Abstract
Background
Dupilumab, an anti-IL4 receptor-α monoclonal antibody, was the first biologic to be approved in Canada for the treatment of Chronic Rhinosinusitis with Nasal Polyps (CRSwNP). In phase III clinical trials, it has demonstrated to be effective in reducing nasal polyp size and the severity of symptoms, improve disease-specific quality of life, and to have an acceptable safety profile. This study aims to present long-term follow-up data on disease-specific sinonasal outcomes of patients with CRSwNP who have been treated with dupilumab for up to 3 years in a real-world setting.
Methods
Retrospective review of electronic medical records of a single Canadian rhinology center evaluating disease-specific sinonasal outcomes that are routinely collected for clinical care. This study included all patients who received dupilumab for the treatment of CRSwNP and who had completed at least one follow-up visit. The Sino-Nasal Outcome Test (SNOT)-22 was used to evaluate treatment symptom improvement.
Results
Ninety-nine patients started dupilumab therapy during the study period. The mean SNOT-22 at the start of therapy was 61.1 (±22.91) At the time of the review, 65 patients had completed 1 year of therapy, 40 had completed 2 years of therapy, and 18 had completed 3 years of therapy. The mean SNOT-22 score at these timepoints was 16.75 (±13.86), 15.02 (±14.40), and 10.22 (±11.56), respectively.
Conclusion
This real-world study shows that in patients with CRSwNP treated with dupilumab, improvement in disease-specific quality of life seen after 1 year continues and can be maintained at 3 years of treatment.

Introduction
Chronic Rhinosinusitis with Nasal Polyps (CRSwNP) is predominantly an inflammatory disease characterized primarily by type 2 inflammation and it is diagnosed by the presence of a combination of clinical symptoms (nasal obstruction, congestion, rhinorrhea, reduction/loss of olfaction, and in some instances, facial pain) as well as the objective finding of nasal polyps. 1 CRSwNP is associated with substantial healthcare costs 2 and has a significant impact on health-related quality of life (HRQoL). 3 The impact on HRQoL is typically even greater in CRSwNP patients with associated comorbidities such as asthma and aspirin-exacerbated respiratory disease (AERD).3,4
Recommended initial treatment for CRSwNP is medical and it includes intranasal corticosteroid spray (INCS) complemented with a short course of oral corticosteroids and adjunct therapies such as saline rinses and antibiotics when warranted. 5 For those who fail medical treatment, surgery is recommended. 5 However, about 20% of patients who undergo surgery will require revision surgery within 5 years; this interval decreases with a higher number of surgeries.6,7
Until recently, treatment options for patients who failed both medical and surgical therapy were limited to additional trials of medical therapies and/or revision surgery. However, over the past 5 years, several biological therapies (dupilumab, mepolizumab, and omalizumab) have been approved for the treatment of nasal polyps in several jurisdictions including the United States, the European Union, and Canada.8-10
Dupilumab, an anti-IL4 receptor-α monoclonal antibody, was the first biologic to be approved in Canada, in August 2020. It was demonstrated to be effective in reducing nasal polyp size and the severity of CRSwNP symptoms and to have an acceptable safety profile. 8 In phase III clinical trials, treatment with dupilumab also resulted in significant improvements in disease-specific quality of life demonstrated by clinically significant improvement in the Sino-Nasal Outcome Test (SNOT)-22. 11
Since the introduction of dupilumab as a treatment option for CRSwNP, several studies have investigated its effectiveness outside of clinical trials. To date, the results of these real-world studies support the findings of the phase III trials with CRS symptom improvement, a reduction of nasal polyp score, and improvement in disease-specific quality of life.12-15 However, currently there is no data available on treatment outcomes beyond 12 months.
This study aims to present long-term follow-up data on disease-specific sinonasal outcomes of patients with CRSwNP who have been treated with dupilumab for up to 3 years.
Methods
Data Sources
We conducted a retrospective review of electronic medical records of a single Canadian rhinology center. We collected data on patient characteristics, diagnosis, history of sinus surgery, medical treatment for CRSwNP, dupilumab start date, SNOT-22, and Asthma Control Questionnaire (ACQ)-6 scores at baseline and during follow-up. Recorded adverse events were also collected. Data were collected and managed in an Excel spreadsheet (Microsoft).
Study Population
Patients diagnosed with CRSwNP who had undergone prior surgery for CRSwNP or who had a medical contraindication to surgery and who had not achieved optimal symptom control with adequate medical therapy were considered for dupilumab therapy at our center. Patients who started therapy and had completed at least one follow-up visit between September 1, 2020, and January 31, 2024, were included in the study.
Reference Outcome
At our center, the SNOT-22 questionnaire is routinely administered to patients with CRSwNP to monitor progress and evaluate treatment outcomes. The minimal clinically important difference for the SNOT-22 is 8.9 points. 16 Patients with CRSwNP with concomitant asthma also routinely complete the ACQ-6. At our center, patients treated with biologics are followed up at approximately 3 and 6 months (12 and 24 weeks) after the start of therapy and then yearly after the first year of therapy.
Statistical Analysis
We present a descriptive analysis of patient characteristics and the changes in SNOT-22 and ACQ-6 scores during the period of this chart review. Categorical variables were summarized using frequency and proportions; continuous variables were summarized using means and standard deviations. Means were compared using t tests. Analysis was performed using Stata 12 (StataCorp LP).
Ethical Statement
All methods were carried out in accordance with relevant guidelines and regulations. The study was approved by the research ethics board (IRB #Pro00057336).
Results
A total of 99 patients started dupilumab and completed at least one follow-up visit during the study period. The mean age for the initial cohort was 53.75 years, and 48 (48%) were female. Eighty-five patients (85%) had a diagnosis of asthma and 37 (37%) had AERD. Ninety-two patients (92%) had undergone surgery for nasal polyps. At the time of the application for dupilumab, all patients were receiving intranasal corticosteroids spray ± saline rinse or budesonide irrigations. The baseline characteristics of the cohort are detailed in Table 1.
Baseline Characteristics.

Abbreviation: AERD, aspirin-exacerbated respiratory disease.
Outcomes With Dupilumab Therapy
Ninety-five patients completed a SNOT-22 questionnaire before the start of dupilumab treatment. The mean baseline score was 61.1 (±22.91). Eighty-four patients completed a SNOT-22 after 28 weeks of treatment. At this timepoint, the mean SNOT-22 score decreased by 37 points to 23.29(±18). At the time of the review, 65 patients had completed 1 year of therapy, 40 had completed 2 years of therapy, and 18 had completed 3 years of therapy. The mean SNOT-22 score at these timepoints was 16.75 (±13.86), 15.02 (±14.40), and 10.22 (±11.56), respectively (Figure 1).

Mean SNOT-22 score in patients with CRSwNP treated with dupilumab. SNOT, Sino-Nasal Outcome Test; CRSwNP, Chronic Rhinosinusitis with Nasal Polyps.
SNOT-22 scores were grouped by domains. 17 The rhinologic symptoms domain (maximum score = 30) at baseline was 20 (±6.24) and decreased by 13 points to 6.82 (±5.19) at the 28 week timepoint. The change was maintained through years 1, 2, and 3 with mean scores of 5.5, 4.9, and 3.3, respectively. The improvement observed in all domains exceeded the MCID 18 at the 28 week timepoint and was maintained in all domains through years 1, 2, and 3. Table 2 shows the SNOT-22 scores stratified by domain for all timepoints.
SNOT-22 Scores Grouped by Domains.

Abbreviation: SNOT, Sino-Nasal Outcome Test.
The individual SNOT-22 items with the highest baseline mean score were “Decreased sense of smell/taste” 4.29, “Nasal obstruction” 3.8, and “Need to blow your nose” 3.46. At the 1 year timepoint, the mean scores for these items decreased to 1.16, 1.06, and 1.04, respectively. At the 2 year timepoint, the mean score for “Decrease sense of smell/taste” was 1.07, for “Nasal obstruction” 0.70, and for “Need to blow your nose” 1.02.
The baseline SNOT-22 score for patients with comorbid asthma was 62.25 and for those without asthma was 54.64. This difference was not statistically (P = .25) significant. Sixty-four patients completed an ACQ-6 questionnaire at baseline. The mean ACQ-6 score at this timepoint was 1.68 (±1.3), representing poorly controlled disease. The ACQ-6 scores at 6 months, 1, 2, and 3 years were 0.66, 0.61, 0.39, and 0.38, respectively. The changes compared to baseline are statistically (P < .05) and clinically significant, given they are greater than 0.5. At baseline, 20 patients (31.2%) had well-controlled asthma (ACQ-6 score 0-0.75), at the 1 year timepoint, 34 patients (64.15%) had well-controlled asthma (see Table 3 and Figure 2).
ACQ-6 Mean Scores in Patients Receiving Dupilumab Therapy for CRSwNP.

Abbreviations: ACQ, Asthma Control Questionnaire; CRSwNP, Chronic Rhinosinusitis with Nasal Polyps.
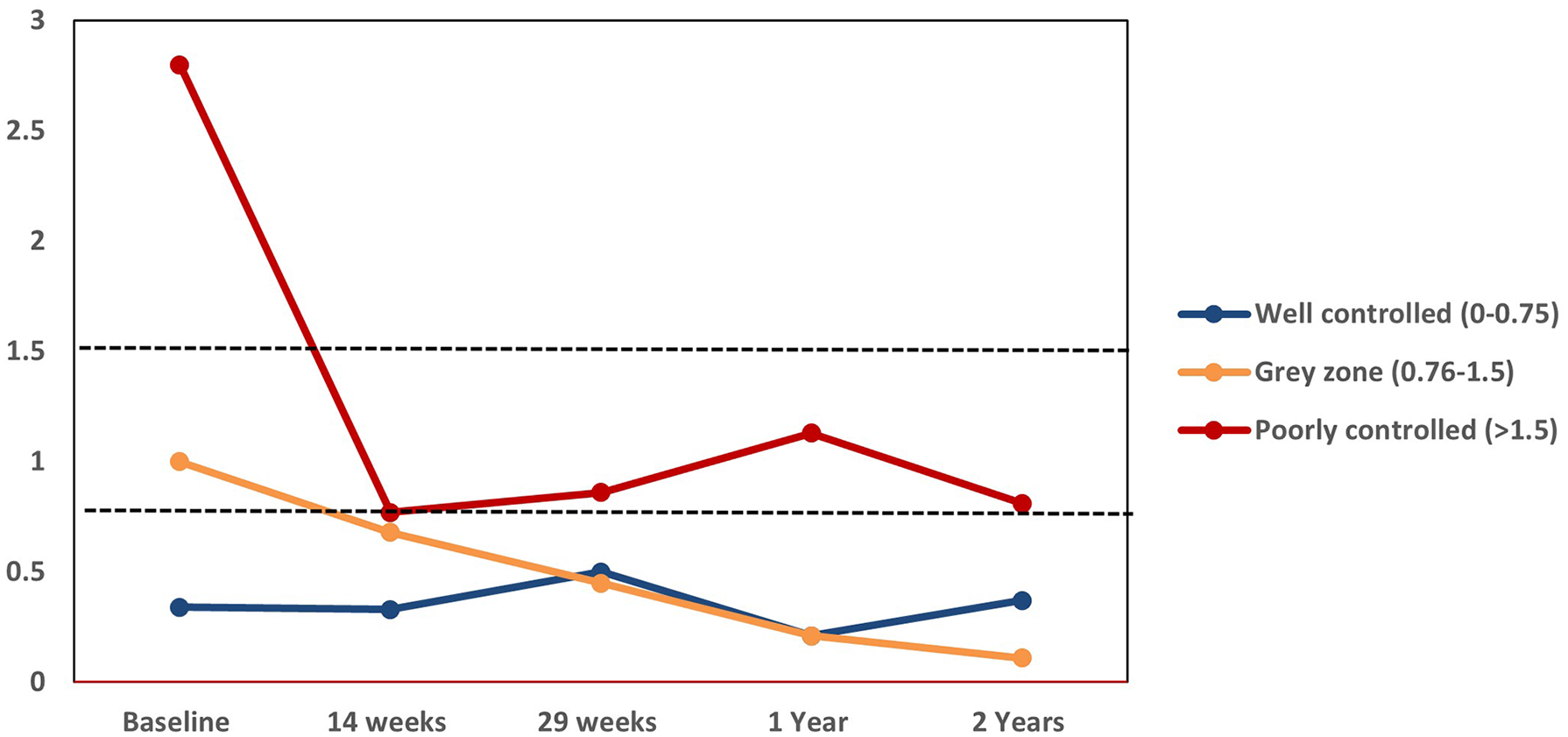
Change in mean ACQ-6 scores with dupilumab therapy based on baseline score severity. Well-controlled asthma (0-0.75), gray zone (0.76-1.5), and poorly controlled asthma (>1.5). ACQ, Asthma Control Questionnaire.
Adverse Events Related to Dupilumab Therapy
Sixteen patients (16%) reported one or more adverse events. The most common adverse event was injection site reaction (7 patients or 7%). These reports were received between 1 and 14 months after the start of dupilumab. The reactions were minor and resolved within a few hours after the injection. Six patients reported arthralgia. The most commonly affected joints were knee, ankle, and shoulder reported by 2 patients each; elbow, jaw, and hands reported by 1 patient. Reports of arthralgia were received between 1 and 13 months after the start of therapy. Five patients stopped dupilumab therapy due to adverse events. Three patients stopped due to new-onset arthralgia. One patient stopped due to subjective “decreased effect,” and one patient stopped due to unspecified side effects that affected their daily life (see Table 4).
Adverse Events.

Dose Modification
To lower the cost of treatment, the dose of dupilumab was changed to 300 mg every 4 weeks for 2 patients who were paying for the medication out of pocket. One of these patients further reduced the dose to 300 mg every 6 weeks on their own. These patients did not report a worsening of symptoms after the dose change.
The dose of dupilumab was changed to 300 mg every 4 weeks for 3 patients who reported adverse events; however, one of these patients went back to the original dose due to worsening of CRSwNP symptoms. One additional patient had the dose changed to 300 mg every 4 weeks at their personal request.
Discussion
Dupilumab has been shown to be effective at improving CRSwNP symptom control and reducing nasal polyp size. 8 It has also been demonstrated to improve disease-specific quality of life 11 ; however, long-term data beyond 52 weeks are not currently available. Here, we expand on our previous report 19 on the use of dupilumab for the treatment of CRSwNP in a real-world cohort at a tertiary rhinology clinic. In this report, we present long-term disease-specific quality of life of patients who have been treated with dupilumab for up to 3 years.
Patients in this study had severe symptoms of CRSwNP before the start of therapy as shown by a high mean baseline SNOT-22 score of 61.1. After 1 year of therapy, the mean SNOT-22 had decreased to 16.75, a change of 44.35 points, indicating both clinically significant and statistically significant (P = .00) improvements from baseline. The improvement seen in this real-world cohort is consistent with the change seen in the Sinus-52 trial 8 where at week 52 those in the dupilumab (every 2 weeks) arm reported a mean SNOT-22 score of 21.67, a decrease of 28 points from the baseline score of 50.16. In our cohort, improvement was maintained at the 2 year timepoint with a mean SNOT-22 score of 15.02. After 3 years of treatment, the mean SNOT-22 was 10.22. This shows that the improvements in disease-specific quality of life are sustained over time. All SNOT-22 domains showed clinically significant improvements that were maintained at the 3 year timepoint. “Decrease of sense of smell/taste” was the individual SNOT-22 item with the highest score at baseline indicating a severe problem, at 4.29. After 1 year of therapy, the mean score decreased to 1.16 (±1.5) and the improvement was also maintained at 3 years. An analysis of the SNOT-22 data of the phase III SINUS studies showed sustained improvements between 24 and 52 weeks of treatment and the authors suggest that the maximum treatment effect may still to be achieved at week 52. 11 In our real-world clinical experience, the mean SNOT-22 scores continued to improve beyond 52 weeks; however, this further decrease in the mean score of the cohort appears not to be clinically significant.
In patients with comorbid asthma, we observed both a clinically and a statistically significant improvement in disease control. The proportion of patients with well-controlled asthma increased from 31% at baseline to 64%, 83%, and 81% after 1, 2, and 3 years of therapy, respectively. This shows that patients treated with dupilumab who have concurrent asthma receive a considerable improvement in their asthma control as a secondary benefit to monoclonal antibody treatment, given the similarities in inflammation between the 2 diseases, further supporting the one airway hypothesis. 20
Our cohort had a higher proportion of patients with comorbid asthma compared to the cohort in the Sinus-24 and Sinus-52 studies, 8 85% and 59%, respectively. In our cohort, patients with comorbid asthma had a higher baseline SNOT-22 score (62.25) than those who did not have asthma (54.64); however, this difference is not statistically significant. The mean ACQ-6 score decreased from 1.68 at baseline to 0.61, 0.39, and 0.38 at 1, 2, and 3 years, respectively.
For patients who initially had poorly controlled asthma at baseline, the group’s ACQ-6 mean score was 0.86, 1.13, and 0.81 at 6 months, 1, and 2 years, respectively. These changes are both clinically significant and statistically significant from baseline. Patients who had well-controlled asthma before initiating dupilumab for their CRSwNP disease continued to have well-controlled asthma. Patients with asthma control in the gray zone at baseline improved with dupilumab therapy for their CRSwNP, to have well controlled asthma within 14 weeks of having initiated therapy (see Figure 2).
In a long-term efficacy study in patients with asthma with or without CRSwNP, dupilumab provided a similar reduction in asthma exacerbations, improved forced expiratory volume in 1 second, and improved ACQ-5 and Asthma Quality of Life questionnaire scores in both groups. 21 These improvements were maintained after 3 years of treatment; however, in this study, CRSwNP was self-reported and the severity of CRSwNP was not measured. Our real-world study shows that asthma control improvements are maintained after 2 years of treatment with dupilumab in patients with confirmed CRSwNP. The improvement in asthma control observed in our real-world study is supported by the shared type 2 inflammatory process that is characteristic of both asthma and CRSwNP.
Our real-world study had a cumulative exposure to dupilumab of 198 patient/years (5125 doses); during the study period, 16 patients reported one or more adverse events. The most common events were injection site reactions and arthralgia. None of the adverse events were considered serious. Five patients stopped dupilumab due to adverse events, three of them were related to arthralgia.
The TRAVERSE study 22 assessed the safety of dupilumab in patients with moderate to severe asthma with cumulative exposure to dupilumab of 431.7 patient/years. This study found that the safety and tolerability of long-term treatment were consistent with the known safety profile of dupilumab and did not find new patterns of incidence of treatment-emergent adverse events. Additional long-term safety data on the CRSwNP population are needed.
Our study had several limitations. Given that dupilumab was first approved in Canada in August 2020, the number of patients with 2 and 3 year data is small. This study does not include certain objective outcomes such as nasal polyp score or Lund-Mackay computed tomography (CT) score that could be correlated to the SNOT-22 scores. However, serial CT scans are not routinely used for assessing improvements in paranasal sinus inflammation secondary to medical therapy. Furthermore, nasal polyp score is not an outcome that determines treatment. It is patient symptom and quality of life concerns which drive patient treatment.
Conclusion
This real-world study shows that in patients with CRSwNP treated with dupilumab, improvement in disease-specific quality of life seen after 1 year continues to improve and can be maintained at 3 years of treatment. Long-term treatment with dupilumab in CRSwNP patients appears to be consistent with the known safety profile generated from shorter patient observation periods of other studies. Larger prospective long-term studies are required to evaluate the extended benefits of this therapy and to monitor the development of late-onset events in this patient population.
Footnotes
Availability of Data and Materials
Data will be made available from the corresponding author on reasonable request.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.J.K.: Site investigator for the LIBERTY NP SINUS-52 study. Prior Advisory Board member (Sanofi), Speaker bureau GSK, Sanofi. Research funding CIHR, PSI. A.L.: no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study was approved by Advarra IRB. Protocol # Pro00057336.
