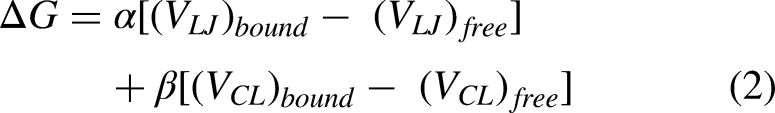Abstract
Skin cancer represents a major public health concern due to high global incidence and mortality rates. Oxidative stress induced by ultraviolet radiation plays a central role in skin carcinogenesis. Natural compounds with chemopreventive properties, such as those derived from Vaccinium meridionale Swartz, have attracted attention for their antioxidant potential. Consequently, this study evaluated the protective effect of V. meridionale against Reactive Oxygen Species (ROS)-induced skin damage in HaCaT keratinocytes. To this end, cells were treated with a water–ethanol extract (0.25–10.0 mg/mL) for 24 h, followed by hydrogen peroxide exposure. Assays assessing cell viability, ROS levels, DNA damage, and cell cycle progression were performed. Additionally, in silico analyses examined interactions between phenolic compounds and proteins involved in oxidative stress regulation, including antioxidant and pro-oxidant targets. Results showed that the extract reduced both ROS accumulation and DNA damage and that cyanidin-3-glucoside may inhibit the pro-oxidant enzyme nitric oxide synthase-2 (NOS2). These findings demonstrate the antioxidant and genoprotective activity of V. meridionale, supporting its potential role in skin cancer prevention.
Introduction
Human skin, particularly the epidermis, is constantly exposed to environmental, chemical, and physical agents that cause cellular stress. Among these factors, solar ultraviolet (UV) radiation is especially harmful because it promotes the production of Reactive Oxygen Species (ROS) and induces DNA damage. These effects can ultimately lead to mutations that increase the risk of skin cancer. 1 According to GLOBOCAN data, skin cancer is among the most frequent malignancies worldwide, accounting for approximately one-third of all cancer diagnoses. 2 Consequently, there is a pressing need to identify chemopreventive agents capable of reducing incidence and providing accessible protective strategies for at-risk populations.
In recent decades, phenolic compounds found in berries have received growing attention due to their bioactive properties and their ability to mitigate oxidative stress. 3 Particularly, species of the Vaccinium genus have demonstrated antiproliferative and pro-apoptotic activity in cancer cells, 4 which highlights their potential as chemopreventive agents. A previous review by our research group reported protective effects of phenolic compounds from Vaccinium berries against ROS-induced skin damage. 5 However, further studies are required to elucidate the underlying molecular mechanisms and to investigate lesser-studied species in greater detail.
One such species is Vaccinium meridionale, commonly known as mortiño, agraz, or Andean berry. It grows wild and is considered a promising crop throughout the Andean region, notably across Colombia, Venezuela, and Ecuador. 6 The fruit contains a diverse array of bioactive compounds, primarily flavonoids (e.g., anthocyanins) and non-flavonoids (e.g., phenolic acids), 7 which exhibit antioxidant, anti-inflammatory, antidiabetic, anticancer, and anti-aging properties. 8
Phenolic compounds in V. meridionale display in vitro antioxidant activity comparable to—or exceeding—that of well-known species such as Vaccinium myrtillus (bilberry) and Vaccinium corymbosum (blueberry).9,10 The species has been widely explored in food applications, particularly as juice, which is consumed for its reported anti-inflammatory and cardioprotective benefits.11,12 Additionally, berry extracts act as natural antioxidants, colorants, and flavor enhancers, supporting the development of functional foods and nutraceutical products.13,14
The diverse phenolic profile of V. meridionale extracts includes high concentrations of anthocyanins and other bioactive molecules recognized for their cardioprotective, antitumor, and anti-inflammatory properties.13,15,16 In vitro studies have demonstrated cytotoxic and antiproliferative activity against colon cancer SW480 and SW620 cell lines. 17 Moreover, the extract has been shown to protect fibrosarcoma HT1080 cells from rotenone-induced loss of viability 18 and to inhibit proliferation in acute myeloid leukemia OCI-AML3 cells, although less effectively than the standard drug doxorubicin. 16
Furthermore, aqueous extracts have exhibited synergistic interactions with chemotherapeutic agents such as 5-fluorouracil, leucovorin, and oxaliplatin, enhancing antiproliferative activity and reducing metastatic potential in cancer cells. 19 While several extraction methods—ranging from aqueous and ethanolic to supercritical fluid extraction10,19,20—confirm that the fruit is a rich source of therapeutic agents, existing evidence remains largely confined to tumor-derived systems. This represents a significant knowledge gap regarding its impact on non-tumorigenic tissues.
To address this gap, we hypothesized that V. meridionale extract may also exert protective effects against ROS-induced skin damage in human keratinocytes. Accordingly, the aim of this study was to evaluate the in vitro cytoprotective capacity of the extract and its ability to mitigate oxidative damage. In addition, an in silico simulation was performed to identify possible interactions between V. meridionale anthocyanins and proteins involved in oxidative stress regulation, including both antioxidant and pro-oxidant targets. The findings provide mechanistic insights into the antioxidant potential of this species and support its integration into skin cancer prevention strategies.
Materials and methods
Preparation of Vaccinium meridionale extract
Fresh V. meridionale fruits were washed with water and disinfected using a 50-ppm aqueous sodium hypochlorite solution for 1 min. Whole fruits were air-dried at room temperature for 24 h and subsequently dehydrated in a Memmert oven (model UN55, Germany) at 40 °C ± 2 °C until the moisture content fell below 7%. The dehydrated material was pulverized using an industrial grinder and sieved to obtain particles ≤250 μm.
Extraction followed a modified protocol adapted from Natolino and Da Porto. 21 Briefly, 300 mg of the powdered sample was mixed with 1.5 mL of a water–ethanol solution (1:1, v/v) and vortexed at 300 rpm for 30 s. Ultrasonic extraction was performed in a GT-SONIC bath (model GT-1730QTS) at 33 kHz and 25 °C. Extraction time was evaluated at 15-min intervals up to 120 min to determine maximum yield. The extracts were centrifuged at 6000 rpm for 10 min at 25 °C. All experiments were conducted in triplicate, and optimal conditions were selected based on the highest total phenolic content.
Chemical characterization of Vaccinium meridionale extract
The total phenolic content of the extract was quantified using the Folin–Ciocalteu method. A gallic acid calibration curve (10, 20, 40, 60, 80, and 100 μg/mL) was prepared, yielding an R2 of 0.9937. The extract was diluted (1:100, v/v) in three solvent samples. The reaction mixture consisted of 2250 μL of distilled water, 300 μL of diluted sample, 300 μL of 10% Na2CO3, and 150 μL of Folin reagent. A blank containing 450 μL of distilled water was prepared in parallel. Mixtures were incubated in darkness at 25 °C for 60 min. Absorbance was measured at 760 nm using an Agilent 8453 UV–Visible spectrophotometer. All measurements were performed in triplicate. Results were expressed as mg of gallic acid equivalents per 100 g of fresh fruit (mg GAE/100 g FF). 22
The total anthocyanin content was determined using the pH differential spectrophotometric method, based on pigment structural changes at pH 1.0 (colored) and pH 4.5 (colorless). Methanolic extracts were diluted in potassium chloride buffer (pH 1.0) and sodium acetate buffer (pH 4.5). Absorbance was recorded at the maximum absorption wavelength (λmax = 515 nm) and at 700 nm. Anthocyanin concentration (C) was calculated according to the Beer–Lambert law (equation (1)):
Where C is the molar concentration, A is the absorbance, ε is the molar absorptivity or extinction coefficient, and L is the path length (cm). The concentration in mg/L was obtained by multiplying by the pigment molecular weight and dilution factor. The total anthocyanin content was calculated using the molecular weight and molar absorptivity of the predominant anthocyanin pigment. 23
Determination of antioxidant capacity
The antioxidant capacity was measured using the Oxygen Radical Absorbance Capacity (ORAC) assay, following the protocol described by Vélez et al. 22 with minor modifications. A Trolox calibration curve (5, 10, 25, 50, 100, 150, and 200 μM) was prepared in 10 mM phosphate buffer (pH 7.4). In a 96-well microplate (Costar, USA), fluorescein, phosphate buffer, Trolox dilutions, and the sample extracts (diluted 1:200 in phosphate buffer) were added sequentially.
The mixture was incubated at 37 °C for 30 min. After incubation, an AAPH solution was added to each well. Fluorescence was measured every 2 min for 2 h. The excitation and emission wavelengths were set at 485 nm (slit width 5 nm) and 520 nm (slit width 13 nm), respectively, using a 1% attenuator. Measurements were performed in triplicate. The results were expressed as μmol TE per g of sample.
Cell culture
Human keratinocytes (HaCaT) derived from normal skin were cultured in Dulbecco's Modified Eagle Medium (DMEM) supplemented with 5% Fetal Bovine Serum (FBS), 100 μg/mL penicillin, and 100 μg/mL streptomycin. Cultures were incubated at 37 °C in a humidified 5% CO2 atmosphere. Subculturing was performed every 2–3 days by seeding approximately 2 × 105 cells in 5 mL of culture medium.
MTT assay
Cells were seeded in 96-well microplates at 1 × 104 cells/well in DMEM supplemented with 5% FBS and maintained under standard culture conditions. Extract treatments were performed in triplicate at the indicated concentrations. After 24 h of exposure, 10 μL of MTT solution (final concentration 0.5 mg/mL) was added to each well, followed by incubation for 2 h in the dark. Formazan crystals were dissolved by adding 100 μL of isopropanol–HCl (0.04 M). Absorbance was measured at 571 nm using an ELISA microplate reader. Cell viability was expressed as a percentage relative to the negative control.
Evaluation of mitochondrial potential
To assess mitochondrial membrane potential and cytoplasmic membrane integrity following treatment with V. meridionale extract (0.16 mg/mL), 0.5 mM H2O2, or their combination, cells were resuspended in 500 μL of Phosphate-Buffered Saline (PBS) containing 50 nM DiOC6. The suspension was incubated in the dark at room temperature for 20 min. A total of 10,000 events were analyzed by flow cytometry.
Detection of mitochondrial ROS
Mitochondrial ROS levels were quantified using the fluorochrome MitoTracker™ Red CMXRos. After treatment with V. meridionale extract (0.16 mg/mL), 0.5 mM H2O2, or their combination, approximately 5 × 105 cells were resuspended in PBS and incubated with the fluorochrome at a final concentration of 3 μM for 30 min. Cells were washed twice with PBS to remove excess dye. Fluorescence was measured using a BD LSRFortessa flow cytometer, and ROS levels were quantified accordingly.
Comet assay
DNA Single-Strand Breaks (SSBs) were evaluated using the alkaline comet assay as described by Tice et al. 24 Briefly, after treatment with V. meridionale extract (0.16 mg/mL), aliquots containing 2 × 104 cells were mixed with Low-Melting-Point Agarose (LMPA, ∼37 °C) and spread onto glass slides. Untreated cells served as the negative control, whereas cells treated with 0.5 mM H2O2 were used as the positive control. Cells were lysed, and DNA was denatured in pH 13 buffer, followed by alkaline electrophoresis. Slides were stained with a fluorescent DNA intercalating agent and visualized under a fluorescence microscope. Images were captured with a digital camera attached to the microscope, and the percentage of DNA in the comet tail was quantified using CaspLab - Comet Assay Software Project (http://casp.sourceforge.net).
Cell cycle analysis
Cell cycle progression was assessed using PI staining. After treatment with V. meridionale extract (0.16 mg/mL), 0.5 mM H2O2, or their combination, HaCaT cells were washed twice with PBS, trypsinized, and fixed in cold 70% ethanol for at least 24 h. Cells were then incubated with RNase (6.7 μg/mL) and PI (5 μg/mL) at room temperature for 30 min. Flow cytometry analysis was performed on 10,000 events using a BD LSRFortessa cytometer. Cell cycle distribution was calculated using FlowJo software.
Molecular docking
An initial molecular docking screening was performed to identify potential interactions involving two anthocyanins reported in V. meridionale: cyanidin-3-glucoside and delphinidin-3-glucoside. Their molecular structures were retrieved from PubChem and energy-minimized using Avogadro 1.2. Five proteins associated with oxidative activity—nitric oxide synthase-2 (NOS2), mitochondrial superoxide dismutase-2 (SOD2), thioredoxin isoform-2 (TXN2), glutathione peroxidase-1 (GPX1), and catalase (CAT)—were selected, 25 and their crystallographic structures were obtained from the RCSB Protein Data Bank. Only structures with resolution below 3 Å and minimal R-free and outlier values were considered.
Docking simulations were carried out using AutoDock Vina with default parameters. The docking grid was positioned at the protein active site based on the location of its native ligand. Affinity values below −7 kcal/mol were considered significant. A second docking study was conducted to further examine the highest-affinity interactions within the complex. The anthocyanins from the initial screening were retained, and chlorogenic acid was incorporated for comparison due to its reported antioxidant activity 26 and its presence in V. meridionale. The inducible NOS2 inhibitor ethyl 4-[(4-methylpyridin-2-yl)amino]piperidine-1-carboxylate (AT2) was used as a reference compound to facilitate comparison of relative binding affinities. All ligands were drawn using ChemSketch, and their 3D structures were optimized using Chimera 1.14.
Given the presence of a heme group in the catalytic center of NOS2, which contains an iron atom not parameterized in the Amber force field, the protein–cofactor complex was reconstructed. The heme group was removed while preserving its coordinates, and the iron atom was extracted. The charges of the organic moiety (−4, considering the anions on carboxylates and pyrroles) were recalculated using Chimera 1.14. The iron topology parameters (+2) were manually entered following Li et al. 27 The protein–cofactor adduct was subjected to a 10 ns Molecular Dynamics (MD) simulation to evaluate its stability. 28 The optimized NOS2 structure was subsequently used for docking in AutoDock Vina. The grid box was centered at x = 54.4, y = 19.6, z = 83.8, with dimensions of 25 × 25 × 25 Å. For each ligand, the conformation with the lowest docking energy was selected for MD studies.
Molecular dynamics simulations
MD simulations were performed using GROMACS 2018.2 28 on the NMRbox server 29 with the protein–ligand complexes exhibiting the highest affinity scores. Molecular topologies for the selected ligands and the cofactor 5,6,7,8-tetrahydrobiopterin were generated using the semi-empirical quantum chemistry program SQM implemented in ACPYPE.30,31 All-atom MD simulations employed the Amber99SB-ILDN force field32–34 and the TIP3P water model. 35
The protein–ligand complexes were solvated in a cubic box with a 1.0 nm water layer on all sides. Na+ and Cl− ions were added randomly to neutralize the system and achieve an ionic concentration of 0.15 M. Each complex underwent energy minimization using the steepest descent method, followed by equilibration with the Berendsen thermostat 36 and the Parrinello–Rahman barostat. 37 A 100 ns MD simulation was then performed for each system.
Throughout the simulation, the Root-Mean-Square Deviation (RMSD) was monitored. In addition, the binding free energy of the protein–ligand complexes was calculated using the Linear Interaction Energy (LIE) equation
38
during the final 30 ns.
For this study, the interaction energy values (VLJ)bound and (VCL)bound were analyzed separately to determine the individual contributions of the protein and the heme group.
Statistical analysis
Analysis of Variance (ANOVA) was conducted using GraphPad Prism software. Post hoc comparisons were performed using Fisher's Protected Least Significant Difference (FPLSD) test. A p-value < 0.05 was considered statistically significant.
Results
Chemical characterization and antioxidant capacity of V. meridionale extract
The ethanolic extract of V. meridionale exhibited a high concentration of bioactive compounds. Total polyphenol content was quantified at 15.89 ± 0.9 mg GAE/100 g, while total monomeric anthocyanins reached 6.06 mg cyanidin-3-glucoside equivalents/100 mL. The extract also demonstrated strong antioxidant activity, as measured by the ORAC assay, with a value of 892.4 ± 129.5 μmol TE/g. These results confirm the extract's potential as a rich source of antioxidants.
Cytoprotective potential of V. meridionale extract
Exposure to the extract alone did not reduce cell viability below 70%, the cytotoxicity threshold defined by ISO 10993-5 (Table 1). This indicates that V. meridionale extract was not cytotoxic under the tested conditions. The cytoprotective potential of the extract against oxidative stress was assessed in HaCaT cells exposed to hydrogen peroxide (H2O2). Treatment with 0.5 mM H2O2 reduced cell viability to 69%, as determined by the MTT assay. In contrast, pretreatment with the extract at concentrations of 0.16 mg/mL and 0.31 mg/mL significantly attenuated H2O2-induced cytotoxicity, demonstrating its protective potential. Figure 1 illustrates the reduction in cell viability following H2O2 exposure and the mitigation observed after extract pretreatment.
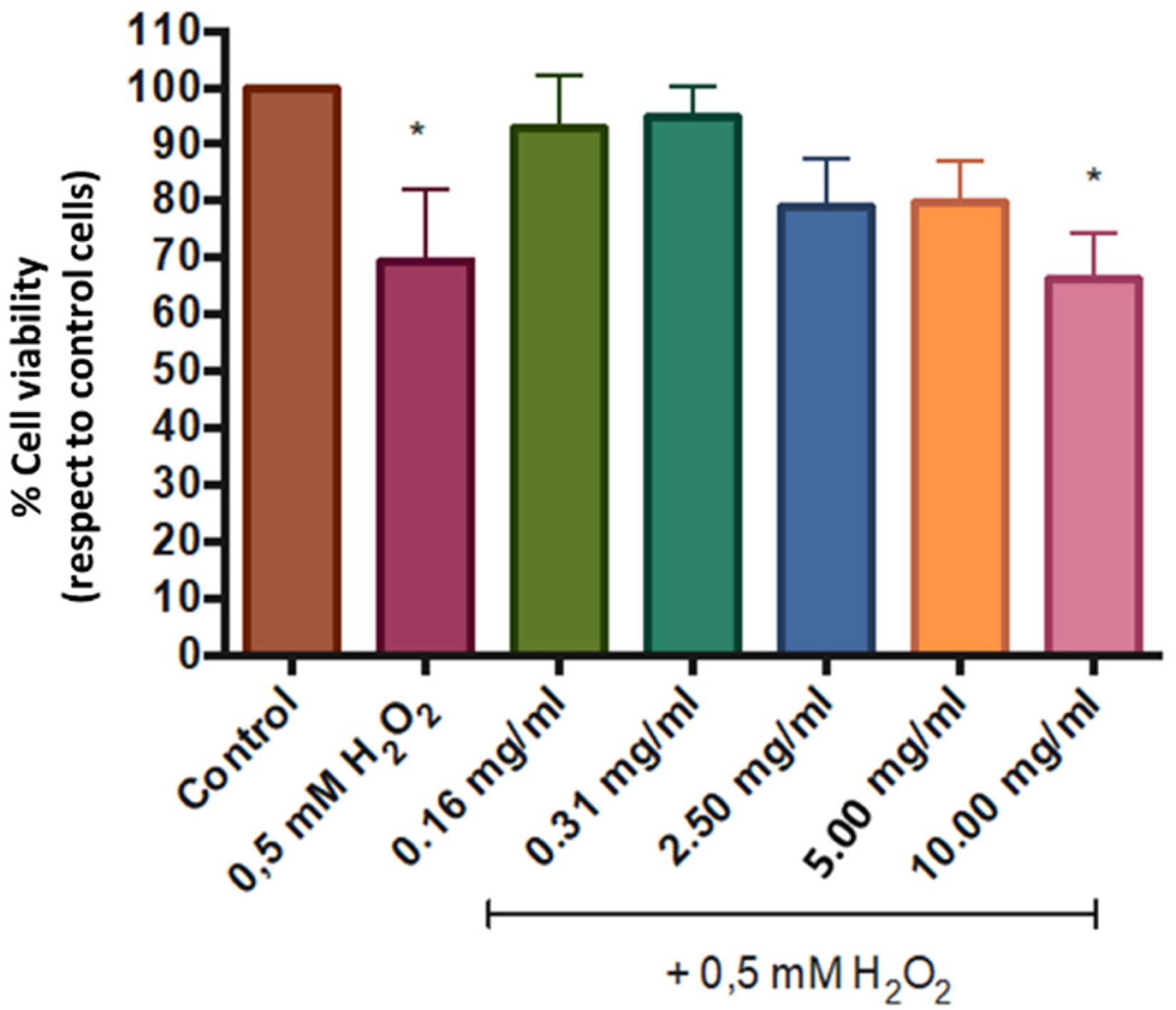
Effect of V. meridionale extract on cell viability. HaCaT cells were pretreated with varying concentrations of the extract for 24 h. The medium was then removed, and cells were exposed to 0.5 mM H2O2 for 1 h. Viability was evaluated using the MTT assay. Untreated cells served as the control. Results are expressed as mean ± SEM from three independent experiments. One-way ANOVA identified significant differences compared with untreated cells (p ≤ 0.05).
Cytotoxicity of V. meridionale extract in HaCaT cells measured by MTT assay.

To further evaluate the protective effect of the lowest extract concentration (0.16 mg/mL), DiOC6 staining assays were performed to assess mitochondrial polarization, a parameter directly related to cell viability. Figure 2 shows representative histograms and quantification of the Mean Fluorescence Intensity (MFI) obtained by flow cytometry after extract pretreatment and subsequent exposure to 0.5 mM H2O2. Treatment with H2O2 alone reduced fluorophore uptake, indicating mitochondrial depolarization and cytotoxicity. In contrast, pretreatment with 0.16 mg/mL of the extract for 24 h appeared to mitigate mitochondrial depolarization. However, due to variability among replicates and the preliminary nature of these in vitro results, the findings should be interpreted as indicative rather than conclusive of a protective effect.
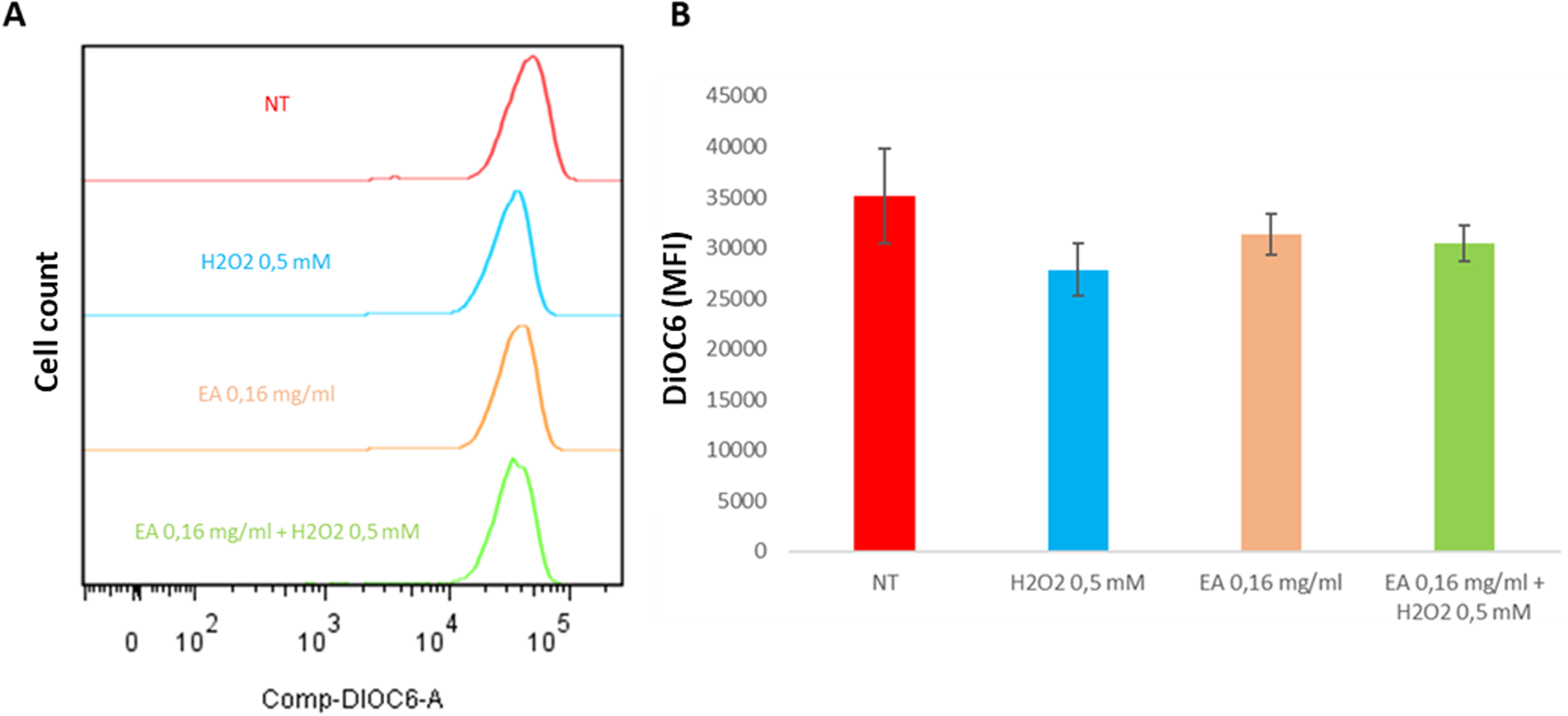
Effect of V. meridionale extract on mitochondrial permeability. HaCaT cells were pretreated with 0.16 mg/mL extract for 24 h. The medium was then removed, and cells were exposed to 0.5 mM H2O2 for 1 h. DiOC6 uptake was subsequently assessed, and fluorescence was quantified by flow cytometry. (a) Representative histograms showing DiOC6 MFI for each treatment. (b) MFI values expressed as mean ± SEM from three independent experiments. One-way ANOVA detected no significant differences compared with untreated cells.
Antioxidant protection of V. meridionale extract
To determine whether the protective effect against H2O2-induced cytotoxicity is associated with antioxidant activity, mitochondrial ROS levels were quantified using the MitoTracker™ Red CMXRos probe—a red fluorescent dye that oxidizes in response to mitochondrial ROS—and analyzed by flow cytometry. As shown in Figure 3, pretreatment with 0.16 mg/mL of V. meridionale extract reduced oxidative stress in HaCaT cells exposed to 0.5 mM H2O2. These observations are based on a single experiment analyzing 10,000 events per condition. Therefore, while the results suggest a trend toward antioxidant protection, they are preliminary and should be interpreted as indicative rather than conclusive.
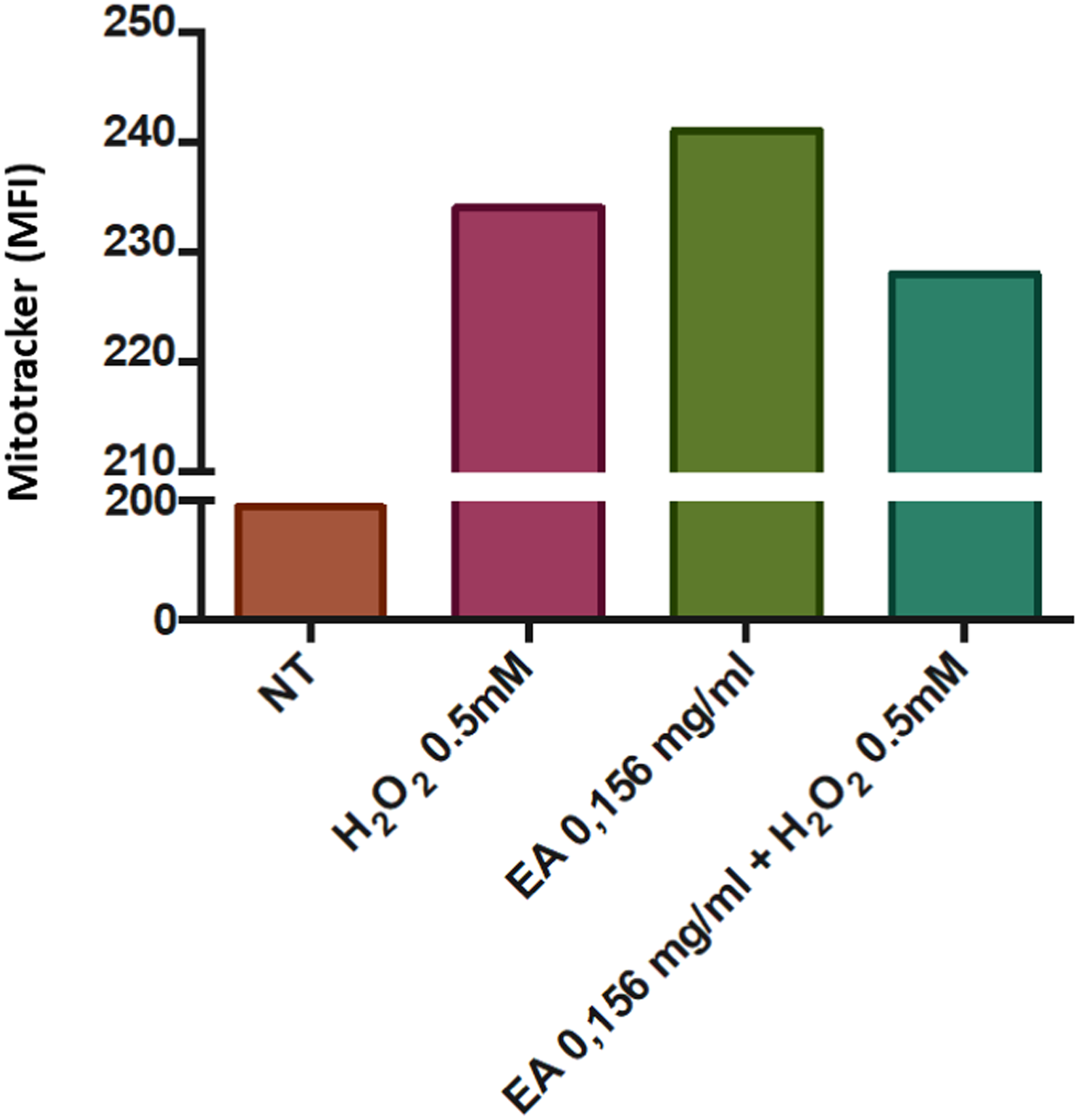
Quantification of mitochondrial ROS. HaCaT cells were pretreated with 0.16 mg/mL extract for 24 h. The medium was then removed, and cells were exposed to 0.5 mM H2O2 for 1 h. Mitochondrial ROS levels were assessed using the MitoTracker™ Red CMXRos probe and quantified by flow cytometry.
Genotoxic protection by V. meridionale extract
To determine whether V. meridionale extract protects against ROS-induced DNA damage, the alkaline comet assay was used to quantify the percentage of DNA in the comet tail in cells treated with 0.5 mM H2O2, with or without pretreatment with 0.16 mg/mL extract. As illustrated in Figure 4, extract pretreatment reduced tail DNA from 12.6% to 10%, indicating attenuation of H2O2-induced SSBs in HaCaT cells.
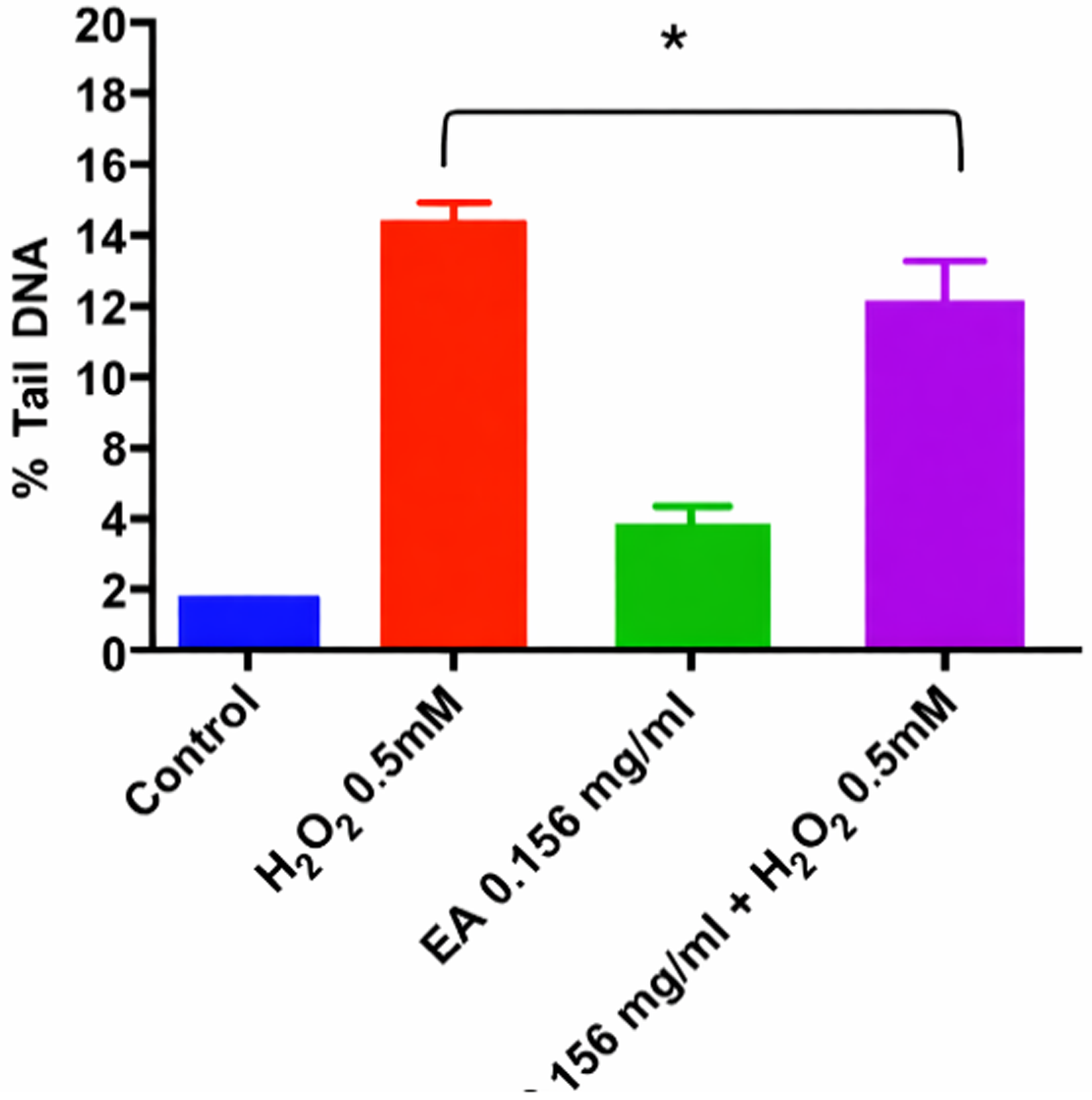
Quantification of DNA damage. HaCaT cells were pretreated with 0.16 mg/mL extract for 24 h. The medium was then removed, and cells were exposed to 0.5 mM H2O2 for 1 h. SSBs were assessed using the alkaline comet assay. Results are expressed as mean ± SEM from three independent data sets. One-way ANOVA revealed a statistically significant difference between H2O2-treated cells and extract pretreated cells (p ≤ 0.05).
As an additional indicator of DNA damage, DNA content was quantified by flow cytometry. As shown in Figure 5, exposure to 0.5 mM H2O2 increased the proportion of cells in the G2/M phase to 29.9%, consistent with activation of DNA damage checkpoints. Conversely, cells pretreated with 0.16 mg/mL V. meridionale extract before H2O2 exposure showed a reduction in G2/M accumulation to 22.2%. Cell cycle distribution was evaluated by PI staining and flow cytometry, analyzing 10,000 events per treatment in a single experiment. Under these conditions, the extract appeared to attenuate H2O2-induced alterations in cell cycle progression.
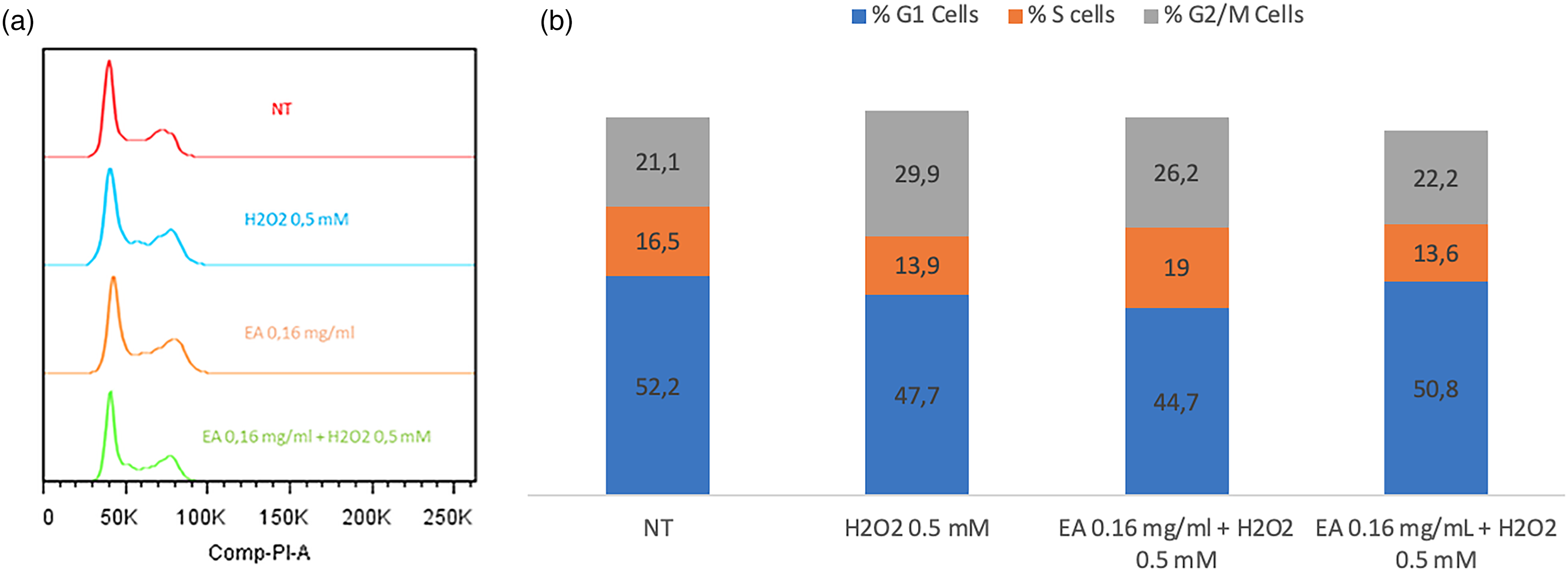
Quantification of DNA content. HaCaT cells were pretreated with 0.16 mg/mL extract for 24 h. The medium was then removed, and cells were exposed to 0.5 mM H2O2 for 1 h. DNA content was assessed using PI staining and flow cytometry. (a) Representative histograms showing cell cycle distribution for each treatment. (b) Percentage of cells in each phase of the cell cycle. Data correspond to a single experiment with 10,000 events acquired per treatment.
In Silico analysis of anthocyanin–protein interactions
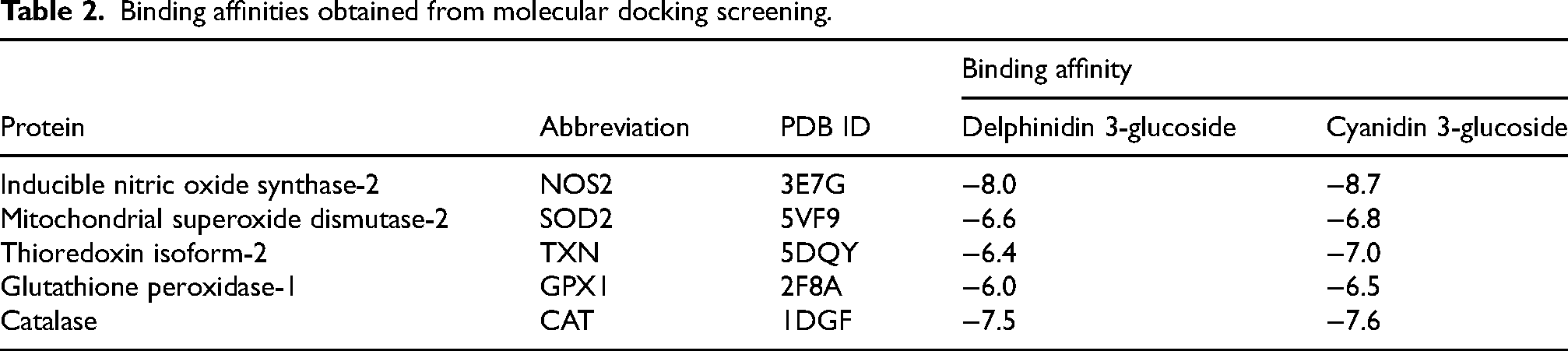
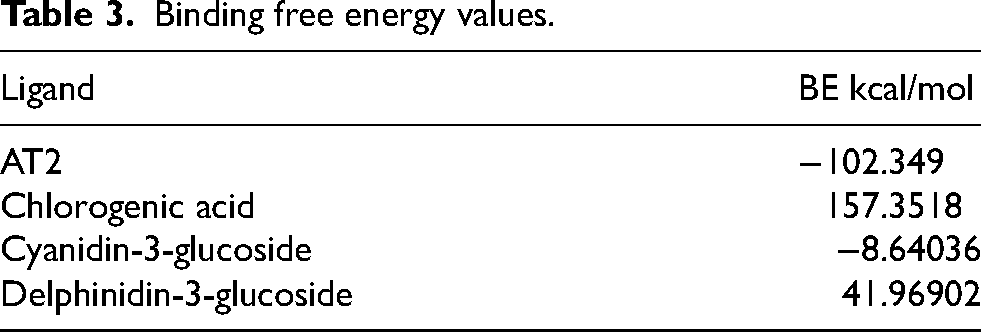
The initial docking screening yielded the binding affinities listed in Table 2 and the binding free energy values presented in Table 3.
Binding affinities obtained from molecular docking screening.
Binding free energy values.
Among the evaluated proteins—which included antioxidant enzymes (SOD2, TXN, GPX1, and CAT) and the pro-oxidant enzyme NOS2—NOS2 exhibited the highest binding affinity. For this reason, subsequent docking analyses focused on its interactions with cyanidin-3-glucoside and delphinidin-3-glucoside, the main phenolic constituents reported in V. meridionale. 39 Both compounds have demonstrated antioxidant activity and potential to modulate oxidative stress. Chlorogenic acid and AT2 were also included for comparative purposes.
The results of the molecular docking, as illustrated in Figure 6, suggest that chlorogenic acid and AT2 establish favorable cation–π interactions between the metallic center in the heme group and their aromatic moieties. In addition, chlorogenic acid forms hydrogen bonds with the heme group and the HB4 cofactor. In contrast, cyanidin-3-glucoside and delphinidin-3-glucoside adopt similar conformations within the NOS2 pocket, stabilized primarily by hydrogen bonding with the HB4 cofactor through hydroxyl groups in the glucoside core. Notably, the iron atom does not appear to contribute to stabilization of these compounds.
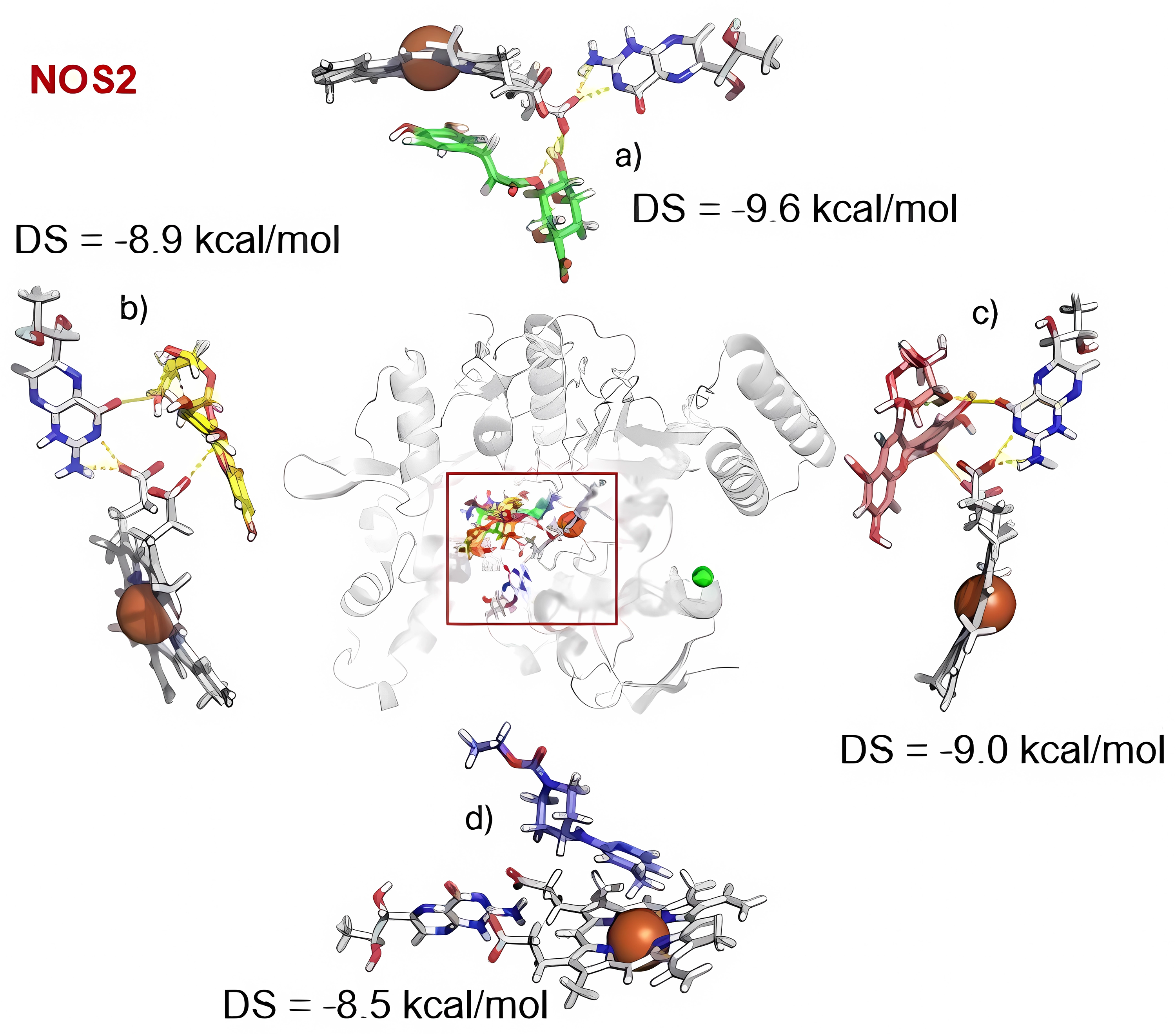
Molecular docking conformations and scores for (a) chlorogenic acid, (b) cyanidin-3-glucoside, (c) delphinidin-3-glucoside, and (d) AT2.
Molecular dynamics simulations
The RMSD behavior was analyzed to determine the stability of the ligands in the protein pocket. After a few initial stabilization frames, all ligands remained stable throughout the simulations (Figure 7), suggesting their potential to inhibit NOS2. However, binding free energy calculations are required to further validate this observation.
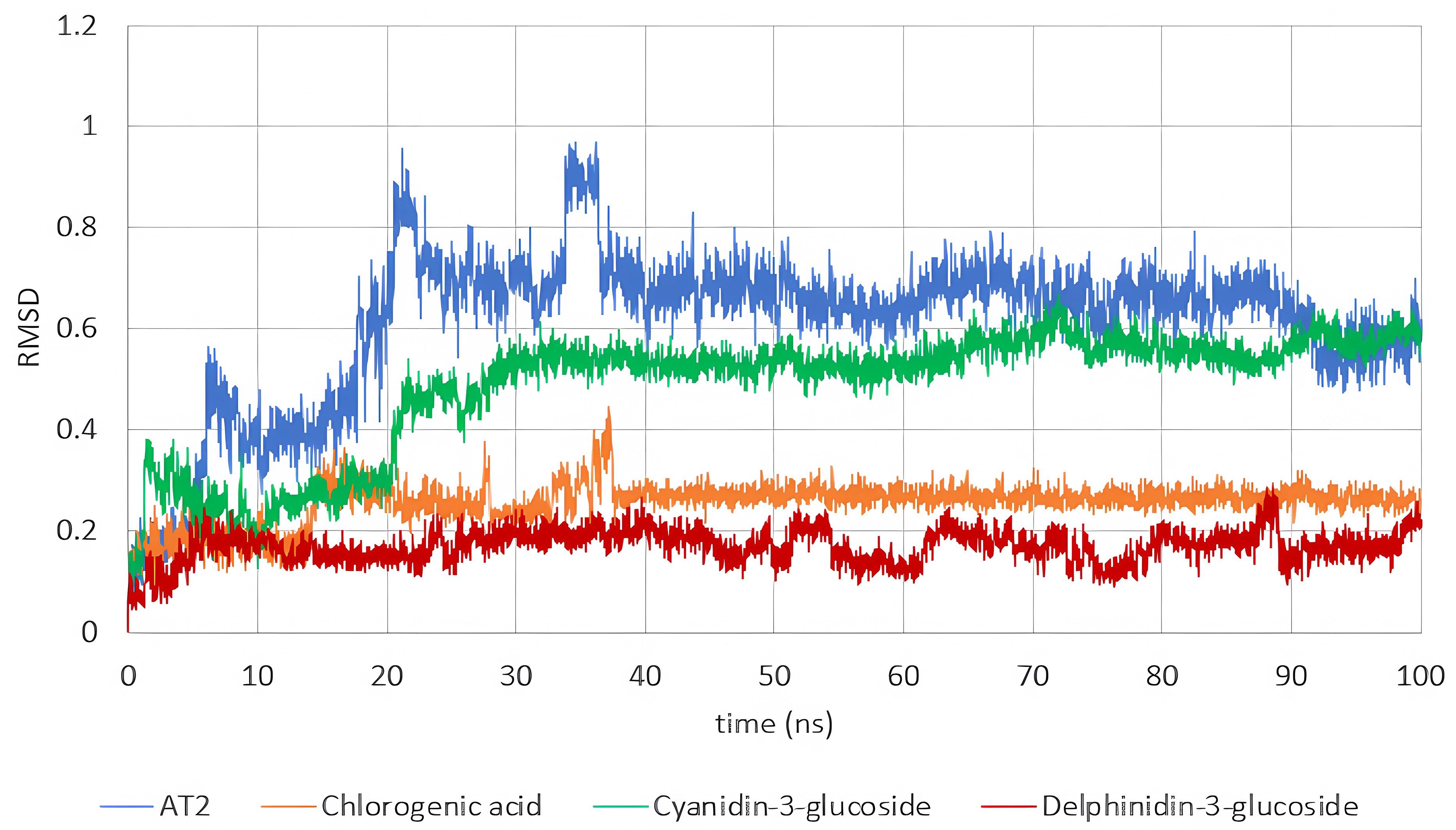
RMSD trajectories for the three V. meridionale ligands and the reference inhibitor (AT2).
Hydrogen bond analysis (Figure 8) revealed notable differences between the reference inhibitor (AT2) and the anthocyanins. Cyanidin-3-glucoside and delphinidin-3-glucoside formed the highest number of hydrogen bonds during the simulations, suggesting that these two ligands have the strongest potential to inhibit NOS2.
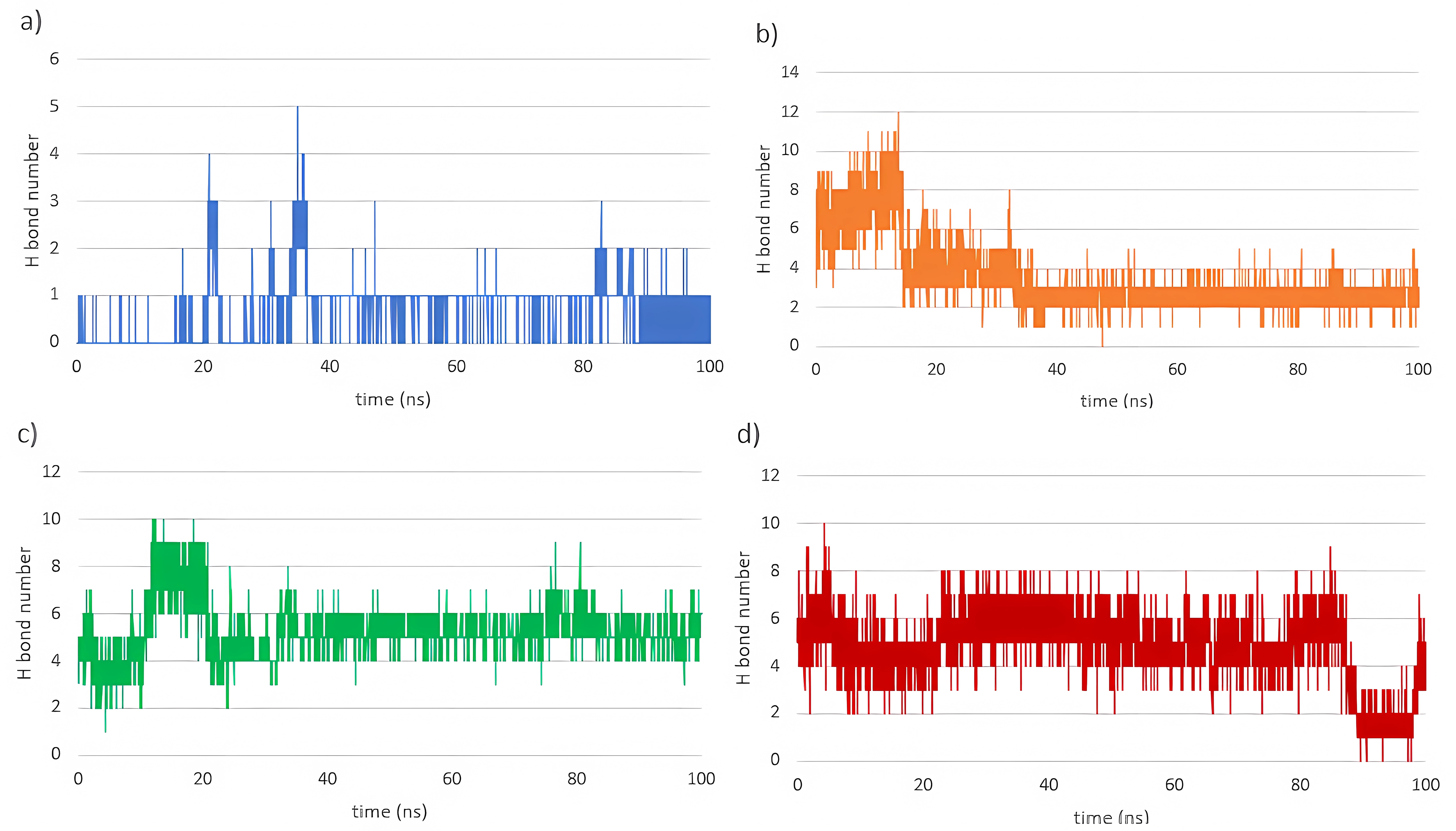
Number of hydrogen bonds formed throughout the simulation: (a) AT2, (b) chlorogenic acid, (c) cyanidin-3-glucoside, and (d) delphinidin-3-glucoside.
Binding free energy calculations for each complex were performed during the final 30 ns of the simulations (Table 2). The results show that AT2 and cyanidin-3-glucoside exhibited favorable binding free energy values, whereas chlorogenic acid and delphinidin-3-glucoside displayed positive values. These findings indicate that although chlorogenic acid and delphinidin-3-glucoside can stabilize within the active site, their high-water solubility, driven by their hydroxyl groups, makes them more stable in the aqueous medium than within the protein.
Among the studied ligands, cyanidin-3-glucoside is the only compound with a favorable binding profile for NOS2 inhibition. This finding prompted a structural comparison with delphinidin-3-glucoside to explain the differences in their behavior. Structurally, the two ligands differ by a single hydroxyl group at position 5′ (Figure 9), which is absent in cyanidin. Considering that high water solubility is a decisive factor in ligand stability within the NOS2 pocket, and that hydroxyl groups enhance solubility in water, the additional hydroxyl group in delphinidin-3-glucoside likely increases its preference for the aqueous medium, thereby reducing its affinity for the protein pocket.
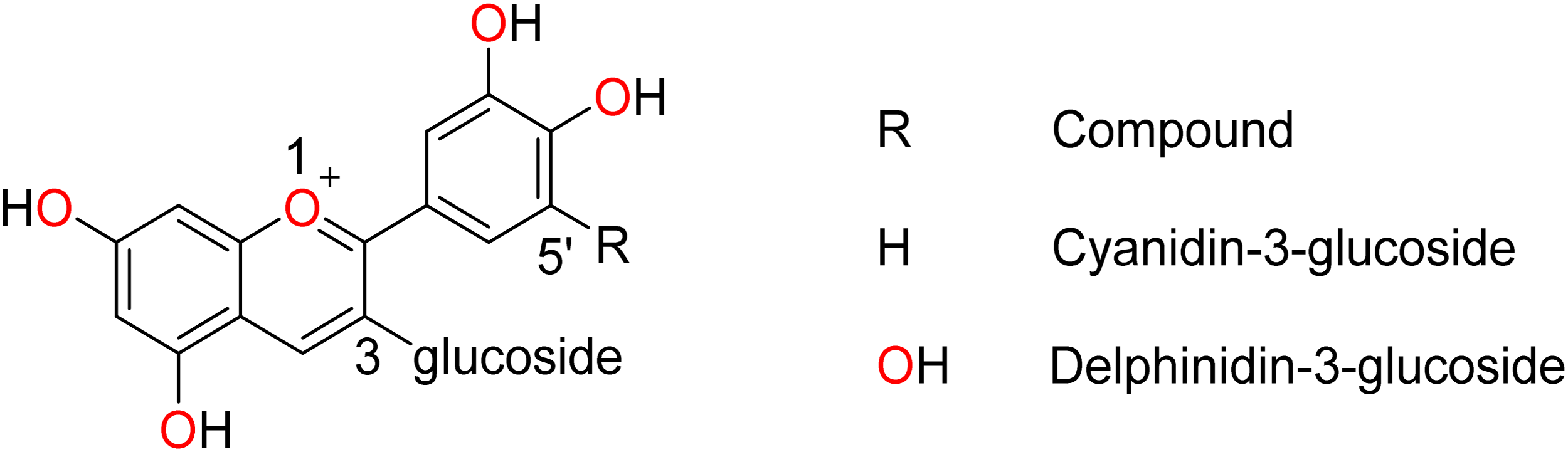
General structure of anthocyanins, highlighting the hydroxyl group difference at position 5’.
Discussion
This study demonstrates the protective effect of V. meridionale extract against oxidative stress in HaCaT human keratinocytes, highlighting its potential as a natural alternative to mitigate ROS-induced damage. In the skin, oxidative stress occurs when prolonged and excessive free radical generation depletes the endogenous antioxidant defense system. This process contributes not only to skin carcinogenesis but is also associated with other dermatological conditions such as psoriasis, vitiligo, and acne vulgaris. 40 Skin cancer is among the most prevalent malignancies worldwide, with incidence rates increasing steadily. According to WHO data, one in three cancer diagnoses corresponds to skin cancer. 2
The primary risk factors for skin cancer include prolonged sun exposure and a history of sunburns. UV radiation promotes carcinogenesis through two main mechanisms: direct DNA damage and oxidative stress. 41 This underscores the need to evaluate chemopreventive agents that are both effective and accessible to populations frequently exposed to UV radiation. Consequently, research on antioxidants with preventive and therapeutic applications has attracted growing attention. In this context, blueberries have been extensively studied for their antioxidant and anti-inflammatory properties. 42
Berries are rich in bioactive compounds, including minerals, vitamins, dietary fiber, and polyphenolic phytochemicals. 43 Among these, species of the Vaccinium genus, such as V. meridionale, exhibit high phenolic and anthocyanin contents, particularly in methanolic and aqueous extracts. For instance, methanolic extracts have shown total phenolic concentrations of 758.6 ± 62.3 mg GAE/100 g fresh weight and anthocyanin levels of 329.0 ± 28.0 mg cyanidin-3-glucoside equivalents/100 g, 7 while aqueous extracts have reported 4409.78 ± 63.05 mg GAE/100 mL and 106.57 ± 1.43 mg cyanidin-3-glucoside equivalents/100 mL. 17
In the literature, the antioxidant capacity of V. meridionale extract has been evaluated using multiple assays, including 1,1-diphenyl-2-picrylhydrazyl (DPPH), 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulpfonic acid) (ABTS), Ferric Ion Reducing Antioxidant Power (FRAP), and ORAC. A methanolic extract, for example, exhibited antioxidant activity of 45.5 ± 2.3 μmol TE/g fresh weight (ABTS) and 87.0 ± 17.8 μmol TE/g fresh weight (FRAP). 7 Another study reported antioxidant activity of a polyphenol-rich extract as ABTS 42.65 μmol TE/g, ORAC 49.56 μmol TE/g, DPPH 53.55 μmol TE/g, and FRAP 70.37 μmol TE/g. 44
In other investigations, methanol pulp extracts showed ORAC values of 250.0 ± 32.0 μmol TE/g fresh weight, 10 while aqueous extracts recorded FRAP values of 476.84 ± 18.81 μmol TE/100 mL and ORAC values of 1131.8 ± 45.3 μmol TE/mL. 11 In the present study, characterization of a water–ethanol (1:1) extract of V. meridionale revealed a total phenolic content of 15.89 ± 0.9 mg GAE/100 g and a total monomeric anthocyanin concentration of 6.06 mg cyanidin-3-glucoside equivalents/100 mL. Antioxidant activity measured by ORAC reached 892.4 ± 129.5 μmol TE/g.
High-Performance Liquid Chromatography (HPLC) analyses have identified specific phenolic compounds in V. meridionale extracts, including chlorogenic acid, caffeic acid, cyanidins, and quercetins, all of which have demonstrated anticancer properties.7,13,45 Chlorogenic acid modulates apoptosis and inhibits tumor cell proliferation. Its potential in cancer prevention and treatment has been linked to molecular mechanisms involving intracellular ROS production, a pathway closely associated with apoptosis induction in cancer cells. Its activity has been documented in breast, colon, lung, and prostate cancer models. 46
Cyanidin-3-glucoside and delphinidin-3-glucoside are potent antioxidants capable of scavenging free radicals, thereby reducing oxidative stress and protecting against cellular damage. These compounds neutralize ROS and contribute to the prevention of chronic diseases such as cardiovascular disorders and cancer. 47 Cyanidin-3-glucoside also exhibits iron-chelating activity, reducing free radical formation. Both anthocyanins protect DNA from oxidative fragmentation, preserving genomic stability and preventing mutations.
In addition, they enhance antioxidant enzyme activity, strengthening cellular defense mechanisms. Anthocyanins may further modulate immune responses, regulate gene expression, and inhibit oxidative DNA damage. 48 They reduce UV-induced oxidative damage in keratinocytes, 49 inhibit collagen degradation and inflammatory responses in human dermal fibroblasts via NF-κB-dependent pathways, 50 and decrease melanogenesis in human melanocytes. 51
Extracts of V. meridionale have demonstrated biological activity in multiple cancer cell models. For instance, an aqueous extract (30% v/v) combined with 5-fluorouracil, leucovorin, and oxaliplatin exerted a greater inhibitory effect on SW480 and SW620 colon cancer cell viability than the drugs alone. The extract reduced SW480 cell adhesion and migration and inhibited SW620 cell invasion. Moreover, the combination treatment enhanced suppression of SW620 migration and invasion compared with the drugs alone and decreased MMP-9 levels in SW620 cells. 19
Further studies have supported the antiproliferative effects of V. meridionale extracts in SW480 and SW620 cell lines. An aqueous extract induced dose- and time-dependent growth inhibition, with IC50 values of 32% for SW480 and 38.4% for SW620 at 48 h. A stronger antiproliferative response was observed in SW480 cells, accompanied by cell cycle arrest in S and G2/M phases. In SW620, cell-cycle arrest occurred in G0/G1, indicative of apoptosis. Apoptotic signaling was confirmed by increased phosphatidylserine exposure in both cell lines, without significant mitochondrial depolarization. 17 Collectively, these findings highlight the antiproliferative and pro-apoptotic activity of V. meridionale extract in cancer cells, 52 as well as its high antioxidant capacity associated with polyphenol content, 53 suggesting its potential use as chemopreventive agent.
Specifically related to skin cancer, certain polyphenols, including those found in V. meridionale, have been reported to induce apoptosis and inhibit cell proliferation, angiogenesis, and cell cycle progression. Additionally, they modulate the expression of inflammatory genes and cytokines in melanoma models. 54 For example, fisetin, a flavonoid present in various fruits, has shown antiproliferative properties against melanoma and non-melanoma skin cancers by disrupting the WNT/β-catenin signaling pathway. 55
The results of the present study suggest that V. meridionale extract may play a protective role against oxidative stress by attenuating ROS accumulation and reducing DNA damage. Pretreatment with 0.16 mg/mL extract for 24 h mitigated H2O2 (0.5 mM)-induced cytotoxicity, an effect associated with decreased mitochondrial ROS production and reduced genomic damage. The decline in mitochondrial ROS levels observed after exposure to the extract is consistent with its chemical antioxidant profile and may partially explain the reduction in single-strand breaks (from 12.6% to 10%) and the lower accumulation of cells in the G2/M phase (from 29.9% to 22.2%) following H2O2 challenge.
Together, these observations indicate that the extract may limit oxidative injury and attenuate the activation of DNA damage checkpoints. However, given that some assays were performed with a single repetition, the evidence should be considered preliminary. Further studies incorporating additional biological replicates and mechanistic exploration are required to confirm the cytoprotective and genoprotective potential of V. meridionale extract.
Notably, similar protective responses have been reported in other Vaccinium species. For instance, fruit extracts previously studied increased the viability of HT1080 cells treated with 50 and 100 µg/mL for 48 h before exposure to rotenone-induced oxidative stress. 18 In Bunea et al., an anthocyanin-rich extract inhibited B16-F10 melanoma cell proliferation in a dose-dependent manner. 56 Similarly, the effects of Bluecrop blueberry extract in HaCat cells were evaluated in, 57 reporting reduced transcription of inflammatory genes associated with ROS production, such as TNF-α, IL-1β, IL-8, and IL-6. Moreover, a mixed berry formulation (wild blueberry, cranberry, raspberry seed, and strawberry) was found to suppress ROS generation and TNF-α-induced VEGF expression in human keratinocytes. 58
Furthermore, UVB radiation (100 mJ/cm2) was reported to reduce HaCaT cell viability, disrupt 53BP1, and alter cytoskeletal organization, decreasing cell stiffness Delphinidin (5 or 10 μM) prevented these effects, with post-treatment showing greater efficacy, which highlights its potential in repairing sun-damaged skin. 59 Likewise, cyanidin was found to mitigate UVA-induced ROS production in immortalized fibroblasts. 49 Cyanidin-3-glucoside, has also been identified as an inhibitor of inducible NOS2, 60 a finding particularly relevant to skin cancer, where oxidative stress and chronic inflammation are key drivers of tumor progression. 61 Excess nitric oxide production due to dysregulated NOS2 activity promotes cell proliferation, angiogenesis, and resistance to apoptosis, all hallmarks of carcinogenesis. Additionally, the interaction between nitric oxide and ROS generates highly reactive species such as peroxynitrite, capable of causing DNA lesions and contributing to mutagenesis. 62
The ability of cyanidin-3-glucoside to inhibit NOS2 suggests a dual protective mechanism, reducing oxidative damage in keratinocytes and chronic inflammation associated with tumor progression. Accordingly, V. meridionale extract, which contains high levels of this compound, emerges as a promising natural chemopreventive agent. Cyanidin-3-glucoside may also shield skin cells from UV-mediated damage, a major risk factor for carcinogenesis. These observations underscore the potential of V. meridionale extract not only in prevention but also as a complementary treatment for precancerous skin lesions and the management of skin malignancies. 63
Computational modeling of NOS2 interactions revealed that, among the tested compounds, only cyanidin-3-glucoside exhibited an acceptable binding affinity, although weaker than that of the reference inhibitor. Nevertheless, its interaction with the active site suggests potential NOS2 modulation that could reduce nitric oxide production. 64 Further analysis indicated that, once within the active site, the compound stabilizes through hydrogen bonding within the catalytic pocket. These findings warrant additional investigation aimed at structural optimization to enhance binding affinity and inhibitory efficacy. In summary, while the compounds do not surpass the efficacy of the reference inhibitor, they represent promising candidates for NOS2 modulation and provide a basis for future chemical refinement.
Few studies have explored the benefits of polyphenolic phytochemicals present in Vaccinium berries and their role in protective response mechanisms against ROS-induced skin damage. A limitation of this study is the absence of in vivo validation, which is necessary to confirm the biological relevance of the findings. Moreover, anthocyanins such as cyanidin-3-glucoside may face challenges related to poor bioavailability and rapid metabolism, potentially limiting their clinical effectiveness. Future research should therefore focus on elucidating specific cellular mechanisms, assessing pharmacokinetics and long-term effects in more complex in vivo models, and evaluating potential applications in dermatological formulations.
Conclusion
These results highlight the potential of V. meridionale as a natural protective agent against oxidative stress-related skin damage and support further research on its application in skin cancer prevention. The in silico analysis demonstrated that cyanidin-3-glucoside exhibits a binding profile consistent with inhibition of the pro-oxidant enzyme NOS2, identifying it as a promising candidate for NOS2 modulation. Taken together, these findings suggest that V. meridionale extracts and their bioactive constituents may serve as valuable components in the development of skincare formulations. Future studies, including in vivo models and clinical trials, are warranted to validate these effects and to explore their translational potential in dermatological applications.
Footnotes
Acknowledgments
The authors gratefully acknowledge the financial support provided by Instituto Tecnológico Metropolitano (ITM) under grant ID P20247. The authors also thank ITM Translation Agency (traducciones@itm.edu.co) for editing the manuscript.
ORCID iDs
Ethical statement
This research was conducted using in vitro models with established cell lines and in silico simulations. No animal models, human subjects, or personal data were involved in this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support of Instituto Tecnológico Metropolitano (ITM) under grant ID P20247. The authors also thank ITM Translation Agency (traducciones@itm.edu.co) for editing the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Not applicable.
Additional information
No additional information is available for this paper.