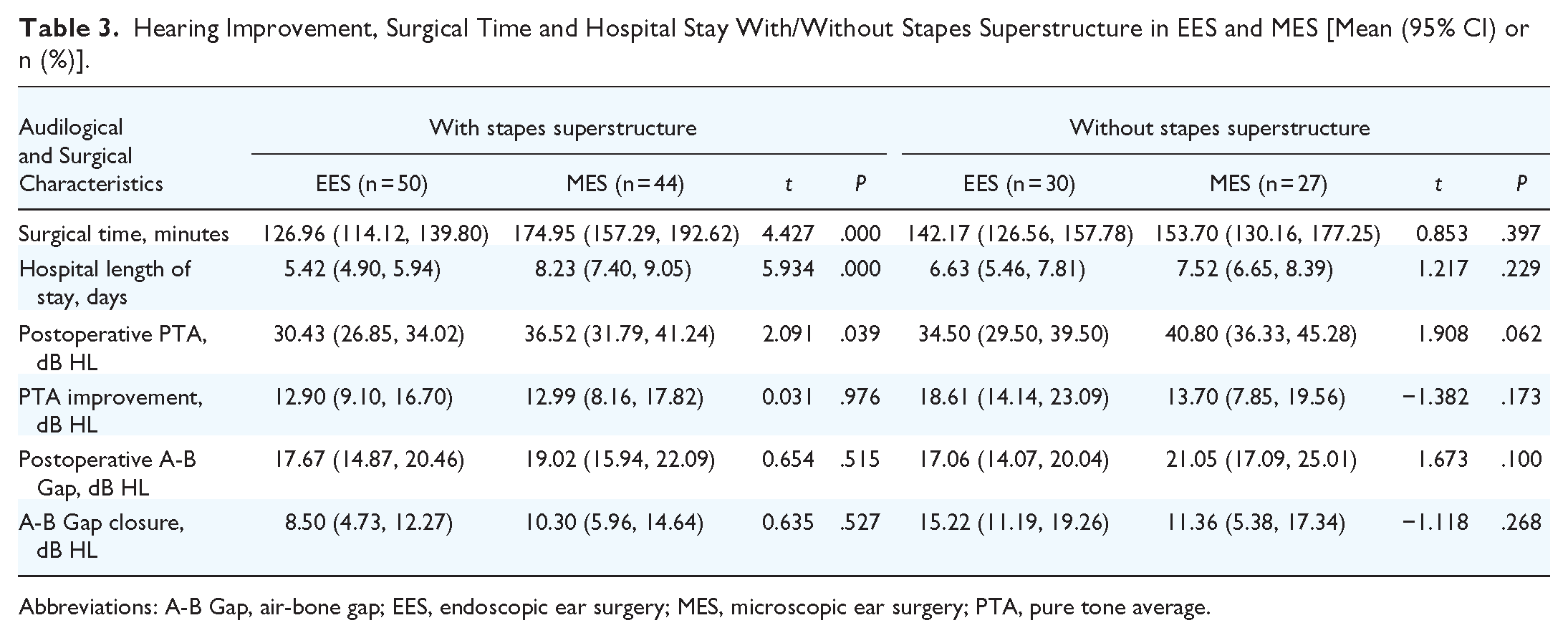
Abbreviations: A-B Gap, air-bone gap; EES, endoscopic ear surgery; MES, microscopic ear surgery; PTA, pure tone average.

Hearing thresholds evaluated with Amsterdam hearing evaluation plots. (A, C) PTA improvement (X-axis) compared with preoperative A-B Gap (Y-axis), (B, D) preoperative BC (X-axis) compared with postoperative BC (Y-axis), (A, B) with stapes superstructure, (C, D) without stapes superstructure. The area between the lines indicated successful surgery with A-B Gap ≤20 dB. PTA, pure tone average; BC, bone conduction; A-B Gap, air-bone gap.
This result was suggested that a tendency toward enhanced hearing improvement with stapes superstructure in EES compared to MES [EES: r = 0.676, Adjusted R2 = 0.446, F(1, 48) = 40.451, P < .001, y = 0.534x−10.257; MES: r = 0.610, Adjusted R2 = 0.358, F(1, 42) = 24.927, P < .001, y = 0.515x−12.489]. Our results indicated a trend toward greater hearing improvement with stapes superstructure in EES when preoperative A-B Gap ≤34 dB HL [r = 0.716, Adjusted R2 = 0.503, F(1, 48) = 50.596, P < .001, y = 0.738x−10.808], while MES demonstrated superior hearing improvement when preoperative A-B Gap ≥34 dB HL [r = 0.717, Adjusted R2 = 0.502, F(1, 42) = 44.344, P < .001, y = 0.849x−14.590].
Our findings indicated that EES may yield greater improvements in hearing outcomes in cases where preoperative PTA is below 79 dB HL, particularly in the absence of stapes superstructure [r = 0.616, Adjusted R2 = 0.357, F(1, 28) = 17.126, P < .001, y = 0.438x−4.637]. In contrast, MES may be more beneficial for patients with preoperative PTA >79 dB HL [r = 0.726, Adjusted R2 = 0.509, F(1, 25) = 27.904, P < .001, y = 0.684x−23.578]. Our findings also showed a trend toward a better hearing improvement in EES without stapes superstructure when preoperative A-B Gap ≤52 dB HL [r = 0.730, Adjusted R2 = 0.516, F(1, 28) = 31.870, P < .001, y = 0.723x−8.123], whereas MES have better hearing improvement when preoperative A-B Gap >52 dB HL [r = 0.752, Adjusted R2 = 0.548, F(1, 25) = 32.511, P < .001, y = 0.925x−18.611] (Figure 5).

Scatter plot and line graphs of (A, C) preoperative PTA (dB) and PTA improvement (dB), (B, D) preoperative A-B Gap (dB) and A-B Gap closures (dB), (A, B) with stapes superstructure, (C, D) without stapes superstructure. PTA, pure tone average; A-B Gap, air-bone gap.
Discussion
Cholesteatoma, a benign lesion, poses a higher risk of complications. MES was commonly used, but EES has become popular since Tarabichi’s success in 1997. 15 EES offers an improved visualization and requires less removal of normal structures for cholesteatoma removal and hearing restoration.
Surgery is the cholesteatoma treatment of choice, but there is controversy about whether microscopic or endoscopic surgery should be used. The Lempert I incision by EES and Lempert II incision by MES both showed similar A-B Gap closure. 16 Both EES and MES can obviously improve hearing level, but EES has better middle ear structural visibility and less post-operative pain compared to MES. 17 Studies showed that both EES and MES are effective for lesion removal and hearing improvement, especially in attic tympanic exposure. The efficacy of hearing improvement should be validated through a standardized multicenter study. Our research examined the impact of EES and MES on hearing improvement in cholesteatoma patients within a large-scale, multicenter setting, with EES demonstrating superior efficacy compared to MES.
This study showed that the success rate of grafts in EES was 8.95% higher than MES, as well as shorter surgical times and shorter hospital length of stays, which demonstrated that EES is an effective way in the surgical treatment of attic cholesteatomas, without the need for retro-auricular incisions. Notably, endaural MES with Shambaugh incision was also shown to be a viable surgical alternative that was minimally invasive and demonstrated a positive impact on wound healing processes, resulting in comparable success rates for grafts and audiological outcomes when compared to EES.18,19
Our study found a low recurrence rate of 3.45% for EES and 6.10% for MES, which may have been due to the short 3-year follow-up period. According to a previous study, cholesteatoma recurrence rates tended to rise over time, with 3.7% in 3 years and 11.8% in 5 years. 20 In this study, we used different angles of endoscopes to effectively visualize the middle ear, facilitating the thorough removal of any hidden cholesteatomas and reconstructing the middle ear ventilation routes to reduce the recurrence. Furthermore, cholesteatoma recurrence rates may be affected by multiple factors, such as surgical technique, type of patients, and mastoid development.
Meta-analysis found lower recurrence rates in EES compared to MES (RR = 0.51), particularly in attic cases (2.6% in EAONO/JOS stage 1 and STAM stage 1), and higher in mastoid cases (OR = 4.12).21,22 The 5-year recurrence rate was 16.4% in pediatric cholesteatoma, with no significant difference between EES and MES, but it was higher at 22.6% in primary acquired pars flaccida cholesteatoma, especially in EAONO/JOS stage 3 (HR = 5.06) and age <7 years (HR = 1.90). 23 The recurrence rate was 9% in EES, significantly lower than canal wall up at 22%, especially when the ossicular chain was preserved or reconstructed. 24 Moreover, Yasuo Mishiro found that canal wall down tympanoplasty recurrence was 3.9%, while intact canal wall tympanoplasty or canal wall reconstruction recurrence was 16.7%. 20
Our study confirmed that both EES and MES lead to improved hearing outcomes. This aligns with previous clinical findings that both EES and MES effectively remove attic cholesteatoma extending to the antrum area (stages Ib and II), and result in postoperative A-B Gap closures of approximately 17 dB and the average healing time of around 43 days. 25 In addition, our study has shown that EES demonstrated superior results in terms of postoperative PTA and A-B Gap compared to MES. Also, our study found that more ears in the EES group had a postoperative A-B Gap ≤20 dB HL compared to the MES group, with a similar trend for a postoperative A-B Gap ≤10 dB HL. This aligns with previous studies showing an average hearing improvement of 10 to 20 dB after tympanoplasty. Furthermore, the healthy or mildly edematous intratympanic mucosa has a great influence on achieving a postoperative A-B Gap of ≤20 dB HL. 26
However, the existing literature generally shows no significant differences between EES and MES when it comes to hearing improvement. For instance, Giuseppe Magliulo found no statistical difference in the average postoperative AC thresholds between EES and MES for patients suffering from chronic suppurative otitis media with attic cholesteatoma after 12.3 months of follow-up. 27 Meta-analysis indicated that EES and MES had similar A-B Gap closure in tympanoplasty with middle ear cholesteatoma, but EES had lower postoperative pain (OR = 0.2).22,28,29 Other meta-analyses have shown comparable audiological outcomes in pediatric cholesteatoma patients treated with either EES or MES.30,31 The discrepancy may be explained by different sample sizes, inclusion criteria, individual differences, and so on.
In our study, EES showed better hearing improvement, shorter surgical times, and shorter hospital length of stays than MES for cases of epitympanic cholesteatoma without stapes involvement. Another important finding was that preoperative PTA and A-B Gap values of AC ≤79 dB and/or A-B Gap ≤52 dB were found to have prognostic value, especially without stapes superstructure, supporting Dornhoffer’s (2001) idea that the malleus handle is crucial in hearing reconstruction. 32 Factors such as mucosal fibrosis, frequent drainage, revision ear surgery, and canal-wall-down mastoidectomy were associated with poorer hearing outcomes.
Therefore, EES offers advantages like less invasiveness, improved hearing, and quicker recovery, but it can be limited by 2D views. 33 Using 3D endoscopy in this surgery has shown to improve anatomical understanding and reduce complications. 34 Furthermore, bleeding in the middle ear can make the surgical site dirty and hard to see. Ways to prevent this include controlled hypotension, local anesthesia, tranexamic acid, and perfusion technique. 35 However, it is important to consider the risk of increased temperature in the middle ear during EES, as it can affect inner ear structures and audiometric outcomes.36,37 To address this issue, cooler light sources should be used instead of xenon.
In addition to the aforementioned drawbacks, it can be concluded that EES for cholesteatoma is a procedure worth widespread adoption due to its demonstrated efficacy, safety profile, and cost-effectiveness. This study is limited by its retrospective design, which may introduce selection bias and confounding factors due to the non-random selection of patients. Subsequent research should aim to validate these findings through multicenter randomized controlled trials (RCTs) to provide more robust evidence.
Conclusion
This study launched a large-scale, multicenter study to compare hearing improvement and determine the prognostic factors between EES and MES for attic cholesteatoma. This study found that EES is more effective for improving hearing in attic cholesteatoma cases, with shorter surgical times and hospital length of stays, particularly when the stapes superstructure is not involved. The range of preoperative PTA and A-B Gap have shown the prognostic value, with the preoperative PTA ≤79 dB and/or A-B Gap ≤52 dB associated with a more favorable prognosis in EES, especially in cases where the stapes superstructure is absent. This may serve as a favorable surgical indication for both EES and MES. Future research should prioritize well-designed multicenter RCTs to confirm the hearing improvement and prognostic factors of EES compared to MES, as retrospective studies have limitations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Featured Clinical Technique of Guangzhou (2023P-TS37, Haidi Yang).
