Abstract
Background
Diagnostic dilemma between clinical Meniere’s disease and radiological endolymphatic hydrops (EH) has emerged since the introduction of hydrops magnetic resonance imaging (MRI). The aim of this study is to explore the potential application of hydrops MRI on diagnosing the EH.
Methods
This review was developed from peer-reviewed articles published in those journals listed on journal of citation reports. The MEDLINE database of the US National Library of Medicine, Scopus, and Google Scholar were used to collect articles based on the guidelines (PRISMA 2020 statement) for reporting reviews.
Results
Initially, 470 articles were retrieved from 1983 to 2023, and 80 relevant articles were ultimately selected. The sensitivity (69%-92%) and specificity (78%-96%) values varied from each laboratory for detecting EH via hydrops MRI, probably due to candidate selection and the grading system employed.
Conclusion
The application of hydrops MRI allows (1) differentiation between EH and sudden sensorineural hearing loss; (2) determination of the affected side of EH; and (3) confirmation of the diagnosis of EH concomitant with other disorders. Notably, not all differentials for EH can be visualized on MR images. One of the existing gaps to be filled is that updated hydrops MRI fails to identify distortion, that is, rupture, collapse, fistula, or fibrosis of the inner ear compartments, akin to what histopathological evidence can demonstrate. Hence, enhanced ultrahigh resolution of hydrops MRI is required for demonstrating fine structures of the inner ear compartments in the future.

Introduction
History Background
The first histopathological report of Meniere’s disease with the hallmark of endolymphatic hydrops (EH) was published in September 1938 by Hallpike and Cairns in the United Kingdom. 1 However, Japanese researchers argued that Yamakawa of Osaka university had already described EH as a pathological correlate in a patient with Meniere’s disease, which was published in the Journal of Japanese Society of Otorhinolaryngology in April 1938, with a German abstract. 2 Paparella et al 3 later suggested that Hallpike and Cairns in 1938 as well as Yamakawa in 1938 independently detected EH in autopsy specimens of Meniere’s disease.
Nevertheless, the first magnetic resonance (MR) images of EH in Meniere’s disease patients dedicated to Nakashima and colleagues of Nagoya university in Japan, 4 although early methodology in 2007 has some limitations. First, an intratympanic contrast injection is rather invasive. Second, a contrast agent injected into the middle ear is off-label use. Third, 1 contrast agent injection is allowed for detecting 1 ear at 1 time. Later, Naganawa et al 5 of the department of radiology at Nagoya University altered the intratympanic injection route into intravenous route. This alteration necessitates delayed scan, conducted after a 4 hour interval, to allow the intravenous contrast agent diffuse into the perilymph and enhance the imaging.
Naganawa et al
5
devised a novel method termed “HYDROPS” technique, which is derived from the following sentence:
On the other hand, the American Academy of the Otolaryngology—Head and Neck Surgery (AAO-HNS) categorized the Meniere’s disease into 4 types in 1995, namely, possible, probable, definite, and certain Meniere’s disease. 10 Of them, certain Meniere’s disease is defined as “definite” Meniere’s disease coupled with histopathological confirmation of EH. Given that Meniere’s disease is a benign disease and the temporal bone of a Meniere’s disease patient is hard to obtain unless the patient has passed away, the term “certain” Meniere’s disease has been eliminated from the category of Meniere’s disease by both the AAO-HNS and the Barany Society in 2015.11,12 However, Nakashima et al 13 suggested that hydrops magnetic resonance imaging (MRI) could be substituted for histopathological evidence, preserving the category of “certain” Meniere’s disease.
Rationale and Objective
Clinically, diagnosis of Meniere’s disease was based on the guidelines proposed by the AAO-HNS and the Barany Society.10,12 Definite Meniere’s disease is defined as 2 or more episodes of vertigo exceeding 20 minutes, tinnitus, or aural fullness, and documented hearing loss in the recent 6 months after excluding other causes.
However, diagnostic dilemma between clinical Meniere’s disease and radiological EH has emerged since the introduction of hydrops MRI.14,15 For instance, if 1 patient clinically diagnosed as Meniere’s disease but hydrops MRI fails to demonstrate EH, it poses a challenge for clinicians, raising questions about either a potential misdiagnosis of Meniere’s disease or limitations in radiological EH detection. Hence, this article provides an overview to correlate radiological EH with Meniere’s disease and its variants. The aim of this study is to explore the potential application of hydrops MRI on diagnosing the EH.
Methods
Study Design
This review was conducted according to the Preferred Reported Items for Systematic Reviews and Meta-Analyses (PRISMA 2020 statement) guidelines. 16 Based on the methodology for conducting a systematic review, the PICO question is described as follows, and the studies were selected following these characteristics:
P (Participants): Patients with a diagnosis of Meniere’s disease and its variants were enrolled.
I (Intervention): An inner ear test battery, hydrops MRI, or temporal bone histopathological study was performed.
C (Comparison): Results of the inner ear test battery were correlated with the degree of EH.
O (Outcome): Estimation of positive rate of EH by hydrops MRI correlates with results of the inner ear test battery. Data analysis of hydrops MRI is sufficient to calculate sensitivity and specificity.
Search Strategy
Through the PubMed database of the US National Library of Medicine, Scopus, and Google Scholar, literature written in English from 1983 to 2023 were initially surveyed by 2 independent reviewers. The search terms from databases comprised “endolymphatic hydrops,” “Meniere’s disease,” and its variants such as
“secondary endolymphatic hydrops,” “delayed endolymphatic hydrops,” “embryopathic endolymphatic hydrops,” or “asymptomatic endolymphatic hydrops.” Preference was given to selecting papers that focused on the diagnosis of Meniere’s disease or endolymphatic hydrops from a radiological or histopathological perspective.
Inclusion Criteria
Meniere’s disease confirmed by hydrops MRI or histopathological evidence.EH (including primary, secondary, delayed, embryopathic, and asymptomatic hydrops) confirmed by hydrops MRI or histopathological evidence.
Exclusion Criteria
Studies unrelated to Meniere’s disease or EH.
Studies without mentioning audiovestibular results.
Studies published in non-English language.
Studies published in non-SCI journals.
Duplicate reports.
Case reports.
Data Extraction
After conducting a bibliographic search in databases, duplicate entries were removed, and articles with titles or abstracts unrelated to the objectives of this review were excluded. Then, each article underwent a thorough review to extract the most relevant information aligned with the objectives of the review. Following the framework of this review, 2 independent clinicians meticulously reviewed the titles and abstracts of selected articles to evaluate their eligibility. There was no discordance between the 2 reviewers.
Data Analysis
Since most patients with inner ear disorders revealed nonspecific inner ear symptoms, that is, vertigo, hearing loss, or tinnitus, clinical criteria coupled with audiovestibular testing is essential for diagnosing Meniere’s disease after excluding other disorders. Studies with equivocal cases between Meniere’s disease and sensorineural hearing loss (SNHL) of sudden onset should be further confirmed by MRI. If not, those studies were also removed. To estimate the diagnostic accuracy of hydrops MRI, sensitivity and specificity values for the detection of EH were investigated.
Certainty Assessment
Quality assessment of the hydrops MRI was performed by 2 trained clinicians. Disputes were resolved by discussion with a third specialist. Articles with a poor quality of EH demonstrated on hydrops MRI were excluded. Only articles showcasing a clear resolution of EH on hydrops MRI were considered for certainty assessment.
Results
Literature Search
Initially, 470 articles were retrieved from 1983 to 2023. Following the exclusion of duplicate reports (n = 29), non-English reports (n = 38), and non-SCI journals (n = 19), 384 records were assessed for eligibility. Subsequently, studies that contained unrelated articles (n = 197), case reports (n = 24), or articles covering a similar topic published in a later period (n = 83) were eliminated from consideration. As a result, 80 relevant articles were ultimately chosen (Figure 1).
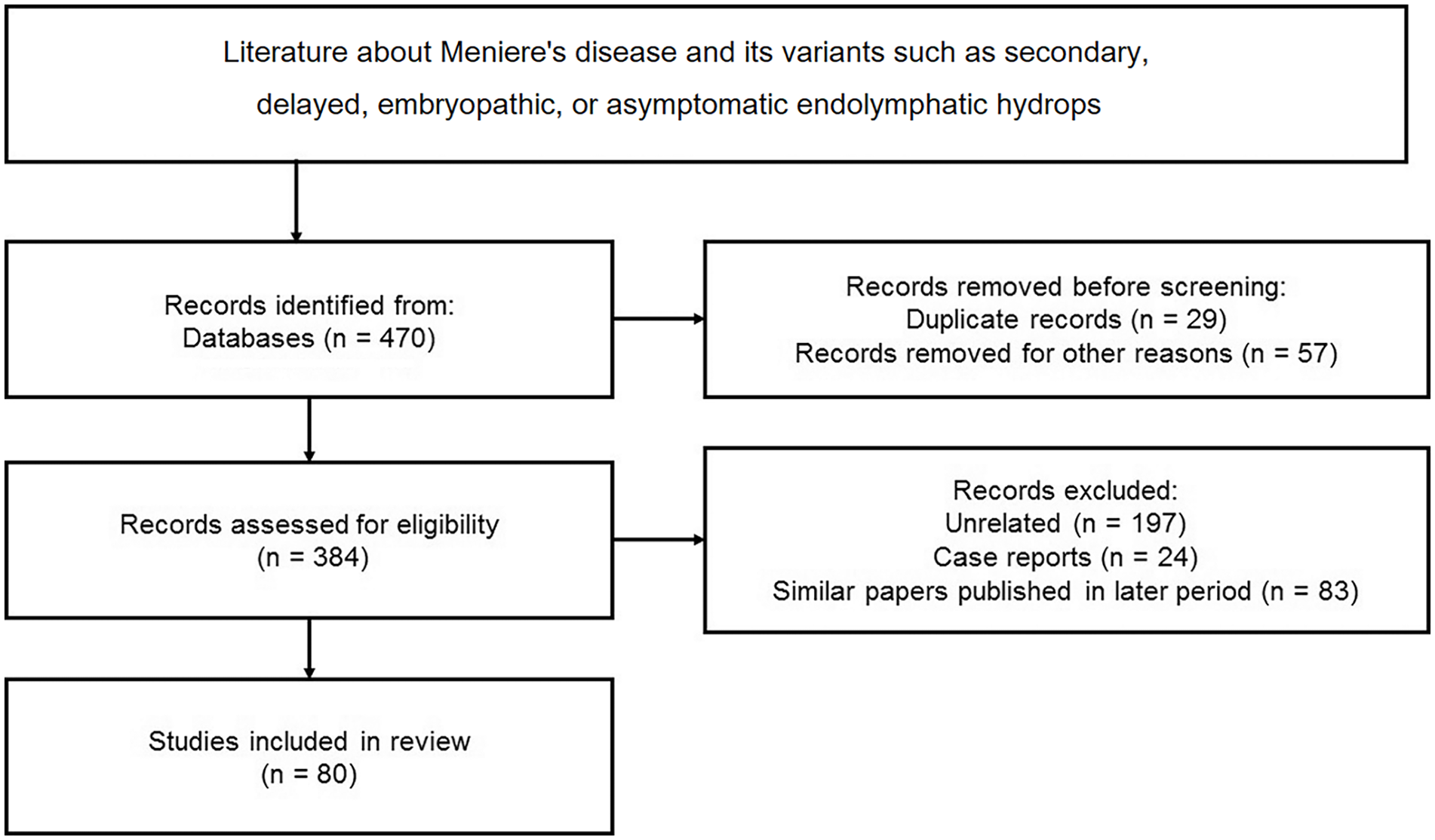
Illustration of how the selection of articles is made based on the guidelines of PRISMA 2020 statement with additional details of the number of studies. PRISMA, Preferred Reported Items for Systematic Reviews and Meta-Analyses.
Grading System Utilized
Grading system for cochlear EH
The grading system of cochlear EH was initially classified into 3 types, namely, no, mild, and significant hydrops by Nakashima et al. 17 Subsequently, Barath et al 7 proposed a 3-grade system, that is, Grades 0 (no hydrops), I (mild hydrops), and II (significant hydrops). Grade 0 means no displacement of the Reissner’s membrane. Grade II indicates that area of the cochlear duct > that of the scala vestibuli, while vice versa is referred to Grade I. Other researchers utilized endolymph volume ratios for assessing the EH.18,19 For instance, Kahn et al 20 categorized cochlear EH as none and Grade I. The latter indicates that the area of the endolymphatic space exceeded the area of the scala vestibuli (Table 1).
Compilation of Sensitivity and Specificity Values for the Detection of Endolymphatic Hydrops Using Hydrops MRI.

Grades of cochlear EH have been correlated with hearing thresholds at low and mid frequency despite which grading system was used.21,22 Like histopathological evidence, cochlear EH of varying severity is noted at various turns. Nakashima et al 17 thus suggested that the highest grade of EH on hydrops MRI is utilized for interpretation, which may not match the responsible site for pure tone average from 500, 1000, 2000, and 3000 Hz. Lin et al 23 suggested using sum of 3 low-frequency (125, 250, and 500 Hz) hearing levels >100 dB to predict positive cochlear EH on hydrops MRI. In contrast, that <100 dB, MRI should be postponed since small-sized EH may be overlooked. Nevertheless, human measurement for grading of EH is prone to variability. One may imagine a future artificial intelligence to calculate the volumes of endolymph and perilymph based on the contrast density gradient between the 2.
Grading system for vestibular EH
The grading system of vestibular EH was firstly proposed by Nakashima et al 17 using the ratio of the area of endolymphatic space to the vestibule (sum of the endolymphatic and perilymphatic spaces), and was categorized into 3 types, namely, no (<33%), mild (33%-50%), and significant (>50%) hydrops. However, this semiquantitative grading system pooled the saccular and utricular hydrops together. Alternatively, another grading system utilized saccule-to-utricle ratio inversion.24,25 Kahn et al 20 separately assessed the saccular and utricular hydrops using a 3-grade system, that is, none, Grades I and II. Following this, Bernaerts et al 9 suggested a 4-grade system for vestibular EH, which has been widely adopted in most laboratories (Table 1). Accordingly, Grade 0 (no vestibular hydrops) means that areas of the saccule and utricle are <50% of that of the vestibule. Grade I indicates area of the saccule ≥ that of the utricle, but they have not yet merged together. Grade II implies confluence of both saccule and utricle with rim enhancement, while Grade III is defined as confluence without rim enhancement (Figure 2).

Male, 31 years, Meniere’s disease, right. Hydrops MRI demonstrates endolymphatic hydrops at the cochlea (C), Grade II; saccule (S), Grade III; and utricle (U), Grade III on the right ear. Left ear, control. Solid arrows: present hydrops; dashed arrow: no hydrops. MRI, magnetic resonance imaging.
The saccular and utricular hydrops could be evaluated by cervical vestibular-evoked myogenic potential (cVEMP) and ocular VEMP (oVEMP) tests, respectively. 26 Katayama et al 27 reported 5 patients with extremely large vestibular EH showing absent cVEMP. Yet, they pooled both saccular and utricular hydrops together to correlate with the cVEMP. Kahn et al 20 reported that there is no correlation between vestibular EH and cVEMP/oVEMP results using loud sound stimuli. However, oVEMP elicited by loud sound may result in small and unreliable responses, especially in comparison to the oVEMP induced by bone vibration stimuli. 28 Later, Lin et al 29 conducted cVEMP and oVEMP tests using bone vibration stimuli, but did not find any significant correlation between cVEMPs and saccular hydrops, nor between oVEMPs and utricular hydrops.
Limitation of Hydrops MRI
The cVEMP/oVEMP results in Meniere’s disease patients consisted of augmented, normal, reduced, and absent responses, depending on individual saccule/utricle is either enlarged (distending to the footplate), normal, reduced, or collapsed contour, respectively. 30 Direct contact between the dilated saccule/utricle and the stapedial footplate may cause either reduced stapedial mobility or dampened acoustic energy transmission to the inner ear. 31 Thus, utricular/saccular EH contacting the oval window may show augmented oVEMP/cVEMP, respectively, which has high specificity and positive predictive value in differentiating Meniere’s disease from other disorders. 31
Nevertheless, Sluydts et al 32 reported that only the highest grades of cochlear and vestibular EH were related to a decline in cochleovestibular function. The reason is, probably because updated MRI fails to identify reduced volume or collapsed wall of the saccule/utricle. Restated, distorted contour of the saccule/utricle may be interpreted as absence of EH on hydrops MRI, resulting in discrepancy between cVEMP/oVEMP and saccular/utricular EH, respectively. To expand the applicability of hydrops MRI in neuro-otology, there is a need for further advancement in techniques that can provide ultrahigh resolution and enhance the visualization of fine structures within the inner ear compartments.
Sensitivity and Specificity Values of Individual Studies
Barath et al 7 showed a sensitivity of 73% and a specificity of 78% for confirming EH in Meniere’s disease patients comprising definite, probable, and possible types. In contrast, Bernaerts et al 9 reported higher sensitivity (80%) and specificity (94%) on hydrops MRI for visualizing EH in definite and probable types, but excluding possible type, Meniere’s disease patients. Lin et al 23 proposed a sensitivity of 69% and a specificity of 83% in MRI for demonstrating EH in primary, secondary, and embryopathic EH patients. Table 1 provides a compilation of the sensitivity (69%-92%) and specificity (78%-96%) values for detecting EH using hydrops MRI from various laboratories, which can differ based on the candidate selection and the grading system employed.
Quality Assessment
In a study of 22 temporal bones of Meniere’s disease donors, Okuno and Sando 33 found that EH predominantly occurs at the cochlea (100%), followed by the saccule (77%), utricle (50%), and semicircular canals (27%). Yet, a surprisingly high rate (95%) of positive vestibular EH on hydrops MRI was reported in Meniere’s disease patients after vertiginous attack, 34 which is opposed to the prevalence of EH in histopathological study. 33 This phenomenon is likely attributed to an inadequate grading system used to define vestibular EH. 35
Discussion
Primary EH (Meniere’s Disease)
Schuknecht and Gulya 36 classified the EH into 2 distinct forms, symptomatic and asymptomatic. Within the symptomatic EH, there are 3 types, that is, primary EH, an idiopathic process termed Meniere’s disease; secondary EH including delayed EH, which is an acquired insult (inflammatory or traumatic); and embryopathic EH as a consequence of an embryopathic process. On the other hand, asymptomatic EH is considered as a quiescent pathological condition, and can only be identified by histopathological study before the development of hydrops MRI.37-39
While EH is considered the hallmark of Meniere’s disease in temporal bone histopathology, autopsy specimens from Meniere’s disease donors have demonstrated additional features such as hydrops rupture, collapse of labyrinthine membranes, and fibrosis in the inner ear compartments.40-42 However, these 3 aforementioned pathological conditions fail to be identified on updated hydrops MRI, leading to a discrepancy between pathological and radiological results. As Meniere’s disease patients with stable symptoms may also exhibit unchanged EH, whereas in some cases, resolution of EH can be observed on hydrops MRI following the recovery of hearing loss. 43 Thus, hydrops MRI may serve an additional biomarker for detecting the dynamic status of Meniere’s disease. 44
Delayed EH
Those patients who had precedent inflammatory or traumatic insult may develop vertiginous attack and/or fluctuating hearing loss in later years and were termed secondary EH.45-48 Delayed EH represents a variant of secondary EH.
The mechanism of delayed EH is proposed as a sufficiently major labyrinthine insult that causes profound SNHL, while preserving vestibular function, leading to delayed atrophy or fibrous obliteration of the endolymph resorption system. 49 Kamei 50 classified the delayed EH into 3 types, namely ipsilateral, contralateral, and bilateral types. The bilateral type has 2 forms: either both ears are affected by the ipsilateral type, or 1 ear affected by the ipsilateral type and the other ear is affected by the contralateral type. The latter form is rarely seen in the past, likely due to a lack of robust evidence. It is until the development of hydrops MRI that various types and forms of delayed EH could be identified.51-53 Since severe type EH was frequently demonstrated in delayed EH patients, the hydrops MRI thus serves not only as a sensitive tool for diagnosing delayed EH, but also as an accurate tool for classifying the affected side of delayed EH, especially the bilateral type. 54 Enrollment of hydrops MRI into the diagnostic algorithm of delayed EH is recommended, because surgical ablation should be reserved for patients with bilateral delayed EH since many delayed EH patients ultimately lose their vestibular function.
Embryopathic EH
Embryopathic EH may occur in a congenitally deaf child concomitant with other anomalies, that is, Mondini anomaly or enlarged vestibular aqueduct. Failure to resorb the endolymph accounts for the hydrops formation, leading to episodic vertigo, and/or fluctuating hearing loss in the following years, mimicking delayed EH. 49
Clinically, positive recruitment phenomenon, increased summating potential to action potential ratio by electrocochleography, or therapeutic diagnosis using osmotic diuretics have provided diagnostic clues for differentiating a hard-of-hearing child with acute SNHL.43,55 Recently advanced hydrops MRI eventually helps confirm the diagnosis of embryopathic EH in a child with profound SNHL experiencing acute exacerbation of residual hearing. 56
Asymptomatic EH
Although EH is most often associated with Meniere’s disease, it may exist without clinically manifest Meniere’s disease, a condition termed asymptomatic EH.38,57 Yamashita and Schuknecht 58 studied 495 temporal bones from 300 donors in whom the otopathological diagnoses were non-Meniere’s disease and found an overall incidence of 15.8% for apical EH, suggesting that apical EH is of neither pathological nor functional significance. Because the interscalar septum at the apical turn can be clearly identified on hydrops MRI, which may explain why apical EH of the cochlea was frequently identified on hydrops MRI. 7 Fortunately, unilateral or bilateral involvement of apical EH could help differentiate whether the apical EH is caused by hydrops itself or normal variant. If apical EH occurs on both ears, normal variant is more likely than pathological EH, while unilateral apical EH may be interpreted as pathological.
Lin et al 38 studied human temporal bones from 17 donors with unilateral Meniere’s disease, and asymptomatic EH on the saccule was identified in 6 (35%) of the unaffected (opposite) ears, indicating that asymptomatic hydrops on the unaffected ear of a Meniere’s disease patient may be a transitional phase between unilateral and bilateral Meniere’s disease. Lu and Young 59 subsequently reported that evolution of Meniere’s disease may progress from unilateral Meniere’s disease (Meniere’s disease 1.0), via unilateral Meniere’s disease coupled with asymptomatic hydrops on opposite ear (Meniere’s disease 1.5), toward bilateral Meniere’s disease (Meniere’s disease 2.0), with the numerical values representing the number of clinically affected ears. Restated, patients with unilateral Meniere’s disease, who exhibit symptomatic EH on the affected ear alongside asymptomatic EH on the opposite (unaffected) ear, should be serially followed by clinical manifestation, audiometry, and hydrops MRI to identify whether the asymptomatic EH progresses to Meniere’s ear.56,60,61
Implications for Practice
In the realm of hydrops MRI, there has been significant progress over the past decade, particularly in terms of imaging techniques, candidate selection, and grading systems. Three advantages of updated hydrops MRI have been noted. First, differentiation between the EH and sudden SNHL can be achieved, because the 2 diseases have significant overlap in symptoms, especially in Meniere’s disease patients with monosymptomatic onset of hearing loss, or a hard-of-hearing child with acute worsening of residual hearing, or an irradiated nasopharyngeal carcinoma (NPC) survivor with acute SNHL. 56 Second, determination of the affected side of EH is either unilateral or bilateral, particularly for cases of delayed or asymptomatic EH. Third, confirmation of EH is concomitant with other disorders. For instance, whether vestibular schwannoma patients had concomitant EH, or whether vertiginous elderly people with underlying systemic diseases had combined with de novo Meniere’s disease.
Notably, not all differentials for EH can be visualized on hydrops MRI, as evidenced by various sensitivity and specificity values for identifying the EH (Table 1). Hence, those Meniere’s disease patients showing (1) hearing recovery (resolution of EH); (2) chronic low-tone hearing loss (collapse of cochlear duct); or (3) immediately after vertiginous attack (rupture of EH) are not candidates for undergoing hydrops MRI. 56
Although some researchers have attempted to conduct MRI without the use of gadolinium contrast media to save the 4 hour waiting period for delayed scan,62,63 the capability to distinguish between artifacts and genuine imaging findings continues to be unsatisfactory. Up to the present time, contrast-enhanced hydrops MRI is still the most reliable method for visualizing the EH. 64 Nevertheless, there is an expectation that noncontrast MRI will eventually become capable of reliably recognizing the endolymphatic space in the future.
Furthermore, like histopathological study in vitro, hydrops MRI serves as a valuable tool for confirming the presence of EH in vivo. However, updated hydrops MRI still struggles to detect distorted contour such as rupture, collapse, or fistula in the inner ear compartments. Enhanced ultrahigh resolution of hydrops MRI is required for meticulously demonstrating fine structures of the inner ear compartments.
Conclusion
The application of hydrops MRI allows (1) differentiation between EH and sudden SNHL; (2) determination of the affected side of EH; and (3) confirmation of the diagnosis of EH concomitant with other disorders. Notably, not all differentials for EH can be visualized on MR images. One of the existing gaps to be filled is that updated hydrops MRI fails to identify distortion, that is, rupture, collapse, fistula, or fibrosis of the inner ear compartments, akin to what histopathological evidence can demonstrate. Hence, enhanced ultrahigh resolution of hydrops MRI is required for demonstrating fine structures of the inner ear compartments in the future.
Footnotes
Author Contributions
YHY: correspondent, data analysis, drafting, final approval, accountability for all aspects of the work. KTL: data analysis, drafting, final approval, accountability for all aspects of the work.
Availability of Data and Materials
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Science Council, Taipei, Taiwan (Grant no. NSTC 112-2314-B418-003), and Good Liver Foundation, Taipei, Taiwan.
Ethical Statement
This study was approved by the institutional review board of National Taiwan University Hospital (202111060RIND), and all patients signed the informed consent to participate.
