Abstract
Background
Cisplatin-based chemoradiation is a standard treatment for many patients with human papillomavirus (HPV)-positive oropharyngeal squamous cell carcinoma (OPSCC), an etiologically distinct subset of head and neck cancer. Although associated with good long-term survival, clinical risk factors for ototoxicity have been understudied in this population. This study aimed to evaluate clinical predictors associated with ototoxicity in HPV-positive OPSCC patients treated with cisplatin chemoradiation.
Methods
This retrospective case-control study included 201 adult patients (>18 years) with histologically confirmed HPV-positive OPSCC who received cisplatin chemoradiation as their primary treatment from 2001 and 2019 at a single tertiary cancer center. Ototoxicity was determined using baseline and follow-up audiometry and the Common Terminology Criteria for Adverse Events v5.0 grading criteria (Grade ≥2). Multivariable logistic regression [adjusted odds ratio (aOR)] identified significant predictors that increased the odds of ototoxicity.
Results
A total of 201 patients [165 males; median (IQR) age, 57 (11) years] were included in the study. The incidence of ototoxicity in the worst ear was 56.2%, with the greatest hearing loss occurring at high frequencies (4-8 kHz), resulting in a loss of 12.5 dB at 4 to 6 kHz and 20 dB at 6 to 8 kHz. High-dose cisplatin administration compared to weekly administration [aOR 4.93 (95% CI: 1.84-14.99), P = .003], a higher mean cochlear radiation dose [aOR 1.58 (95% CI: 1.12-2.30), P = .01], smoking history [aOR 2.89 (95% CI: 1.51-5.63), P = .001], and a 10 year increase in age [aOR 2.07 (95% CI: 1.25-3.52), P = .006] were each independently associated with increased odds of ototoxicity.
Conclusions
Clinical predictors of ototoxicity in HPV-positive OPSCC patients treated with cisplatin-based chemoradiation include the use of a high-dose cisplatin regimen, higher cochlear radiation doses, a history of smoking, and older age. With the rising incidence of this malignancy in Western countries and overall improved survivorship, our research motivates future studies into risk stratification and earlier interventions to mitigate and reduce the risk of ototoxicity.

Keywords
Background
Cisplatin, as a potent radiosensitizer, is a widely used chemotherapeutic drug combined with radiation for locally advanced head and neck squamous cell carcinomas (HNSCC). 1 Unfortunately, a significant proportion of chemoradiation therapy (CRT) patients (between 17% and 88%) can develop irreversible sensorineural ototoxicity, depending on the hearing loss criteria used.2,3 Such hearing loss can be profound and have significant impacts on social and vocational activities as well as overall quality of life.4,5 Human papillomavirus (HPV)-related oropharyngeal squamous cell carcinoma (OPSCC) is a distinct subgroup of HNSCC that has increased in incidence in Western countries. 6 Due to the increased chemo-radiosensitivity of this malignancy, survival rates for HPV-positive OPSCC are high, with 5 year survival rates between 83% and 92%. 7 Clinically, patients with HPV-positive OPSCC tend to be younger, have less exposure to alcohol and tobacco, and have a higher education status.8,9 With the excellent prognosis and increased survivorship of this cancer group, there has been increased emphasis placed on recognizing and addressing treatment-related toxicities for patients with HPV-positive OPSCC.
To date, cisplatin CRT-related ototoxicity has been studied in the general HNSCC population, with the incidence, characteristics of hearing loss, and clinical risk factors previously described. 10 However, most of these studies were conducted in non-OPSCC populations, such as carcinomas of the nasopharynx, which have differing treatments and patient profiles compared to HPV-positive OPSCC. 11 Given the rise in incidence and survivorship of this growing patient population, the objective of this study was to evaluate the characteristics and clinical risk factors associated specifically with ototoxicity in HPV-positive OPSCC patients treated with cisplatin-based chemoradiotherapy.
Methods
Study Design and Patient Population
A retrospective case-control study was conducted on adult patients diagnosed with head and neck cancer between 2001 and 2019 at Princess Margaret Cancer Centre (PM) in Toronto, Canada. Patients were prospectively recruited to the Head and Neck Translational Research Study at PM. 12 Inclusion criteria included adult patients (>18 years) with histologically confirmed HPV-positive squamous cell carcinoma of the oropharynx receiving cisplatin CRT with curative intent as their primary treatment. 13 Patients with incomplete audiometric testing (no baseline and follow-up audiogram) or patients with initial curative treatment that did not include cisplatin CRT were excluded from the final cohort. See Figure 1 for a flow diagram outlining the inclusion and exclusion criteria utilized to arrive at the final analytic sample.

Eligibility diagram of the cohort. CTCAE, Common Terminology Criteria for Adverse Events; HNC, head and neck cancer; HNSCC, head and neck squamous cell carcinoma; HPV, human papillomavirus.
Patient Demographics and Tumor Characteristics
Demographic and epidemiological information (age, sex, smoking/drinking status), clinical information [disease site, Tumor, Node, Metastasis stage at diagnosis (American Joint Committee on Cancer (AJCC) seventh edition)], HPV status, treatment (cisplatin cumulative dose, cisplatin dosing regimen, and mean cochlear radiation dose, which was calculated by averaging the total cochlear radiation dose in both ears), histology, and audiometric data (baseline and follow-up audiograms) were obtained from demographic questionnaires, electronic medical records, and the PM Head and Neck Anthology of Outcomes repository of clinical information. 12 Drinking status was based on a previously described classification system. 15 “Light” drinkers were defined as consuming less than 10 standard drinks per week, “moderate” drinkers were defined as consuming between 10 and 20 standard drinks per week, and “heavy” drinkers were defined as consuming 20 or more standard drinks per week.
HPV status was determined using p16 immunohistochemistry, where strong and diffuse nuclear and cytoplastic staining in >70% of tumor cells would be indicated as p16-positive and hence HPV-positive. In case of equivocal p16 staining, polymerase chain reaction molecular testing was performed to confirm the presence of high-risk HPV. HPV serology was subsequently utilized if immunohistochemistry was not performed. The study protocol was approved by the Research Ethics Board of Princess Margret Cancer Centre.
Patient Treatment Factors
The mean cochlear radiation dose was calculated by averaging the total gray (Gy) dose to each ear, which was extracted from archived radiation therapy plans. Cisplatin dose scheduling was retrospectively collected during the chart review process. Patients were subdivided into a “high-dose” or “weekly” category depending on the scheduling of cisplatin chemotherapy through the duration of their treatment. Following PM guidelines, patients who were classified into the “high-dose” schedule were administered 75 to 100 mg/m2 of cisplatin at weeks 1, 4, and 7 of radiotherapy treatment. Patients classified into the “weekly” regimen were administered a weekly dose of ~40 mg/m2 of cisplatin for the duration of their treatment. 16
Audiometric Evaluation and Ototoxicity Classification
Audiometry data, including internally performed or conducted at an external center, were retrospectively collected. All included study patients undergoing cisplatin CRT at PM had a pre- and post-treatment audiogram, with some patients having additional audiometric testing based on subjective changes in hearing. All patients underwent pure-tone audiometry testing with air conduction thresholds measured at standard frequencies ranging between 0.25 and 8 kHz. Hearing tests with missing measurements at specific frequencies were imputed from the lowest neighboring available frequency measurements. This imputation approach ensured a conservative estimate of the missing value, reflecting the least amount of change. Ototoxicity grading was then determined using the Common Terminology Criteria for Adverse Events v5.0 (CTCAE), with hearing loss status defined as a threshold shift of ≥25 dB averaged at 2 contiguous test frequencies in at least 1 ear (Grade ≥2). Patients with a threshold shift <25 dB (Grade ≤1) averaged at 2 contiguous test frequencies in at least 1 ear were classified as controls.
Statistical Analysis
Descriptive statistics were used to describe patient demographic and clinical characteristics between cases and controls. Medians and interquartile ranges were calculated for continuous variables, and frequencies and percentages were reported for categorical variables. Univariable and multivariable logistic regressions were used to evaluate the association between cisplatin CRT (cisplatin regimen, cisplatin dose, and mean cochlear radiation dose received per side) and hearing loss in at least 1 ear (CTCAE Grade ≥2), controlling for confounding variables determined a priori via previous literature, including age at diagnosis, sex, cancer stage, drinking status, smoking status, renal function [estimated glomerular filtration rate (eGFR)], months of audiometric follow-up, and baseline pure tone average (PTA) obtained by averaging audiometry results at 4, 6, and 8 kHz in both ears.10,17,18 Multivariable logistic regression was utilized to test each independent predictor variable with adjustment for other predictor variables. Augmented backward variable elimination was used where the least significant variables with a P value greater than .2 were eliminated from the model unless their exclusion resulted in a significant change of 10% or more in other estimates. Variables removed in order included drinking status, cancer stage, renal function, and sex. 19 A priori variables of primary interest (cisplatin cumulative dose) were included in the final model due to clinical significance. All statistical tests were 2-sided, and a P value of .05 was used to define statistical significance. Statistical analyses were carried out using R (version 4.1.0). 20
Results
Cohort Demographics
In the final cohort, 201 HPV-positive OPSCC patients were included. Baseline demographic, treatment, and clinical variables are provided in Table 1. All patients were treated with cisplatin concurrent with radiation, with the majority diagnosed with stage IV disease at the time of treatment (n = 192, 96%). The median cumulative cisplatin dose received was 198 (IQR 68) mg/m2, and dosing schedules were divided between high-dose (n = 194, 87%) and weekly (n = 27, 13%). The median cochlear dose of radiation was 12.3 (IQR 12.1) Gy.
Clinical Characteristics of HPV-Positive OPSCC Cohort Treated With Cisplatin Chemoradiation.

Abbreviations: dB, decibel; Gy, gray; “High” dosing, administered 75 to 100 mg/m2 of cisplatin at weeks 1, 4, and 7 of radiotherapy treatment; HPV, human papillomavirus; IQR, interquartile range; OPSCC, oropharyngeal squamous cell carcinoma; PTA, pure tone average calculated from 4, 6, and 8 kHz; “Weekly” dosing, administered a weekly dose of ~40 mg/m2 of cisplatin for the duration of their treatment.
Mean cochlear radiation dose was calculated by averaging the total radiation dose delivered to each ear.
Audiometric follow-up refers to the length of time between baseline and the most recent follow-up audiogram test.
Audiometric Evaluation
The incidence of cisplatin chemoradiation-associated ototoxicity was 56.2% (n = 113). The median baseline PTA (averaged at 4, 6, and 8 kHz) and length of posttreatment audiometric follow-up were 26.7 dB (IQR 24.2) and 8 months (IQR 6.1), respectively. The distributions of hearing loss across frequencies by ear are illustrated in Figure 2. Hearing loss was greatest at the highest frequencies between 4 and 8 kHz, with an average PTA increase of 12.5 and 20 dB at 4 to 6 and 6 to 8 kHz in the worst ear, respectively.

Degree of hearing loss (measured in decibels, dB) across frequencies (measured in kilohertz, kHz) for each ear.
Association of Clinical Variables With Cisplatin Chemoradiation-Induced Ototoxicity
The results of the univariable and multivariable regression models are presented in Table 2. The cisplatin regimen was strongly associated with ototoxicity. Specifically, patients with high-dose cisplatin, compared to a weekly regimen, had 4.9 times the odds of experiencing ototoxicity [adjusted odds ratio (aOR) 4.93 (95% CI: 1.84-14.99), P = .003] after adjusting for confounders. In addition to a high cisplatin dosing regimen, a higher mean cochlear radiation dosage [aOR 1.58 (95% CI: 1.12-2.30), P = .01, per 10 Gy increase] was significantly associated with a higher odds of ototoxicity. Having smoked (previous or current smoker) was associated with a higher odds of ototoxicity [aOR 2.89 (95% CI: 1.51-5.63), P = .001]. Finally, an increase in age was independently associated with a higher odds of ototoxicity [aOR 2.07 (95% CI: 1.25-3.52), P = .006]. The cumulative cisplatin dose was not significantly associated with hearing loss in this study population.
Univariable and Multivariable Analyses of Predictors Associated With Ototoxicity in HPV-Positive OPSCC Patients Treated With Cisplatin Chemoradiation.
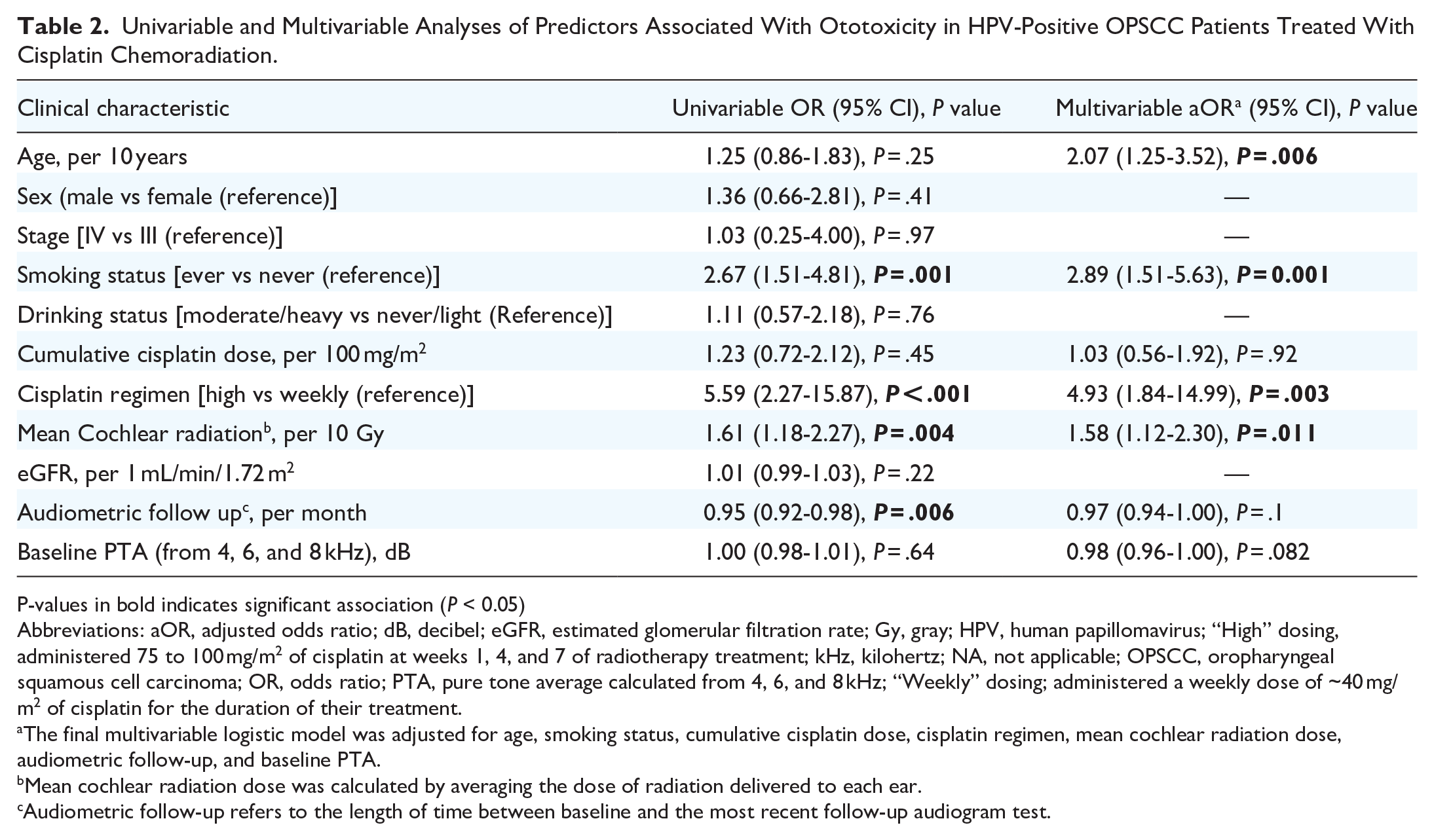
P-values in bold indicates significant association (P < 0.05)
Abbreviations: aOR, adjusted odds ratio; dB, decibel; eGFR, estimated glomerular filtration rate; Gy, gray; HPV, human papillomavirus; “High” dosing, administered 75 to 100 mg/m2 of cisplatin at weeks 1, 4, and 7 of radiotherapy treatment; kHz, kilohertz; NA, not applicable; OPSCC, oropharyngeal squamous cell carcinoma; OR, odds ratio; PTA, pure tone average calculated from 4, 6, and 8 kHz; “Weekly” dosing; administered a weekly dose of ~40 mg/m2 of cisplatin for the duration of their treatment.
The final multivariable logistic model was adjusted for age, smoking status, cumulative cisplatin dose, cisplatin regimen, mean cochlear radiation dose, audiometric follow-up, and baseline PTA.
Mean cochlear radiation dose was calculated by averaging the dose of radiation delivered to each ear.
Audiometric follow-up refers to the length of time between baseline and the most recent follow-up audiogram test.
Discussion
Although hearing loss is a well-established adverse outcome of cisplatin CRT therapy, its incidence and risk factors have yet to be described in HPV-positive OPSCC populations.10,11,21 In our study, we report that over 56% of individuals treated with cisplatin CRT developed clinically significant ototoxicity. Multiple clinical factors were found to be independently associated with ototoxicity in patients with HPV-positive OPSCC, including age, smoking status, high-dose cisplatin regimen, and cochlear radiation dose. Previous research in head and neck cancer that has reported on cisplatin CRT-related hearing loss did so across heterogeneous cohorts with varying anatomical sites and treatment modalities. As the demographics, pathophysiology, treatment, and treatment outcomes of HPV-positive OPSCC differ largely from those of traditional head and neck cancer patients, the findings of this study further contribute to our understanding of this highly significant and common adverse effect of cisplatin CRT in a growing group of cancer survivors.
Our analysis identified the schedule of cisplatin administration as an important factor associated with ototoxicity. Patients placed on a weekly dosing regimen had lower odds of developing ototoxicity when compared to patients who received a bolus dosing schedule. The total cumulative cisplatin dose was not associated with any differential risk of hearing loss. In a recent large study involving 2900 HNSCC patients by Bauml et al., high-dose cisplatin scheduling was associated with an increased risk of hearing loss without providing any additional survival advantage compared to low-dose weekly scheduling. 22 Another study, which analyzed 328 audiograms, found that a weekly cisplatin regimen over a 6 week course was associated with less hearing impairment than a high-dose biweekly regimen, despite both schedules having equivalent dose intensities. 23 The observed hearing benefits of weekly dosing over biweekly dosing may be due to the inner ear’s ability to recover between doses or the possibility of a threshold effect where lower doses of cisplatin are less likely to reach toxic levels in the inner ear. 24 However, the underlying mechanism is not fully understood and requires further investigation. Weekly dosing has demonstrated noninferior efficacy with an improved toxicity profile in other head and neck malignancies, such as laryngeal and hypopharyngeal carcinomas. 25 Prospective trials in HPV-OPSCC have yet to be conducted. 26
A higher mean cochlear radiation dose was significantly associated with increased cisplatin CRT-related ototoxicity, which is in line with the literature. 27 A recent systematic review of patients with head and neck cancer reported a higher incidence of hearing loss with cochlear radiation doses greater than 47 Gy. 10 This is higher than the 13.1 Gy median cochlear radiation dose (calculated from the average radiation dose to both ears) observed in our study; however, the meta-analysis by Theunissen et al included studies that utilized radiation therapy alone as well as combined with cisplatin chemotherapy. 10 Both radiation exposure and cisplatin have been demonstrated to be independently associated with damage to the inner ear, with purported synergistic effects.28,29 Comparing the effects of radiation therapy and cisplatin CRT treatment approaches, Hitchock et al demonstrated that cochlear radiation doses as low as 10 Gy resulted in clinically significant hearing impairment when cisplatin chemotherapy was given concurrently with radiation therapy. 29 In contrast, when radiation therapy was used alone, a minimum dose of 40 Gy was required to cause a similar degree of hearing impairment. 29 In addition, the majority of studies and corresponding participants included in the meta-analysis were from non-OPSCC subsites, which restricts the interpretability of these results in relation to cisplatin CRT for oropharyngeal cancers, particularly given the close proximity of the oropharynx to inner ear structures.10,17 As cisplatin CRT has become a primary treatment regimen for HPV-positive OPSCC, our findings support the importance of further research into the synergistic ototoxic effects of cisplatin CRT to redefine formerly established thresholds that previously often only considered unimodal interventions.
Hearing loss was detected across all frequencies tested, with the most significant deterioration at higher frequencies, mainly at 6 to 8 kHz. These results are comparable to those from a randomized control trial conducted by Wong et al that reported higher frequencies (4 kHz and over) to be more severely affected than lower speech frequencies following cisplatin CRT. 30 The proposed progression of cisplatin-associated cochlear damage is thought to occur predominantly through apoptosis of cells at the base of the cochlea, which is responsible for high-frequency detection, and progresses toward cells at the cochlear apex, which is responsible for low-frequency detection. 21 Although we detected minimal hearing loss in frequencies below 6 kHz, which encompass the range for human speech perception, the higher frequency range is believed to help with speech discrimination and in understanding speech in noisy environments. 31 The consequences of ototoxicity range from emotional disturbance to social isolation and vocational limitations.4,32 Sensorineural hearing loss in older adults is significantly linked to decreased mood, increased depressive symptoms, poorer interpersonal relationships, and most recently dementia.33,34 The negative sequalae associated with hearing loss are further magnified in HPV-positive OPSCC patients, as they are younger in age, have longer survivorship, and are more likely to return to work and social functioning.5,7
This study has some important limitations and considerations. First, audiometric evaluations were not consistently scheduled and administered, resulting in variable follow-up periods between baseline and posttreatment audiograms. This variability may have biased the phenotyping of hearing loss, as ototoxicity related to cisplatin chemoradiation can develop many months to years after treatment, and our study may not have identified this subset of patients with hearing loss.35,36 All patients in our cohort had pre-treatment audiograms and at least 1 follow-up audiogram, mitigating the phenotyping bias of preexisting hearing loss. This contrasts with most of the current literature, which reports findings from a mixed cohort with many patients missing baseline audiograms.21,37 In addition, only 49% of all HPV-positive OPSCC patients treated with cisplatin CRT within the study period had adequate audiometric testing, although a large percentage, 56%, of those tested had clinically significant hearing loss. Similarly, a large retrospective review by Santucci et al found that only 57% of HNC patients treated with cisplatin underwent pretreatment audiograms, and 61% reported otologic complaints post cisplatin treatment. 21 Ontario guidelines recommend that all patients being administered cisplatin chemotherapy have audiogram testing at “baseline and as clinically indicated”; efforts to increase audiometry rates could improve long-term quality of life and survivorship.7,38 Second, our study did not incorporate other clinical factors that may influence the risk of ototoxicity, such as previous noise-induced hearing loss, concomitant ototoxic medications, and relevant neurologic and peripheral vestibular conditions. 39 Third, staging information was from the previous seventh AJCC criteria, which does not incorporate the significant modifications made in the eighth edition to differentiate HPV-positive OPSCC. Furthermore, individual genetic susceptibility to cisplatin-induced ototoxicity could not be explored in our analysis, despite being widely described in both adults and pediatrics, due to a lack of pharmacogenomic studies in HPV-positive OPSCC patients.40,41
To the best of our knowledge, we present the first study examining clinical predictors of hearing loss exclusively in HPV-positive OPSCC patients treated with cisplatin CRT. With the rising incidence of HPV-positive OPSCC in developed countries and overall better survivorship, it is becoming increasingly important to identify risk factors associated with treatment-induced ototoxicity. Such information can be utilized for earlier interventions to mitigate and reduce the risk for hearing loss, such as a change to a less ototoxic platinum alternative or the use otoprotectants during treatment. For example, administration of sodium thiosulfate has been shown to significantly lower the risk of developing hearing loss without a significant effect on treatment efficacy or overall survival. 42 We anticipate that our findings will inform future studies on the development and implementation of surveillance strategies to assess hearing loss risk during CRT for patients with locally advanced HPV-positive OPSCC.
Conclusion
In our HPV-OPSCC cohort, a high-dose cisplatin administration schedule compared to a weekly administration schedule, a higher mean cochlear radiation dose, smoking history (current or past), and older age were each independently associated with increased odds of ototoxicity. The cumulative cisplatin dose was not significantly associated with ototoxicity. It is important for forthcoming investigations to explore risk stratification and surveillance strategies aimed at mitigating the adverse effects of ototoxicity within this expanding demographic of long-term survivors.
Footnotes
Acknowledgements
None.
Author Contributions
The study was designed by JWL and GL. Analysis and interpretation of data was conducted by LJZ, JJWL, and SA. SA and JWL were responsible for drafting the manuscript. Critical revision was provided by DPG and GL. All the authors have read and approved the final manuscript.
Availability of Data and Materials
The datasets generated and/or analyzed during the current study are not publicly available due to patient confidentiality and sensitive information but are available from the corresponding author on reasonable request.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JWL was supported by the Harry Barberian Scholarship Award from the Department of Otoloaryngology-Head and Neck Surgery, University of Toronto. This study was supported by the Princess Margaret Cancer Centre Head & Neck Translational Program, with philanthropic funds from the Wharton Family, Joe’s Team, and Gordon Tozer.
Ethics Approval and Consent to Participate
No private personal information was identifiable in the data. The study protocol was approved by the Research Ethics Board of Princess Margret Cancer Centre.
