Abstract
In this edition of the Huntington's Disease Clinical Trials Update, we expand on the ongoing phase I clinical trial of ALN-HTT02 from Alynlam Pharmaceuticals. We also report on the SAGE-718 (also known as dalzanemdor) program from Sage Therapeutics, with results of the phase II DIMENSION study and the recent termination of the open-label phase III PURVIEW study. Additionally, we discuss recent developments in the regulatory pathway for AMT-130, following discussions between uniQure and the U.S. Food and Drug Administration regarding key aspects of accelerated approval. Finally, we provide a comprehensive listing of all currently registered and ongoing clinical trials in Huntington's disease.
Plain language summary
In this edition we expand on the ongoing phase I clinical trial of ALN-HTT02 from Alynlam Pharmaceuticals. We also report on the SAGE-718 (also known as dalzanemdor) program from Sage Therapeutics, with results of the phase II DIMENSION study and the recent termination of the open-label phase III PURVIEW study. Additionally, we discuss recent developments in the regulatory pathway for AMT-130, following discussions between uniQure and the U.S. Food and Drug Administration regarding key aspects of accelerated approval. Finally, we provide a comprehensive listing of all currently registered and ongoing clinical trials in Huntington's disease.
Introduction
The Clinical Trials Update is a regular feature devoted to highlighting ongoing and recently completed clinical trials in Huntington's disease (HD). Clinical trials previously reviewed in this section are listed in Table 1.
Clinical trials previously reviewed by the Huntington's Disease Clinical Trials Update.
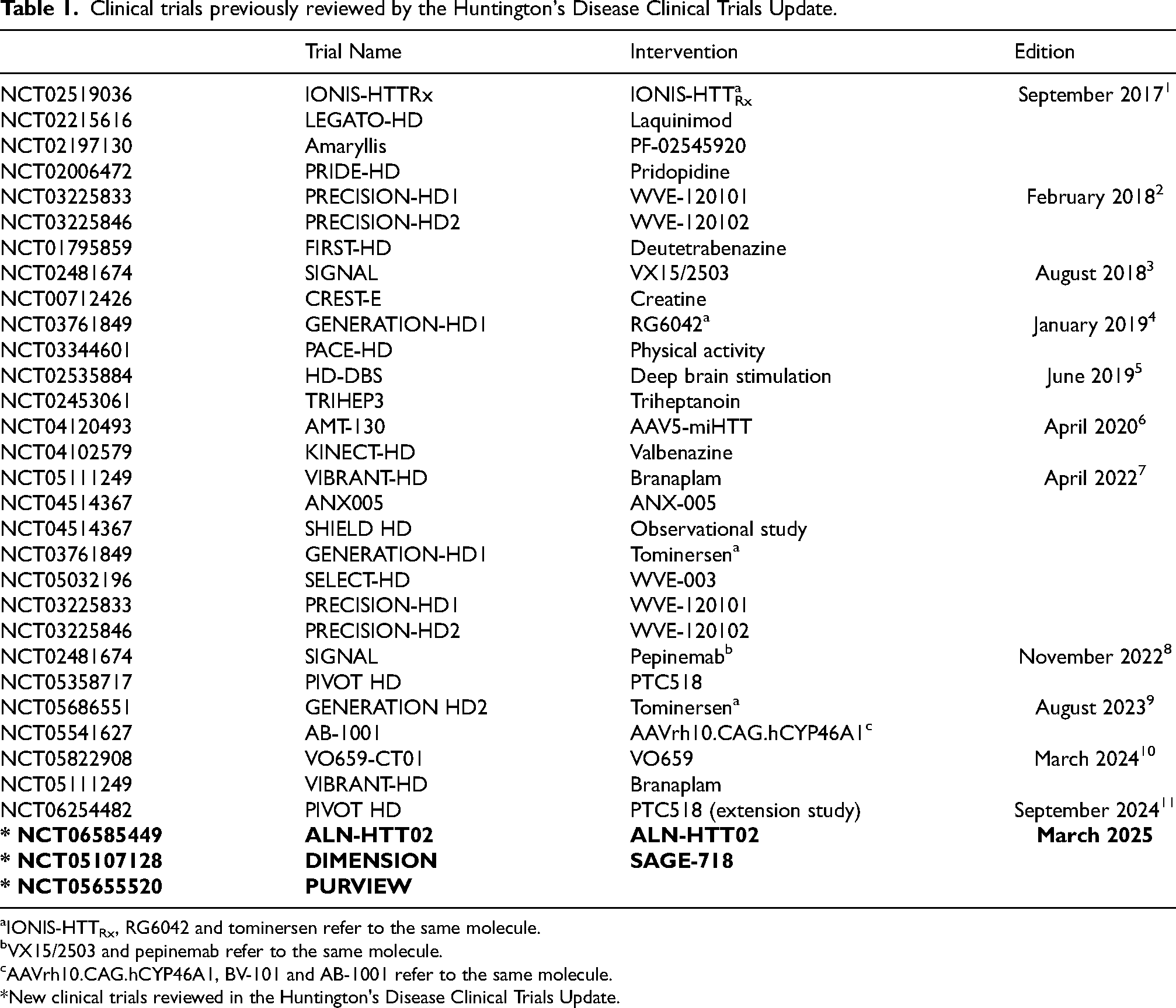
IONIS-HTTRx, RG6042 and tominersen refer to the same molecule.
VX15/2503 and pepinemab refer to the same molecule.
AAVrh10.CAG.hCYP46A1, BV-101 and AB-1001 refer to the same molecule.
New clinical trials reviewed in the Huntington's Disease Clinical Trials Update.
In this edition, we expand on the phase I clinical trial of ALN-HTT02 (NCT06585449) 12 from Alynlam Pharmaceuticals. We also report on the program studying SAGE-718 (also known as dalzanemdor) from Sage Therapeutics, with results of the phase II DIMENSION study (NCT05107128) 13 and the recent termination of the open-label phase III PURVIEW study (NCT05655520). 14 Additionally, we discuss recent developments in the regulatory pathway for the huntingtin-lowering gene therapy AMT-130, following discussions between its sponsor uniQure and the U.S. Food and Drug Administration regarding key aspects of accelerated approval. 15 Finally, we provide a comprehensive listing of all currently registered and ongoing clinical trials in Huntington's disease.
We tabulate all currently registered and ongoing clinical trials in Tables 2–4. For further details on the methodology used, please refer to the first edition of this series. 1
Pharmacological clinical trials registered at the World Health Organization (WHO) International Clinical Trials Research Platform (ICTRP) for people with HD since the first edition of the “Clinical Trials Corner”.
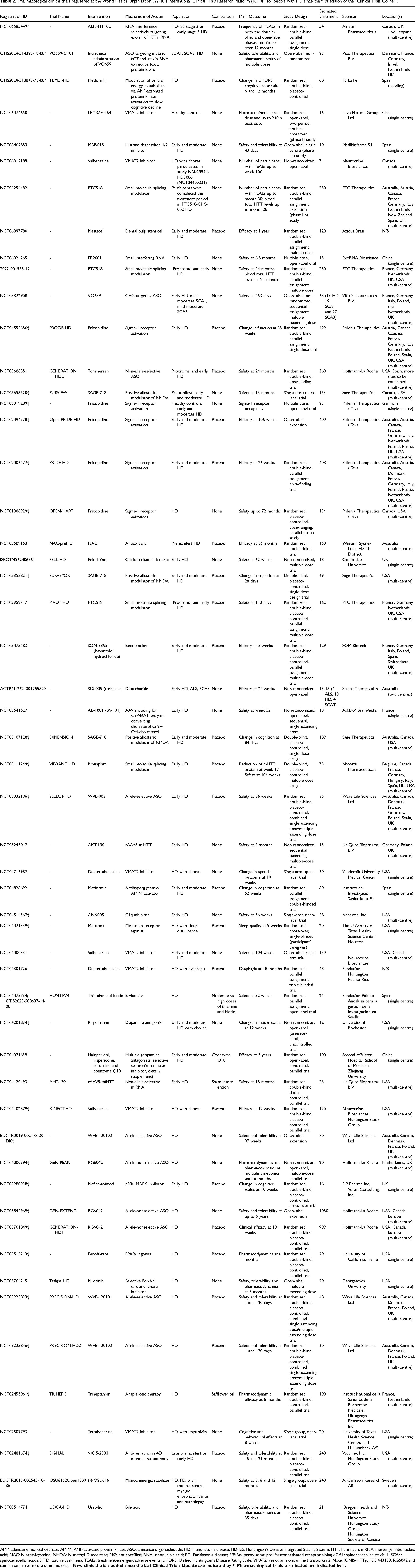
AMP: adenosine monophosphate; AMPK: AMP-activated protein kinase; ASO: antisense oligonucleotide; HD: Huntington's disease; HD-ISS: Huntington's Disease Integrated Staging System; HTT: huntingtin; mRNA: messenger ribonucleic acid; NAC: N-acetylcysteine; NMDA: N-methyl-D-aspartate; N/S: not specified; RNA: ribonucleic acid; PD: Parkinson's disease; PPARα: peroxisome proliferator-activated receptor alpha; SCA1: spinocerebellar ataxia 1; SCA3: spinocerebellar ataxia 3; TD: tardive dyskinesia; TEAEs: treatment-emergent adverse events; UHDRS: Unified Huntington's Disease Rating Scale; VMAT2: vesicular monoamine transporter 2. Note: IONIS-HTTRx, ISIS 443139, RG6042 and tominersen refer to the same molecule.
Invasive non-pharmacological clinical trials registered at the WHO ICTRP for people with HD since the first edition of the “Clinical Trials Corner”.
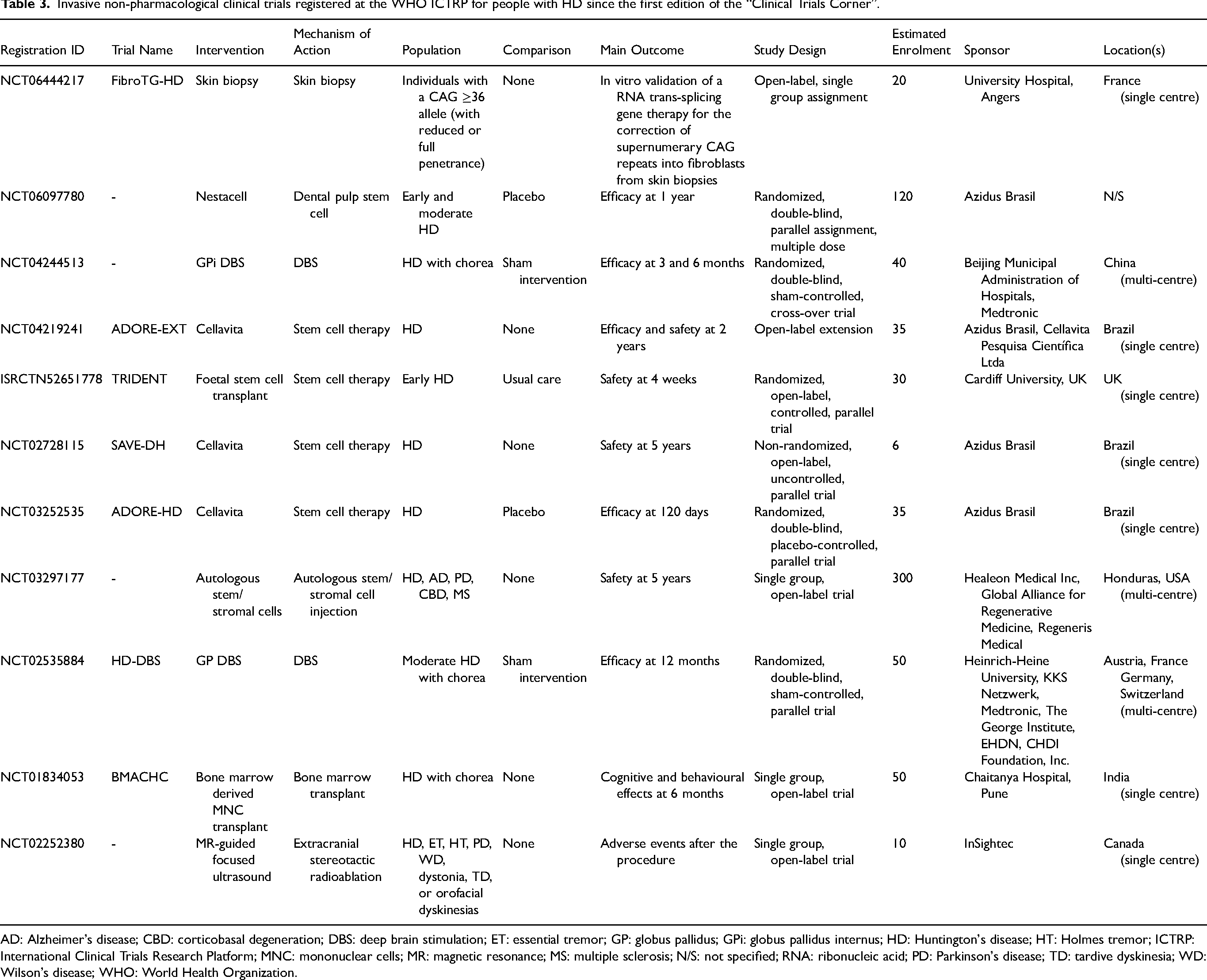
AD: Alzheimer's disease; CBD: corticobasal degeneration; DBS: deep brain stimulation; ET: essential tremor; GP: globus pallidus; GPi: globus pallidus internus; HD: Huntington's disease; HT: Holmes tremor; ICTRP: International Clinical Trials Research Platform; MNC: mononuclear cells; MR: magnetic resonance; MS: multiple sclerosis; N/S: not specified; RNA: ribonucleic acid; PD: Parkinson's disease; TD: tardive dyskinesia; WD: Wilson's disease; WHO: World Health Organization.
Non-invasive non-pharmacological clinical trials registered at the WHO ICTRP for people with HD since the first edition of the “Clinical Trials Corner”.
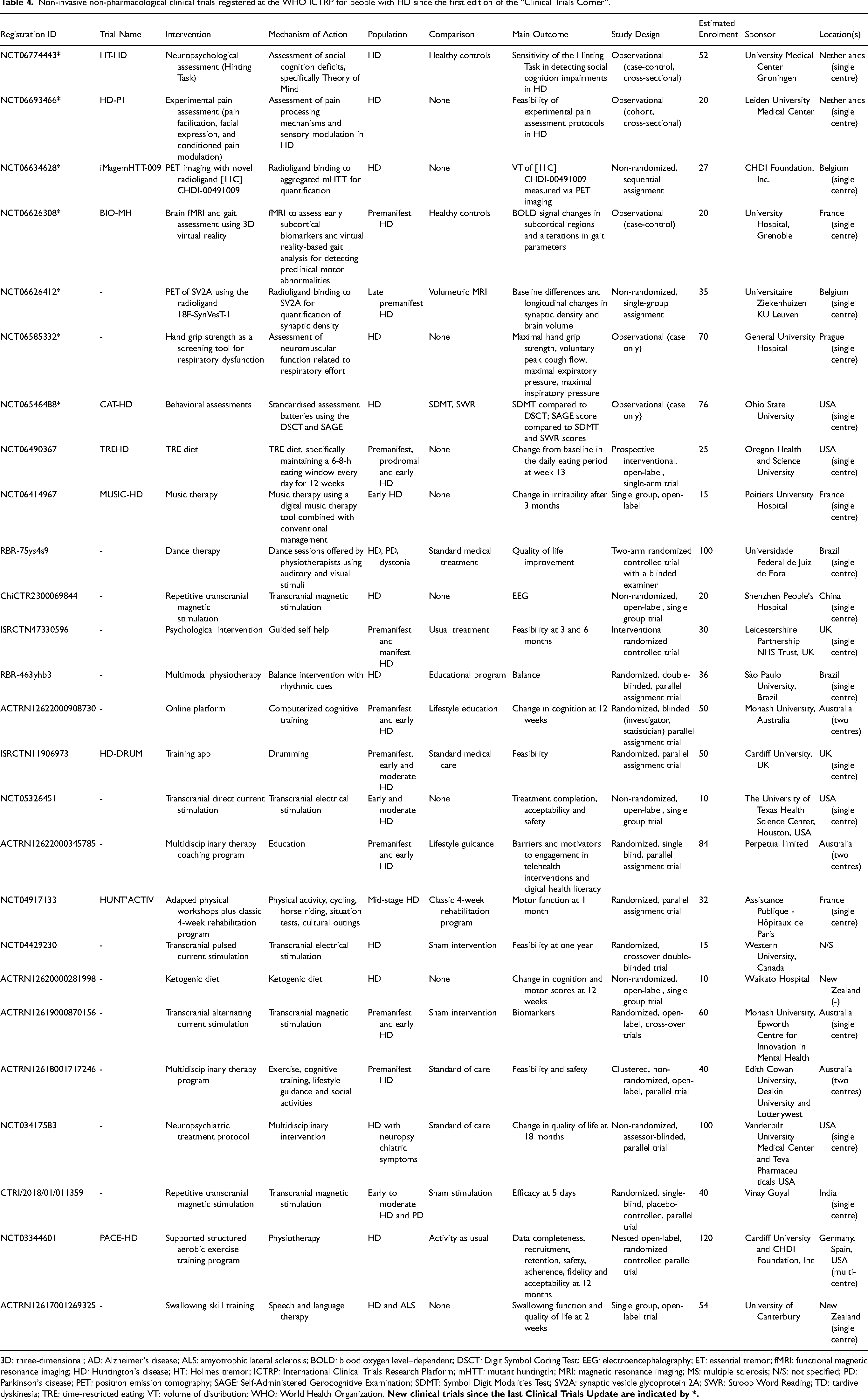
3D: three-dimensional; AD: Alzheimer's disease; ALS: amyotrophic lateral sclerosis; BOLD: blood oxygen level–dependent; DSCT: Digit Symbol Coding Test; EEG: electroencephalography; ET: essential tremor; fMRI: functional magnetic resonance imaging; HD: Huntington's disease; HT: Holmes tremor; ICTRP: International Clinical Trials Research Platform; mHTT: mutant huntingtin; MRI: magnetic resonance imaging; MS: multiple sclerosis; N/S: not specified; PD: Parkinson's disease; PET: positron emission tomography; SAGE: Self-Administered Gerocognitive Examination; SDMT: Symbol Digit Modalities Test; SV2A: synaptic vesicle glycoprotein 2A; SWR: Stroop Word Reading; TD: tardive dyskinesia; TRE: time-restricted eating; VT: volume of distribution; WHO: World Health Organization.
If you would like to draw attention to specific trials, please feel free to email us at: mena.farag@ucl.ac.uk and e.wild@ucl.ac.uk.
Ongoing clinical trials
A list of all registered clinical trials is given in Tables 2, 3 and 4.
ALN-HTT02 (NCT06585449) 12
ALN-HTT02 is a synthetic double-stranded RNA interference therapy designed to target exon 1 of HTT mRNA, leading to a non-selective reduction of both mutant (mHTT) and wild-type HTT proteins including, in principle, the exon 1 missplice variant sometimes known as HTT1a. Conjugated with C16 to enhance CNS delivery, ALN-HTT02 is administered intrathecally and expected to have a half-life consistent with twice-yearly or even less frequent dosing. Participants who receive placebo during the double-blind phase will have the option to receive one dose of ALN-HTT02 in an open-label extension.
The primary outcome measure is the frequency of adverse events (AEs) over 12 months in both the double-blind and open-label study phases. Secondary outcome measures include changes in cerebrospinal fluid (CSF) mHTT levels, as well as CSF, plasma and urine concentrations of ALN-HTT02, all assessed over 12 months in both study phases.
Recruitment for the clinical trial commenced on 14th October 2024, with an estimated study completion date of 5th July 2028. Currently, the study is actively being conducted across two locations, in the UK and Canada, and is expected to expand globally.
The first program to utilise Alnylam's proprietary C16-siRNA technology was ALN-APP (NCT05231785), 17 in collaboration with Regeneron, designed to target amyloid precursor protein (APP) for the potential treatment of Alzheimer's disease (AD) and cerebral amyloid angiopathy (CAA). The phase I clinical trial employed a double-blind, placebo-controlled, single ascending dose approach with the aim to assess the safety, tolerability, pharmacokinetic and pharmacodynamic effects of ALN-APP in patients with early-onset AD; 12 the ALN-HTT02 trial has a similar design. Interim results for ALN-APP, announced by Alnylam Pharmaceuticals in a press release on 25th October 2023, 18 reported that single doses demonstrated sustained pharmacodynamic activity for up to 10 months post-administration. In addition, the treatment led to marked reductions in Aβ42 and Aβ40, key proteins implicated in progression of AD and CAA. From a safety perspective, among the 20 patients randomised to receive ALN-APP or placebo, one subject experienced two mild AEs deemed related to both the study drug and procedure, but no serious AEs or deaths were reported. 19 These interim results for ALN-APP represent the first-in-human translation of an RNA interference therapeutic for neurodegenerative diseases, with the ongoing ALN-HTT02 clinical trial in HD expected to improve understanding of the approach and potential therapeutic benefits of this strategy.
Completed clinical trials
SAGE-718 (NCT05107128; NCT05655520)13,14
The primary outcome measure was the change from baseline in the Symbol Digit Modalities Test (SDMT) score, with higher scores indicating greater cognitive processing speed. Secondary outcomes included changes from baseline in clinical, cognitive and functional assessments. Functional independence was assessed using the UHDRS Independence Scale (IS), where higher scores indicated greater function. Cognitive performance was assessed through the Trail Making Test Part B of the MoCA and the One Touch Stockings of Cambridge (OTS) task, with longer completion times reflecting greater impairment. Motor timing accuracy was measured using the Paced Tapping Test (PTAP), where higher scores indicated improved precision. Functional capacity in daily life was assessed using the HD Everyday Functioning (Hi-DEF) Home Subdomain score, with lower scores reflecting better functional abilities. The Clinical Global Impression – Severity (CGI-S) Cognitive Status Subdomain score was used to assess cognitive symptom severity, with higher scores indicating greater impairment. Safety and tolerability were also assessed as a secondary outcome measure by monitoring the incidence of treatment-emergent AEs (TEAEs).
Recruitment for the clinical trial began on 10th February 2022 and was completed on 3rd October 2024. The study was conducted across 45 sites.
Breaking news
In this section we provide brief updates about ongoing or recently terminated clinical trials.
The
On 10th December 2024, uniQure provided an optimistic update on its discussions with the U.S. Food and Drug Administration (FDA) regarding key aspects of the accelerated approval pathway for AMT-130 in HD. 15 During a Regenerative Medicine Advanced Therapy (RMAT) Type B meeting held in late November 2024, the FDA agreed that data from the ongoing phase I/II studies, compared against an external natural history control, could serve as the primary evidence for a Biologics License Application (BLA) submission under the Accelerated Approval pathway. The FDA also endorsed the use of composite UHDRS (cUHDRS) 24 as an intermediate clinical endpoint, which is a composite measure integrates scores from multiple assessments, including the UHDRS TFC, Total Motor Score (TMS), SDMT and Stroop Word Reading (SWR). The FDA also acknowledged that reductions in CSF neurofilament light chain (NfL) could serve as supportive evidence of therapeutic benefit, given supportive evidence with reductions in CSF NfL levels observed in treated participants at 24 months relative to baseline. 25
While it cannot be assumed that statements made in a RMAT meeting with a single sponsor will automatically apply to other programs, it is highly encouraging to learn of regulatory acceptance in principle for both cUHDRS and NfL. FDA previously rejected the use of cUHDRS as a primary efficacy endpoint, but its use in place of TFC is expected to reduce clinical trial participant number requirements by over 50%. Similarly, NfL is widely seen as a reliable indicator of neuronal damage and rescue in other CNS indications, and has been used to inform regulatory decisions (e.g., for tofersen in amyotrophic lateral sclerosis); it could be highly valuable for therapeutic development for it to receive similar consideration in HD.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SJT receives research grant funding from the CHDI Foundation, Vertex Pharmaceuticals, the UK Medical Research Council, the Wellcome Trust and the UK Dementia Research Institute that receives its funding from DRI Ltd, funded by the UK MRC, Alzheimer's Society, and Alzheimer's Research UK. EJW is supported by CHDI Foundation, Inc. EJW reports grants from CHDI Foundation, and F. Hoffmann-La Roche Ltd.
Declaration of conflicting interests
MF was a sub-investigator in the ALN-HTT02 (NCT06585449), GENERATION-HD2 (NCT05686551), PTC518 (NCT05358717; NCT06254482) and uniQure AMT-130 (NCT05243017) trials.
SJT has undertaken consultancy services for Annexon, Alphasights, Alnylam Pharmaceuticals Inc., Atalanta Pharmaceuticals (SAB), F. Hoffmann-La Roche Ltd/Genentech, Guidepoint, Horama, Locanobio, LoQus23 Therapeutics Ltd (SAB), Novartis Pharma, PTC Therapeutics, Sanofi, Spark Therapeutics, Takeda Pharmaceuticals Ltd, Triplet Therapeutics (SAB), University College Irvine and Vertex Pharmaceuticals Incorporated. All honoraria for these consultancies were paid through the offices of UCL Consultants Ltd, a wholly owned subsidiary of University College London. SJT has a patent Application number 2105484.6 on the FAN1-MLH1 interaction and structural analogues licensed to Adrestia Therapeutics. SJT was an investigator in the ALN-HTT02 (NCT06585449), IONIS HTTRx (NCT02519036), IONIS HTTRx OLE (NCT03342053), GENERATION-HD1 (NCT03761849), Roche Natural History Study (NCT03664804), Roche GEN-EXTEND (NCT03842969), PTC518 (NCT05358717; NCT06254482), SHIELD-HD (NCT04406636) and uniQure AMT-130 (NCT05243017) trials.
EJW has undertaken consultancy/advisory board work with Hoffman La Roche Ltd, Triplet Therapeutics, Takeda, Vico Therapeutics, Voyager, Huntington Study Group, Teitur Trophics, EcoR1 Capital, PTC Therapeutics, Alnylam, Annexon Biosciences, Remix Therapeutics and Skyhawk Therapeutics. All honoraria for these consultancies were paid through the offices of UCL Consultants Ltd, a wholly owned subsidiary of University College London. EJW was an investigator in the ALN-HTT02 (NCT06585449), Amaryllis (NCT02197130), LEGATO-HD (NCT02215616), IONIS HTTRx (NCT02519036), IONIS HTTRx OLE (NCT03342053), GENERATION-HD1 (NCT03761849), Roche Natural History Study (NCT03664804), Roche GEN-EXTEND (NCT03842969), Roche GEN-PEAK (NCT04000594), PTC518 (NCT05358717; NCT06254482), uniQure AMT-130 (NCT05243017) and VIBRANT-HD (NCT05111249) trials.
SJT and EJW are Editorial Board Members of this journal but were not involved in the peer review process of this article nor had access to any information regarding its peer review.
Data availability statement
Data sharing not applicable as no datasets generated and/or analysed for this study.
