Abstract
Background
The high incidence of pediatric acute otitis media (AOM) makes the implications of overdiagnosis and overtreatment far-reaching. Quality indicators (QIs) for AOM are limited, drawing from generalized upper respiratory infection QIs, or locally developed benchmarks. Recognizing this, we sought to develop pediatric AOM QIs to build a foundation for future quality improvement efforts.
Methods
Candidate indicators (CIs) were extracted from existing guidelines and position statements. The modified RAND Corporation/University of California, Los Angeles (RAND/UCLA) appropriateness methodology was used to select the final QIs by an 11-member expert panel consisting of otolaryngology—head and neck surgeons, a pediatrician and family physician.
Results
Twenty-seven CIs were identified after literature review, with an additional CI developed by the expert panel. After the first round of evaluations, the panel agreed on 4 CIs as appropriate QIs. After an expert panel meeting and subsequent second round of evaluations, the panel agreed on 8 final QIs as appropriate measures of high-quality care. The 8 final QIs focus on topics of antimicrobial management, specialty referral, and tympanostomy tube counseling.
Conclusions
Evidence of variable and substandard care persists in the diagnosis and management of pediatric AOM despite the existence of high-quality guidelines. This study proposes 8 QIs which compliment guideline recommendations and are meant to facilitate future quality improvement initiatives that can improve patient outcomes.

Keywords
Background
Acute otitis media (AOM) is a common pediatric condition characterized by middle ear effusion (MEE) and acute signs and symptoms of inflammation. 1 It can manifest by a viral and/or bacterial infection that presents as otalgia, otorrhea, and/or nonspecific symptoms such as fever, irritability, tugging of the ear, or poor feeding in younger children. 2 AOM is generally self-limiting, but its recurrence and frequency entail a significant healthcare utilization burden. 3 Reports estimate that AOM is one of the leading causes for medical consultation in the pediatric population, affecting approximately 50% to 85% of children before the age of 3. 4 The American Academy of Pediatrics estimates that there are more than 5 million cases of AOM annually, resulting in over $1 billion USD in healthcare costs per annum in the United States alone.5 -10
The cost associated with increased healthcare utilization by AOM spans multiple specialties, including family medicine, pediatrics, emergency medicine, and otolaryngology—head and neck surgery. With multiple points of contact for AOM patients in the healthcare system, inconsistent care has been demonstrated. 8 Inconsistencies in diagnosis and management can lead to poor patient care, but also has the potential for far-reaching global impacts on antimicrobial resistance.11,12 AOM has become the most common reason for antimicrobial prescriptions and has been associated with a high prevalence of resistant pathogens isolated from the nasopharynx and the middle ear.13 -15 Despite these challenges and practice variations, measurement tools of quality care in the management of AOM are lacking.
Healthcare quality aims to increase the likelihood of desired health outcomes and is a measurable construct that can be broken down into 6 key areas: safety, effectiveness, patient centeredness, timeliness, efficiency, and equity. 16 In addition, the Donabedian model is a widely accepted framework used to create measurable Quality Indicators (QIs) in healthcare based on the 3 domains of structure, process, and outcome measures. 17 Used as a tool, QIs help facilitate resource prioritization, practice accountability, practice comparisons, and quality improvement monitoring.18,19
The lack of standardized QIs for AOM care provides an opportunity for improvement by creating a universal performance measurement system. The benefits extend to patients, providers, and the overall healthcare system due to AOM’s significant prevalence and healthcare burden, in addition to aiding providers in their care and optimizing best practices. As such, we sought to develop pediatric AOM QIs to facilitate quality improvement efforts focusing on patient quality of life, providers’ diagnostic and management accuracy, treatment optimization, in addition to building a foundation for future quality improvement efforts in the management of this disease.
Materials and Methods
An expert panel was developed which consisted of: 9 pediatric otolaryngologists, 1 pediatrician, and a family physician, with both academic and community experience. Panelists were solicited from the pediatric working group of the Canadian Society of Otolaryngology—Head and Neck Surgery, with names brought forward based on clinical and research experience in AOM diagnosis and management and demonstrated interest in quality improvement. Otolaryngology participants were selected to ensure geographic diversity, reflecting potential regional differences in practice types and patient populations. Recognizing that pediatricians and family physicians are often the first point of contact for this patient population, the 9-member panel was expanded to include a pediatrician and family physician to facilitate multidisciplinary perspectives. Candidate indicators (CI) were derived by 2 authors (EM and JC) using an adapted guideline-based approach that also allowed for the use of both high-quality guidelines and position statements. 20 These included the American Academy of Pediatrics Clinical Practice Guideline on The Diagnosis and Management of Acute Otitis Media, the American Academy of Otolaryngology—Head & Neck Surgery Clinical Practice Guidelines for Tympanostomy Tubes in Children, and the Canadian Paediatric Society Position Statement on Management of Acute Otitis Media in Children Six Month of Age and Older were utilized.8,21,22 These 3 resources were determined by the expert panel to be comprehensive, of clinical relevance, and high quality, with guidelines meeting AGREE II criteria. 23
Inclusion criteria were children aged 6 months to 18 years of age. Children with anatomic abnormalities including cleft palate, conditions with craniofacial abnormalities, immune deficiencies, and the presence of cochlear implants were excluded. CIs were named and typed based on the 3 dimensions of the Donabedian Model. 18 To facilitate real-world measurement, a numerator and denominator was constructed, along with risk adjustment criteria and specific exclusion criteria. To assist panelists in evaluations, existing literature for each CI was provided and variations between guiding documents was summarized.8,21,22
The modified RAND Corporation/University of California, Los Angeles (RAND/UCLA) appropriateness methodology was used to select the final QIs. 24 This consisted of 2 rounds of anonymous, independent ratings by panel members for each CI. A teleconference occurred between the 2 rounds of ratings to discuss the results of the first round, areas of disagreement, and to bring forward CIs that may have importance in AOM management that were not originally extracted. Panelists were asked to provide a global score utilizing a 9-point Likert scale which was informed by the CIs validity, reliability, and feasibility of measurement. QIs deemed appropriate measures of quality care received a median score of 7 to 9, and agreement among panelists, meaning no more than 3 panel members rated the CI outside the 3-point region (7-9) containing the median.18,19,24
Results
A total of 28 CIs were obtained for evaluation (Table 1). Twenty-seven process CI were extracted from the 3 practice guidelines. One process CI was derived by the expert-panel between the 2 rounds of ratings which recommended no antimicrobial therapy for persistent MEE after AOM. No structure or outcome CIs were extracted.
All Candidate Indicator.

Abbreviations: AAFP, American Academy of Family Physicians; AAP, American Academy of Pediatrics; AOM, acute otitis media; MEE, middle ear effusion; OME, otitis media with effusion; TM, tympanic membrane; TTO, tympanostomy tube otorrhea.
After the first period of evaluations, 4 CIs were viewed as appropriate with agreement among all panelists with panelist agreement ranging from 73% to 91% (Table 2). The panel chose to exclude the other 23 CIs for a variety of reasons, including concerns of poor inter-rater reliability of patient history and physical examination findings, inability to measure CIs due to a lack of infrastructure, and failure to generalize the CI geographically or across specialties due to lack of medication/device access or clinical skill (eg, pneumatic otoscopy). The follow-up teleconference highlighted the results of the first rounds of ratings, and then focused on areas of disagreement to help guide the panel into the second round of voting. After a discussion on the geographic variability in antimicrobial resistance patterns, the consensus was to refrain from making specific dosing recommendations for antibiotics. Instead, practitioners should select an appropriate dosage strength based on the local context.
Results From First Round of Voting; CIs Deemed Appropriate With Agreement.

Scores based on a 9-point Likert scale.
Abbreviations: AOM, acute otitis media; CI, candidate indicators.
After the second round of evaluations, the panel deemed 3 of the original guideline-derived CIs that had disagreement, along with the panel-derived CI, to be appropriate QIs, with panelist agreement ranging from 91% to 100% (Table 3). This provided a total of 8 QIs (Table 4) to be appropriate indicators of quality care. Several CIs had an overall median assessment score of being deemed appropriate (score between 7 and 9) yet were not accepted as QIs due to disagreement with reasons defined on Table 5.
Additional CIs Deemed Appropriate With Agreement Following Second Round of Voting.

Scores based on a 9-point Likert scale.
Abbreviations: AOM, acute otitis media; CI, candidate indicator; MEE, middle ear effusion; TTO, tympanostomy tube otorrhea.
Summary of Final QIs.
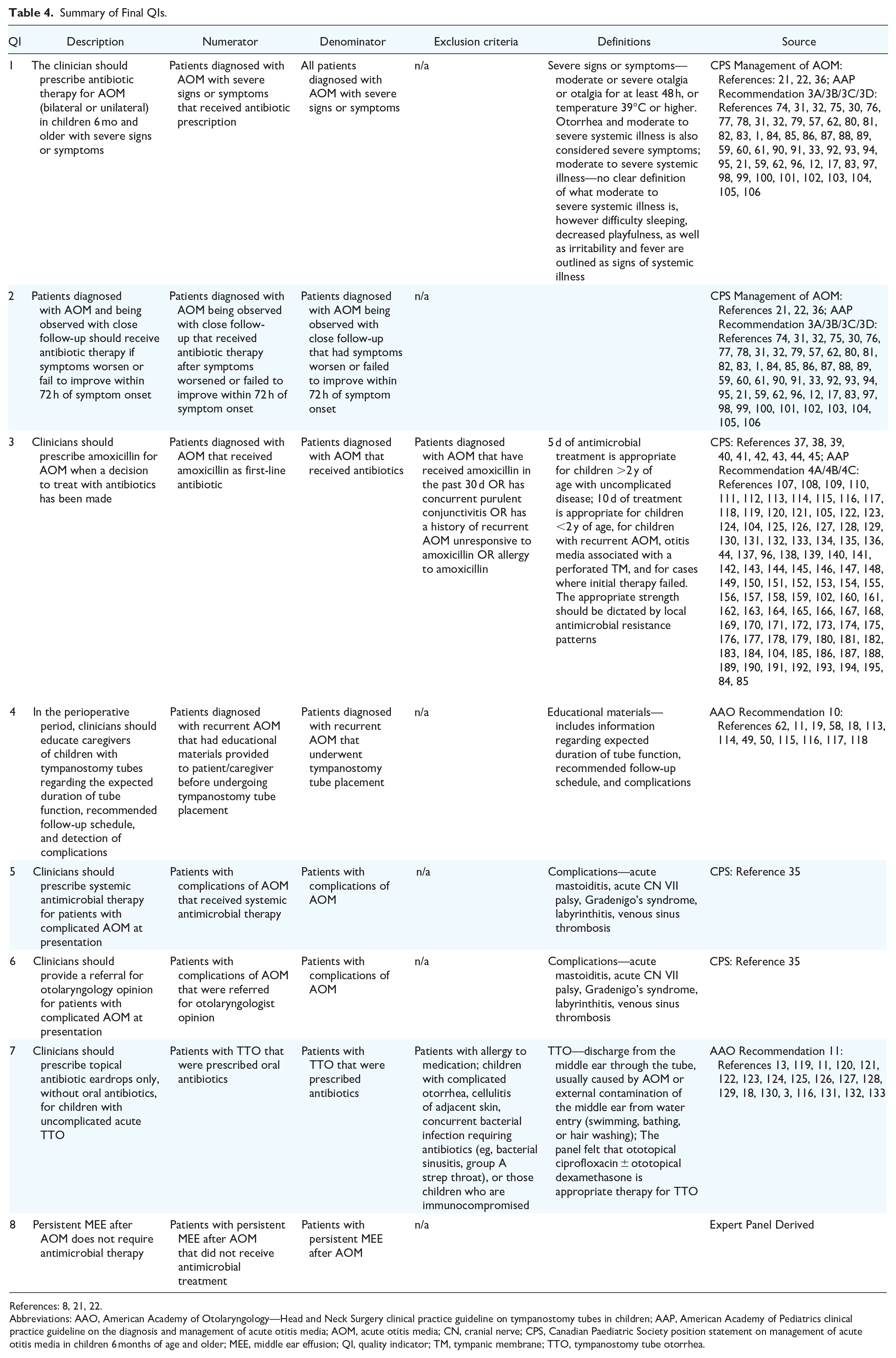
References: 8, 21, 22.
Abbreviations: AAO, American Academy of Otolaryngology—Head and Neck Surgery clinical practice guideline on tympanostomy tubes in children; AAP, American Academy of Pediatrics clinical practice guideline on the diagnosis and management of acute otitis media; AOM, acute otitis media; CN, cranial nerve; CPS, Canadian Paediatric Society position statement on management of acute otitis media in children 6 months of age and older; MEE, middle ear effusion; QI, quality indicator; TM, tympanic membrane; TTO, tympanostomy tube otorrhea.
Candidate Indicators Deemed Appropriate but With Disagreement.

Scores based on a 9-point Likert scale.
Abbreviations: AOM, acute otitis media; MEE, middle ear effusion; QI, quality indicator; TM, tympanic membrane; OME, otitis media with effusion; WHO, World Health Organization.
Discussion
As healthcare systems seek to improve resource efficiency and quality care, it is imperative to set quality standards with measurable outcomes as a foundation for patient care. It has been suggested that guideline adherence for AOM are suboptimal. 25 Studies in recent years from countries including the United States, United Kingdom, Italy, Sweden, Israel, Greece, and Turkey showed overall low to moderate guideline compliance rates for healthcare providers including general practitioners, pediatricians, and otolaryngologists.26 -31 Deviations from guidelines have been significant with estimations showing greater than 80% of the diagnosed cases of AOM being treated immediately with antibiotics, out of step with guideline recommendations.26,32 -34 There are legitimate reasons why strict adherence to guideline recommendations may not occur, for example, when level I evidence is lacking and different interpretations of the literature exist, or when the context of a clinical encounter sufficiently varies from what a guideline describes. Such legitimate barriers to guideline adherence can similarly be found in QI adoption. Recognizing this reality, however, does not justify complete abolition of quality measurement and improvement efforts. Utilizing an inclusive, iterative, and thoughtful approach to QI design and implementation can improve buy-in from providers and better capture the desired elements of quality being measured.
QIs for AOM are limited, drawing from generalized upper respiratory infection QIs, or locally developed benchmarks. 35 With minimal initiatives in quality improvement for AOM management, we sought to build the foundation for such a prevalent condition. As such, the QIs were generated through a rigorous evidence-based methodology to account for a broad spectrum of management strategies to assess the quality of care for patients with AOM. We believe that future quality improvement initiatives for AOM can be strongly facilitated by the 8 QIs developed in this study.
We have found that a guideline-based approach works well for QI development due to the high-quality evidence used in their creation; however, such a focus on clinical practice recommendations does result in a greater propensity toward process indicators.36,37 With a lack of structure and outcome QIs, future scientific exploration of these quality measures for AOM exists, for example, exploring performance thresholds of health-related quality of life tools such as Otitis Media-6. 38
To our knowledge, there are currently no existing standardized North American QIs for AOM management. Previous QIs have been developed in Europe with a focus on accurate diagnoses through clinical evaluation of factors like eardrum mobility, along with evaluating antibiotic prescribing practices.39,40 In reviewing the QIs developed in this study, there are notable differences in terms of clinical recommendations, such as the preference toward amoxicillin rather than penicillin V in European QIs. Furthermore, while European QIs prioritized the evaluation of eardrum mobility for quality improvement measurement, the panel in this study felt that other areas of clinical care should take precedent. These differences are likely the result of variations between North American and European care environments and demonstrate the importance of undergoing an evidence-based process of developing QIs that maximize their relevance and utility for North American use. The QIs presented in this study introduce measures that can be evaluated through a majority of the Institute of Medicine’s 6 quality domains. 16 Specifically, these QIs provide evaluation in areas of safety, effectiveness, timeliness, patient-centeredness, and efficiency. Equity care measures provide an opportunity for future creation.
Working closely with care providers to thoughtfully implement QIs in a way that recognizes the complexity and variability of healthcare ecosystems will be paramount, necessitating an iterative process as field experience is gained and literature evolves. To effectively evaluate QIs, healthcare systems require detailed data that at present are often lacking or inaccurate. Quality care improvements will necessitate detailed, accessible, and self-sustaining measurement methods from providers and healthcare systems to ultimately obtain substantive outcome improvements for patients. Despite challenges, QIs serve as a prerequisite for measuring the quality of care provided to patients and therefore warrant funding and initiatives that can overcome barriers to use. This will serve to help increase the likelihood of desired healthcare outcomes for patients, which is the ultimate goal of all quality improvement efforts.
Limitations
The authors acknowledge the international variability in healthcare systems and quality perception that may affect the appropriateness of the proposed QIs and the necessity for local adjustments, therefore one of the key limitations of the final QIs proposed in this study may not be generalizable to all healthcare practice settings. However, the 28 CIs (Table 4) originally extracted can be used as a reference for future groups interested in similar QI development. The authors also recognize that the composition of the panel may have affected the results of this study. Our panel was mainly comprised of otolaryngologists with 1 pediatrician and family physician. Including other important stakeholders such as patients, infectious disease specialists, nurse practitioners, and other allied healthcare professionals in future studies would be beneficial and potentially result in different outcomes. The authors feel that this would be considered complimentary, and not mutually exclusive to our efforts and may highlight a better understanding between different specialty practices and clinical environments. The authors also acknowledge that these QIs have not yet undergone real-world validity testing. It is important that future studies consider small scale real-world testing of these QIs prior to large-scale system implementation.
Conclusions
This study proposes 8 QIs for the management of patients with AOM. With our development of QIs for the diagnosis and management of AOM, the authors of the present study aim to empower practitioners and healthcare systems to measure the quality of care provided to patients. Setting this standard will further support accountability, regulation, and accreditation, while also leading to prioritization of quality improvement initiatives.
Supplemental Material
sj-docx-1-ohn-10.1177_19160216241248538 – Supplemental material for Proposed Quality Indicators for Aspects of Pediatric Acute Otitis Media Management
Supplemental material, sj-docx-1-ohn-10.1177_19160216241248538 for Proposed Quality Indicators for Aspects of Pediatric Acute Otitis Media Management by Justin Cottrell, Amirpouyan Namavarian, Jonathan Yip, Paolo Campisi, Neil K. Chadha, Ali Damji, Paul Hong, Sophie Lachance, Darren Leitao, Lily H. P. Nguyen, Natasha Saunders, Julie Strychowsky, Warren Yunker, Jean-Philippe Vaccani, Yvonne Chan, John R. de Almeida, Antoine Eskander, Ian J. Witterick and Eric Monteiro in Journal of Otolaryngology - Head & Neck Surgery
Footnotes
Acknowledgements
None.
Author Contributions
JC, EM, JY, PC, YC, JRDA, AE, and IJW were involved in the project idea inception and design. JC and EM were involved in completing the search, conducting the panel discussion, and finalizing the quality indicators (QIs) for AOM. NKC, AD, PH, SL, DL, LHPN, NS, JS, WY, and JPV were involved in the expert panel rating and discussion for QIs. AN and JC were involved in the manuscript writing and AN, JC, EM, JY, PC, YC, JRDA, AE, and IJW were involved in the manuscript editing. All authors read and approved the final manuscript.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
