Abstract
Background
Patients with oropharyngeal squamous cell carcinoma (OPSCC) treated with radiation-based therapy suffer from short- and long-term toxicities that affect quality of life (QOL). Transoral robotic surgery (TORS) has an established role in the management of early OPSCC but adjuvant treatment is often indicated postoperatively due to the high incidence of nodal metastasis associated with advanced human papillomavirus (HPV)-related OPSCC. To overcome the need for adjuvant radiation therapy (RT), neoadjuvant chemotherapy followed by TORS and neck dissection (ND) is proposed. This study aimed to assess if QOL in HPV-associated OPSCC receiving neoadjuvant chemotherapy followed by TORS and ND returns to baseline within 12 months of completing treatment.
Methods
A 12 month longitudinal study was carried out at McGill University Health Centre in Montreal, Canada, among a convenience sample of patients with American Joint Committee on Cancer Seventh Edition stage III and IVa HPV-related OPSCC who were treated with neoadjuvant chemotherapy followed by TORS and ND. QOL data were obtained pretreatment and at 1, 3, 6, and 12 months following treatment completion using the European Organisation for Research and Treatment of Cancer Core and Head and Neck extension modules. Paired t tests and mixed models for repeated measures analysis were used to assess changes in QOL from baseline to 12 months postoperatively and over time, respectively.
Results
Nineteen of 23 patients (median age 58 years) who received the study treatment fulfilled the eligibility criteria. OPSCC subsites were palatine tonsil (n = 12) and base of tongue (n = 7). All 19 patients were treated per protocol and none required adjuvant RT as per pathology review and protocol requirements at a postoperative multidisciplinary team tumor board discussion. No significant differences were found when comparing 12 month QOL follow-up scores to pretreatment scores in measures that would likely be affected by RT [eg, swallowing (P = .7), social eating (P = .8), xerostomia (P = .9)].
Conclusion
In HPV-related OPSCC, neoadjuvant chemotherapy followed by TORS and ND as definitive treatment is associated with excellent QOL outcomes. Postoperative QOL scores returned to baseline by 3 months and were maintained for all measures, indicating a return to normal function.

Introduction
The incidence of human papillomavirus (HPV)-related oropharyngeal squamous cell carcinoma (OPSCC) has been rising over the past 20 years, becoming the most prevalent subtype of head and neck cancer in the western world.1,2 Patients diagnosed with HPV-related OPSCC are demographically and biologically different from those with typical head and neck squamous cell carcinomas as they are generally healthier and younger.2 -4 Sixty to seventy percent of HPV-related OPSCC patients have positive nodal disease at presentation, identified during imaging workup, or in surgical pathology specimens necessitating treatment escalation. 4 Typically patients present after identifying a nonpainful neck mass while being otherwise aymptomatic.5,6 Despite the presence of lymphadenopathy, the prognosis of HPV-related OPSCC is favorable given that this cancer is treatment sensitive.7,8 HPV-positive OPSCC patients have higher rates of overall survival (82.4% vs 57.1%) and 3 year progression-free survival (73.7% vs 43.4%) than HPV-negative OPSCC patients. 9 Given the minimal symptom burden at diagnosis as well as the high rates of overall long-term survival, preservation of high quality of life (QOL) is a therapeutic goal.
The current standard of care treatment for early stage HPV-related OPSCC as per the National Cancer Comprehensive Network guidelines is definitive radiation therapy (RT) or surgery followed by adjuvant therapy if adverse features are found on pathology. 10 For more advanced disease, patients are typically treated with concurrent chemoradiotherapy (CRT) or surgical resection with adjuvant RT/CRT depending on pathologic risk stratification. Although associated with excellent oncologic outcomes, there is significant long-term morbidity with both standard RT and cytotoxic therapy, including xerostomia, dental loss and poor oral health, dysphagia, risk of gastrostomy tube dependency, as well as upper alimentary tube stenosis.9,11 -13 Due to the excellent prognosis of HPV-associated OPSCC, survivors are living for decades with these posttreatment toxicities.9,14
Transoral robotic surgery (TORS) was approved for the treatment of OPSCC by the Food and Drug Administration in 2009. 15 This technology allows head and neck surgeons to perform minimally invasive resections through the oral cavity and is effective for providing excellent oncologic results while preserving speech and swallowing function.16 -18 TORS has been proposed in the context of treatment de-intensification as it is less invasive than the traditional open surgical access to the oropharynx. In many patients treated with primary TORS, adjuvant RT or CRT is indicated because of the presence of nodal metastases, extracapsular extension, or a close/positive surgical resection margin. Clinical trials are underway searching for de-intensification regimens that maintain the current oncological outcomes while minimizing treatment toxicity.
To reduce the need for post-TORS adjuvant RT, neoadjuvant chemotherapy prior to definitive TORS and neck dissection (ND) has demonstrated competitive survival outcomes to that of CRT, quoting a 5 year disease-free survival rate of 96.1%.16,19,20 Clinical experience suggests that avoidance of adjuvant RT/CRT results in QOL at 12 month posttreatment that returns to pretreatment scores. The study aim is to determine whether patient-reported QOL scores return to baseline values by following data obtained over a 12 month period following treatment.
Materials and Methods
Ethics approval was obtained from the McGill University Health Centre (MUHC) research ethics board REB (MP-37-2018-3568). This 12 month longitudinal study was conducted at the MUHC in Montreal, Canada, from January 2017 to July 2018. Eligible patients were between 18 and 80 years of age, diagnosed with American Joint Committee on Cancer (AJCC) Seventh Edition stage III or IVa HPV-related OPSCC, treatment naïve and without evidence of distant metastases (M0), ≤5 nodal metastasis on radiology without gross extranodal extension (ENE) into the surrounding soft tissue on imaging, Eastern Cooperative Oncology Group (ECOG) performance status <2, and without history of head and neck malignancy or any other malignancy within the past 5 years. Irregularity at the nodal margin, suggesting ENE, was not an exclusion criterion. To be included in the study, patients were required to complete a minimum of 2 of a possible 5 QOL questionnaires. QOL was obtained using the European Organisation for Research and Treatment of Cancer (EORTC) Core Quality of Life questionnaire and the disease-specific head and neck cancer module, the EORTC QLQ-C30, and H&N35.21 -23 Patients completed the questionnaires at pretreatment (baseline), and at 1, 3, 6, 12 posttreatment, where treatment completion is defined as the date of surgical resection.
Prior to study participation, all potential participants were discussed in a multidisciplinary tumor board for therapeutic recommendations. For patients who met eligibility criteria for treatment with neoadjuvant chemotherapy followed by TORS and selective ND (nodal levels 2, 3, and 4), both this option and standard of care RT/CRT were discussed and offered. Written consent was obtained prior to registration and treatment.
The neoadjuvant chemotherapy regimen consisted of 3 cycles of docetaxel 75 mg/m2 and cisplatin 75 mg/m2.16,19,20 Adequate primary site TORS resection is based on pre-chemotherapy extent and margins of the tumor. Tattooing of baseline margins was performed in cases where the tumor extends outside of the anatomical unit of tonsil or ipsilateral base of tongue (BOT). The laterality of ND performed was guided by radiological distribution of nodal disease and location of primary tumor. Patients with BOT tumors underwent bilateral ND independent of radiological findings. Patients with tonsil primary and extension to the soft palate or the BOT for more than 1 cm also underwent bilateral ND. All patients were re-presented at the tumor board meeting postoperatively for review of pathological analysis. Adjuvant radiotherapy was offered if indicated, that is, for positive margins not suitable for revision surgery, ≥3 nodes with viable cancer cells following neoadjuvant chemotherapy, and pathological ENE.
Statistical analysis was performed using R.v.3.4.1 software (The R Foundation). Descriptive statistics were used to describe the study population. Proportions were calculated for categorical variables. Continuous data were presented as mean and standard deviations (SDs) or 95% confidence interval (CI). For each outcome measure, 2 statistical analyses were performed. Paired t tests were used to assess differences in scores from baseline to 12 months. Repeated measures mixed-effects models were used to assess whether the mean population scores differed over time.24 -26 Multiple comparisons were adjusted using Tukey post hoc tests for all outcome measures. Statistical significance was set at P < .05.
Results
Between January 2017 and July 2018, 23 patients underwent neoadjuvant chemotherapy followed by TORS and selective ND, 19 of whom fulfilled eligibility criteria and were included in this study. Of the excluded patients, 2 did not complete pretreatment surveys and 2 did not complete a minimum of 2 surveys. Nineteen patients completed the questionnaire at baseline, 16 at 1 month, 13 at 3 months, 16 at 6 months, and 18 at 12 months. Baseline demographic data show that the majority of patients were male (n = 17) and the median age was 58 years, with an interquartile range of 48 to 79 years (Table 1). The primary tumor site was at the palatine tonsil in 12 patients and BOT in 7 patients. The AJCC Seventh edition clinical tumor (T) staging within the cohort was T1 (26.3%), T2 (68.4%), T3 (0), and T4 (5.2%). The AJCC Seventh edition clinical nodal (N) staging was N0 (10.5%), N1 (21.1%), N2a (10.5%), and N2b (57.8%). The AJCC Seventh edition overall stage was stage III in 31.5% and stage IVa in 68.5%. The AJCC Eighth edition overall stage was stage I in 42.1%, stage II in 52.6%, and stage III in 5.3%. Patients with T1-2N0 disease were not eligible for this treatment regimen and, therefore, are excluded. All 19 patients were treated with neoadjuvant chemotherapy (docetaxel and cisplatin) followed by TORS and ND. One patient only completed 2 cycles of neoadjuvant chemotherapy due to a leg cellulitis following trauma. Similar numbers of patients required unilateral (n = 10, 52.7%) and bilateral (n = 9, 47.3%) ND.
Baseline and Treatment Characteristics of HPV-Associated OPSCC Patients.

Abbreviations: AJCC, American Joint Committee on Cancer; HPV, human papillomavirus; ND, neck dissection; NG, nasogastric; OPSCC, oropharyngeal squamous cell carcinoma; PEG, [Percutaneous endoscopic gastrostomy tube; SD, standard deviation; TORS, transoral robotic surgery.
Postoperative pathology results reported no residual cancer in the primary site surgical specimen in 15 patients (78.9%). Of the remaining 4 patients with ypT1 disease, there was an 87% reduction in primary site volume. Clear margins (defined as no tumor at inked margin) at the primary tumor site were achieved in 2 ypT1 patients while 2 ypT1 patients underwent margin revision surgery at the primary site. The first underwent revision surgery for focal disease less than 1 mm from the specimen edge and had no cancer in the second specimen. His initial specimen also demonstrated lymphovascular invasion and perineural invasion and he recurred in the neck at 20 months posttreatment. He was treated with adjuvant CRT and is currently free of disease for 37 months. The other patient had crushed/cauterized carcinoma in situ at the initial margin and underwent follow-up surgery that also yielded carcinoma in situ at the revision margin. After a tumor board discussion, the decision was made with the patient to observe given that this area shows no current lesion or mass and was in a location easy to survey. He recurred at the primary site and in the parotid at 16 months post-initial treatment. He has been successfully salvaged with additional surgery (parotidectomy) and CRT and is free of disease for 47 months. No other patient required adjuvant RT. A nasogastric (NG) feeding tube was placed for all patients for postoperative nutritional support in hospital and was utilized for an average of 8.9 days (SD 5.6 days). No patient required a gastrostomy tube during treatment or during the 12 month follow-up period. There were no severe (grade 3+) treatment-related adverse events during the study period, as per the Cooperative Group Common Terminology Criteria for Adverse Events.
QOL Outcomes
QOL was assessed using the EORTC QLQ-C30 and EORTC QLQ-H&N35 (Tables 2 and 3, respectively). Changes over time in QOL scores are illustrated in Figure 1, where select subdomains and symptom scores from the EORTC QLQ-30 and EORTC H&N-35 are shown, demonstrating the expected postoperative deterioration and return to baseline by 3 to 6 months.
EORTC QLQ-30 Scores of HPV-Associated OPSCC Patients Treated With Neoadjuvant Chemotherapy and TORS With ND Across the 5-Time Assessment Points.
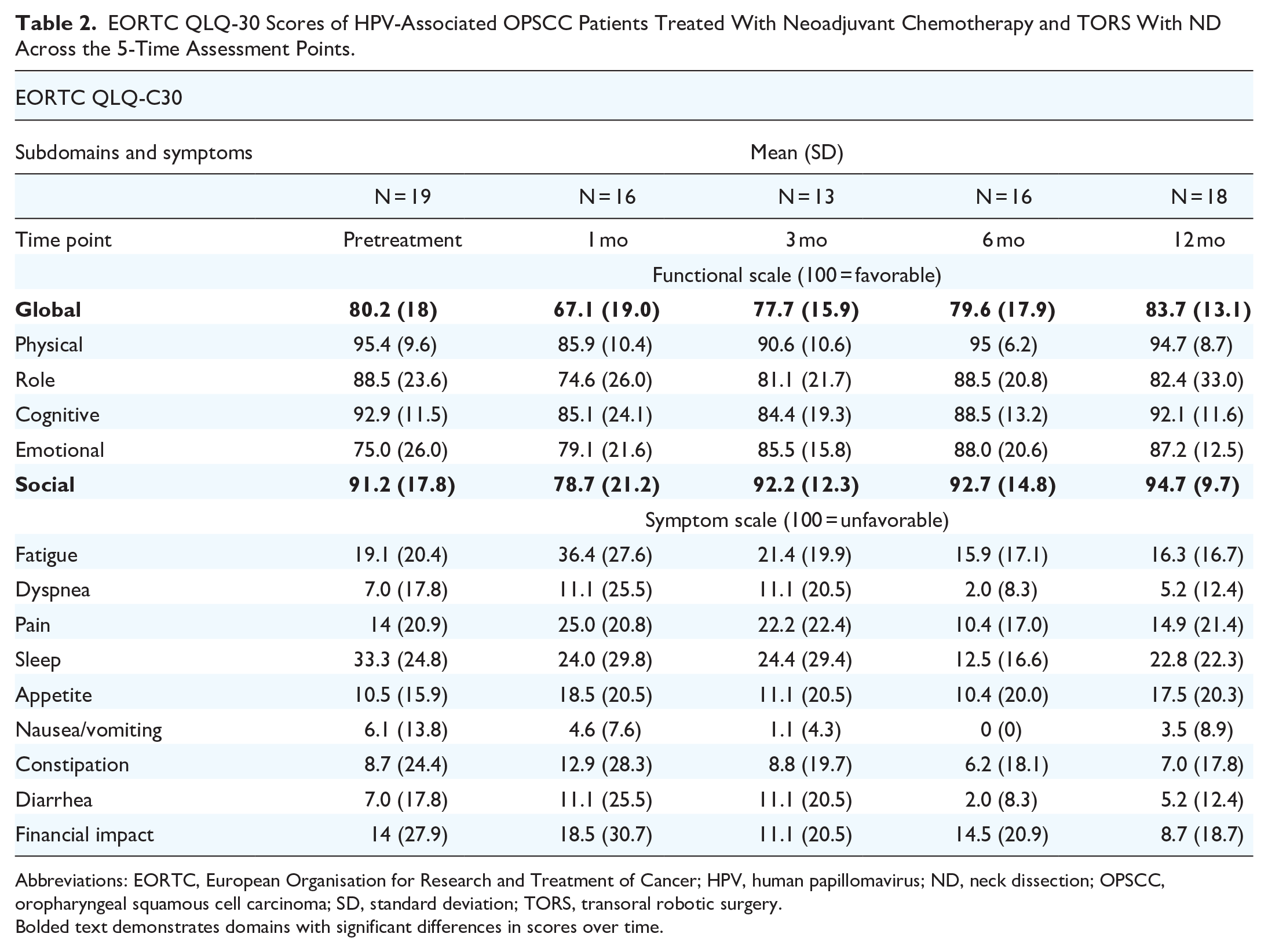
Abbreviations: EORTC, European Organisation for Research and Treatment of Cancer; HPV, human papillomavirus; ND, neck dissection; OPSCC, oropharyngeal squamous cell carcinoma; SD, standard deviation; TORS, transoral robotic surgery.
Bolded text demonstrates domains with significant differences in scores over time.
EORTC QLQ-H&N35 Scores in HPV-Associated OPSCC Patients Treated With Neoadjuvant Chemotherapy and TORS With ND Across the 5-Time Assessment Points.
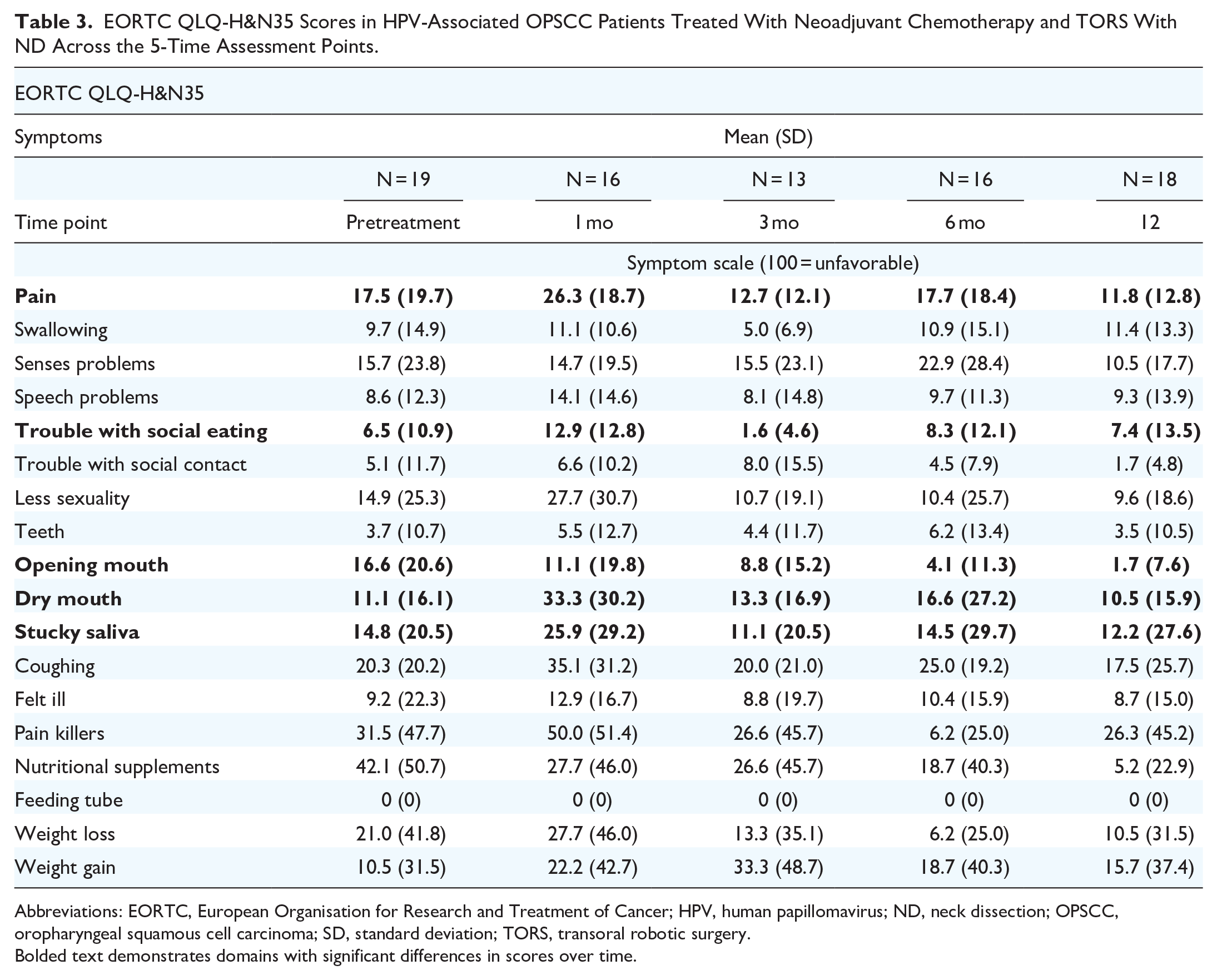
Abbreviations: EORTC, European Organisation for Research and Treatment of Cancer; HPV, human papillomavirus; ND, neck dissection; OPSCC, oropharyngeal squamous cell carcinoma; SD, standard deviation; TORS, transoral robotic surgery.
Bolded text demonstrates domains with significant differences in scores over time.

The change of EORTC QLQ-30 and EORTC H&N-35 quality of life scales over time in 19 patients with HPV-associated OPSCC treated with neoadjuvant chemotherapy and TORS. Higher scores for the global health status and functioning scales denote a better level of functioning (ie, a better state of the patient), while higher scores on the symptom and single-item scales indicate a higher level of symptoms (ie, a worse state of the patient). Shaded area in graphs represents 95% CI. EORTC, European Organisation for Research and Treatment of Cancer; HPV, human papillomavirus; OPSCC, oropharyngeal squamous cell carcinoma; TORS, transoral robotic surgery; CI, confidence interval.
Global QOL
There was no significant difference between global QOL at baseline versus 12 months postoperatively [mean of differences −1.85, 95% CI (−11.45, 7.74), P = .7]. There were significant differences between global QOL scores over time (P < .001). At 1 postoperative month, global QOL was significantly lower than baseline [−15.3, 95% CI (−27.4, −3.2), P = .004]. However, from 3 months postoperatively and onward, there were no significant differences of global QOL scores compared to baseline scores (P > .05).
Social Functioning
There was no significant difference between social functioning at baseline versus 12 months postoperatively [mean of the differences −3.70, 95% CI (−14.17, 6.76), P = .5]. There were differences in social functioning scores over time (P = .008). Social functioning scores decreased 1 month postoperatively compared to baseline [−16.1, 95% CI (−29.4, −2.95), P = .007]; however, all other scores were not significantly different over time (P > .05).
Swallowing Difficulties
There was no significant difference between swallowing scores at baseline versus 12 months postoperatively [mean of the differences 1.74, 95% CI (−11.64, 8.16), P = .7]. There was no significant difference in swallowing scores over time (P = .56).
Senses Problems
There was no significant difference between swallowing scores at baseline versus 12 months postoperatively [mean of the differences −5.56, 95% CI (−9.30, 20.42), P = .5]. There was no significant difference between senses problem scores over time (P = .64).
Social Eating
There was no significant difference between swallowing scores at baseline versus 12 months postoperatively [mean of the differences −0.92, 95% CI (−10.45, 8.59), P = .8]. There were no significant differences between social eating scores over time (P = .08).
Sticky Saliva
There was no significant difference between swallowing scores at baseline versus 12 months postoperatively [mean of the differences −2.72, 95% CI (−14.34, 19.79), P = .7]. There was no sticky saliva significant difference through the study period (P = .50).
Dry Mouth
There was no significant difference between swallowing scores at baseline versus 12 months postoperatively [mean of the differences 0.65, 95% CI (−10.56, 11.87), P = .9]. There were significant differences between dry mouth scores over time (P = .013). Dry mouth scores at 1 month postoperative are significantly higher (more bothersome) than baseline scores [21.3, 95% CI (1.47, 41.1), P = .028] and 12 months postoperative scores [22.0, 95% CI (2.5, 41.5), P = .017]. However, all other scores were not significantly different over time (P > .05), suggesting that by 3 months from surgery most patients recover from dry mouth.
Discussion
This longitudinal study investigates QOL following neoadjuvant chemotherapy and TORS and ND for locally advanced and/or regionally involved HPV-related OPSCC (ie, AJCC Seventh edition stage III and IVa). Given the minimally symptomatic baseline status, younger profile of HPV-associated OPSCC patients, and high survival rates compared to HPV-negative OPSCC patients, patient-reported QOL is an essential component of treatment decision-making.
Overall, this project demonstrated very favorable QOL results with a neoadjuvant chemotherapy approach followed by surgery. Functionally, patients did not require long-term supplemental feeding as NG tubes were removed at approximately 9 days. Patient-reported QOL scores returned to baseline by 6 months for all domains and remained stable at 12 months. There was worsening in global QOL, social functioning, and dry mouth QOL at 1 month postoperatively as expected from the acute toxicity of the treatment. However, all rapidly returned to baseline values by 3 months and remained stable at 12 postoperative months.
The standard of care treatment for stage III and IV (AJCC Seventh edition) OPSCC is concurrent CRT or surgery with adjuvant RT/CRT. 10 In a previous study assessing outcomes 1 year post-therapy, 77 advanced OPSCC patients were treated with CRT (n = 56) or surgery with adjuvant RT (n = 21). 27 Over half of these patients report persistent xerostomia. This contrasts with our findings where dry mouth has returned to baseline values by 3 months and stabilized until 12 months (P = .50). Patients receiving CRT in another study reported clinically significant deteriorations in 9/13 domains of the EORTC QLQ-H&N35 at 12 months. 27 In the current study, for the patients undergoing neoadjuvant chemotherapy and TORS, all domains returned to baseline. In a systematic review and meta-analysis of OPSCC survivors 12 to 200 months posttreatment, the patients had worse dry mouth, sticky saliva, and mouth opening as opposed to their reference baseline values. 28 Compared to scores prior to treatment, OPSCC survivors had a moderate clinically important deteriorations in sticky saliva and a large clinically important deterioration in dry mouth. This meta-analysis noted that average (decreased) QOL scores 1 year posttreatment were comparable among those undergoing organ preservation strategies with CRT or primary surgery, where a majority required adjuvant RT. This decrease in QOL is expected in both cohorts as they both receive radiotherapy and its related locoregional sequalae persist. There were limited data of surgical patients who did not receive adjuvant therapy but a qualitative synthesis was performed and demonstrated better QOL in this group, which is a cohort most similar to the 19 patients within this cohort. A direct comparison between all treatment modalities was not performed given cancer stage and comorbidities are potential confounders. 28 Another systematic review compared outcomes after radiotherapy versus TORS and, although oncological outcomes were similar with both modalities, TORS appeared to achieve better functional results. 29 The authors note that among the 44 included studies, there were no randomized studies comparing TORS and RT. As well, a large proportion of TORS patients within the review were treated with adjuvant RT (26%) or CRT (41%), challenging the ability to directly compare surgery and RT.
Clinical trials employing TORS include ORATOR (NCT01590355), ORATOR2 (NCT03210103), ECOG-3311 (NCT01898494), and the AVOID trial (NCT02159703), among others.30 -34 A similarity among these trials is that the majority of patients enrolled still require adjuvant RT postoperatively and suffer the short-term and long-term side effect profiles of both modalities. In the ORATOR study, patients were randomized to primary TORS and ND with risk-based adjuvant RT or CRT or definitive RT/CRT based on nodal status at diagnosis. 30 Seventy-six percent (26/34) of patients in the surgical arm required adjuvant treatment, with 16/34 receiving adjuvant RT and 10/34 receiving adjuvant CRT. 30 The study reported statistically superior swallowing-related QOL in the RT group at 1 year, although this finding was not clinically significant. The synergistic locoregional adverse effects of adjuvant RT in the TORS arm limits the direct comparison of the surgery versus RT cohorts and diminishes the importance of the statistical difference between dysphagia QOL results. However, the study demonstrates that when adjuvant RT/CRT is used following TORS, there does not seem to be worsening in patient-reported swallowing outcomes (P = .76). In a follow-up study, there were no significant differences in total swallowing-related QOL scores at 2 or 3 years when comparing the surgery and RT groups. 35 However, longitudinal analyses using linear mixed-effects models identified a statistically difference favoring the RT arm (P = .049). ORATOR2 randomized 31 patients to the surgical arm of whom 21 patients underwent adjuvant RT, with a median dose of 52 Gy.30,31 Although swallowing-related QOL scores at 1 year were similar between groups (P = .85), the patients suffered significant treatment toxicity, including 3 deaths in the surgical group leading to early closure of the trial and limiting the power in statistical comparisons. In ECOG-3311, patients are stratified by their postoperative pathology and 339 of the 377 enrolled patients received adjuvant RT or CRT with RT dosages ranging between 50 and 66 Gy. 33 This trial used patient-reported outcomes measures to assess overall QOL and swallowing-related QOL. This group found that there is an initial decline in scores postoperatively for all patients but those who did not require adjuvant RT or who received adjuvant RT without concomitant chemotherapy recovered to baseline. The cohort requiring adjuvant CRT had improved QOL scores but did not recover their pretreatment baseline. The initial decline is expected, and is seen in our patients, as well. Most clinical trials using TORS still require adjuvant RT, a large contributor to posttreatment adverse effects. These trials contrast our protocol which avoided RT in 17/19 patients. Those who underwent salvage CRT are those with worrisome pathological features (lymphovascular invasion and perineural invasion or carcinoma in situ at the resection site) but have been free of disease for over 3 years.
The treatment protocol that our 19 study patients underwent, the paradigm of systemic escalation (neoadjuvant chemotherapy) followed by locoregional de-escalation (surgical consolidation), is being carried out in an ongoing clinical trial (NeCTORS, NCT02760667). There are several advantages of NeCTORS compared to standard of care surgery with adjuvant RT or CRT. The systemic therapy intensification addresses possible micrometastases, an important element given distant metastatic disease is the main cause of failure in HPV-related OPSCC patients. 36 As it relates to locoregional treatment, the neoadjuvant chemotherapy results in significant downstaging of the primary tumor and nodal metastasis allowing for definitive TORS and ND, eliminating the need for adjuvant RT or CRT based on trial treatment protocol in vast majority of patients (>95%).9,22 Finally, this regimen allows RT to be reserved for salvage.
The treatment approach utilizes radical locoregional de-escalation by avoiding adjuvant RT in additional to TORS.16,20 This protocol prevents secondary RT-induced effects on all major and minor salivary glands within the upper aerodigestive tract, avoids lymphedema, preserves the muscles of mastication, and short-term oropharyngeal swallowing dysfunction caused from surgical resection improves with time. Studies of patients undergoing TORS with RT have demonstrated poorer swallowing function recovery and worse swallowing efficiency, based on oral transit time, increased residue, narrower cricopharyngeal opening, as compared to surgery-only controls who had improvement in swallowing between 3 and 12 months posttreatment.37 -39 Other research exploring the effects of adjuvant treatments on dysphagia-related QOL also note unfavorable findings.40 -43 Specifically, a cross-sectional study of OPSCC patients undergoing TORS found that those not necessitating adjuvant therapy (n = 10) had significantly better Xerostomia Index scores and dysphagia-related QOL in the composite, physical, and emotional subdomains of the MD Anderson Dysphagia Inventory compared to those receiving adjuvant RT (n = 30) and CRT (n = 22). 40 Post-RT, dysphagia may be due to fibrosis causing poor tongue base mobility and poor contact to the posterior pharyngeal wall, impaired laryngeal elevation, and reduced vestibule and true vocal cord closure during the swallow. 44
There are several limitations to the present study. Factors including single-institution, small sample size, selection bias of eligibility/fitness for surgery, and all surgeries performed by one highly experienced surgeon limit the generalizability of the findings. Transient chemotherapy-related adverse effects were unlikely to be captured as the first follow-up questionnaires were distributed after the surgical intervention, between 7 and 9 weeks after chemotherapy. This study also lacks comparison to patients treated with standard of care therapies.
Conclusion
In this small sample of patients treated with neoadjuvant chemotherapy followed by TORS and ND without adjuvant therapy, QOL measures reach a nadir at 1 month then rapidly return to baseline within 3 months and remain stable over the year from treatment. HPV-related OPSCC patients present with minimal symptom burden at diagnosis and their high QOL is preserved using this treatment paradigm. Within our institution, a larger cohort of patients receiving this treatment paradigm is currently being monitored in a phase II clinical trial for oncologic and QOL outcomes (NeCTORS, NCT02760667).
Footnotes
Acknowledgements
None.
Author Contributions
Concept and study design (A.D., M.J.S., N.S.); acquisition, analysis, or interpretation of data (all authors); drafting of manuscript (A.D., J.A.S., T.S., M.J.S., M.A.M., N.S.); critical revision of the manuscript (all authors).
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The authors declare that they have no competing interests.
Funding
This study was supported by a grant from the Cedar Cancer Foundation and the Research Institute of McGill University Health Centre.
Ethics Approval and Consent to Participate
Ethics approval for the study was obtained from the McGill University Health Centre research ethics board REB (MP-37-2018-3568).
